Abstract
Accumulating evidence suggests a potential role of transient receptor potential vanilloid 1 (TRPV1) channels in inflammatory and cancer-related pain. However, the role of TRPV1 in the maintenance of neuropathic pain remains elusive. The current study investigated the effects of transient Trpv1 gene silencing using a small interference RNA (siRNA) on neuropathic pain induced by chronic constriction injury (CCI) of the sciatic nerve in rats. Seven days after CCI, the TRPV1 siRNA was intrathecally administered (5 µg/15 µl, once daily for 2 days). TRPV1 and Ca2+/calmodulin-dependent protein kinase II (CAMKII) expression and extracellular signal-regulated kinase (ERK) phosphorylation in the spinal cord were detected using western blotting. The thresholds to mechanical and thermal stimuli were determined before and after intrathecal TRPV1 siRNA administration. TRPV1 and CAMKII expression and ERK2 phosphorylation in the spinal cord were upregulated after CCI. Intrathecal administration of the TRPV1 siRNA not only attenuated behavioural hyperalgesia but also reduced the expression of TRPV1 and CAMKII, as well as ERK2 phosphorylation. Based on these results, silencing of the TRPV1 gene in the spinal cord attenuates the maintenance of neuropathic pain by inhibiting CAMKII/ERK2 activation and suggests that TRPV1 represents a potential target in pain therapy.
Similar content being viewed by others
Introduction
Neuropathic pain is defined as pain caused by a lesion or disease of the somatosensory nervous system, which is characterised by aberrant spontaneous pain, alterations in pain perception, and stimulus-evoked pain symptoms that are typically manifested as hyperalgesia and allodynia1. The treatment results in neuropathic pain are often disappointing due to the complex molecular and cellular mechanisms2,3, and therefore, neuropathic pain often becomes chronic and debilitating, ultimately affecting the patients’ productivity and quality of life4,5. Neuropathic pain is estimated to affect approximately 30% of the population and has become a serious global health burden6,7.
Transient receptor potential vanilloid 1 (TRPV1), a non-selective cation channel expressed by sensory neurons, functions to perceive external stimuli, including heat or capsaicin8,9,10. TRPV1 is also activated by an acidic pH, a plethora of chemicals of plant origin, and several toxins11,12,13,14. During the past decade, the role of TRPV1 in pain processing has been extensively investigated15,16,17,18,19,20,21,22. Under pathophysiological conditions, the sensitisation of TRPV1 channels reduces their activation threshold and, therefore, increases the sensitivity to painful or normally non-painful stimuli (hyperalgesia and allodynia, respectively)23,24,25,26,27. However, the role of spinal TRPV1 in the maintenance of neuropathic pain remains elusive. In a study of a rat model of peripheral nerve injury by Kanai Y et al., spinal TRPV1 expression was upregulated in response to mechanical allodynia28. However, in a model of partial spinal nerve ligation, no differences in behavioural hyperalgesia between TRPV1 null and wild-type mice were observed29. Moreover, the TRPV1 antagonist capsazepine reversed the mechanical hyperalgesia in a guinea pig model of partial SNL, with no effect on neuropathic pain in mice or rats30. These results suggest that TRPV1 may possess different functions in different species and different experimental models.
Extracellular signal-regulated kinases (ERK, including ERK1 and ERK2) are widely expressed intracellular signalling molecules. They are involved in functions such as the regulation of meiosis, mitosis, and postmitotic functions in differentiated cells. Recent studies have identified important roles for ERK in neuronal plasticity and the modulation of pain processing, including neuropathic and inflammatory pain31,32. Following peripheral nerve injury, innocuous stimuli (such as a gentle touch) are capable of evoking ERK activation in the dorsal horn neurons33. Furthermore, the inhibition of ERK activation by an MEK inhibitor has been shown to inhibit inflammatory pain hyperalgesia34, reduce the nociceptive behaviour induced by the ankle bend test in monoarthritic rats35, and suppress brain derived neurotrophic factor expression in the spinal cord36. ERK is activated by phosphorylation in the spinal cord following paw incision and participates in morphine analgesia37,38. Furthermore, TRPV1 was proposed to act as an upstream driver of ERK activation39.
Calmodulin (CaM) is one of the most abundant and well characterised Ca2+ sensor proteins. It controls a variety of cellular events, such as gene transcription, protein phosphorylation, nucleotide metabolism, and ion transport40. An elevated cytosolic Ca2+ concentration increases the binding of Ca2+ to CaM, and the resulting Ca2+-CaM interaction leads to the activation of several protein kinases, including CaM-dependent kinases (CaMKs)41. CAMKII is involved in ERK activation and thus plays a role in pain processing42,43,44,45.
However, the impact of spinal TRPV1 knockdown on the maintenance of neuropathic pain induced by chronic constriction injury (CCI) of the sciatic nerve, as well as the underlying molecular mechanisms among TRPV1, CAMKII, and ERK, are not fully understood. Therefore, the present study investigated the role of spinal TRPV1 using transient gene silencing with a small interfering RNA (siRNA) in a behavioural hyperalgesia model induced by CCI in rats. Silencing of the Trpv1 gene attenuated the maintenance of neuropathic pain by inhibiting CAMKII expression and ERK2 phosphorylation.
Results
Behavioural pain hypersensitisation induced by CCI
CCI successfully induced neuropathic pain in rats, consistent with our previous reports46,47,48. Within 3 days after CCI, the rats developed a stable neuropathic state. Animals guarded their ipsilateral hindpaw but appeared otherwise healthy with well-groomed coats and normal weight gain. Seven days after CCI, their ipsilateral hindpaw exhibited a significantly decreased mechanical and thermal threshold compared to their contralateral hindpaw or to the hindpaws in the sham group, indicating increased sensitivity to both mechanical and thermal stimulation (Fig. 1A,B, respectively; P < 0.001, n = 10, t-test).
Behavioural assessment of neuropathic pain induced by loose ligation of the sciatic nerve in rats. (A) Chronic constriction injury induced significant mechanical hyperalgesia, as denoted by a decreased paw withdrawal threshold to mechanical stimuli on the ipsilateral side. (B) Chronic constriction injury induced significant thermal hyperalgesia, as evidenced by the decreased paw withdrawal latency on the ipsilateral side. Data are presented as means ± SEM, and error bars represent the SEM. ***P < 0.001 compared with the sham group or the contralateral side (t-test, n = 10 rats per group). Contra: contralateral; Ipsi: ipsilateral; CCI: chronic constriction injury.
CCI increased TRPV1 and CAMKII expression, as well as ERK phosphorylation, in the spinal dorsal horn
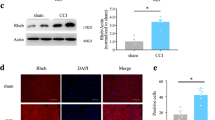
We examined TRPV1 expression in the spinal dorsal horn using western blotting. The expression of the TRPV1 protein was enriched in the anterior cingulate cortex, dorsal horn, and the dorsal root ganglion (data not shown). Next, we investigated the alterations in the levels of the TRPV1, CAMKII and ERK/phosphorylated ERK (pERK) proteins following CCI. The unilateral constriction injury of the sciatic nerve induced a significant increase in TRPV1 (P = 0.031, analysis of variance [ANOVA], n = 5; Fig. 2A,B) and CAMKII (P = 0.018, ANOVA, n = 4; Fig. 2A,B) expression compared to the sham group. Similarly, CCI increased pERK2 levels in the ipsilateral spinal dorsal horn (P = 0.023, ANOVA, n = 4; Fig. 2C,E) without affecting the pERK1 levels (Fig. 2C,D).
Increased expression levels of TRPV1, CAMKII, and phosphorylated ERK in the ipsilateral dorsal horn induced by chronic constriction injury. (A) The loose ligation of the sciatic nerve significantly increased TRPV1 and CAMKII expression in the ipsilateral spinal dorsal horn 7 days after CCI. (B) Bar chart showing the protein content relative to the sham group. The levels of TRPV1 and CAMKII in the ipsilateral spinal dorsal horn of the CCI group were significantly higher than those in the sham group (P = 0.031 for TRPV1; P = 0.018 for CAMKII). (C) Loose ligation of the sciatic nerve significantly increased ERK phosphorylation in the ipsilateral spinal dorsal horn. (D,E) Bar charts depicting the protein content relative to that in the sham group. A significantly higher level of phosphorylated ERK2 was observed in the ipsilateral spinal dorsal horn of the CCI group than in the ipsilateral spinal dorsal horn of the sham group (P = 0.023). GAPDH or tubulin served as a loading control and was run on the same blot. *P < 0.05 compared with the sham group (ANOVA, n = 4–5 rats per group). Error bars represent the SEM. CCI: chronic constriction injury; Contra: contralateral to CCI.
TRPV1 siRNA attenuated the behavioural hypersensitisation induced by CCI
We administered a TRPV1 siRNA in vivo to downregulate the spinal expression of the TRPV1 protein and then measured pain behaviours to examine the role of TRPV1 in the maintenance of neuropathic pain. Mechanical and thermal threshold were determined before and after the intrathecal administration of the TRPV1 siRNA. The administration of the TRPV1 siRNA (5 μg/15 µl) once daily for two days significantly attenuated the mechanical and thermal hyperalgesia on days 1 to 4 post-transfection compared to the CCI group or the CCI + polyethylenimine (PEI) control group, as well as compared to the pre-transfection baseline (Fig. 3; P < 0.01 or 0.05, ANOVA, n = 5–6).
The intrathecal injection of the TRPV1 siRNA in vivo attenuated the mechanical and thermal hyperalgesia induced by chronic constriction injury. (A) The administration of the TRPV1 siRNA (5 μg/15 μl) once daily for two days significantly increased the paw withdrawal threshold to mechanical stimuli on days 1 to 4 post-transfection compared to the CCI + PEI control group or to the pre-injection baseline. (B) The TRPV1 siRNA significantly increased the paw withdrawal latency in response to thermal stimuli on days 1 to 4 post-transfection compared to results in the CCI + PEI control group or the pre-injection baseline. **P < 0.01 and *P < 0.05 compared with the PEI group results; ##P < 0.01 and #P < 0.05 compared with the pre-injection baseline results (ANOVA, n = 5–6 rats per group). Error bars represent the SEM. PEI: polyethylenimine; CCI: chronic constriction injury.
TRPV1 siRNA reduced the spinal TRPV1 and CAMKII expression, as well as ERK phosphorylation
We administered the TRPV1 siRNA in vivo to downregulate the spinal expression of the TRPV1 protein and then determined CAMKII expression and ERK phosphorylation to investigate the possible mechanism by which TRPV1 mediated CCI-induced neuropathic pain. We performed western blotting to detect protein levels 24 hours post-siRNA administration. The TRPV1 siRNA markedly reduced TRPV1 expression compared to PEI alone in both naive (Fig. 4A,B; P = 0.001, ANOVA, n = 5) and CCI (Fig. 4C,D; P = 0.003, n = 4, t-test) animals. Similarly, the TRPV1 siRNA decreased the CAMKII expression (P = 0.026, n = 3, t-test; Fig. 4E,F) and ERK2 phosphorylation (P = 0.002, n = 6, Mann-Whitney U test; Fig. 4G,H) in the enlargement segments of the lumbar spinal cord without affecting the level of ERK1 phosphorylation (Fig. 4G,H).
Reduced TRPV1 and CAMKII expression, as well as decreased ERK phosphorylation, in the spinal dorsal horn following TRPV1 siRNA administration. Western blots showing the reduced expression of the TRPV1 protein in the spinal dorsal horn 24 hours after the intrathecal injection of the TRPV1 siRNA (5 μg/15 μl) once daily for 2 days, in both naive (A) and CCI (C) rats. (B,D) Bar chart depicting the level of TRPV1 protein relative to that in the group administered PEI alone; the level of TRPV1 in the spinal dorsal horn was significantly reduced. (E) Western blots showing the reduced expression of CAMKII following the intrathecal injection of the TRPV1 siRNA compared to the results in the CCI + PEI group. (F) Bar chart presenting the protein content relative to the CCI + PEI group; the CAMKII level was significantly reduced. (G) Western blots showing the reduced level of phosphorylated ERK2 following the intrathecal injection of the TRPV1 siRNA in comparison to the results in the CCI + PEI group. (H) Bar chart depicting the protein content relative to the CCI + PEI group; the level of phosphorylated ERK2 was significantly reduced in the CCI + siRNA group. Tubulin or GAPDH served as the loading control and was run on the same blot. TRPV1 levels were significantly reduced in the siRNA group compared with those in the PEI group in naive rats. **P = 0.001 compared with those in the PEI group (ANOVA, n = 5 rats per group). TRPV1 levels were significantly reduced in the CCI + siRNA group compared with those in the CCI + PEI group in CCI rats. **P = 0.003 compared with those in the CCI + PEI group (t-test, n = 4 rats per group). The level of CAMKII in the ipsilateral spinal dorsal horn of the CCI + siRNA group was significantly lower than that in the CCI + PEI group. *P = 0.026 compared with that in the CCI + PEI group (t-test, n = 3 rats per group). The level of phosphorylated ERK2 in the ipsilateral spinal dorsal horn of the CCI + RNAi group was significantly lower than that of the CCI + PEI group. **P = 0.002 compared with that in the CCI + PEI group (Mann-Whitney U test, n = 6 rats per group). Error bars represent the SEM. PEI: polyethylenimine; CCI: chronic constriction injury.
Discussion
TRPV1 is sensitised by several stimuli, including capsaicin, noxious heat, and an acidic pH8,9,13. Accumulating evidence suggests a potential role for TRPV1 in inflammatory and cancer-related pain29,49,50. However, its role in neuropathic pain has not yet been fully elucidated. Therefore, the present study investigated in a rat CCI model the contributions of spinal TRPV1 proteins to neuropathic pain. The unilateral constriction injury of the sciatic nerve induced a significant increase in TRPV1 expression compared to the sham group, consistent with previous observations28. This finding prompted us to hypothesise that spinal TRPV1 may be involved in behavioural hyperalgesia. The intrathecal administration of TRPV1 siRNA resulted in the expected marked downregulation of spinal TRPV1 expression that was accompanied by a significant increase in the threshold for mechanical and thermal stimuli.
The regional expression and function of TRPV1 in the rodent spinal cord have been previously explored26,28,29,51. Doly et al. previously reported the expression of the TRPV1 protein and mRNA in the spinal dorsal horn52. Consistent with these findings, our immunoblotting results confirmed that the TRPV1 protein was enriched in the dorsal horn and the dorsal root ganglion. The spinal dorsal horn plays a pivotal role in the processing and transmission of peripheral noxious stimuli26,53. Therefore, the abundance of TRPV1 in the spinal horn implies that TRPV1 may be involved in the mechanisms of processing nociceptive stimuli.
A variety of methods have been employed to disrupt TRPV1 functions and to clarify the role of spinal TRPV1 in neuropathic pain, including gene silencing and antisense technology; however, the results remain inconclusive54,55. An adeno-associated virus expression system was recently developed to knock down spinal TRPV1 expression in a mouse model of neuropathic pain56. However, concerns regarding the associated cell toxicity or neuronal dysfunction persisted57. PEI is a non-virus polymer that forms a complex with the siRNA and facilitates its transfection. It was first reported by Tan et al. in 200558, and the results were successfully replicated by Zhou et al. in 201059. The siRNA-PEI polymer complexes have proven to be a highly efficient and easy-to-use system for the in vivo administration of siRNAs. Therefore, this PEI-based non-viral delivery method may represent a potentially powerful tool to maximise in vivo the function of administered siRNAs with potential use in clinical practice57. To the best of our knowledge, this study is the first to report successful spinal TRPV1 gene silencing in rats with neuropathic pain induced by peripheral nerve injury. The behavioural assessment revealed that spinal TRPV1 knockdown attenuated the pain-induced hyperalgesia, suggesting a critical role for spinal TRPV1 in the maintenance of neuropathic pain induced by CCI.
To date, the molecular mechanism by which TRPV1 regulates neuropathic pain has not been clearly elucidated. ERK, a member of the mitogen-activated protein kinase (MAPK) family, has been implicated in modulating various pain modalities, including neuropathic and inflammatory pain, and has been proposed as an alternative target for pain therapy2,60. Calmodulin is one of the most abundant and well characterised Ca2+ sensor proteins. According to Cao et al., TRPV1 induces Ca2+ influx, which in turn activates various Ca2+-dependent kinases (such as CAMKII) to initiate the MAPK signalling cascade in the spinal dorsal horn neurons34. Following CCI, TRPV1 and pERK2 levels were significantly increased without significantly affecting the pERK1 level in the present study. As shown in a previous study by Xu et al., increased ERK2 phosphorylation in the dorsal horn neurons is required for the development of inflammatory pain hyperalgesia61. Due to the high sequence similarity between ERK1 and ERK2 proteins and the lack of a specific ERK2 antiserum, immunohistochemical techniques are not usually able to distinguish between ERK1 and ERK2 expression. Nevertheless, Xu et al. indicated that immunohistochemical staining in the medial superficial dorsal horn most likely reflects ERK2 expression61. Therefore, in the present study we used western blotting to explore the activation of ERK1/2 following CCI and TRPV1 silencing. In parallel with the behavioural analgesia, the TRPV1 siRNA also reduced the levels of CAMKII and pERK2. These results imply the involvement of a TRPV1-dependent ERK2 phosphorylation pathway.
Conclusions
Our results revealed increased spinal levels of TRPV1 and ERK phosphorylation in a rat model of neuropathic pain. The TRPV1 siRNA attenuated the behavioural hyperalgesia and decreased the CAMKII and pERK2 levels. The results obtained here revealed the molecular crosstalk between the spinal TRPV1 and CAMKII/ERK2 signalling. However, these findings do not exclude the possible involvement of other signalling molecules. Future studies are required to confirm our results and to further dissect the molecular pathways that regulate the development and maintenance of neuropathic pain. Nevertheless, our results support the hypothesis that spinal TRPV1 may be an alternative target to treat neuropathic pain.
Materials and Methods
Animals
Male Wistar rats (~200 g) were obtained from the Animal Centre of the Chinese Academy of Sciences and housed in groups of two per cage with water and food available ad libitum. The animals were allowed 3 days to acclimate to their surroundings before starting any experiments. The animals were maintained on a 12-h light/12-h dark cycle, with lights on at 08:00 am. All animal experiments were approved by the Animal Care Committee at Zhejiang University in accordance with the ethical guidelines for the investigation of experimental pain in animals62. All efforts were made to minimise the number of animals used and their suffering.
Intrathecal catheterisation
Rats were anaesthetised with pentobarbital (60 mg/kg) via intraperitoneal injection, and a polyethylene-10 (Becton-Dickinson, San Jose, MD, USA) catheter was inserted into the subarachnoid space at the lumbar enlargement level of the spinal cord, as described in our previous report63. The animals were allowed to recover for 3 days before being randomly assigned to their respective groups. Rats showing postoperative neurological deficits were excluded from the study.
CCI-induced neuropathic pain
A neuropathic pain model was induced as described in previous studies and our recent reports47,48,64. Briefly, under isoflurane anaesthesia (5% for induction, 2.5% for maintenance), the left sciatic nerve was exposed, and the surrounding connective tissue was removed. Three ligations were placed around the nerve with 4–0 chromic gut (CCI group, n = 10). A typical twitch of the hind paw was observed upon successful nerve constriction. The sham-operated animals (sham group; n = 10) underwent an identical surgical procedure without sciatic nerve ligation. All animals received antibiotics (penicillin 0.5 ml, 96 mg/ml, via a hypodermic injection) to reduce the possibility of infection.
In vivo RNA interference
A smart pool of siRNAs targeting TRPV1 was designed by Dharmacon RNAi Technology (CO, USA). The siRNA pool was combined with branched PEI reagent (ExGen 500; Fermentas, Waltham, MA, USA) according to the manufacturer’s protocol. Due to its higher biological safety, we opted to use a non-virus delivery system in this study. After the establishment of the neuropathic pain model (7 days post-CCI), a dose of 5 μg/15 µl siRNA was intrathecally administered once daily for two days followed by an injection of 10 µl saline to flush the catheter. Behavioural tests were performed before and 1, 2, 3, 4, 5, and 8 days after the in vivo transfection. TRPV1 protein expression was analysed one day after the intrathecal administration of TRPV1 siRNA or PEI reagent by western blotting.
Behavioural assessment
All animals underwent a behavioural assessment to determine the paw withdrawal threshold in response to mechanical stimuli before and after in vivo transfection (n = 5–6) as described by our group and others37,47,65. Briefly, animals were placed in a cage with a wire mesh floor and allowed time to explore and groom until they settled (30 min). An electronic von Frey anesthesiometer (Model 2390, IITC/Life Science Instruments, Woodland Hills, CA, USA) with a flexible probe was applied to the plantar surface of the injured hind paws. Brisk withdrawal or paw flinching was considered a positive response. The mechanical threshold is the maximum force of the von Frey hair triggering the withdrawal of the hindpaw. Each animal was measured three times, and the average values from the three trials were calculated.
The thermal threshold to noxious heat stimuli was determined using an analgesia apparatus (Model 33B, IITC/Life Science Instruments) as described previously47,48,63,66. Briefly, rats (n = 5–6) were placed in a Plexiglas chamber on a glass plate under which a lightbox was located. A radiant heat stimulus was applied by aiming a beam of light through a hole in the light box onto the heel of each hind paw through the glass plate. The light beam was turned off when the rat lifted its foot. The time between the illumination with the light beam and the foot lift was defined as the thermal threshold. Each trial was repeated three times at 5-min intervals. A cut-off time of 20 s was used to prevent tissue damage.
Immunoblot analysis
Immunoblotting was performed as described in our previous reports47,48,67,68. Briefly, rats were decapitated under deep anaesthesia. The spinal lumbar enlargement was quickly excised and divided into ipsilateral and contralateral halves, and then, further divided into dorsal and ventral quadrants. The ipsilateral and contralateral dorsal quadrants were homogenised in an ice-cold homogenisation buffer (P0013; Beyotime Institute of Biotechnology, Jiangsu, China). The homogenates were centrifuged at 10000 × g for 10 min at 4 °C, the supernatants were collected, and the protein concentrations were determined using a Micro BCA protein assay reagent kit (23235; Thermo Fisher Scientific Inc., Waltham, MA, USA). Proteins in the supernatants were separated on 8% SDS-PAGE gels and transferred to a polyvinylidene difluoride membrane. Proteins bound to the membrane were stained with a Ponceau S solution (P0022; Beyotime Institute of Biotechnology) to determine the transfer quality. Membranes were blocked with 5% skim milk for 2 h at room temperature and incubated with primary antibodies in an antibody buffer containing Tween-20 (50 µl/100 ml) overnight at 4 °C. The primary antibodies were directed against the following proteins: TRPV1 (1:2000; Alomone Labs., Jerusalem, Israel), CaMKII (1: 1000; Abcam, MA, USA), pERK (1:1000; Millipore, CA, USA), ERK (1:1000; Cell Signaling Technology, Danvers, MA, USA), and β-tubulin (1:2000; Beyotime Institute of Biotechnology) or GAPDH (1: 5000; Kang Chen Bio-tech, Shanghai, China). Next, the membranes were washed and incubated with peroxidase-conjugated secondary antibodies for 2 hours at room temperature. Membranes were washed thoroughly, and immunolabelling was visualised with the SuperSignal West Femto Maximum Sensitivity Substrate (34095; Pierce, NJ, USA) and captured using the ChemiDoc XRS system (Bio-Rad Laboratories Inc., Hercules, CA, USA). The intensities of protein bands on the blots were quantified using Quantity One 4.62 software (Bio-Rad Laboratories Inc.).
Statistical analysis
The pain threshold and protein abundance were statistically analysed by performing a one- or two-way ANOVA followed by the least significant difference test for multiple comparison test. The unpaired Student’s t-test was used for comparisons between two groups. Non-parametric data were analysed with the Mann-Whitney U test. Data are presented as the mean ± standard error of the mean (SEM) and were analysed using SPSS software (IBM Corp., Armonk, NY, USA). A P value of less than 0.05 was considered statistically significant.
Data Availability
All data generated or analysed during this study are included in this published article. The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Finnerup, N. B. et al. Neuropathic pain: an updated grading system for research and clinical practice. Pain 157, 1599–1606 (2016).
Ji, R. R., Xu, Z. Z. & Gao, Y. J. Emerging targets in neuroinflammation-driven chronic pain. Nat Rev Drug Discov 13, 533–548 (2014).
Ji, R. R., Chamessian, A. & Zhang, Y. Q. Pain regulation by non-neuronal cells and inflammation. Science 354, 572–577 (2016).
Peirs, C. & Seal, R. P. Targeting Toll-like receptors to treat chronic pain. Nat Med 21, 1251–1252 (2015).
Hohmann, S. W. et al. The G2A receptor (GPR132) contributes to oxaliplatin-induced mechanical pain hyperalgesia. Sci Rep 7, 446 (2017).
von Hehn, C. A., Baron, R. & Woolf, C. J. Deconstructing the neuropathic pain phenotype to reveal neural mechanisms. Neuron 73, 638–652 (2012).
Gilron, I., Jensen, T. S. & Dickenson, A. H. Combination pharmacotherapy for management of chronic pain: from bench to bedside. Lancet Neurol 12, 1084–1095 (2013).
Caterina, M. J. et al. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389, 816–824 (1997).
Clapham, D. E. TRP channels as cellular sensors. Nature 426, 517–524 (2003).
Vandewauw, I. et al. A TRP channel trio mediates acute noxious heat sensing. Nature 555, 662–666 (2018).
Yang, B. H. et al. Activation of vanilloid receptor 1 (VR1) by eugenol. J Dent Res 82, 781–785 (2003).
Xu, H., Blair, N. T. & Clapham, D. E. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in a vanilloid-independent mechanism. J Neurosci 25, 8924–8937 (2005).
Siemens, J. et al. Spider toxins activate the capsaicin receptor to produce inflammatory pain. Nature 444, 208–212 (2006).
Song, J. et al. TRPV1 Activation in Primary Cortical Neurons Induces Calcium-Dependent Programmed Cell Death. Exp Neurobiol 22, 51–57 (2013).
Bolcskei, K. et al. Investigation of the role of TRPV1 receptors in acute and chronic nociceptive processes using gene-deficient mice. Pain 117, 368–376 (2005).
Patapoutian, A., Tate S Fau - Woolf, C. J. & Woolf, C. J. Transient receptor potential channels: targeting pain at the source. Nat Rev Drug Discov 8, 55–68 (2009).
Kim, Y. H. et al. TRPV1 in GABAergic interneurons mediates neuropathic mechanical allodynia and disinhibition of the nociceptive circuitry in the spinal cord. Neuron 74, 640–647 (2012).
Mickle, A. D., Shepherd, A. J. & Mohapatra, D. P. Nociceptive TRP Channels: Sensory Detectors and Transducers in Multiple Pain Pathologies. Pharmaceuticals (Basel) 9 (2016).
Marwaha, L. et al. TRP channels: potential drug target for neuropathic pain. Inflammopharmacology 24, 305–317 (2016).
Takayama, Y., Furue, H. & Tominaga, M. 4-isopropylcyclohexanol has potential analgesic effects through the inhibition of anoctamin 1, TRPV1 and TRPA1 channel activities. Sci Rep 7, 43132 (2017).
Liao, H. Y., Hsieh, C. L., Huang, C. P. & Lin, Y. W. Electroacupuncture Attenuates CFA-induced Inflammatory Pain by suppressing Nav1.8 through S100B, TRPV1, Opioid, and Adenosine Pathways in Mice. Sci Rep 7, 42531 (2017).
Zhang, F. et al. Heat activation is intrinsic to the pore domain of TRPV1. Proc Natl Acad Sci USA 115, E317–E324 (2018).
Jordt, S. E. et al. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 427, 260–265 (2004).
Moran, M. M., McAlexander Ma Fau - Biro, T., Biro T Fau - Szallasi, A. & Szallasi, A. Transient receptor potential channels as therapeutic targets. Nat Rev Drug Discov 10, 601–620 (2011).
Julius, D. TRP channels and pain. Annu Rev Cell Dev Biol 29, 355–384 (2013).
Vriens, J., Nilius, B. & Voets, T. Peripheral thermosensation in mammals. Nat Rev Neurosci 15, 573–589 (2014).
Basso, L. & Altier, C. Transient Receptor Potential Channels in neuropathic pain. Curr Opin Pharmacol 32, 9–15 (2017).
Kanai, Y., Nakazato E Fau - Fujiuchi, A., Fujiuchi A Fau - Hara, T., Hara T Fau - Imai, A. & Imai, A. Involvement of an increased spinal TRPV1 sensitization through its up-regulation in mechanical allodynia of CCI rats. Neuropharmacology 49, 977–984 (2005).
Caterina, M. J. et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 288, 306–313 (2000).
Walker, K. M. et al. The VR1 antagonist capsazepine reverses mechanical hyperalgesia in models of inflammatory and neuropathic pain. J Pharmacol Exp Ther 304, 56–62 (2003).
Zhuang, Z. Y., Gerner P Fau - Woolf, C. J., Woolf Cj Fau - Ji, R.-R. & Ji, R. R. ERK is sequentially activated in neurons, microglia, and astrocytes by spinal nerve ligation and contributes to mechanical allodynia in this neuropathic pain model. Pain 114, 149–159 (2005).
Liu, C. C. et al. Interferon alpha inhibits spinal cord synaptic and nociceptive transmission via neuronal-glial interactions. Sci Rep 6, 34356 (2016).
Gao, Y. J. & Ji, R. R. Light touch induces ERK activation in superficial dorsal horn neurons after inflammation: involvement of spinal astrocytes and JNK signaling in touch-evoked central sensitization and mechanical allodynia. J Neurochem 115, 505–514 (2010).
Cao, H. et al. Activation of extracellular signal-regulated kinase in the anterior cingulate cortex contributes to the induction and expression of affective pain. J Neurosci 29, 3307–3321 (2009).
Cruz, C. D., Neto, F. L., Castro-Lopes, J., McMahon, S. B. & Cruz, F. Inhibition of ERK phosphorylation decreases nociceptive behaviour in monoarthritic rats. Pain 116, 411–419 (2005).
Obata, K. et al. Differential activation of extracellular signal-regulated protein kinase in primary afferent neurons regulates brain-derived neurotrophic factor expression after peripheral inflammation and nerve injury. J Neurosci 23, 4117–4126 (2003).
Li, N. N. et al. Dezocine Antagonizes Morphine Analgesia upon Simultaneous Administration in Rodent Models of Acute Nociception. Pain Physician 20, E401–E409 (2017).
Horvath, R. J., Landry, R. P., Romero-Sandoval, E. A. & DeLeo, J. A. Morphine tolerance attenuates the resolution of postoperative pain and enhances spinal microglial p38 and extracellular receptor kinase phosphorylation. Neuroscience 169, 843–854 (2010).
Zhuang, Z. Y., Xu H Fau - Clapham, D. E., Clapham De Fau - Ji, R.-R. & Ji, R. R. Phosphatidylinositol 3-kinase activates ERK in primary sensory neurons and mediates inflammatory heat hyperalgesia through TRPV1 sensitization. J Neurosci 24, 8300–8309 (2004).
Ikura, M. & Ames, J. B. Genetic polymorphism and protein conformational plasticity in the calmodulin superfamily: two ways to promote multifunctionality. Proc Natl Acad Sci USA 103, 1159–1164 (2006).
Schmitt, J. M., Guire, E. S., Saneyoshi, T. & Soderling, T. R. Calmodulin-dependent kinase kinase/calmodulin kinase I activity gates extracellular-regulated kinase-dependent long-term potentiation. J Neurosci 25, 1281–1290 (2005).
Song, L., Xiao, W. & Templeton, D. M. Low-concentration heparin suppresses ionomycin-activated CAMK-II/EGF receptor- and ERK-mediated signaling in mesangial cells. J Cell Physiol 224, 484–490 (2010).
Banerjee, C. et al. Role of calmodulin-calmodulin kinase II, cAMP/protein kinase A and ERK 1/2 on Aeromonas hydrophila-induced apoptosis of head kidney macrophages. PLoS Pathog 10, e1004018 (2014).
Nakanishi, M. et al. Acid activation of Trpv1 leads to an up-regulation of calcitonin gene-related peptide expression in dorsal root ganglion neurons via the CaMK-CREB cascade: a potential mechanism of inflammatory pain. Mol Biol Cell 21, 2568–2577 (2010).
Leo, M. et al. Cisplatin-induced neuropathic pain is mediated by upregulation of N-type voltage-gated calcium channels in dorsal root ganglion neurons. Exp Neurol 288, 62–74 (2017).
Yang, J. X. et al. Caveolin-1 in the anterior cingulate cortex modulates chronic neuropathic pain via regulation of NMDA receptor 2B subunit. J Neurosci 35, 36–52 (2015).
Huang, L. E. et al. N-methyl D-aspartate receptor subtype 2B antagonist, Ro 25-6981, attenuates neuropathic pain by inhibiting postsynaptic density 95 expression. Sci Rep 8, 7848 (2018).
Lin, J. P. et al. Dexmedetomidine Attenuates Neuropathic Pain by Inhibiting P2X7R Expression and ERK Phosphorylation in Rats. Exp Neurobiol 27, 267–276 (2018).
Davis, J. B. et al. Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia. Nature 405, 183–187 (2000).
Karai, L. et al. Deletion of vanilloid receptor 1-expressing primary afferent neurons for pain control. J Clin Invest 113, 1344–1352 (2004).
Eckert, W. A., 3rd, Julius, D. & Basbaum, A. I. Differential contribution of TRPV1 to thermal responses and tissue injury-induced sensitization of dorsal horn neurons in laminae I and V in the mouse. Pain 126, 184–197 (2006).
Doly, S., Fischer J Fau - Salio, C., Salio C Fau - Conrath, M. & Conrath, M. The vanilloid receptor-1 is expressed in rat spinal dorsal horn astrocytes. Neurosci Lett 357, 123–126 (2004).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Christoph, T. et al. Silencing of vanilloid receptor TRPV1 by RNAi reduces neuropathic and visceral pain in vivo. Biochem Biophys Res Commun 350, 238–243 (2006).
Christoph, T. et al. Antinociceptive effect of antisense oligonucleotides against the vanilloid receptor VR1/TRPV1. Neurochem Int 50, 281–290 (2007).
Hirai, T. et al. Intrathecal AAV serotype 9-mediated delivery of shRNA against TRPV1 attenuates thermal hyperalgesia in a mouse model of peripheral nerve injury. Mol Ther 22, 409–419 (2014).
Aigner, A. Delivery systems for the direct application of siRNAs to induce RNA interference (RNAi) in vivo. J Biomed Biotechnol 2006, 71659 (2006).
Tan, P. H., Yang, L. C., Shih, H. C., Lan, K. C. & Cheng, J. T. Gene knockdown with intrathecal siRNA of NMDA receptor NR2B subunit reduces formalin-induced nociception in the rat. Gene Ther 12, 59–66 (2005).
Zhou, D., Chen, M. L., Zhang, Y. Q. & Zhao, Z. Q. Involvement of spinal microglial P2X7 receptor in generation of tolerance to morphine analgesia in rats. J Neurosci 30, 8042–8047 (2010).
Ma, W. & Quirion, R. The ERK/MAPK pathway, as a target for the treatment of neuropathic pain. Expert Opin Ther Targets 9, 699–713 (2005).
Xu, Q., Garraway Sm Fau - Weyerbacher, A. R., Weyerbacher Ar Fau - Shin, S. J., Shin Sj Fau - Inturrisi, C. E. & Inturrisi, C. E. Activation of the neuronal extracellular signal-regulated kinase 2 in the spinal cord dorsal horn is required for complete Freund’s adjuvant-induced pain hyperalgesia. J Neurosci 28, 14087–14096 (2008).
Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 16, 109–110 (1983).
Yao, Y. X., Jiang, Z. & Zhao, Z. Q. Knockdown of synaptic scaffolding protein Homer 1b/c attenuates secondary hyperalgesia induced by complete Freund’s adjuvant in rats. Anesthesia and analgesia 113, 1501–1508 (2011).
Bennett, G. J. & Xie, Y. K. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33, 87–107 (1988).
Sun, R. et al. Hippocampal activation of microglia may underlie the shared neurobiology of comorbid posttraumatic stress disorder and chronic pain. Mol Pain 12 (2016).
Hargreaves, K., Dubner, R., Brown, F., Flores, C. & Joris, J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32, 77–88 (1988).
Yao, Y. X. et al. Spinal synaptic scaffolding protein Homer 1b/c regulates CREB phosphorylation and c-fos activation induced by inflammatory pain in rats. Neurosci Lett 559, 88–93 (2014).
Yang, Y. et al. Delayed activation of spinal microglia contributes to the maintenance of bone cancer pain in female Wistar rats via P2X7 receptor and IL-18. J Neurosci 35, 7950–7963 (2015).
Acknowledgements
We thank Professor Yu-Qiu Zhang for her kind assistance with the animal experiments. We also thank Nature Research Editing, Medjaden Bioscience Limited, and Editage (https://wkonline.editage.com) for providing language editing services. This work was supported by grants from the National Natural Science Foundation of China (grants No. 81471127, 30972846, and 81600954) and the Department of Education of Zhejiang Province (grant No. Y201636423).
Author information
Authors and Affiliations
Contributions
Y.-X.Y. and S.-M.Z. designed the study; Y.-X.Y. analysed the data and drafted the manuscript; S.-H.G., L.-E.H., J.-P.L. and M.-Y.S. performed the experimental procedures; C.-Q.C., N.-N.L. and X.Z. provided technical assistance; Y.Y. helped in designing the study and analysing the data. All authors approved the final version of this manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, SH., Lin, JP., Huang, LE. et al. Silencing of spinal Trpv1 attenuates neuropathic pain in rats by inhibiting CAMKII expression and ERK2 phosphorylation. Sci Rep 9, 2769 (2019). https://doi.org/10.1038/s41598-019-39184-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39184-4
This article is cited by
-
HBO treatment enhances motor function and modulates pain development after sciatic nerve injury via protection the mitochondrial function
Journal of Translational Medicine (2023)
-
Optogenetic modulation of electroacupuncture analgesia in a mouse inflammatory pain model
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.