Abstract
Pseudo-occlusion (PO) of the cervical internal carotid artery (cICA) can be caused by distal ICA occlusion. We explored the clinical impact of PO after mechanical thrombectomy (MT). Patients who underwent MT to treat distal ICA occlusions between July 2012 and March 2018 were reviewed. A cICA-PO was defined as when single phase computed tomography angiography (CTA) revealed a gradual decline in contrast above the level of the carotid bulb. We investigated the relationship between a cICA-PO and outcome; we also explored the association between successful recanalization and outcome. Among 71 patients, 40 (56.3%) exhibited cICA-PO and more likely to experience poor outcomes (80.0% vs. 25.8%, P < 0.001), hemorrhagic transformation (32.5% vs. 9.6%, P = 0.01), and a lower rate of successful recanalization (65.0% vs. 90.3%, P = 0.014) than the non-PO group. In binary logistic regression, a cICA-PO was independently associated with a poor outcome (odds ratio, 4.278; 95% CI, 1.080–33.006; P = 0.045). In the cICA-PO group, all patients who failed recanalization (n = 15) experienced poor outcomes, as did 69.2% of patients in whom recanalization was successful (P = 0.018). cICA-POs are common and have worse outcomes than non-PO patients. Patients with cICA-POs are more likely to exhibit poor outcomes after MT, particularly when recanalization fails.
Similar content being viewed by others
Introduction
Recent randomized controlled trials have shown that intra-arterial mechanical thrombectomy (MT) effectively and safely reduces morbidity and mortality in patients who present with acute ischemic strokes caused by large-vessel occlusions1,2,3,4,5. In the cited studies, computed tomography angiography (CTA) was most commonly used to detect and localize large artery occlusions.
A pseudo-occlusion (PO) of the cervical internal carotid artery (cICA) mimics a true occlusion in CTA but the artery is patent when digital subtraction angiography (DSA) is performed during endovascular treatment6. The underlying cause of a cICA-PO is sluggish or absent contrast flow caused by distal occlusion of the intracranial vasculature7. Several previous studies have reported that cICA-POs develop in 6–15% of patients with anterior circulation strokes and in almost 50% of those with distal ICA occlusions7,8,9. Recently, several studies have used various CTA protocols featuring delayed-phase imaging, or four-dimensional CTA after CT perfusion, to enhance diagnostic accuracy6,8,10,11,12,13. Here, we explored the relationships between cICA-POs in patients with acute distal ICA occlusions and the clinical outcomes after MT.
Methods
Study population and clinical assessment
We retrospectively reviewed consecutive patients with acute ischemic strokes who underwent reperfusion therapy between July 2012 and March 2018. Our data collection protocol was approved by the Institutional Review Board of Ajou University hospital (approval no. AJIRB-MED-MDB-18-301) and implemented in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments. The need for written informed consent was waived by the Institutional Review Board, given the retrospective nature of the study. We enrolled patients who underwent non-contrast CT (NCCT), CTA, DSA, catheter exploration, and MT; exhibited complete distal ICA occlusion (from the paraophthalmic to the terminal ICA segment) on DSA; and had baseline modified Rankin scale (mRS) scores ≤2. Patients for whom NCCT or CTA data were of poor quality, and those with true cICA tandem occlusions or atherosclerotic steno-occlusive changes of cICA evident in DSA, were excluded. We collected demographic, clinical, laboratory, and imaging data including age; sex; comorbidities such as hypertension, diabetes mellitus, coronary heart disease, hyperlipidemia, and/or atrial fibrillation; smoking history; time from stroke onset to puncture; time from puncture to final recanalization or angiography; National Institutes of Health Stroke Scale (NIHSS) score; hemorrhagic transformation status; 3-month mRS score; and stroke cause as determined by the Trial of ORG 10172 in Terms of the Acute Stroke Treatment (TOAST) classification.
The primary outcome parameter was the modified Rankin scale (mRS) score at 3 months after mechanical thrombectomy; a poor outcome was defined as a 3-month score of 4–6. The secondary outcome parameter was successful recanalization using the modified Thrombolysis in Cerebral Infarction (mTICI) scale (“successful” was defined as an mTICI score of 2b or 3) Hemorrhagic transformation (type 1 or 2 parenchymal hemorrhage) was identified via susceptibility-weighted magnetic resonance imaging or CT during follow-up14.
Image acquisition and analyses
Our institutional stroke imaging protocol features NCCT of the head (5 mm slice thickness; 120 kV) followed by contrast-enhanced head CT (5 mm slice thickness; 120 kV). Single-phase CT angiography (100 kV, 180 mA; 10 mm slice thickness, 2 mm axial reconstruction increment) was performed using an automatic bolus-tracking technique from the aortic arch to the vertex; contrast was intravenously injected by a power injector at 5 mL/s followed by a saline flush. All CT procedures featured automated tube current modulation; we obtained multiplanar, maximum-intensity projection reformations. Endovascular procedures were performed using a biplane angiographic unit (Integris Allura; Philips Healthcare, Best, the Netherlands). An 8-F balloon-tipped guidance catheter was positioned in the common carotid artery or the proximal cICA prior to initial angiographic evaluation. Then the ICA was explored using a 0.014-inch guidewire within a 0.021-inch microcatheter. cICA patency (and hence PO status) following ICA recanalization was confirmed by complete angiography.
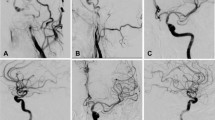
Two experienced interventional neuroradiologists independently reviewed the CTA images to detect cICA-POs, which were considered present when a gradual contrast decay (a flame-shaped leading contrast edge) was evident in the ICA above the level of the carotid bulb, in the absence of plaque around the bulb, and the vessel was normal according to angiographic exploration but a distal ICA occlusion was apparent. Subsequently, DSA images were re-examined to determine whether the cICA was in fact patent (Fig. 1). Axial angiography was used to derive collateral CTA scores of 0 to 5, based on single-phase CTA images of the ischemic and asymptomatic hemispheres: Grade 5, normal or increased prominence/extent of pial vessels; Grade 4, slightly reduced prominence/extent; Grade 3, moderately reduced prominence/extent; Grade 2, decreased prominence/extent, and regions with no vessels; Grade 1, only a few vessels; Grade 0, no vessels15.
Example of a cervical ICA pseudo-occlusion on CTA with a non-corresponding cause of apparent occlusion found on DSA. (A) Pseudo-occlusion of the left cervical ICA in a 69-year-old woman. CTA shows a sharp, flame-shaped leading contrast edge at the level of the cervical ICA, with T-occlusion present. (B) DSA of this patient shows contrast moving slowly upward and finally a patent ICA (C) after mechanical thrombectomy of the distal ICA occlusion.
Statistical analyses
Clinical characteristics and imaging features are reported as means ± standard deviations or medians for continuous variables and as proportions for categorical variables. The independent-samples two-tailed t-test or the Mann–Whitney U-test was used (as appropriate) to compare continuous variables when the data were normally distributed; Fisher’s exact test was used to compare dichotomous variables. Parameters with P-values < 0.10 in univariate analyses were included in binary logistic regression. A P-value < 0.05 was considered to reflect statistical significance. All analyses were performed using SPSS ver. 22.0 (IBM Corp., Armonk, NY, USA).
Results
Of 85 patients with distal ICA occlusions, we excluded 14 with true cICA tandem occlusions (n = 8), atherosclerotic steno-occlusive changes of cICA evident in DSA (n = 5), and poor-quality CTA data (n = 1). Thus, we ultimately included 71 patients with a mean age 70 ± 14 years; 41 (57.7%) were male. The median NIHSS score on admission was 19 (interquartile range 16–21). Of all patients, 56.3% (n = 40) exhibited cICA-POs combined with distal ICA occlusions. The evaluations of the two readers were in complete agreement.
Patients with cICA-POs (n = 40) exhibited a higher rate of atrial fibrillation (82.5% vs. 58.0%, P = 0.024), a higher median NIHSS score on admission (19 vs. 16, P = 0.043), and a lower rate of successful first-pass recanalization (22.5% vs. 48.3%, P = 0.023), compared to non cICA-PO patients (n = 31). After MT, patients with cICA-POs exhibited a lower rate of successful recanalization (65.0% vs. 90.3%, P = 0.014), a higher rate of hemorrhagic transformation (32.5% vs. 9.6%, P = 0.01), and worse outcomes (80.0% vs. 25.8%, P < 0.001) than non cICA-PO patients (Fig. 2). There were no significant between-group differences in any other variable including the median ASPECTS score, median CTA collateral score, stroke onset to door time, or door to recanalization time (Table 1).
The distribution of the modified Rankin scale (mRS) scores. The scores range from 0 to 6, with 0 indicating no symptoms, 1 no clinically significant disability, 2 slight disability (patient is able to look after own affairs without assistance, but is unable to perform previous activities), 3 moderate disability (patient requires some help but is able to walk unassisted), 4 moderately severe disability (patient is unable to attend to bodily needs without assistance and unable to walk unassisted), 5 severe disability (patient requires constant nursing care and attention), and 6 death. The mRS score at 3 months according to the presence of cervical ICA pseudo-occlusion (cICA-PO). There were more patients with poor outcomes (mRS 4–6) in the cICA-PO group.
Binary logistic regression analyses revealed that the baseline CT ASPECTS score (odds ratio [OR], 1.605; 95% confidence interval [CI], 1.091–2.360; P = 0.016) and cICA-PO positivity (OR, 4.278; 95% CI, 1.080–33.006; P = 0.045) were independently associated with poor outcomes at 3 months (Table 2).
Of the 40 patients with cICA-POs, 26 were successfully recanalized after MT, whereas 14 were not. All of the latter patients experienced poor outcomes, whereas 69.2% (n = 18) of successfully recanalized patients experienced poor outcomes (P = 0.018) (Table 3). Of the 71 patients, 56 exhibited proximal involvement of the M1 segment attributable to carotid-distal ICA T- or L-type occlusions; 55.4% (56/34) experienced poor outcomes. Fifteen patients had pure, distal ICA I-type occlusions lacking M1 segment involvement and nine of these 15 patients (60%) experienced poor outcomes; the prognoses did not significantly differ between the two groups (P = 0.747). In the PO subgroup, 29 patients evidenced M1 involvement; 22 of these 29 patients (72.5%) experienced poor outcomes. Of 11 patients lacking M1 involvement, nine (81.8%) had poor outcomes. The “poor outcome” rate did not significantly differ between the two groups (P = 0.687).
Discussion
More than half (approximately 56%) of patients with distal ICA occlusions exhibited cICA-POs in arterial-phase CTA scans. The basic cause of PO remains unclear; the suggested mechanisms include hemodynamic insufficiency and luminal collapse triggered by acute embolism of the distal intracranial artery16. Patients with cICA-POs often have thrombi extending below the level of the posterior communicating artery, thus occluding that artery. Such patients tend to have larger clots (filling the terminal ICA segment) than those without cICA-POs (who usually exhibit contrast-filling of the posterior communicating artery, triggering adequate, antegrade, Willisian circle collateral flow)7,8,12,17,18. In other words, circle collateral flow affects cICA-PO status as determined via CTA. In addition, sluggish flow may be associated with poor collateral circulation and enlargement of the original clot, compromising successful recanalization and outcomes. Those with larger clots experience lower rates of recanalization and poorer outcomes19.
Endovascular treatment has become the gold standard for those with large-vessel occlusion strokes; rapid treatment and successful revascularization are important predictors of good outcomes after acute ischemic stroke16. Accordingly, any condition that delays or impedes recanalization negatively impacts care; it is important to image such conditions effectively. We found that those with cICA-POs tended to fail first-pass recanalization, require longer procedural times, and exhibit a lower final recanalization rate after MT than other patients. Reperfusion time was delayed, causing more brain damage. The hemorrhagic transformation rate was significantly higher in cICA-PO patients than in non cICA-PO patients (32.5% vs. 9.6%); thus, the poor outcome rate was higher in the former group. A cICA-PO in a patient with a distal ICA occlusion is a useful, surrogate imaging marker of outcome.
Patients with cICA-POs experienced lower rates of successful first-pass (22.5% vs. 48.3%) and final (65.0% vs. 90.3%) recanalization than those lacking cICA-POs. In the former group, when recanalization failed, the 3-month mRS was poor for all patients. When recanalization was successful, 69.2% of patients experienced poor outcomes; approximately 31% of patients did not. cICA-PO patients may require more aggressive approaches toward rapid recanalization.
An earlier work13 reported the outcomes of patients who received reperfusion therapy without mechanical thrombectomy (intravenous thrombolysis alone in 86 of 143 patients, 60.1%); all of our patients received thrombectomy. Grossberg et al.9 reported that, of 21 patients who exhibited PO on CTA, seven (33%) did not show PO on intra-arterial angiography; we evaluated patients who exhibited pseudo-occlusions on both CTA and intra-arterial catheter angiography. We found that, in the MT group, the PO sign was clinically significant. Patients with cICA-POs tended to fail first-pass recanalization, required longer procedural times, and exhibited lower final recanalization rates after MT, compared to other patients.
In patients with acute ischemic strokes associated with ipsilateral cICA non-attenuation in single-phase CTA, even specialized radiologists may not reliably distinguish true cervical occlusions from POs. In many studies, the interobserver agreement was low; CTA and DSA interpretations often differed. Our study included only cICA-POs in patients with normal DSA of the cICA. Therefore, the cICA-PO diagnostic accuracy was not a problem.
This study had some limitations. First, the single-center retrospective design of the work may feature selection bias. Second, all CT, CTA, and DSA data were interpreted simultaneously in a non-blinded manner, conceivably introducing bias. Finally, the sample size of the cICA-PO group was relatively small; we further subdivided this group into those who experienced successful and those who had non-successful recanalization. A future study should enroll more patients.
Conclusions
cICA-PO is common in patients with acute ischemic strokes caused by distal ICA occlusion and is associated with worse outcomes compared to non-PO patients. Rapid recanalization is essential and more active approaches for obtaining complete recanalization may be required by cICA-PO patients.
References
Campbell, B. C. et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N. Engl. J. Med. 372, 1009–1018 (2015).
Jovin, T. G. et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N. Engl. J. Med. 372, 2296–2306 (2015).
Berkhemer, O. A. et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N. Engl. J. Med. 372, 11–20 (2015).
Saver, J. L. et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med. 372, 2285–2295 (2015).
Goyal, M. et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N. Engl. J. Med. 372, 1019–1030 (2015).
Kim, J. J., Dillon, W. P., Glastonbury, C. M., Provenzale, J. M. & Wintermark, M. Sixty-four-section multidetector CT angiography of carotid arteries: a systematic analysis of image quality and artifacts. AJNR Am. J. Neuroradiol. 31, 91–99 (2010).
Wareham, J., Crossley, R., Barr, S. & Mortimer, A. Cervical ICA pseudo-occlusion on single phase CTA in patients with acute terminal ICA occlusion: what is the mechanism and can delayed CTA aid diagnosis? J Neurointerv Surg (2018).
Kappelhof, M. et al. Accuracy of CT Angiography for Differentiating Pseudo-Occlusion from True Occlusion or High-Grade Stenosis of the Extracranial ICA in Acute Ischemic Stroke: A Retrospective MR CLEAN Substudy. AJNR Am. J. Neuroradiol. 39, 892–898 (2018).
Grossberg, J. A. et al. Cervical Carotid Pseudo-Occlusions and False Dissections: Intracranial Occlusions Masquerading as Extracranial Occlusions. Stroke 48, 774–777 (2017).
Diouf, A. et al. Cervical Internal Carotid Occlusion versus Pseudo-occlusion at CT Angiography in the Context of Acute Stroke: An Accuracy, Interobserver, and Intraobserver Agreement Study. Radiology 286, 1008–1015 (2018).
Prakkamakul, S., Pitakvej, N., Dumrongpisutikul, N. & Lerdlum, S. Mid-cervical flame-shaped pseudo-occlusion: diagnostic performance of mid-cervical flame-shaped extracranial internal carotid artery sign on computed tomographic angiography in hyperacute ischemic stroke. Neuroradiology 59, 989–996 (2017).
Lee, S. U. et al. Differentiating Carotid Terminus Occlusions into Two Distinct Populations Based on Willisian Collateral Status. J. Stroke 18, 179–186 (2016).
Chen, Z. et al. Pseudo-Occlusion of the Internal Carotid Artery Predicts Poor Outcome After Reperfusion Therapy. Stroke 49, 1204–1209 (2018).
Zhang, J., Yang, Y., Sun, H. & Xing, Y. Hemorrhagic transformation after cerebral infarction: current concepts and challenges. Ann Transl Med 2 (2014).
Menon, B. K. et al. Multiphase CT Angiography: A New Tool for the Imaging Triage of Patients with Acute Ischemic Stroke. Radiology 275, 510–520 (2015).
Saver, J. L. et al. Time to Treatment With Endovascular Thrombectomy and Outcomes From Ischemic Stroke: A Meta-analysis. JAMA 316, 1279–1288 (2016).
Akpinar, S., Gelener, P. & Yilmaz, G. Aetiologies of internal carotid artery pseudo-occlusions in acute stroke patients: what neurointerventionalists can expect. Br. J. Radiol. 90, 20160352 (2017).
Marquering, H. A. et al. Carotid pseudo-occlusion on CTA in patients with acute ischemic stroke: a concerning observation. Clin. Neurol. Neurosurg. 115, 1591–1594 (2013).
Bhatia, R. et al. Low rates of acute recanalization with intravenous recombinant tissue plasminogen activator in ischemic stroke: real-world experience and a call for action. Stroke 41, 2254–2258 (2010).
Acknowledgements
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2017R1D1A1B03033880) and a grant from the Central Medical Service (CMS) Research Fund.
Author information
Authors and Affiliations
Contributions
All authors contributed to study conception and design. Material preparation, data collection and analysis, review, and editing were performed by Woo Sang Jung, Jin Soo Lee, Sten Solander, and Jin Wook Choi. The first draft of the manuscript was written by Woo Sang Jung; all authors commented on the draft. All authors have read and approved of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jung, W.S., Lee, J.S., Solander, S. et al. Pseudo-Occlusion of the Internal Carotid Artery in Acute Ischemic Stroke: Clinical Outcome after Mechanical Thrombectomy. Sci Rep 10, 2832 (2020). https://doi.org/10.1038/s41598-020-59609-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-59609-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.