Abstract
Glycoprotein 2[GP2] is a specific target of pancreatic autoantibodies[PAbs] in Crohn’s disease(CD) and is involved in gut innate immunity processes. Our aim was to evaluate the prevalence and prognostic potential of PAbs in primary sclerosing cholangitis(PSC). Sixty-five PSC patients were tested for PAbs by indirect immunofluorescence and compared with healthy (n = 100) and chronic liver disease controls(CLD, n = 488). Additionally, a panel of anti-microbial antibodies and secretory (s)IgA levels were measured, as markers of bacterial translocation and immune dysregulation. PAbs were more frequent in PSC(46.2%) compared to controls(healthy:0% and CLD:4.5%), [P < 0.001, for each]. Occurrence of anti-GP2 antibody was 30.8% (20/65) and was exclusively of IgA isotype. Anti-GP2 IgA positive patients had higher sIgA levels (P = 0.021). With flow-cytometry, 68.4% (13/19) of anti-GP2 IgA antibodies were bound with secretory component, suggesting an active retro-transportation of anti-GP2 from the gut lumen to the mucosa. Anti-GP2 IgA was associated with shorter transplant-free survival [PLogRank < 0.01] during the prospective follow-up (median, IQR: 87 [9–99] months) and remained an independent predictor after adjusting for Mayo risk score(HR: 4.69 [1.05–21.04], P = 0.043). These results highlight the significance of gut-liver interactions in PSC. Anti-GP2 IgA might be a valuable tool for risk stratification in PSC and considered as a potential therapeutic target.
Similar content being viewed by others
Introduction
Primary sclerosing cholangitis (PSC) is a chronic cholestatic liver disease characterized by persistent, progressive biliary inflammation and fibrosis. Clinical manifestations and progression of the disease are heterogeneous and lack reliable biomarkers. Consequently, patients with PSC cannot be stratified appropriately1. Until now only a clinical score, the Mayo risk score, has been proved useful in risk assessment of the disease2. While the etiology of the disease is poorly understood, a dysregulated interplay of key genetic and immunologic pathways with the microbiome appears to contribute to the development of PSC and associated inflammatory bowel diseases (IBD)3. A more detailed exploration of derangement in gut-liver axis might lead to assist the development of disease specific therapy. There is an unmet need for effective medical treatments for PSC, and to date, the only curative therapy is liver transplantation reserved for those with end-stage liver disease4.
Enhanced formation of autoantibodies against acinar cells of the exocrine pancreas have long been described in Crohn’s disease (CD) and considered one of the various serological hallmarks of the disease5. Recently, the target antigens of pancreatic autoantibodies (PAbs) have been identified: the pancreatic major glycoprotein of the zymogen granule membrane (GP2, also referred to as MZGP2) and CUB and zona pellucida-like domain-containing protein 1 (CUZD1)6,7. Further investigations revealed that GP2 plays an important role in intestinal immune responses8,9. Previously, we demonstrated in a large cohort of patients with IBD10, that the presence of PAbs was associated with concomitant PSC and also with faster disease progression in patients with CD. Interestingly, anti-GP2 and anti-CUZD1 antibodies predicted different features of disease progression and notably, only IgA but not IgG antibodies were related to the development of complications. In addition, various other target specific auto- and anti-microbial IgA antibodies were associated with pathologic bacterial translocation in cirrhosis, and thus the development complications11,12. The gut-associated lymphoid tissue (GALT) plays a central role in the IgA antibody formation. Occurrence of serological antibodies directed against gut innate immune proteins or intestinal microorganisms could be a clue of an excessive mucosal immune response due to extended microbial challenge or dysregulation. Association between serological antibodies linked to enteric bacteria and the progressive disease course in PSC is a reasonable hypothesis13,14.
IgA is an important humoral component of intestinal mucosal immunity. During transportation of the molecule through the epithelial cell to the mucosal surface, a cleaved fragment of the polymeric IgA receptor (secretory component, SC) remains covalently attached to the dimeric IgA molecule and this complex is referred to as secretory IgA (sIgA). The molecule has a dual function. It plays a role in neutralizing microbial antigens and in preventing interaction with the intestinal epithelium, a mechanism called immune exclusion. Secretory IgA is also involved in the retro-transportation of antigens to GALT that has important immunological functions15. After retro-transportation of sIgA molecules, they reach the systemic circulation via the thoracic duct and thus can be detectable in sera. In healthy individuals, however, the percentage of SC containing IgA in terms of the total IgA pool is less than 1%.
The aims of this study were to investigate1: the prevalence and type of target specific PAbs in a mixed cohort of pediatric and adult PSC patients with and without IBD2; the association between PAbs and both clinical and laboratory characteristics of the disease3; whether the presence of PAbs is associated with a progressive disease course in PSC; and4 the possible mechanisms related to the formation of PSC-associated PAbs.
Results
Frequency of target-specific PAbs in PSC
Frequencies of IgA and IgG isotype PAbs in PSC and various control groups comprising healthy blood-donors and both IBD and CLD patients are summarized in Table 1. A total of 46.2% (30/65) of PSC patients were positive for either one of the two target-specific PAbs (anti-CUZD1 and anti-GP2), with a significant difference compared to healthy controls (0.0%, P < 0.001) and all disease control groups (CD: 26.8%, P < 0.001 and UC: 7.6%, P < 0.001 or aLC: 6.7%, chrHCV: 4.2% and PBC: 4.9%, P < 0.001 for all CLD). Distribution of the two types of PAbs in PSC was equal, 30.8% (20/65) for each. One-third of the positive cases (10/30) showed double positivity (anti-CUZD1 and anti-GP2). Anti-GP2 antibody positivity was exclusively IgA isotype, while anti-CUZD1 antibodies were of both IgA and IgG isotypes.
Association between target-specific PAbs and clinical or laboratory characteristics of PSC
We analyzed clinical and laboratory characteristics of PSC patients according to their PAb status and summarized these results in Table 2.
PAbs demonstrated no association with gender and younger age at diagnosis. Several laboratory and clinical parameters, indicating more severe disease were significantly higher in the presence of anti-GP2 IgA antibody. Patients with anti-GP2 IgA antibodies had a significantly shorter disease duration (4 [2–7] vs. 7 [3–10] years, P = 0.009), and increased Mayo risk score. All liver enzymes were significantly elevated, while albumin level was decreased in anti-GP2 IgA positive cases compared to anti-GP2 negative ones. There was an elevated occurrence of cirrhosis in anti-GP2 IgA-positive patients (35% vs. 11.1%, P = 0.022). CUZD1 IgA-positivity was associated with shorter disease duration and higher ALP, while CUZD1 IgG-positivity with lower albumin level. There was no difference in Mayo risk score according to anti-CUZD1 antibody status. The presence and subtype of concomitant IBD were similar according to PAb status.
Significance of target-specific PAbs in the risk of progressive disease course in PSC
Seven patients underwent prior orthotopic liver transplantation (OLTx) during the follow-up period. In all cases the indication was the development of end-stage liver disease. Six patients died due to liver-related complications, three out of six deaths occurred after OLTx, therefore the composite end-point (OLTx and/or liver-related death) occurred in a total of 10 patients. One patient died due to acute myocardial infarction, this case was censored at time of event. Median follow-up from inclusion was 2632 [IQR: 286–3022] days. Development of colon cancer occurred in 2, while biliary tract cancer in 1 patient. Nine of involved patients had at least one episode of cholangitis.
We analyzed the association of clinical variables and the different PAbs with poor disease outcome. In Kaplan-Meier analysis, the median time to OLTx and/or liver-related death was 490 days (IQR: 49–1033). Mayo risk score and the presence of cirrhosis (PLog-rank < 0.001 and =0.007, respectively), but not gender (PLog-rank = 0.441), age at onset (PLog-rank = 0.884), disease location (PLog-rank = 0.722) or concomitant IBD (PLog-rank = 0.432) were significantly associated with faster disease progression (Supplementary Figure 1 and Supplementary Table 1).
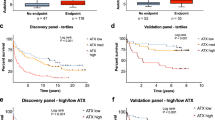
Positivity for IgA isotype anti-GP2 antibody (PLog-rank = 0.008), but not for IgA or IgG isotype anti-CUZD1 antibodies (PLog-rank = 0.335 and 0.998, respectively) predicted OLTx and/or liver-related death, see Fig. 1. Accordingly, univariate Cox regression analysis (Supplementary Table 1) revealed anti-GP2 IgA-positivity as a predictor for poor disease outcome (HR: 5.15 [1.33–19.97], P = 0.018), that remained an independent predictor after adjusting for Mayo risk score in multivariate Cox-regression analysis (HR: 4.69 [1.05–21.04], P = 0.043). A similar tendency was found when we adjusted for the presence of cirrhosis, instead of Mayo risk score (HR: 3.74 [0.9–15.55], P = 0.07). In sensitivity analysis, positivity for anti-GP2 IgA was associated with poor outcome in the subgroup of patients with adult onset PSC (PLog-rank = 0.034) and concomitant IBD (PLog-rank < 0.001), but not in the subgroup of patients with pediatric onset PSC (PLog-rank = 0.098) and no concomitant IBD (PLog-rank = 0.666).
Progressive disease course in primary sclerosing cholangitis (PSC) according to the presence of different target-specific anti-pancreatic antibodies. Patients positive for anti-GP2 IgA have higher cumulative probability of disease progression defined by need for OLTx and/ or death compared to those negative for anti-GP2 IgA antibody. (A) Neither anti-CUZD1 IgA and nor anti-CUZD1 IgG were associated with the progressive disease course (B) GP2: glycoprotein 2, CUZD1: CUB and zona pellucida-like domain-containing protein 1.
Significance of classic serological markers in the risk of progressive disease course in PSC
We also determined the frequency of classic serological markers in our PSC cohort. A total of 83.1% (54/65) and 28.1% (18/64) of PSC patients were positive for IgA/IgG atypical P-ANCA and ASCA and significantly different from the healthy controls (4%, P < 0.001 and 16%, P = 0.04). Regarding Ig isotypes, atypical P-ANCA was primarily IgG, while ASCA was equally IgG or IgA: 83.1% vs. 40% and 18.8% vs. 21.9%, respectively. Furthermore, 18.8% and 9.4% of the patients were positive for IgA type anti-OMP PlusTM and EndoCab antibody, respectively. However, the frequency of these latter serological antibodies did not differ from those of healthy controls (20% and 4.7%).
Association of these antibodies with poor disease outcome was also assessed in Kaplan-Meier analysis. Neither IgG nor IgA isotypes of these serological antibodies were associated with the risk of the progressive disease course nor were corresponding IgA isotypes, see Fig. 2.
Progressive disease course in primary sclerosing cholangitis (PSC) according to the presence of various classic serological markers. Neither IgA and nor IgG atypical P-ANCA (A) ASCA (B) anti-OMP (C) and EndoCab antibodies (D) were associated with the progression of disease. ASCA: anti-Saccharomyces cerevisiae antibodies, P-ANCA: perinuclear anti-neutrophil cytoplasmic antibody, EndoCab: endotoxin-core antibody.
Anti-GP2 IgA antibody status is associated with increased secretory IgA concentration
Serum levels of total sIgA were significantly higher in patients with PSC compared to healthy controls (median [IQR], 96 [73–180] vs. 30 [21–42] μg/ml, P < 0.001). Furthermore, anti-GP2 IgA positive cases had higher sIgA levels compared to anti-GP2 IgA negative ones (149 [86–246] vs. 89 [71–118] μg/ml, P = 0.021). No difference in serum sIgA levels were observed in case of other serological antibodies (Table 3).
Characterization of GP2 antibodies
Finally, we characterized anti-GP2 IgA antibodies in patients with PSC and Crohn’s disease in a flow-cytometry subtyping assay. The presence of secretory component (SC) on GP2 IgA antibodies was 68.4% in PSC (13/19) similar to the frequency (75%,9/12) observed in patients with CD without PSC, see Fig. 3.
Discussion
Previously, we observed high frequency of PSC in anti-GP2-positive CD patients10. We, as well as others16 also reported associations of novel target-specific PAbs (anti-GP2 and anti-CUZD1) with various aspects of progressive disease course in CD patients. Interestingly only IgA, but not IgG isotype antibodies were related to the development of complications in that prospective, large-scale study10. Therefore, we aimed to investigate the prevalence and clinical importance of different target-specific PAbs in a prospective PSC cohort with respect to different immunoglobulin isotypes.
Simultaneously with a research group from Germany, we demonstrated enhanced IgA type anti-GP2 antibody formation in PSC17,18. We used the same cell-based IIF test with an identical cut-off value. As shown elsewhere, IIF test appears to detect anti-GP2 IgA in patients with PSC better in contrast to ELISA10. The IIF test used in this study detects the interaction of IgA with glycosylphosphatidylinositol (GPI)-anchored GP2 on the surface of HEK239 cells6. This might provide a unique epitope structure of GP2 which could be different from one of GP2 adsorbed to ELISA solid phases. A careful comparison with the findings of Jendrek et al. published as a full paper recently19 revealed however, some differences. In our PSC patient cohort, occurrence of anti-GP2 IgA was lower (30.8% vs. 48.7%), while that of anti-CUZD1 antibodies was higher (IgA: 18.5% vs. 9.4% and IgG: 20% vs. 6.3%). The reason for these differences is not fully understood, but is most probably attributed to the composition of the patient populations. First, the Mayo risk score reflecting disease severity was higher in their cohort. Second, the occurrence of biliary tract cancer was 12.3%, while none of our patients had CCA at baseline. Third, in that study anti-GP2 IgA positivity was not exclusively associated with PSC, but rather with the presence of large bile duct diseases, irrespectively of their malignant or benign character based on findings in their disease control groups. Finally, in our study, we used an extensive CLD group with various etiology as disease controls in contrast to the study of Jendrek et al. Neither patients with small bile duct disease (primary biliary cholangitis, PBC n = 102), nor patients with alcoholic liver cirrhosis showed increased PAb frequency. This later finding is particularly intriguing, because in cirrhosis we found high frequency of various serological antibodies, mainly of IgA isotype, such as ASCA (38.5%) or ANCA (52.2%)11,12 that are also frequent in PSC.
Exploring patient characteristics at enrolment, the presence of anti-GP2 IgA-positivity was associated with a more severe disease phenotype. Consistently, anti-GP2 IgA predicted faster disease progression during follow-up, even after adjusting for the Mayo risk score or the presence of cirrhosis. In our study, both liver-related death and OLTx were considered as equal endpoints, since they represent the development of end-stage liver disease as the result of the progressive fibrosis. In the parallel study of Jendrek et al., anti-GP2 IgA-positivity also identified a subgroup of patients with high mortality. Notably, poor survival was primarily attributed to cholangiocarcinoma (CCA) and not to the development of end-stage liver disease in their cohort. In our cohort, only 1 patient (1.5%) developed biliary tract cancer during follow-up, therefore evaluation of PAbs regarding the development of CCA was not possible. This CCA rate is lower than the reported 8–13.2% in other studies20,21, but equals with those ones from Israel (2.1%) and the Netherlands with corresponding follow-up times in recent reports22,23. Supporting clinical evidence for the anti-GP2 IgA - fibrosis linkage in PSC might be the observation that anti-GP2 IgA was more prevalent in CD patients with stricturing disease in previous reports10,16,24.
Formation of anti-GP2 antibody represents the breakdown of tolerance towards the GP2 protein. Glycoprotein 2, an innate immunity protein released from the exocrine pancreas into the intestinal lumen, is also expressed on the apical membrane surface of M-cells. There GP2 can interact with FimH-positive bacteria bearing type-1 fimbriated pyli. GP2-mediated transcytosis is necessary for the initiation of antigen-specific mucosal immune responses against this type of bacterial antigen25. Interestingly we showed, that there is an enhanced immunological response to a wide range of other enteric bacterial antigens in PSC as well. These were as follows: phosphopeptidomannan (ASCA), different endotoxins (EndoCab) and multiple Gram-negative and Gram-positive proteins (OMP). In autoimmune liver disorders, atypical P-ANCA was reported to be directed against human β-tubulin isotype-5 (TTB-5), a protein which shares an extraordinary high structural homology with the microbial cell division protein (FtsZ). FtsZ is present in almost all bacteria in the intestinal microflora. Therefore, occurrence of P-ANCA has been considered a sign of immunological response to enteric bacteria13. However, in our study, serological response to these bacterial antigens was not associated with disease progression in PSC.
To explore the possible link between mucosal immunity and the development of severe phenotype in PSC, the serum levels of total sIgA were measured. A novel finding of our study is that in PSC, the total sIgA levels were significantly elevated, namely three-fold higher, compared to healthy controls. These results indicate an elevated retro-transport of sIgA from the gut mucosal surface in PSC. An additional two-fold elevation of total sIgA levels were found in anti-GP2 IgA-positive cases that was not seen in the presence of other serological anti-microbial antibodies or ANCA. These results suggest that retro-transport is specifically further enhanced in patients with anti-GP2 IgA-positivity. Flow-cytometry based characterisation of anti-GP2 IgA-positive samples revealed that SC was present on these molecules in up to 68.4% of cases. This high percentage of SC strongly indicates that after secretion to the gut lumen, anti-GP2 IgA antibodies are also retro-transported across the mucosal epithelium. Retro-transported sIgA molecules, however, are not “lonely” particles, but rather exist in partnership with their antigens. Since antigen-coupled sIgA molecules are those that have high affinity to their epithelial receptors26, anti-GP2 IgA antibodies grab GP2-coated FimH-positive bacteria as companions. In this way enhanced retro-transport of anti-GP2 IgA might increase microbial overload in the mucosal compartment and perpetuate antigen-induced signalling. Memory T lymphocytes primed in the inflamed gut and homing to the biliary tract via aberrantly expressed adhesion molecules plays a fundamental role in the extension of gut inflammation to the biliary tract27. Interestingly, FimH has been recently identified as a ligand of Toll-like receptor (TLR)-428. Sustained TLR4 activation leads to enhanced fibrosis through TGF-beta signaling29. At the same time, FimH elicits an immune response with enhanced type I Interferon production that has been linked to disease amplification in autoimmunity30. These mechanisms could serve an explanation of how the breakdown of tolerance towards GP2 in the gut is associated with the development of enhanced fibrosis, and thus disease progression in the liver. The GP2—FimH axis deserves further exploration in the pathogenesis of PSC and might also be a highly intriguing issue from the therapeutic point of view. GP2 has a high structural and functional homology with uromodulin (Tamm-Horsfall protein in the urinary tract)9. Recombinant vaccine against the adhesion protein of FimH is already under development in recurrent urinary tract infections. Furthermore mannose-derived FimH antagonists, also hold promise as a novel treatment for UTIs and Crohn’s disease31,32,33.
In summary, the presence of target-specific PAbs is associated with PSC and its more severe phenotype, but not with other CLDs including cholestatic liver disease with small bile duct involvement. The findings of our prospective referral cohort study indicate that anti-GP2 IgA may be a useful additional serological tool in the stratification of PSC patients and is associated with the progressive disease course. In addition, occurrence of IgA type anti-GP2 antibody may serve as an additional clue towards the significance of gut-liver interactions in the disease course of PSC.
Methods
Patient population
We performed an observational cohort study among adult and pediatric PSC patients recruited in Hungarian referral hepatology centers (Hungarian Autoimmune Liver Disease Study Group). This study population is equal with the patient cohort published previously34, with exception that 4 patients were excluded due to prior liver transplantation in this study. In total 65 well-characterized PSC patients with a complete clinical follow-up (adult: 55 [male/female: 39/16], median age at presentation: 28 years [IQR: 19–37], median disease duration: 6 years [2–10] and children: 10 [male/female: 7/3], median age at presentation: 10 years [IQR: 6–11], disease duration: 5 [1.5–7.5]) were included between January, 2006 and December, 2007. Clinical characteristics of patients at enrolment are presented in Table 4.
Diagnosis of PSC was based on clinical, biochemical, serological and cholangiographic (magnetic resonance or endoscopic imaging) features or, when indicated, on histological findings35. Patients with any concomitant malignant disease were excluded. Blood samples and detailed description of clinical phenotypes were obtained at inclusion. Clinical data were determined by thorough review of patients’ medical records, which had been collected in a uniform format. Medical records that documented disease phenotype (age at onset of disease, duration [time elapsed from diagnosis until study inclusion], type of PSC – large duct or small duct), presence and type of concomitant IBD, presence of overlap syndrome, presence of cirrhosis and portal hypertension related complications (e.g. ascites, encephalopathy, oesophageal varices or variceal bleeding), prior orthotopic liver transplantation (OLTx), co-morbidities and medication (e.g. ursodeoxycholic acid, steroid, immunosuppressive and/or biological therapy) at inclusion were retrospectively analyzed for the period prior to the prospective follow-up. At enrolment, revised Mayo risk score was calculated2 and biochemical analyses were performed using standard routine laboratory protocols for tests including platelet count, creatinine, total bilirubin, albumin, international normalized ratio (INR) of prothrombin time, aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and γ-glutamyl-transferase (GGT).
Phenotypical characterization of PSC patients during prospective follow-up
Phenotypical characterization of this patient cohort was performed previously and is provided similarly as the publication34. PSC patients were enrolled into a prospective follow-up study, where treating physicians registered laboratory data, imaging and endoscopic findings, medical treatment, date and type of complications (cirrhosis, colorectal cancer, biliary tract cancer: cholangiocarcinoma [CCA] gallbladder cancer [GBC] or cholangitis) during regular outpatient follow-up visits and inpatient stays. In Hungary, a follow-up visit is usually scheduled for every 6 months at a specialized hepatology center (the actual interval varies between 3–6 months). Collected data were transferred and stored in a database for analysis. On December 1st 2015, all patients’ charts and data were reviewed and updated for the data points mentioned above. Adverse outcome was defined as need for OLTx and/or liver-related death (composite end-point). Follow-up for a particular patient was terminated if adverse outcome occurred or there was no further record available. One case with a non-liver related death (myocardial infarction) was censored at the time of event.
Control groups
Healthy controls and two disease control groups (patients with inflammatory bowel diseases [IBD] and chronic liver diseases [CLD]) were included.
The healthy control group consisted of 100 age-matched individuals (male/female: 45/55, age: 30 years [21–40]) selected from consecutive blood donors in Debrecen. Control subjects did not have any known gastrointestinal or liver diseases.
The inflammatory bowel disease control group consisted of a previously reported patient cohort (CD: 257, male/female: 108/148, age: 25 [19–33] years and ulcerative colitis [UC]: 170, male/female: 78/92, age: 34 [23–44] years)10.
The chronic liver disease control group consisted of patients with primary biliary cholangitis without cirrhosis (PBC, n = 102, male/female: 4/98, age: 59 [52–66] years), chronic hepatitis C virus (chrHCV, n = 119, male/female: 50/69, age: 55 [47–65] years) and alcoholic liver cirrhosis (aLC, n = 267, male/female: 147/120, age: 58 [51–66] years). The diagnosis of PBC was based on biochemical evidence of cholestasis, serum anti-mitochondrial antibodies (AMA) and/or PBC-specific AMA-M2 positivity, compatible histology, and the exclusion of extrahepatic cholestasis36. The diagnosis of chronic HCV was based on positive HCV ribonucleic acid, elevated liver function tests (2xULN for more than 6 months) and compatible liver biopsy, if available. The diagnosis of cirrhosis was based on clinical, biochemical, imaging, and, when available, histological data.
Serological Analysis
Blood samples were obtained at enrolment from each patient and were frozen at −70 °C until testing. All serological assays were performed in a blinded fashion without prior knowledge of the patient’s clinical information.
Detection of target specific serological antibodies
Autoantibodies
IgA and IgG type anti-GP2 and anti-CUZD1 antibodies as well as atypical perinuclear anti-neutrophil cytoplasmic antibodies (P-ANCA) were detected in sera using commercially available cell-based IIFT according to the test instructions [CIBD Mosaic, EUROIMMUN Medizinische Labordiagnostika AG, Lübeck, Germany]. A specific fluorescence at a dilution of 1:10 or higher was considered positive for anti-GP2 and anti-CUZD1, while 1:32 or higher for atypical P-ANCA as recommended by the manufacturer. Detailed description of the protocols has been reported previously10,12 and given in the Supporting Material.
Anti-microbial antibodies
Anti-Saccharomyces cerevisiae (ASCA) (IgA and IgG, US FDA-cleared) and anti-OMP Plus (IgA) (research use only) antibodies were detected by commercially available sandwich enzyme-linked immunosorbent assays (ELISA) employing a specially purified preparation of Saccharomyces cerevisiae antigen and a mixture of multiple bacterial proteins derived from two species of intestinal bacteria (one Gram-positive and one Gram-negative) respectively, with a cut-off positivity at 25 U for all (QUANTA Lite®, Inova Diagnostics, San Diego, CA) as recommended by the manufacturer. Endotoxin core IgA antibody (EndoCAb) directed against a mixture of incomplete endotoxins of 4 different species (Pseudomonas aeruginosa, Salmonella typhimurium, Escherichia coli and Klebsiella aerogenes) was detected by commercially available ELISA (Hycult Biotechnology, Uden, Netherlands). The cut-off positivity was 195 AU/mL, defined as values exceeding the 95th percentile level of the healthy control group.
Detection of serological antibody of mucosal origin
Secretory subtype of total IgA (sIgA) was detected by an in-house sandwich ELISA in PSC and healthy controls. Detailed description of this experiment is presented in the Supporting Material.
Characterization of IgA type anti-GP2 antibodies
We used GP2-coated bead-based in-house flow cytometric immunoassay to determine the presence of secretory component (SC) on anti-GP2 IgA antibodies in sera. Detailed description of this experiment is presented in the Supporting Material.
The IgA subtype analysis was performed in anti-GP2 IgA-positive sera of PSC (n = 19) and Crohn’s disease patients without PSC (n = 14) by IIFT and was also verified by ELISA test (anti-GP2 IgA, GA Generic Assays, Dahlewitz/Berlin, Germany). Anti-GP2 IgA-negative sera of healthy subjects (n = 20) served as controls.
Ethical considerations
The study protocol was approved by the Regional and Institutional Research Ethics Committee of University of Debrecen and the National Scientific and Research Ethics Committee (DEOEC-RKEB/IKEB 5306–9/2011, 3515–2011, 3885/2012/EKU [60/PI/2012], 3880/2012/EKU [59/PI/2012]). Each patient was informed of the nature of the study and gave informed consent in writing. All methods were performed in accordance with the relevant guidelines and regulations2,6,35,36,37.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Statistical methods
Variables were tested for normality using Shapiro Wilk’s W test. Continuous variables were summarized as medians (interquartile range [IQR, lowest 25% - highest 25%]) and were compared with the Mann-Whitney U test. Categorical variables were compared with χ2 test or Fisher’s exact test, as appropriate. Kaplan–Meier analysis was used to determine association between serological antibodies and adverse disease outcome (OLTx and/or liver-related mortality). Differences in observed survival were assessed by the log-rank test. The association of categorical clinical variables or serological antibodies with adverse disease outcome during the follow-up was evaluated by univariate Cox-regression analysis. Multivariate analyses were performed to adjust for the Mayo risk score or presence of cirrhosis with forced entry method. Associations are given as hazard ratio [HR] with 95% confidence intervals [CI]. For statistical analysis and graphical presentation, the SPSS v.22.0 (SPSS, Chicago, IL), and GraphPad Prism 6 (San Diego, CA) programs were used. A two-sided probability value of < 0.05 was considered to be statistically significant.
References
Lazaridis, K. N. & LaRusso, N. F. Primary Sclerosing Cholangitis. N. Engl. J. Med. 375, 1161–1170 (2016).
Kim, W. R. et al. A revised natural history model for primary sclerosing cholangitis. Mayo Clin. Proc. 75, 688–694 (2000).
Eksteen, B. The Gut-Liver Axis in Primary Sclerosing Cholangitis. Clinics in Liver Disease 20, 1–14 (2016).
Sirpal, S. & Chandok, N. Primary sclerosing cholangitis: diagnostic and management challenges. Clin. Exp. Gastroenterol. 10, 265–273 (2017).
Roggenbuck, D. et al. Crohn’s disease specific pancreatic antibodies: clinical and pathophysiological challenges. Clin. Chem. Lab. Med. 52, 483–494 (2014).
Komorowski, L. et al. Autoantibodies against exocrine pancreas in Crohn’s disease are directed against two antigens: the glycoproteins CUZD1 and GP2. J. Crohns. Colitis 7, 780–790 (2013).
Roggenbuck, D. et al. Identification of GP2, the major zymogen granule membrane glycoprotein, as the autoantigen of pancreatic antibodies in Crohn’s disease. Gut 58, 1620–1628 (2009).
Schierack, P. et al. Species-specific and pathotype-specific binding of bacteria to zymogen granule membrane glycoprotein 2 (GP2). Gut 64, 517–519 (2015).
Roggenbuck, D. et al. Glycoprotein 2 antibodies in Crohn’s disease. Adv. Clin. Chem. 60, 187–208 (2013).
Papp, M. et al. Rediscovery of the Anti-Pancreatic Antibodies and Evaluation of their Prognostic Value in a Prospective Clinical Cohort of Crohn’s Patients: The Importance of Specific Target Antigens [GP2 and CUZD1]. J. Crohns. Colitis 9, 659–668 (2015).
Papp, M. et al. Presence of anti-microbial antibodies in liver cirrhosis–a tell-tale sign of compromised immunity? PLoS One 5, e12957 (2010).
Papp, M. et al. High prevalence of IgA class anti-neutrophil cytoplasmic antibodies (ANCA) is associated with increased risk of bacterial infection in patients with cirrhosis. J. Hepatol. 59, 457–466 (2013).
Terjung, B. et al. p-ANCAs in autoimmune liver disorders recognise human beta-tubulin isotype 5 and cross-react with microbial protein FtsZ. Gut 59, 808–816 (2010).
Papp, M. & Lakatos, P. L. Serological studies in inflammatory bowel disease: how important are they? Curr. Opin. Gastroenterol. 30, 359–364 (2014).
Mantis, N. J., Rol, N. & Corthesy, B. Secretory IgA’s complex roles in immunity and mucosal homeostasis in the gut. Mucosal Immunol. 4, 603–611 (2011).
Michaels, M. A. et al. Pancreatic Autoantibodies Against CUZD1 and GP2 Are Associated with Distinct Clinical Phenotypes of Crohn’s Disease. Inflamm. Bowel Dis. 21, 2864–2872 (2015).
Jendrek, S. T. et al. SAT-387 - Autoreactive Iga Antibodies against the Pancreatic Major Glycoprotein 2 are Associated with Primary Sclerosing Cholangitis and Related Biliary Tract Cancer. J. Hepatol. 64, S647 (2016).
Papp, M. et al. Target-specific anti-pancreatic antibodies are frequent in patients with primary sclerosing cholangitis and associated with poor disease outcome. In Falk Symposia 201 - Gut-Liver Interactions: From IBD to NASH 33–34 (2016).
Jendrek, S. T. et al. Anti-GP2 IgA autoantibodies are associated with poor survival and cholangiocarcinoma in primary sclerosing cholangitis. Gut 66, 137–144 (2017).
Broome, U. et al. Natural history and prognostic factors in 305 Swedish patients with primary sclerosing cholangitis. Gut 38, 610–615 (1996).
Farrant, J. M. et al. Natural history and prognostic variables in primary sclerosing cholangitis. Gastroenterology 100, 1710–1717 (1991).
Boonstra, K. et al. Population-based epidemiology, malignancy risk, and outcome of primary sclerosing cholangitis. Hepatology 58, 2045–2055 (2013).
Yanai, H. et al. Prognosis of primary sclerosing cholangitis in israel is independent of coexisting inflammatory bowel Disease. J. Crohns. Colitis 9, 177–184 (2015).
Degenhardt, F. et al. Serologic Anti-GP2 Antibodies Are Associated with Genetic Polymorphisms, Fibrostenosis, and Need for Surgical Resection in Crohn’s Disease. Inflamm. Bowel Dis. 22, 2648–2657 (2016).
Hase, K. et al. Uptake through glycoprotein 2 of FimH(+) bacteria by M cells initiates mucosal immune response. Nature 462, 226–230 (2009).
Duc, M., Johansen, F.-E. & Corthesy, B. Antigen binding to secretory immunoglobulin A results in decreased sensitivity to intestinal proteases and increased binding to cellular Fc receptors. J. Biol. Chem. 285, 953–960 (2010).
Pollheimer, M. J., Halilbasic, E., Fickert, P. & Trauner, M. Pathogenesis of primary sclerosing cholangitis. Best Pract. Res. Clin. Gastroenterol. 25, 727–739 (2011).
Mossman, K. L. et al. Cutting edge: FimH adhesin of type 1 fimbriae is a novel TLR4 ligand. J. Immunol. 181, 6702–6706 (2008).
Seki, E. et al. TLR4 enhances TGF-[beta] signaling and hepatic fibrosis. Nat Med 13, 1324–1332 (2007).
Hall, J. C. & Rosen, A. Type I interferons: crucial participants in disease amplification in autoimmunity. Nat. Rev. Rheumatol. 6, 40–49 (2010).
Mydock-McGrane, L. K., Cusumano, Z. T. & Janetka, J. W. Mannose-derived FimH antagonists: a promising anti-virulence therapeutic strategy for urinary tract infections and Crohn’s disease. Expert Opin. Ther. Pat. 26, 175–197 (2016).
Dreux, N. et al. Point Mutations in FimH Adhesin of Crohn’s Disease-Associated Adherent-Invasive Escherichia coli Enhance Intestinal Inflammatory Response. PLoS Pathog. 9, e1003141 (2013).
Gouin, S. G. Repurposing Escherichia coli antiadhesives in Crohn’s disease. Future Med. Chem. 8, 1903–1906 (2016).
Tornai, T. et al. Gut barrier failure biomarkers are associated with poor disease outcome in patients with primary sclerosing cholangitis. World J. Gastroenterol. 23, 5412–5421 (2017).
Martins, E. B. & Chapman, R. W. Sclerosing cholangitis. Curr. Opin. Gastroenterol. 17, 458–462 (2001).
Kaplan, M. M. & Gershwin, M. E. Primary biliary cirrhosis. N. Engl. J. Med. 353, 1261–1273 (2005).
Papp, M. et al. Evaluation of the combined application of ethanol-fixed and formaldehyde-fixed neutrophil substrates for identifying atypical perinuclear antineutrophil cytoplasmic antibodies in inflammatory bowel disease. Clin. Vaccine Immunol. 16, 464–470 (2009).
Acknowledgements
The authors are very much grateful to Eva Tömöri for her technical assistance. Maria Papp was supported by the János Bólyai Research Scholarship of Hungarian Academy of Sciences (BO/00232/17/5) and Scholarship of Leader Scientist and Tutor of University of Debrecen. Tamás Tornai was supported through the New National Excellence Program of the Ministry of Human Capacities (ÚNKP-16–3). This study was supported by IOIBD Research Grant (2011–2013) and the Research Grant of National Research Development and Innovation Office (K115818/2015/1).
Author information
Authors and Affiliations
Contributions
P.M., T.T. and T.I. made the concept and designed the present study. T.T., S.N., V.G., P.G., P.A., S.Z.F., L.P.L., P.M. made the acquisition of data. T.D., A.R., A.S.Z.P., P.M. made the experimental work. P.M., T.T., G.L.N., D.G., K.F., A.S.Z.P. made the analysis and interpretation of the data. P.M., T.T. drafted the article. A.S.Z.P., G.L.N., D.R., K.F. revised the article critically for important intellectual content. All authors approved the final version of the manuscript to be submitted.
Corresponding author
Ethics declarations
Competing Interests
Gary L. Norman is an employee of Inova Diagnostics, an in-vitro diagnostic manufacturer. Kai Fechner is an employee of EUROIMMUN Medizinische Labordiagnostika AG, an in-vitro diagnostic manufacturer. Prof. Dr. Dirk Roggenbuck is an employee and owns shares of GA Generic Assays GmbH, an in-vitro diagnostic manufacturer.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tornai, T., Tornai, D., Sipeki, N. et al. Loss of tolerance to gut immunity protein, glycoprotein 2 (GP2) is associated with progressive disease course in primary sclerosing cholangitis. Sci Rep 8, 399 (2018). https://doi.org/10.1038/s41598-017-18622-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-18622-1
This article is cited by
-
The microbiota and the gut–liver axis in primary sclerosing cholangitis
Nature Reviews Gastroenterology & Hepatology (2023)
-
Antigen presentation, autoantibody production, and therapeutic targets in autoimmune liver disease
Cellular & Molecular Immunology (2021)
-
The search for the Holy Grail: autoantigenic targets in primary sclerosing cholangitis associated with disease phenotype and neoplasia
Autoimmunity Highlights (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.