Abstract
Alcohol use is common, imposes a staggering burden on public health, and often resists treatment. The central extended amygdala (EAc)—including the bed nucleus of the stria terminalis (BST) and the central nucleus of the amygdala (Ce)—plays a key role in prominent neuroscientific models of alcohol drinking, but the relevance of these regions to acute alcohol consumption in humans remains poorly understood. Using a single-blind, randomized-groups design, multiband fMRI data were acquired from 49 social drinkers while they performed a well-established emotional faces paradigm after consuming either alcohol or placebo. Relative to placebo, alcohol significantly dampened reactivity to emotional faces in the BST. To rigorously assess potential regional differences in activation, data were extracted from unbiased, anatomically predefined regions of interest. Analyses revealed similar levels of dampening in the BST and Ce. In short, alcohol transiently reduces reactivity to emotional faces and it does so similarly across the two major divisions of the human EAc. These observations reinforce the translational relevance of addiction models derived from preclinical work in rodents and provide new insights into the neural systems most relevant to the consumption of alcohol and to the initial development of alcohol abuse in humans.
Similar content being viewed by others
Introduction
Alcohol use is common (nearly three-quarters of Americans consumed some form of ethanol in the past year and, among them, 17.5% met criteria for an alcohol use disorder), contributes to a variety of adverse outcomes, and imposes a rapidly growing burden on public health and the economy1,2,3, highlighting the need to understand the acute impact of alcohol consumption on the human brain.
Evidence gleaned from animal models highlights the potential importance of the central extended amygdala (EAc), including the bed nucleus of the stria terminalis (BST) and the central nucleus of the amygdala (Ce)4,5 (Fig. 1). The BST and the Ce show similar patterns of connectivity, cellular composition, neurochemistry, and gene expression, and both are critical for triggering defensive responses to threat4,6,7,8,9. Through dense projections to downstream effector regions, these regions play an important role in prioritizing the processing of salient social cues, such as facial expressions of emotion10,11, and shaping social interactions12,13. The EAc also plays a key role in prominent neuroscientific models of alcohol-drinking14,15,16,17,18, with work in rodents indicating that alcohol acutely dampens EAc reactivity19,20,21,22,23.
Human EAc. The EAc (magenta) encompasses the BST (encircling the anterior commissure) and the Ce (within the dorsal portion of the amygdala proper). The BST and the Ce are anatomically interconnected via the ventral amygdalofugal pathway and the stria terminalis, as indicated by deterministic tractography (gold). Both regions are poised to orchestrate responses to emotionally salient stimuli via dense projections to downstream effector regions. Portions of this figure were adapted from ref.71. Abbreviations—BL, basolateral nucleus of the amygdala; BM, basomedial nucleus of the amygdala; BST, bed nucleus of the stria terminalis; Ce, central nucleus of the amygdala; La, lateral nucleus of the amygdala; Me, medial nucleus of the amygdala.
The acute impact of alcohol on BST or Ce function in humans remains unclear. To date, imaging research has focused on the role of the amygdala proper, with several small-scale studies reporting evidence suggestive of dampened reactivity to emotional faces, particularly those expressing fear or anger (Table 1)24,25,26,27. None directly examined either the BST or the Ce, despite their central role in prominent models of alcohol consumption. Here, we used a novel combination of approaches to rigorously assess the impact of acute alcohol consumption on EAc reactivity for the first time. Using a single-blind, randomized-groups design and ecologically relevant dosing (Table 2), fMRI data were acquired from 49 psychiatrically healthy social drinkers while they performed an fMRI-optimized emotional-faces/places paradigm after consuming alcohol or placebo. The choice of paradigm was rooted in work demonstrating that the amygdala is robustly activated by emotional faces, particularly those depicting expressions of fear28,29,30,31. This has motivated the use of similar paradigms in work focused on the development of anxiety and mood disorders32,33 and the acute impact of alcohol and pharmaceutical (e.g. benzodiazepine) interventions24,25,26,27,34,35, as well as large-scale neuroimaging initiatives (e.g., Human Connectome Project, UK BioBank)36,37. Several methods served to enhance neuroanatomical resolution, including a multiband pulse sequence and advanced co-registration and spatial normalization techniques38 (Table 2). Recently developed, anatomically defined regions-of-interest (ROIs)39,40 made it possible to directly compare the hypothesized dampening effects of alcohol in the BST and the Ce in an unbiased manner. Understanding the acute consequences of alcohol for EAc function is important. It would clarify whether models of substance abuse derived from work in rodents—a species that diverged from the ancestors of modern humans ~75 million years ago41—are relevant to human alcohol consumption14,15. It also promises to inform our understanding of work linking variation in EAc function to the emergence of alcohol abuse42,43 and to provide insight into the EAc’s role in recreational drinking.
Method
Subjects
A total of 61 individuals between the ages of 21 and 35 years were recruited from the community as part of a larger study. All had experience with the highest study dose of alcohol used in the present study (~4–5 standard drinks) within the past 12 months, normal or corrected-to-normal color vision, and reported the absence of lifetime alcohol or substance-related problems, lifetime neurological symptoms, current psychiatric diagnosis or treatment, pervasive developmental disorder or very premature birth, or a medical condition that would contraindicate either acute alcohol consumption or MRI. Twelve subjects were excluded from analyses due to unusable T1-weighted datasets (n = 3), technical problems with the scanner (n = 1), incidental neurological findings (n = 2), inadequate behavioral performance (>2 SDs below the mean; n = 3), or excessive motion artifact (n = 3; see below), yielding a final sample of 49 subjects (46.9% female; Table 2 in the main report). All procedures were approved by the University of Maryland Institutional Review Board and carried out in accordance with the relevant guidelines and regulations. Subjects provided informed written consent.
Overview and General Procedures
Subjects abstained from alcohol and other substances for 24 hours and food/drink for 3 hours prior to the session. At the start of the session, initial sobriety was confirmed using a standard breath assay (Alcosensor IV Breathalyzer; Intoximeters Inc., St. Louis, MO). Subjects were randomly assigned (stratified by sex and race/ethnicity) to receive an alcoholic or placebo beverage, which was consumed just prior to scanning. The decision to employ a between-subjects design was motivated by work underscoring the relatively low test-retest stability of fMRI measures of amygdala reactivity, which renders it suboptimal for randomized cross-over designs44,45. Blood alcohol level (BAL) was assessed immediately before and after scanning. Subject status was continuously monitored using an MRI-compatible eye-tracker. At the end of the session, subjects estimated the number of standard alcoholic drinks that they had consumed.
Alcohol/Placebo Procedures
Well-established procedures were used for administering alcohol or placebo46. Consistent dosing was achieved using a formula that uses height, weight, age, and sex to produce the target BAL of ~0.09% ~30 minutes after the completion of beverage consumption (range: 0.06–0.12%; Table 2)47,48. Alcoholic beverages contained a mixture of juice and 100-proof vodka. To control absorption, subjects consumed 3 equal doses over 30 minutes. The placebo group received a similar beverage, with distilled water replacing the vodka. Subjects assigned to the alcohol (or placebo) group observed the experimenter pouring the vodka (or distilled water) from a vodka bottle. The placebo manipulation was reinforced by floating 3 ml of bitters and 3 ml of vodka on the surface of the beverage and delivering a minute amount of aerosolized vodka to the rim of the beverage containers outside the subject’s view. Immediately following consumption of the third beverage, BAL was assessed and subjects were scanned. BAL was re-assessed immediately following the final scan (inter-assessment period: M = 70 min, SD = 6.0 min), as in prior work49. On average, subjects in the placebo group estimated that they consumed ~2 drinks, confirming the efficacy of the placebo manipulation (Table 2).
Emotional-Faces/Places Paradigm
To assess the impact of acute alcohol administration on EAc function, imaging data were acquired while subjects performed a simple, fMRI-optimized, continuous-performance task. Building on work by our group38,50 and many others24,25,26,27,28,29,30,31,32,33,34,35,51 demonstrating the utility of emotional face paradigms for probing amygdala reactivity—particularly when compared to low-level perceptual control stimuli—subjects viewed alternating blocks of either emotional faces (8 blocks) or places (9 blocks). The use of a block design enhances detection power and mitigates potential concerns about alcohol-induced changes in the shape of the hemodynamic response function (HRF)52,53. Block length (~16.3 s) was also optimized to detect differential blood oxygen level-dependent (BOLD) signals across the two conditions52,53. To maximize signal strength and homogeneity and minimize potential neural habituation52,53,54, each block consisted of 16 brief presentations of faces or places (~1.02 s/image). During face blocks, subjects discriminated (two-alternative/forced-choice) between fearful (75% trials) and emotionally neutral facial expressions (25% trials) presented in a pseudorandomized order. This design choice was aimed at reducing monotony and minimizing potential habituation of the amygdala54. Face stimuli were adapted from prior work by Gamer and colleagues55,56 and included standardized images of unfamiliar male and female adults displaying unambiguous fearful or neutral expressions. To maximize the number of models and mitigate potential habituation, images were derived from several well-established databases: Ekman and Friesen’s Pictures of Facial Affect57, the FACES database58, the Karolinska Directed Emotional Faces database (http://www.emotionlab.se/resources/kdef), and the NimStim Face Stimulus Set (https://www.macbrain.org/resources.htm). Color images were converted to grayscale, brightness normalized, and masked to occlude non-facial features (e.g., ears, hair). During place blocks, subjects discriminated between suburban residential buildings (i.e. houses; 75%) and urban commercial buildings (i.e. skyscrapers; 25%). Grayscale place stimuli were adapted from prior work59,60. Responses were made using an MRI-compatible, fiber-optic response pad (MRA, Washington, PA).
MRI Data Acquisition
MRI data were acquired using a Siemens Magnetom TIM Trio 3 Tesla scanner and 32-channel head-coil. Sagittal T1-weighted anatomical images were acquired using a MPRAGE sequence (TR = 1,900 ms; TE = 2.32 ms; inversion time = 900 ms; flip angle = 9°; sagittal slice thickness = 0.9 mm; in-plane = 0.449 × 0.449 mm; matrix = 512 × 512; field-of-view = 230 × 230). To enhance resolution, a multi-band sequence was used to collect a total of 286 oblique-axial echo planar imaging (EPI) volumes during the faces/places task (multiband acceleration = 6; TR = 1,000 ms; TE = 39.4 ms; flip angle = 36.4°; slice thickness = 2.2 mm, number of slices = 60; in-plane resolution = 2.1875 × 2.1875 mm; matrix = 96 × 96). Images were collected in the oblique axial plane (approximately −20° relative to the AC-PC plane) to minimize susceptibility artifacts. To enable fieldmap correction, two oblique-axial spin echo (SE) images were collected in each of two opposing phase-encoding directions (rostral-to-caudal and caudal-to-rostral) at the same location and resolution as the functional volumes (i.e., co-planar; TR = 7,220 ms; TE = 73 ms).
MRI Data Preprocessing
Given our focus on the EAc, methods were optimized to minimize spatial normalization error and other potential sources of noise. All MRI data were visually inspected before and after processing for quality assurance purposes.
Anatomical Data Processing: Methods are similar to those described in other recent reports by our group38,40. T1 images were brain-extracted (‘skull-stripped’) using a multi-tool approach40. Brain-extracted T1 images were normalized to the MNI152 template using the high-precision diffeomorphic approach implemented in SyN61. The mean of the normalized T1 images is depicted in Supplementary Figure S1. FSL was used to create a fieldmap and undistorted SE image.
Functional Data Processing: The first 3 volumes of each EPI scan were removed. Remaining volumes were de-spiked and slice-time corrected using AFNI62. For co-registration of the functional and anatomical images, an average EPI image was created. The average image was simultaneously co-registered with the corresponding T1-weighted image in native space and corrected for geometric distortions using the boundary-based registration method implemented in FSL and the previously created fieldmap, undistorted SE image, and T1 image. Spatial transformations were concatenated and applied to the functional data in a single step. The transformed images were re-sliced (2-mm3), smoothed (6-mm), and filtered (0.0078125-Hz high-pass). To assess residual motion artifact, the variance of volume-to-volume displacement of a selected voxel in the center of the brain (x = 5, y = 34, z = 28) was calculated using the motion-corrected EPI data. Subjects (n = 3) with extreme motion variance (>2SDs above the mean) were excluded from analyses.
fMRI Modeling: At the first level (single-subject), the emotional-faces/places task was modeled using a boxcar function with place blocks serving as the implicit baseline63. Block onsets were modeled as nuisance variates using two additional event-related predictors. All predictors were convolved with a canonical HRF. Prior research in relatively large samples has failed to uncover alcohol-induced changes in EAc blood flow, mitigating concerns about gross hemodynamic differences64. Additional nuisance variates included motion and physiological noise estimates. To attenuate physiological noise, white matter (WM) and cerebrospinal fluid (CSF) time-series were identified by thresholding the tissue prior images distributed with FSL. The EPI time-series was orthogonalized with respect to the first 3 right eigenvectors of the data covariance matrix from the WM and CSF compartments65.
Reactivity to emotional faces (i.e., the main effect of Stimulus: Emotional Faces vs. Places) was assessed using a voxelwise one-sample t test controlling for potential nuisance variance in mean-centered age and sex. The impact of alcohol administration was assessed using a voxelwise two-sample t test controlling for mean-centered age and sex, equivalent to testing the Stimulus (Emotional Faces vs. Places) × Treatment (Alcohol vs. Placebo) interaction.
Hypothesis Testing Strategy
The major aim of the study was to test the hypothesized dampening effects of acute alcohol administration on EAc reactivity to emotional faces.
EAc Region-of-Interest (ROI) Analyses: The Stimulus × Treatment interaction was rigorously thresholded at p < 0.05 familywise error (FWE) corrected for the extent of the EAc ROI, as in prior work by our group66. The EAc ROI encompassed the amygdala, substantia innominata/sublenticular extended amygdala (SI/SLEA), and BST bilaterally40,67. Consistent with recent recommendations4,6, the ROI was created using the Mai and Harvard-Oxford atlases68,69,70,71,72 and included the probabilistic BST ROI developed by Theiss and colleagues (p > 0%)39 and the Harvard-Oxford probabilistic amygdala. Using this as a starting point, voxels in the region of the SI/SLEA was manually added in the coronal plane of the 1-mm MNI152 template, working from rostral to caudal, and confirmed in the other planes. At intermediate levels of the amygdala’s rostral-caudal axis, where the BST was no longer visible, the SI/SLEA was limited to voxels dorsal to the amygdala and ventral to the putamen and pallidum. SI/SLEA voxels were included until the head of the hippocampus was clearly visible. Voxels in neighboring regions of the accumbens, caudate, putamen, pallidum, thalamus, and ventricles (Harvard-Oxford atlas, p > 50%) were excluded using a Boolean ‘NOT.’ The resulting bilateral EAc ROI was decimated to 2-mm3 (Supplementary Figure S2; total: 1,205 voxels; 9,640 mm3). Significant clusters (p < 0.05, whole-brain FWE corrected) outside the EAc are reported on an exploratory basis for voxelwise analyses of the Condition (Emotional Faces vs. Places) and Stimulus × Treatment effects.
Unbiased Comparison of the BST and Ce: In order to test the whether the BST and the Ce differ in their sensitivity to the hypothesized dampening effects of alcohol in an unbiased manner, we extracted and averaged standardized contrast coefficients using anatomically defined, a priori ROIs39,40 (Supplementary Figure S3). A general linear model was used to compare the impact of Treatment and Hemisphere on regional reactivity to emotional faces. Significant interactions were decomposed using simple effects. The Group effect is reported using the Welch-Satterthwaite correction (FW-S). A power analysis revealed that a minimum of 44 subjects is required to achieve 95% power to detect a Stimulus × Treatment interaction with a between-subjects design (as in the present study) and an estimated effect size of Cohen’s d = 1.13 (reported in ref.25) at p < 0.05, uncorrected.
Results
Behavior
On average, subjects were highly accurate at performing the simple discrimination tasks (M = 86.8%, SD = 7.9). Nevertheless, performance was ~8% lower in the alcohol (M = 83.2%, SD = 8.2) compared to the placebo group (M = 91.1%, SD = 4.9; FW-S(1,47) = 15.98, p < 0.001), consistent with prior work73. Subjects were ~4% more accurate when performing the places (M = 88.8%, SD = 8.8) compared to the faces discrimination (M = 84.4%, SD = 8.4; F(48) = 22.37, p < 0.001), but the Group × Condition interaction was not reliable (F(1,47) = 0.24, p = 0.63). As noted below, control analyses indicated that these modest differences in performance were not the primary determinant of alcohol-related differences in neural reactivity.
The Dorsal Amygdala is Sensitive to Emotional Faces
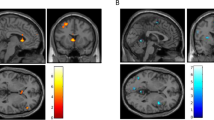
Within the EAc, emotional faces were associated with significant activation of the dorsal amygdala, bilaterally (p < 0.05, FWE-corrected; Left: t = 12.59, volume = 1,032 mm3; x = −20, y = −10, z = −14; Right: t = 12.22, volume = 1,368 mm3; x = 22, y = −8, z = −16; Fig. 2a and Supplementary Table S1), consistent with prior work10. As shown in Supplementary Figure S4, the amygdala cluster overlapped the anatomically defined Ce ROI, with the left and right peaks lying in the dorsocaudal region where the Ce, medial, and basomedial nuclei abut.
The impact of acute alcohol administration on reactivity to emotional faces in the central extended amygdala. (a) Consistent with prior work, voxelwise regression analyses revealed significant activation to emotional faces in the dorsal amygdala (p < 0.05, FWE corrected for the volume of the anatomically defined EAc region-of-interest; total volume: 1,205 voxels; 9,640 mm3). Inset indicates the location of the coronal slice. Significant clusters within the EAc ROI (Supplementary Figure S2) are depicted here. For additional results, see Supplementary Figures S4 and S5 and Supplementary Tables S1 and S2. (b) Voxelwise analyses revealed a significant reduction in reactivity to emotional faces in the region of the left BST in the alcohol compared to the placebo group (same threshold; equivalent to testing the Stimulus × Treatment interaction). The left half of the panel depicts the BST cluster. The right half depicts the BST (green) in the corresponding section of the human brain atlas71. Note the similar appearance of several key landmarks, including the fornix and lateral ventricle (white), as well as the optic tract and anterior commissure (gold). Upper left inset indicates the location of the coronal slice. Upper right inset depicts the myeloarchitecture (Weigert fiber stain) of this region in the atlas. The left BST was the only significant cluster in EAc-focused or whole-brain analyses. For additional results, see Supplementary Figure S6 and Supplementary Table S3. (c) For illustrative purposes, barplot depicts mean standardized regression coefficients extracted from the peak voxel in the BST cluster for the alcohol (light green) and placebo (dark green) groups. Hypothesis testing was performed on a voxelwise basis (corrected for multiple comparisons). Error bars indicate the standard error of the mean. Portions of this figure were adapted with permission from ref.71. Abbreviations—ac, anterior commissure; BST, bed nucleus of the stria terminalis; Cd, caudate; EAc, central division of the extended amygdala; FWE, family-wise error; fx, fornix; GPe, external globus pallidus; GPi, internal globus pallidus; L, left hemisphere; LV, lateral ventricle; OT, optic tract; Pu, putamen; R, right hemisphere; SVC, small volume correction.
On an exploratory basis, we also computed a series of whole-brain analyses. Results indicated that the dorsal amygdala and fusiform cortex (‘fusiform face area’) were significantly more sensitive to emotional faces, whereas the parahippocampal cortex (‘parahippocampal place area’) was significantly more sensitive to places, as expected74,75 (p < 0.05, FWE-corrected; Supplementary Figure S5 and Supplementary Table S2).
Alcohol Dampens BST Reactivity
Within the EAc, acute alcohol administration was associated with a significant reduction in left BST reactivity to emotional faces (Stimulus × Treatment: p < 0.05, FWE-corrected; t = 5.46, volume = 104 mm3; x = −8, y = −2, z = 0; Fig. 2b–c and Supplementary Table S3). As shown in Supplementary Figure S6, the left BST cluster overlapped the anatomically defined BST ROI. The Stimulus × Treatment interaction was not significant in the amygdala at this threshold. Exploratory whole-brain analyses revealed no additional clusters. Control analyses performed using a performance-matched sub-sample (n = 15/group) yielded similar results, suggesting that the dampening effects of alcohol on BST reactivity are not due to group differences in performance.
Alcohol Exerts Similar Effects in the Ce and the BST
To assess potential regional differences in EAc activation in an unbiased manner76, standardized contrast coefficients (i.e., emotional faces vs. places) were extracted from the left and right BST and Ce using anatomically defined, a priori ROIs, as shown in the upper portion of Fig. 3 (Ce: cyan; BST: green). A mixed-model GLM was then used to compare the impact of Treatment and Hemisphere on regional reactivity to emotional faces. Analyses revealed greater activation to faces in the Ce compared to the BST (Region: F(1,47) = 32.99, p < 0.001), consistent with recent high-resolution imaging research77. Analyses also revealed a significant alcohol-dampening effect across regions (Group: FW-S(1,47) = 3.93, p = 0.05). Other omnibus effects were not significant (ps > 0.15). Control analyses performed using a performance-matched sub-sample yielded similar results. Collectively, these observations indicate that alcohol acutely dampens EAc reactivity to emotional faces, it does so similarly in the BST and Ce, and these effects are not an artifact of group differences in task engagement.
The impact of acute alcohol administration on the two major divisions of the EAc. Barplot depicts mean regression coefficients associated with the emotional-faces/places task for the anatomically defined Ce and BST ROIs for each group. The Ce was significantly more reactive to emotional faces, relative to the BST (p < 0.001). On average, subjects randomly assigned to the alcohol group showed significantly less reactivity to emotional faces, relative to those in the placebo group (p = 0.05; equivalent to testing the Stimulus × Treatment interaction). The Treatment × Region interaction was not significant (p = 0.88), suggesting that the Ce and BST are similarly sensitive to acute alcohol dampening. Error bars indicate the standard error of the mean. Abbreviations—EAc, central extended amygdala; ROI, region of interest.
Discussion
Recent epidemiological work indicates that the United States is facing a growing alcohol use crisis78, yet the neural circuitry most relevant to human alcohol consumption has remained unclear. Leveraging a placebo-controlled, randomized-groups design, our voxelwise results demonstrate for the first time that alcohol acutely dampens BST reactivity to emotional faces (Fig. 2). Analyses performed using unbiased, anatomically defined ROIs revealed similar patterns of reduced reactivity in the BST and the Ce (Fig. 3). Control analyses indicated that these results were not an artifact of group differences in performance. Collectively, these findings indicate that acute alcohol intoxication dampens reactivity to emotional faces and it does so similarly across the major divisions of the EAc.
The present findings are broadly consistent with models of alcohol drinking derived from preclinical research in mice and rats14,15,16,17. This work strongly implicates both divisions of the EAc in the anxiety-reducing consequences of alcohol19,23. Alcohol robustly engages the BST and the Ce, as indexed by elevated expression of the immediate early gene c-fos20. Acute alcohol consumption is associated with reduced behavioral signs of anxiety and increased c-fos induction in the BST and Ce21. While the molecular consequences of alcohol are complex, alcohol acutely inhibits excitatory (i.e., glutamatergic) neurotransmission across the EAc and increases inhibitory (i.e., GABA) neurotransmission in the Ce79,80,81. Other work indicates that EAc microcircuits play a critical role in excessive drinking82,83, consistent with evidence implicating the EAc in withdrawal-induced signs of anxiety and stress-induced substance use14. Although these observations highlight the importance of specific cell types and circuits within and between the Ce and BST for alcohol consumption in rodents, the relevance of these discoveries to human drinking and disease has remained unclear. The present results, which underscore the similar consequences of acute alcohol consumption across the EAc, increase our confidence that the pathways identified in rodent models are broadly conserved across species and can guide the development of improved treatments84. The development of integrative animal models that combine focal perturbations of the EAc with the same kinds of paradigms and imaging techniques routinely used in human studies would allow a more complete and detailed synthesis of these distinct bodies of research4,85.
Future Challenges
Although the present study affords new insights into the acute impact of alcohol on the human brain, several limitations and challenges merit comment. First, while single-blind designs are routinely used in acute alcohol challenge studies86, use of a double-blind design would eliminate potential experimenter-expectancy biases. Second, the present study used static images of fearful (75%) and emotionally neutral (25%) faces to probe EAc reactivity. Although fearful faces do not elicit robust signs of fear or anxiety10 (e.g. potentiation of the startle reflex) and are less ecologically valid than dynamic expressions of emotion87, they are widely used in neuroimaging research, rated as more threatening and arousing than neutral or happy faces, and associated with increased behavioral caution88,89,90,91. Fearful faces also promote vigilance; the mere presentation of fearful faces produces persistent increases in visual sensitivity, boosts the resolution of visual processing, and enhances the efficiency of attentional search10. Vigilance is thought to be mediated by circuits emanating from the EAc11 and, once elicited, increases the likelihood of experiencing more extreme or pervasive states of distress10,92. Notably, recent neuroimaging research indicates that individuals with elevated amygdala reactivity to fearful faces are more likely to abuse alcohol in the future, during and following exposure to negative life events (e.g. significant academic, financial, health, or relationship problems)42,43. The present results reinforce the possibility that this prospective association reflects attempts to downregulate, normalize, or self-medicate neural circuits centered on the EAc. Testing this hypothesis is an important challenge for future research. Assessing whether our conclusions generalize to more intense cues, such as threat-of-shock, represents another important avenue. The use of stimuli that elicit robust signs of anxiety (e.g. startle potentiation) would dovetail with work in rodent models, enhancing the likelihood of successful bi-directional translation85,93. Combined with more naturalistic measures of stress-induced drinking in the laboratory or field (e.g., using ecological momentary assessment), this approach might provide a means of stratifying at-risk populations or patients into the subset for whom negative reinforcement circuits are most relevant to intervention.
Conclusions
Existing treatments for excessive alcohol consumption are far from curative94,95, highlighting the need for a deeper understanding of the underlying neural and motivational systems. The present results demonstrate that alcohol acutely dampens EAc reactivity in humans, providing an important source of validation for models of alcohol drinking derived from preclinical research in rodents. The use of a relatively large sample, placebo-controlled between-groups design, ecologically relevant dosing, fMRI-optimized task, best practices for the acquisition and processing of functional neuroimaging data, and unbiased ROI analytic approach enhances our confidence in the clinical and translational significance of these results. More broadly, these observations provide insights into some of the neural systems most relevant to the consumption of alcohol and the initial development of alcohol abuse in humans.
Data Availability
Key statistical maps and regions-of-interest are available at NeuroVault.org (http://neurovault.org/collections/4414/).
References
Grant, B. F. et al. Prevalence of 12-month alcohol use, high-risk drinking, and DSM-IV alcohol use disorder in the United States, 2001–2002 to 2012–2013: Results from the National Epidemiologic Survey on Alcohol and Related Conditions. JAMA psychiatry 74, 911–923 (2017).
Forouzanfar, M. H. et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. The Lancet 388, 1659–1724 (2016).
Rehm, J. et al. Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. The Lancet 373, 2223–2233 (2009).
Fox, A. S. & Shackman, A. J. The central extended amygdala in fear and anxiety: Closing the gap between mechanistic and neuroimaging research. Neuroscience letters.
Avery, S. N., Clauss, J. A. & Blackford, J. U. The human BNST: Functional role in anxiety and addiction. Neuropsychopharmacology 41, 126–141, https://doi.org/10.1038/npp.2015.185 (2016).
Shackman, A. J. & Fox, A. S. Contributions of the central extended amygdala to fear and anxiety. Journal of Neuroscience 36, 8050–8063 (2016).
Inman, C. S. et al. Human amygdala stimulation effects on emotion physiology and emotional experience. Neuropsychologia, https://doi.org/10.1016/j.neuropsychologia.2018.03.019.
Feinstein, J. S., Adolphs, R., Damasio, A. & Tranel, D. The human amygdala and the induction and experience of fear. Current biology 21, 34–38 (2011).
Ahrens, S. et al. A Central Extended Amygdala Circuit That Modulates Anxiety. Journal of Neuroscience 38, 5567–5583 (2018).
Shackman, A. J. et al. The neurobiology of anxiety and attentional biases to threat: Implications for understanding anxiety disorders in adults and youth. Journal of Experimental Psychopathology 7, 311–342 (2016).
Davis, M. & Whalen, P. J. The amygdala: vigilance and emotion. Mol Psychiatry 6, 13–34 (2001).
Adolphs, R. What does the amygdala contribute to social cognition. Annals of the New York Academy of Sciences 1191, 42–61 (2010).
Shackman, A. J. et al. Dispositional negativity: An integrative psychological and neurobiological perspective. Psychological Bulletin 142, 1275–1314 (2016).
Wise, R. A. & Koob, G. F. The development and maintenance of drug addiction. Neuropsychopharmacology 39, 254 (2014).
Koob, G. F. & Volkow, N. D. Neurobiology of addiction: a neurocircuitry analysis. The Lancet Psychiatry 3, 760–773 (2016).
Volkow, N. D., Koob, G. F. & McLellan, A. T. Neurobiologic advances from the brain disease model of addiction. New England Journal of Medicine 374, 363–371 (2016).
Ron, D. & Barak, S. Molecular mechanisms underlying alcohol-drinking behaviours. Nat Rev Neurosci 17, 576–591, https://doi.org/10.1038/nrn.2016.85 (2016).
Goode, T. D. & Maren, S. Common neurocircuitry mediating drug and fear relapse in preclinical models. Psychopharmacology (Berl). https://doi.org/10.1007/s00213-018-5024-3 (in press).
Pandey, S. C. et al. Effector immediate-early gene arc in the amygdala plays a critical role in alcoholism. Journal of Neuroscience 28, 2589–2600 (2008).
Leriche, M., Mendez, M., Zimmer, L. & Berod, A. Acute ethanol induces Fos in GABAergic and non-GABAergic forebrain neurons: a double-labeling study in the medial prefrontal cortex and extended amygdala. Neuroscience 153, 259–267 (2008).
Sharko, A. C., Kaigler, K. F., Fadel, J. R. & Wilson, M. A. Individual differences in voluntary ethanol consumption lead to differential activation of the central amygdala in rats: relationship to the anxiolytic and stimulant effects of low dose ethanol. Alcohol Clin Exp Res 37(Suppl 1), E172–180, https://doi.org/10.1111/j.1530-0277.2012.01907.x (2013).
Sharko, A. C., Kaigler, K. F., Fadel, J. R. & Wilson, M. A. Ethanol-induced anxiolysis and neuronal activation in the amygdala and bed nucleus of the stria terminalis. Alcohol 50, 19–25, https://doi.org/10.1016/j.alcohol.2015.11.001 (2016).
Pandey, S. C., Zhang, H., Roy, A. & Misra, K. Central and medial amygdaloid brain-derived neurotrophic factor signaling plays a critical role in alcohol-drinking and anxiety-like behaviors. J Neurosci 26, 8320–8331, https://doi.org/10.1523/JNEUROSCI.4988-05.2006 (2006).
Gilman, J. M., Ramchandani, V. A., Crouss, T. & Hommer, D. W. Subjective and neural responses to intravenous alcohol in young adults with light and heavy drinking patterns. Neuropsychopharmacology 37, 467–477 (2012).
Gilman, J. M., Ramchandani, V. A., Davis, M. B., Bjork, J. M. & Hommer, D. W. Why we like to drink: a functional magnetic resonance imaging study of the rewarding and anxiolytic effects of alcohol. Journal of Neuroscience 28, 4583–4591 (2008).
Padula, C. B. et al. Alcohol attenuates activation in the bilateral anterior insula during an emotional processing task: a pilot study. Alcohol and alcoholism 46, 547–552 (2011).
Sripada, C. S., Angstadt, M., McNamara, P., King, A. C. & Phan, K. L. Effects of alcohol on brain responses to social signals of threat in humans. Neuroimage 55, 371–380 (2011).
Costafreda, S. G., Brammer, M. J., David, A. S. & Fu, C. H. Y. Predictors of amygdala activation during the processing of emotional stimuli: a meta-analysis of 385 PET and fMRI studies. Brain research reviews 58, 57–70 (2008).
Fusar-Poli, P. et al. Functional atlas of emotional faces processing: a voxel-based meta-analysis of 105 functional magnetic resonance imaging studies. J Psychiatry Neurosci 34, 418–432 (2009).
Sergerie, K., Chochol, C. & Armony, J. L. The role of the amygdala in emotional processing: a quantitative meta-analysis of functional neuroimaging studies. Neurosci Biobehav Rev 32, 811–830, https://doi.org/10.1016/j.neubiorev.2007.12.002 (2008).
Hariri, A. R., Tessitore, A., Mattay, V. S., Fera, F. & Weinberger, D. R. The amygdala response to emotional stimuli: a comparison of faces and scenes. Neuroimage 17, 317–323 (2002).
Stein, M. B., Simmons, A. N., Feinstein, J. S. & Paulus, M. P. Increased amygdala and insula activation during emotion processing in anxiety-prone subjects. Am J Psychiatry 164, 318–327, https://doi.org/10.1176/appi.ajp.164.2.318 (2007).
Swartz, J. R., Knodt, A. R., Radtke, S. R. & Hariri, A. R. A neural biomarker of psychological vulnerability to future life stress. Neuron 85, 505–511 (2015).
Del-Ben, C. M. et al. Effects of diazepam on BOLD activation during the processing of aversive faces. Journal of Psychopharmacology 26, 443–451 (2012).
Paulus, M. P., Feinstein, J. S., Castillo, G., Simmons, A. N. & Stein, M. B. Dose-dependent decrease of activation in bilateral amygdala and insula by lorazepam during emotion processing. Archives of General Psychiatry 62, 282–288 (2005).
Miller, K. L. et al. Multimodal population brain imaging in the UK Biobank prospective epidemiological study. Nat Neurosci 19, 1523–1536, https://doi.org/10.1038/nn.4393 (2016).
Barch, D. M. et al. Function in the human connectome: task-fMRI and individual differences in behavior. Neuroimage 80, 169–189, https://doi.org/10.1016/j.neuroimage.2013.05.033 (2013).
Smith, J. F., Hur, J., Kaplan, C. M. & Shackman, A. J. The impact of spatial normalization for functional magnetic resonance imaging data analyses revisited. bioRxiv (2018).
Theiss, J. D., Ridgewell, C., McHugo, M., Heckers, S. & Blackford, J. U. Manual segmentation of the human bed nucleus of the stria terminalis using 3T MRI. Neuroimage 146, 288–292, https://doi.org/10.1016/j.neuroimage.2016.11.047 (2017).
Tillman, R. M. et al. Intrinsic functional connectivity of the central extended amygdala. Human brain mapping 39, 1291–1312 (2018).
Gibbs, R. A. et al. Evolutionary and biomedical insights from the rhesus macaque genome. Science 316, 222–234, https://doi.org/10.1126/science.1139247 (2007).
Nikolova, Y. S., Knodt, A. R., Radtke, S. R. & Hariri, A. R. Divergent responses of the amygdala and ventral striatum predict stress-related problem drinking in young adults: possible differential markers of affective and impulsive pathways of risk for alcohol use disorder. Mol Psychiatry 21, 348–356, https://doi.org/10.1038/mp.2015.85 (2016).
Elsayed, N. M. et al. Trajectories of alcohol use initiation and risk to develop an alcohol use disorder during adolescence: A role for stress and amygdala activity. BioRxiv (2017).
Lois, G., Kirsch, P., Sandner, M., Plichta, M. M. & Wessa, M. Experimental and methodological factors affecting test-retest reliability of amygdala BOLD responses. Psychophysiology, e13220, https://doi.org/10.1111/psyp.13220.
Cannon, T. D., Cao, H., Mathalon, D. H. & Gee, D. G. & NAPLS Consortium. Reliability of an fMRI paradigm for emotional processing in a multisite longitudinal study: Clarification and implications for statistical power. Hum Brain Mapp 39, 599–601, https://doi.org/10.1002/hbm.23875 (2018).
Shokri-Kojori, E., Tomasi, D., Wiers, C. E., Wang, G.-J. & Volkow, N. D. Alcohol affects brain functional connectivity and its coupling with behavior: greater effects in male heavy drinkers. Molecular psychiatry 22, 1185 (2017).
Curtin, J. J. & Fairchild, B. A. Alcohol and cognitive control: implications for regulation of behavior during response conflict. Journal of abnormal psychology 112, 424 (2003).
Bradford, D. E., Shapiro, B. L. & Curtin, J. J. How bad could it be? Alcohol dampens stress responses to threat of uncertain intensity. Psychological science 24, 2541–2549 (2013).
Trim, R. S. et al. Acute ethanol effects on brain activation in low- and high-level responders to alcohol. Alcohol Clin Exp Res 34, 1162–1170, https://doi.org/10.1111/j.1530-0277.2010.01193.x (2010).
Stout, D. M., Shackman, A. J., Pedersen, W. S., Miskovich, T. A. & Larson, C. L. Neural circuitry governing anxious individuals’ mis-allocation of working memory to threat. Scientific Reports 7, 8742 (2017).
Villalta-Gil, V. et al. Convergent individual differences in visual cortices, but not the amygdala across standard amygdalar fMRI probe tasks. Neuroimage 146, 312–319, https://doi.org/10.1016/j.neuroimage.2016.11.038 (2017).
Maus, B., van Breukelen, G. J., Goebel, R. & Berger, M. P. Optimization of blocked designs in fMRI studies. Psychometrika 75, 373–390 (2010).
Henson, R. Efficient experimental design for fMRI. Statistical parametric mapping: The analysis of functional brain images, 193–210 (2007).
Plichta, M. M. et al. Amygdala habituation: a reliable fMRI phenotype. NeuroImage 103, 383–390 (2014).
Gamer, M., Schmitz, A. K., Tittgemeyer, M. & Schilbach, L. The human amygdala drives reflexive orienting towards facial features. Current Biology 23, R917–R918 (2013).
Scheller, E., Büchel, C. & Gamer, M. Diagnostic features of emotional expressions are processed preferentially. PloS one 7, e41792 (2012).
Ekman, P. & Friesen, W. Pictures of Facial Affect (Palo Alto, CA: Consulting Psychologists). (1976).
Ebner, N. C., Riediger, M. & Lindenberger, U. FACES—A database of facial expressions in young, middle-aged, and older women and men: Development and validation. Behavior research methods 42, 351–362 (2010).
Choi, J. M., Padmala, S. & Pessoa, L. Impact of state anxiety on the interaction between threat monitoring and cognition. NeuroImage 59, 1912–1923 (2012).
Choi, J. M., Padmala, S. & Pessoa, L. Counteracting effect of threat on reward enhancements during working memory. Cognition and Emotion 29, 1517–1526 (2015).
Avants, B. B. et al. A reproducible evaluation of ANTs similarity metric performance in brain image registration. Neuroimage 54, 2033–2044, https://doi.org/10.1016/j.neuroimage.2010.09.025 (2011).
Cox, R. W. AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Computers and Biomedical Research 29, 162–173 (1996).
O’Craven, K. M. & Kanwisher, N. Mental imagery of faces and places activates corresponding stimulus-specific brain regions. Journal of cognitive neuroscience 12, 1013–1023 (2000).
Tolentino, N. J. et al. Alcohol effects on cerebral blood flow in subjects with low and high responses to alcohol. Alcoholism: Clinical and Experimental Research 35, 1034–1040 (2011).
Behzadi, Y., Restom, K., Liau, J. & Liu, T. T. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. Neuroimage 37, 90–101 (2007).
Shackman, A. J. et al. Heightened extended amygdala metabolism following threat characterizes the early phenotypic risk to develop anxiety-related psychopathology. Molecular psychiatry (2016).
Yilmazer-Hanke, D. M. In The human nervous system (eds Mai, J. K. & Paxinos, G.) 759–834 (Academic Press, 2012).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980 (2006).
Frazier, J. A. et al. Structural brain magnetic resonance imaging of limbic and thalamic volumes in pediatric bipolar disorder. American Journal of Psychiatry 162, 1256–1265 (2005).
Goldstein, J. M. et al. Hypothalamic abnormalities in schizophrenia: sex effects and genetic vulnerability. Biological psychiatry 61, 935–945 (2007).
Mai, J. K., Majtanik, M. & Paxinos, G. Atlas of the human brain. (Academic Press, 2015).
Makris, N. et al. Decreased volume of left and total anterior insular lobule in schizophrenia. Schizophrenia research 83, 155–171 (2006).
Trim, R. S. et al. Acute Ethanol Effects on Brain Activation in Low‐and High‐Level Responders to Alcohol. Alcoholism: clinical and experimental research 34, 1162–1170 (2010).
Kanwisher, N. Functional specificity in the human brain: a window into the functional architecture of the mind. Proceedings of the National Academy of Sciences 107, 11163–11170 (2010).
Sabatinelli, D. et al. Emotional perception: meta-analyses of face and natural scene processing. Neuroimage 54, 2524–2533 (2011).
Poldrack, R. A. et al. Scanning the horizon: towards transparent and reproducible neuroimaging research. Nature Reviews Neuroscience 18, 115–126 (2017).
Sladky, R. et al. Unsmoothed functional MRI of the human amygdala and bed nucleus of the stria terminalis during processing of emotional faces. Neuroimage 168, 383–391, https://doi.org/10.1016/j.neuroimage.2016.12.024 (2018).
Schuckit, M. A. Remarkable increases in alcohol use disorders. JAMA psychiatry 74, 869–870 (2017).
Gilpin, N. W., Herman, M. A. & Roberto, M. The central amygdala as an integrative hub for anxiety and alcohol use disorders. Biological psychiatry 77, 859–869 (2015).
Silberman, Y. & Winder, D. G. Ethanol and corticotropin releasing factor receptor modulation of central amygdala neurocircuitry: an update and future directions. Alcohol 49, 179–184 (2015).
Lovinger, D. M. & Kash, T. L. Mechanisms of neuroplasticity and ethanol’s effects on plasticity in the striatum and bed nucleus of the stria terminalis. Alcohol research: current reviews 37, 109 (2015).
de Guglielmo, G. et al. Optogenetic characterization of CeA CRF pathways in alcohol dependence. bioRxiv, 134759 (2017).
Augier, E. et al. A molecular mechanism for choosing alcohol over an alternative reward. Science 360, 1321–1326 (2018).
Hyman, S. E. Back to basics: luring industry back into neuroscience. Nat Neurosci 19, 1383–1384, https://doi.org/10.1038/nn.4429 (2016).
Markou, A., Chiamulera, C., Geyer, M. A., Tricklebank, M. & Steckler, T. Removing obstacles in neuroscience drug discovery: the future path for animal models. Neuropsychopharmacology 34, 74 (2009).
Kaye, J. T., Bradford, D. E., Magruder, K. P. & Curtin, J. J. Probing for neuroadaptations to unpredictable stressors in addiction: Translational methods and emerging evidence. Journal of Studies on Alcohol and Drugs 78, 353–371 (2017).
Zinchenko, O., Yaple, Z. A. & Arsalidou, M. Brain responses to dynamic facial expressions: A normative meta-analysis. Front Hum Neurosci 12, 227, https://doi.org/10.3389/fnhum.2018.00227 (2018).
Blairy, S., Herrera, P. & Hess, U. Mimicry and the judgment of emotional facial expressions. Journal of Nonverbal behavior 23, 5–41 (1999).
Grillon, C. & Charney, D. R. In the face of fear: anxiety sensitizes defensive responses to fearful faces. Psychophysiology 48, 1745–1752 (2011).
Wieser, M. J. & Keil, A. Fearful faces heighten the cortical representation of contextual threat. NeuroImage 86, 317–325 (2014).
Tipples, J. Caution follows fear: Evidence from hierarchical drift diffusion modelling. Emotion 18, 237–247 (2018).
Grupe, D. W. & Nitschke, J. B. Uncertainty and anticipation in anxiety: an integrated neurobiological and psychological perspective. Nature Reviews Neuroscience 14, 488 (2013).
Shackman, A. J. & Fox, A. S. Getting serious about variation: Lessons for clinical neuroscience. Trends in Cognitive Sciences.
Willenbring, M. L. Gaps in clinical prevention and treatment for alcohol use disorders: Costs, consequences, and strategies. Alcohol research: current reviews 35, 238 (2014).
Miller, W. R., Walters, S. T. & Bennett, M. E. How effective is alcoholism treatment in the United States? Journal of studies on alcohol 62, 211–220 (2001).
Acknowledgements
Authors acknowledge critical feedback from E. Bernat, L. Pessoa, M. Roesch, and two anonymous reviewers and assistance from K. DeYoung, L. Friedman, M. Gamer, B. Nacewicz, M. Stockbridge, S. Padmala, P. Spechler, R. Tillman, D. Tromp and personnel from the Affective and Translational Neuroscience Laboratory and Maryland Neuroimaging Center. This work was supported by the University of California, Davis; University of Maryland, College Park; University of Wisconsin—Madison; and National Institutes of Health (DA040717 and MH107444). Development of the NimStim Face Database was overseen by N. Tottenham and supported by the MacArthur Foundation.
Author information
Authors and Affiliations
Contributions
C.M.K., A.J.S. and J.F.S. designed the imaging study based on procedures originally developed and refined by D.E.B. and J.J.C. for psychophysiological research. C.M.K. and J.F.S. collected data. J.H., C.M.K. and J.F.S. processed data. J.H., C.M.K., A.J.S. and J.F.S. analyzed data. J.H., A.J.S., A.S.F. and J.F.S. interpreted data. J.H., C.M.K., J.F.S. and A.J.S. wrote the paper. A.J.S. created figures. J.H., J.F.S. and A.J.S. created tables. A.S.F., D.E.B. and J.J.C. provided theoretical guidance. A.J.S. funded and supervised all aspects of the study. All authors contributed to reviewing and revising the paper and approved the final version.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hur, J., Kaplan, C.M., Smith, J.F. et al. Acute alcohol administration dampens central extended amygdala reactivity. Sci Rep 8, 16702 (2018). https://doi.org/10.1038/s41598-018-34987-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-34987-3
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.