Abstract
Dengue virus (DENV) infection in neuronal cells was speculated to trigger neuropathy. Herein, we determined the blockade of DENV infection by targeting endocytic pathways in vitro and in vivo. In DENV-infected mouse brains, we previously showed that viral proteins are expressed in neuronal cells around the hippocampus with accompanying neurotoxicity. DENV caused infection, including entry, double-stranded (ds)RNA replication, protein expression, and virus release, followed by cytotoxicity in the mouse neuronal Neuro-2a cell line. Pharmacologically blocking clathrin-mediated endocytosis of the DENV retarded viral replication. Targeting vacuolar-type H+-ATPase (V-ATPase)-based endosomal acidification effectively blocked the DENV replication process, but had no direct effect on viral translation. Blockade of the clathrin- and V-ATPase-based endocytic pathways also attenuated DENV-induced neurotoxicity. Inhibiting endosomal acidification effectively retarded DENV infection, acute viral encephalitis, and mortality. These results demonstrate that clathrin mediated endocytosis of DENV followed by endosomal acidification-dependent viral replication in neuronal cells, which can lead to neurotoxicity.
Similar content being viewed by others
Introduction
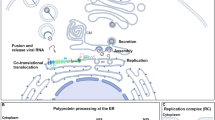
Dengue virus (DENV), a positive-sense single-stranded RNA virus of the family Flaviviridae, causes approximately 50 million infections per year1. The DENV genome encodes structural proteins (capsid (C), pre-membrane (prM), and envelope (E) proteins) and nonstructural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5) that are involved in viral infection and pathogenesis2. Through putative receptors, such as heparan sulfate, mannose receptor, dendritic cell-specific ICAM-grabbing non-integrin (CD209), heat shock protein 90/70, phosphatidylserine receptors, etc., DENV causes various infections that are both cell-specific and receptor-dependent3. Different serotypes of DENV cause clathrin-dependent and -independent endocytosis, and various cell types of the DENV infection entry pathway occur through a non-classical endocytic pathway, independent of caveolae and lipid rafts4. Following viral receptor-mediated endocytosis accompanied by vacuolar-type hydrogen (H+)-ATPase (V-ATPase)-mediated endosomal acidification, viral RNA can be uncoated from the enveloped virus and released into the cytoplasm for protein translation and genome replication3. Targeting the endocytic pathways of DENV infection, including viral entry/fusion and uncoating, confers a wide range of antiviral effects3, 5, 6.
Flaviviral encephalitis, including Japanese encephalitis, West Nile encephalitis, St. Louis encephalitis, Murray Valley encephalitis, etc., is well recognized in endemic regions, clinically characterized by a reduced level of consciousness associated with seizures, poliomyelitis-like paralysis, and parkinsonian movement disorders7. The emergence of neurotropic arboviruses, such as Zika virus, chikungunya virus, and DENV, is associated with acute neurological complaints8, 9. In severe dengue infection, symptoms include plasma leakage, mucosal bleeding, neurological dysfunction, and multiple organ failure1, 2. Although DENV is not a characteristic neurotropic virus, dengue is widely considered to be one of the leading causes of neurological manifestations, including encephalitis, encephalopathy, dengue-associated neuromuscular complications, and dengue-associated neuro-ophthalmic complications10,11,12. However, the pathogenesis of dengue-associated neurological complications is not fully understood due to a lack of appropriate evaluations in previous studies. A retrospective 2-year study reported that dengue patients may show various persistent neurological complications13. DENV is rapidly replicated and causes viremia during the acute phase of infection, and it was speculated that acute viral encephalitis and central nervous system (CNS) inflammation may facilitate dengue-associated neurological complications.
Intracranial infection with non-adapted DENV induces lethality in immunocompetent mice following limb paralysis, seizures, and encephalitis14,15,16. Expressions of viral antigens, double-stranded (ds)RNA, and virus particles are found in DENV-infected brains. Importantly, in vivo DENV infection in mouse brains causes neuronal damage in the pyramidal layer of Cornu Ammonis (CA) areas of the hippocampus14, 16. Based on viral antigen identification, neurons can be infected by DENV; however, viral receptors and infectious routes have not been well addressed. The targeting of dopamine receptors, which are expressed in hippocampal neurons17, retarded DENV infection in vitro 18, 19 and in vivo 19. Studies showed induction of neuronal cell death, particularly apoptosis, by DENV infection both in vitro 20,21,22,23 and in vivo 14, 16, 24. This study attempted to investigate the infectious route of DENV in neuronal cells and evaluate potential antiviral strategies by targeting endocytic pathways of DENV infection in vitro and in vivo.
Results
DENV initiates infection in neuronal cells in vitro, including viral entry, RNA replication, protein expression, and viral release
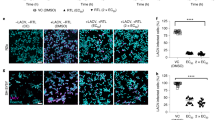
In DENV-infected brains of 7-day ICR mice16, we noted that NS3-positive cells were present within the Iba-1-negative cell population, suggesting the ability of DENV to infect non-microglia in vivo. Previous work15, 24 showed that DENV can infect neuronal cells in vivo; however, the effects of DENV on neuronal cells remain poorly understood. To demonstrate infection efficacy in Neuro-2a cells, we performed fluorescent DENV staining followed by fluorescent imaging (Fig. 1A) and a flow cytometric analysis (Fig. 1B). Results showed viral binding/entry at 2 h post-inoculation. To confirm this finding, we used confocal microscopy to evaluate the intracellular localization of fluorescence-stained DENV in Neuro-2a cells (Fig. 1C). To investigate DENV replication in neuronal cells, we used immunostaining, which demonstrated significant viral dsRNA expression in DENV-infected Neuro-2a cells (p < 0.05, Fig. 1D). We also performed Western blotting (Fig. 1E) and a quantitative polymerase chain reaction (qPCR) (Fig. 1F). Our results confirmed viral NS1 protein expression in Neuro-2a cells 24 h post-infection. A plaque assay, which was performed to determine viral replication and release, showed significant (p < 0.05) infection of Neuro-2a cells by DENV (Fig. 1G).
Dengue virus (DENV) serotype 2 PL046 (DENV2) causes infection, including viral entry, double-stranded (ds)RNA replication, viral protein expression, virus release, and neurotoxicity, in the Neuro-2a murine neuronal cell line. (A) Fluorescence, (B) flow cytometric, and (C) three-dimensional confocal image analyses showed Neuro-2a cells carrying Alexa-594 labeled (red) DENV2 at 2 h post-infection. Phalloidin (green) staining indicates actin. (D) Immunocytochemistry and the relative mean fluorescence intensity (MFI) of viral dsRNA (green) at 24 h post-infection. (E) The Western blot analysis showed viral nonstructural protein1 (NS1) expression at 24 h post-infection. The relative ratio to β-actin is shown. (F) A qPCR showed the time-kinetic mRNA expression of NS1, and (G) plaque assays showed the level of viral replication at 24 h post-infection. (H) Cell morphology, (I) MTT, and (J) lactate dehydrogenase (LDH) assays showed cell growth, viability, and cytotoxicity, respectively, in DENV-infected cells for the indicated times and different multiplicities of infection (MOIs). For all images and blots, representative data were selectively obtained from three individual experiments. DAPI staining indicates nuclei (blue). For the flow cytometric analysis, the percentage of positive cells is shown. All quantitative data are shown as the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001. ns, not significant.
Following infection, DENV can cause neurotoxicity as shown by histopathological changes and apoptotic staining16. Changes in cell morphology, cell growth, and cytotoxicity were used to evaluate the effects of DENV on neuronal cells. Microscopic observations revealed a significant change in Neuro-2a cells after DENV infection (Fig. 1H). The MTT (Fig. 1I) and lactate dehydrogenase (LDH; Fig. 1J) assays showed that DENV caused cell growth inhibition and cytotoxicity, particularly when cells were incubated with a high multiplicity of infection (MOI). These results indicated that DENV can infect neuronal cells and cause neurotoxicity in vitro.
D2R mediates DENV2 infection in Neuro-2a cells
Currently, the targeting of dopamine receptors retards DENV infection in vitro 18, 19 and in vivo 19, suggesting a potential role of dopamine receptor-mediated DENV infection. We performed immunostaining that revealed expression of dopamine receptor D2 (D2R), but not D4R, in Neruo-2a cells (Supplemental Fig. 1A). Additionally, immunostaining showed the expression of D2R in isolated NeuN-positive hippocampal neurons, a neuronal nuclear antigen that is commonly used as a biomarker for neurons (Supplemental Fig. 1B). Fluorescent DENV staining followed by fluorescent imaging showed significant viral binding/entry in isolated hippocampal neurons (Supplemental Fig. 1C). Plaque assays confirmed that significant DENV replication occurred in isolated hippocampal neurons (Supplemental Fig. 1D).
To verify the essential role of D2R in mediating DENV infection, we used metoclopramide (MCP) to pharmacologically decrease DENV binding/entry (Supplemental Fig. 1E) and viral replication (Supplemental Fig. 1F) in Neuro-2a cells, as respectively shown by fluorescent imaging and plaque assays. These findings indicated that DENV caused infection of neuronal cells in a D2R-mediated manner.
DENV2 infects Neuro-2a cells via clathrin-mediated endocytosis
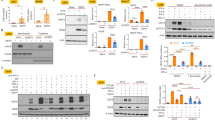
DENV infects cells via distinct entry pathways for DENV internalization, including clathrin-mediated and clathrin-independent endocytosis3, 25, 26. However, no further evidence has revealed the endocytic pathway of DENV in neuronal cells. To investigate the involvement of (clathrin-mediated) endocytosis, the pharmacological inhibitors, CPZ and Pitstop 2, were utilized as previously described4. We performed fluorescent DENV staining followed by fluorescent imaging (Fig. 2A) and a flow cytometric analysis (Fig. 2B). Treatment with CPZ and Pitstop 2 had significantly reduced viral binding/entry in Neuro-2a cells at 2 h post-inoculation. In confirmation of this finding, immunostaining demonstrated a significant decrease in viral dsRNA expression in DENV-infected Neuro-2a cells co-treated with CPZ and Pitstop 2 (Fig. 2C). We also performed plaque assays to study viral replication and release, and results showed significant (p < 0.01) inhibition in DENV-infected Neuro-2a cells (Fig. 2D). Treatment with CPZ and Pitstop 2 did not affect cell growth or cytotoxicity, but they caused an inhibition of acidification following DENV infection. These results show clathrin-regulated DENV binding/entry followed by viral replication in neuronal cells in vitro.
Inhibition of clathrin-mediated endocytosis partly reduces dengue virus serotype 2 (DENV2) infection in Neuro-2a cells. Neuro-2a cells were inoculated with Alexa-594-labeled DENV2 (at a multiplicity of infection of 1) for 2 h in the presence of the clathrin inhibitors, chlorpromazine (CPZ; 5 ng/ml) and Pitstop2 (30 µM). (A) Fluorescent microscopy and (B) flow cytometry were respectively used to measure the expression and percentage of cells carrying fluorescent DENV2. (C) Immunocytochemistry and the relative mean fluorescence intensity (MFI) of viral dsRNA (green) 24 h post-infection. (D) Plaque assays show the level of viral replication 24 h post-infection. For all images, representative data were selectively obtained from three individual experiments. Quantitative data are depicted as the mean ± SD. *p < 0.05, **p < 0.01, and ***p < 0.001. ns, not significant.
Blockade of endosomal acidification attenuates DENV2 infection in Neuro-2a cells
Once the dengue viral receptor mediates endocytosis, V-ATPase facilitates endosomal acidification for an uncoating of the viral genome3. A previous study showed that the dengue prM protein interacts with V-ATPase to facilitate viral entry and egression27. We used BafA1 and ConA, which are inhibitors of V-ATPase28, to decrease endosomal acidification. Immunostaining demonstrated a blockade of viral dsRNA expression in DENV-infected Neuro-2a cells pretreated with BafA1 (p < 0.05, Fig. 3A). We also performed Western blotting, a qPCR, and plaque assays to respectively confirm that inhibiting endosomal acidification reduced viral NS1 protein expression (Fig. 3B), NS1 gene expression (Fig. 3C), and viral replication (Fig. 3D) in Neuro-2a cells at 24 h post-infection. No changes in cell morphology (Fig. 3E), cell growth, or cytotoxicity were observed after drug treatment followed by DENV infection at 24 h post-infection. Treatment with BafA1, ConA, and lysosomotropic agents, such as chloroquine and NH4Cl, reduced acidification. These results indicate that inhibiting endosomal acidification attenuated DENV infection in neuronal cells.
Inhibition of endosomal acidification effectively abolishes dengue virus serotype 2 (DENV2) infection in vitro. Neuro-2a cells were inoculated with DENV2 (at a multiplicity of infection of 1) for 24 h in the presence of the V-ATPase inhibitor, bafilomycin A1 (BafA1; 100 nM) or concanamycin A (ConA; 50 nM). (A) Immunocytochemistry and the relative mean fluorescence intensity (MFI) of viral dsRNA (green). (B) Western blotting analysis shows viral NS1 and NS4B expressions. The relative ratio to β-actin is shown. (C) qPCR shows the mRNA expression of NS1. (D) Plaque assays show the level of viral replication. (E) Cell morphology of DENV2-infected cells. For all images, representative data were selectively obtained from three individual experiments. Quantitative data are depicted as the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001. ns, not significant.
Inhibition of endosomal acidification reduces DENV2 infection independent of translation
We confirmed the inhibitory role of endosomal acidification only in viral genome uncoating but not in other steps of the viral cell cycle, by using firefly luciferase activity in BHK-D2-Fluc-SGR-Neo-1 cells, and found that treatment with BafA1 caused no direct inhibitory effects on viral translation or replication (Fig. 4A) or cytotoxicity in cells (Fig. 4B). However, consistent with results in Neuro-2a cells, BafA1 caused blockade of DENV2 replication in parental BHK-21 cells, as shown by plaque assays (Fig. 4C). These results indicate that the blockade of endosomal acidification had no direct effects on viral inhibition via alterations of viral translation and replication.
Inhibition of endosomal acidification does not repress firefly luciferase activity in BHK-D2-Fluc-SGR-Neo-1 cells. (A) Luciferase activity and (B) lactate dehydrogenase (LDH) assays in bafilomycin A1 (BafA1; 100 nM)-treated parental BHK-21 and BHK-D2-Fluc-SGR-Neo-1 cells (replicons) 24 h post-treatment. (C) Plaque assays show the level of viral replication 24 h post-infection in dengue virus serotype 2 (DENV2) (at a multiplicity of infection of 1)-infected BHK-21 cells with or without BafA1 (100 nM) at 0.5 h of pretreatment. Quantitative data are depicted as the mean ± SD of three independent experiments. ***p < 0.001. ns, not significant.
Targeting endocytic pathways reduces in vitro neurotoxicity induced by DENV infection
Infection with DENV causes neuronal cell apoptosis20,21,22,23. To analyze roles of endocytic pathways involved in DENV infection in neuronal cells, we showed that pretreatment with CPZ, Pitstop2 (for blocking clathrin-mediated endocytosis), BafA1, and ConA (for inhibiting acidification) effectively abolished DENV-induced changes in cell morphology (Fig. 5A), cell growth inhibition (Fig. 5B), and cytotoxicity (Fig. 5C). Treatment with MCP (for inhibiting D2R) was used as a positive control, as D2R mediates DENV binding/entry. These results indicate the involvement of endocytic pathways in DENV-induced neurotoxicity in vitro.
Pharmacologically targeting endocytic pathways reduces dengue virus serotype 2 (DENV2)-induced neurotoxicity in vitro. Following pretreatment with chlorpromazine (CPZ; 5 ng/ml), Pitstop2 (30 µM), metoclopramide (MCP; 10 µM), BafA1 (100 nM), or concanamycin A (ConA; 50 nM), (A) cell morphology, (B) MTT, and (C) lactate dehydrogenase (LDH) assays respectively show cell growth, viability, and cytotoxicity in DENV (at a multiplicity of infection of 10)-infected cells at 72 h. For all images, representative data were selectively obtained from three individual experiments. All quantitative data are shown as the mean ± SD of three independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001. ns, not significant.
Inhibiting endosomal acidification decreases DENV infection, neural impairment, and mortality in suckling mice
Our previous animal model of DENV infection used immunocompetent mice of the ICR strain to induce viral replication in the brain causing acute encephalitis16. BafA1 was administered at days 0 and 1 post-infection. We exploited a series of methods, including a Western blot analysis of the NS1 and NS4B viral proteins (Fig. 6A) and plaque assays for detecting virus replication (Fig. 6B). According to our results, DENV caused significant infection and replication in mouse brains at 7 days post-infection, as BafA1 inhibited viral protein expression and replication. Mice with neurological changes were then evaluated as previously described16. We monitored time-dependent changes in clinical scores, which were graded according to the severity of illness as follows: 0 for healthy; 1 for minor illness, including weight loss, reduced mobility, and a hunchback body orientation; 2 for limbic seizures; 3 for moving with difficulty and anterior limb or posterior limb weakness; 4 for paralysis; and 5 for death. A significant increase in clinical scores (Fig. 6C) had occurred in DENV-infected mice compared to mock-infected mice by 7 days post-infection. The survival rate of DENV-infected mice had decreased by days 8 or 9 post-infection, and all of the mice had died by days 9 or 10 post-infection (Fig. 6D). Either co- or post-treatment with BafA1 significantly reduced DENV-induced disease progression and mortality. The data indicated that inhibiting endosomal acidification abolished encephalitic DENV infection in our model, leading to neural impairment following viral replication.
Inhibiting endosomal acidification increases the survival rate in suckling mice during dengue virus (DENV) infection. Seven-day-old ICR suckling mice were inoculated with DENV2 by concurrent intracranial and intraperitoneal injections with or without bafilomycin A1 (BafA1; 1 mg/kg) co-treatment or post-treatment. (A) Western blot analysis of viral NS1 or NS4B protein expression and (B) plaque assays of DENV replication in the brain of ICR suckling mice at 7 days post-infection. Values are presented as the mean ± SD (n = 4~7). ***p < 0.001. Additionally, time-kinetic changes in clinical scores (C) and survival rates (D) were measured (n = 4 or 5). *p < 0.05.
Discussion
Neurological complications of DENV infection are now classified as some of the hallmarks of severe dengue. In addition to our in vitro study showing DENV-induced MOI- and time-dependent neurotoxicity, our previous study indicated that DENV caused in vivo infection in neuronal cells among hippocampal lesions and induced neuronal cell apoptosis16. We further showed that a clathrin-regulated endocytic pathway controls viral entry at an early step of infection in neuronal cells. Following endocytosis, lysosomal acidification is essential for DENV genome uncoating and replication in infected neuronal cells. Regarding no further studies showing the blockade of endocytic pathways for anti-DENV actions in mice, this study demonstrated that inhibiting the endocytic pathways of DENV infection decreased viral replication and attenuated DENV-induced neurotoxicity as well as acute viral encephalitis.
In general, neurological complications associated with dengue diseases are unusual. Treatment with PCZ, a D2R antagonist that has been approved for treating nausea, vomiting, and headaches in humans, confers anti-dengue effects in vitro and in vivo 19. As to viral entry, PCZ can also act as an inhibitor of clathrin-mediated endocytosis. The roles of PCZ in targeting D2R and clathrin during DENV infection need further investigation. In addition to PCZ, the anti-psychotics, CPZ and Pitstop 2, were used in this study to confirm the essential role of clathrin-mediated endocytosis in DENV entry. CPZ is also an antagonist of D2R29. We showed that D2R was expressed in DENV-infected neuronal cells and mediated DENV entry as a viral receptor. All of these studies indicated that neuronal cells can be targets of DENV infection through potential viral receptor D2R-mediated and clathrin-regulated viral entry. Serotype 2 of DENV was used in our work as well as in a previous study19; however, further studies have to dissect the different routes of clathrin-dependent and -independent endocytosis by other, different serotypes of DENV4. Importantly, according to in vivo and in vitro immunostaining, D2R was expressed in hippocampal neurons, consistent with a previous study17. The pathogenic effects of DENV-infected hippocampal neurons remain unclear in dengue encephalitis. The affinity of viral infection and neurotoxicity, especially in the hippocampal regions, is of interest for further studies.
In addition to developing vaccines and viral inhibitors that precisely target viral proteins, which are essential for viral binding/entry, replication, and assembly/release, identifying specific virus-host interactions, such as viral receptors, endocytic pathways, and viral assembly, could be useful for anti-infective therapies3, 5, 6. Although the search for antivirals to combat DENV infection is critical, there are no currently accepted antiviral drugs for treating dengue patients. Previous studies showed that the administration of chloroquine, a lysosomotropic agent, exerts a modest antiviral effect by interfering with endosomal fusion and furin-dependent virus maturation in vitro 30 and in vivo 31. It was speculated that chloroquine and its analogue, hydroxychloroquine, could be used to treat low pH-dependent viruses, such as dengue, chikungunya, influenza, and Ebola, at the initial phase of infection32, 33. However, a randomized controlled trial of chloroquine failed to inhibit viremia, antigenemia, and cytokine or T cell responses in dengue patients34. Further studies may be needed to evaluate its therapeutic efficacy and treatment route.
By small interfering RNA screening, several human membrane trafficking genes that mediate the infectious entry of DENV were identified35, 36. Importantly, knockdown of V-ATPase reduced DENV infection and replication in both arthropod cells36 and human cells35. Studies in arthropod vectors or cells showed blockade of the endocytic pathway of DENV infection by BafA1, an inhibitor of V-ATPase, thereby reducing viral replication and transmission36,37,38. According to our study, targeting V-ATPase-based endosomal acidification using BafA1 and ConA, which bind to the proteolipid ring of the V0 domain of V-ATPase28, resulted in a notable antiviral response against DENV replication in vitro and in vivo. Consistent with a previous study as demonstrated in vitro 35, our study further showed both in vitro and in vivo antiviral effects of BafA1 treatment. These findings provide evidence to strengthen the preclinical importance of BafA1-based anti-dengue therapy.
Viral infections require an acidic pH for infectivity, generally during the process of viral genome uncoating following endocytosis. BafA1 can block V-ATPase-based endosomal acidification, and the blockade of endocytic pathways by BafA1 treatment can be demonstrated by detecting viral genome uncoating, protein expression, replication, and virus release39. In our study, treatment with BafA1 significantly decreased DENV RNA replication, protein expression, and virus release in vitro and in vivo. These results confirmed the importance of endocytic pathways for DENV infection. However, the antiviral efficiency of BafA1 could also include targeting the autophagic process40, as autophagy-regulated energy production is required during DENV replication41. However, in this study, as demonstrated using a stable luciferase reporter DENV, BafA1 treatment did not affect viral translation, which suggests that its antiviral action presumably occurs by means other than by directly affecting viral replication-associated autophagy. Furthermore, V-ATPases are not only found within membranes of endosomes, lysosomes, and secretory vesicles, but are also found in plasma membranes28. It was speculated that BafA1 treatment may also affect upstream and downstream aspects of the endocytic pathways of DENV infection, such as viral entry or the secretion of infectious virions.
Neurotoxicity can be caused by DENV infection in brains of experimental mice14, 16, 24, and DENV infection has caused fatalities in dengue patients42. Several in vitro studies showed the ability of DENV infection to trigger neuronal cell death20,21,22,23. We confirmed the cytotoxic effects caused by DENV infection in neuronal cells in vivo and in vitro. After either a long time post-infection or a higher MOI, DENV caused neuronal cell growth inhibition accompanied by cytotoxicity. In response to viral replication, cellular stress, including endoplasmic reticular stress, oxidative stress, and cytotoxic factor release (such as tumor necrosis factor-α), may promote the apoptosis of neuronal cells. A direct effect of DENV infection on cytotoxicity in neuronal cells was speculated in vitro; however, a bystander effect such as CNS inflammation, which is probably caused by DENV infection of microglia and astrocytes, may also contribute to neurotoxicity in vivo.
In conclusion, targeting the endocytic pathways of DENV infection can reduce viral replication and cytotoxicity in neuronal cells in vitro and in vivo. These findings shed light on the development of antiviral therapeutics against DENV infection, particularly in the brain. In addition to DENV, targeting an Axl-mediated clathrin-based endocytic pathway conferred protection against the emerging flavivirus Zika infection in human glial cells43. Similar to flaviviruses, endosomal acid-dependent viral entry may also determine Zika membrane fusion during early infection and could be targeted for further antiviral therapy. For flaviviral encephalitis, in addition to a direct cytotoxic effect on neuronal cells, immunopathogenic issues, including innate and adaptive immune responses in the CNS, are also involved in encephalitic development44. Questions have been raised by this study that require further examination, including the involvement and interaction of DENV-infected cells, post-infection effects of neuronal cells, and other host factors involved in DENV infection, neurotoxicity, and dengue encephalitis.
Methods
Ethics statement
Animal studies of this project were performed according to the Animal Protection Act of Taiwan, and all protocols according to guidelines established by the Ministry of Science and Technology, Taiwan were approved by the Laboratory Animal Care and Use Committee of National Cheng Kung University (IACUC #104062).
Cells, virus strains, and reagents
Mouse Neuro-2a cells (ATCC, CCL131) were grown on plastic in RPMI medium 1640 (RPMI; Invitrogen Life Technologies, Rockville, MD) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Invitrogen Life Technologies). Baby hamster kidney (BHK)-21 cells (ATCC, CCL10) and Aedes albopictus C6/36 cells (ATCC, CRL1660) were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen Life Technologies). DENV2 PL046, a Taiwanese human isolate obtained from the Centers for Disease Control in Taiwan, was propagated in C6/36 cells. Viral titers were determined by plaque assays using the BHK-21 cell line. Reagents and antibodies used in these studies were as follows: chlorpromazine (CPZ) and bafilomycin A1 (BafA1) (Cayman Chemical, Ann Arbor, MI); 4,6-diamidino-2-phenylindole (DAPI), acridine orange, dimethyl sulfoxide (DMSO), concanamycin A (ConA), MCP, and a mouse monoclonal antibody (mAb) specific for β-actin (Sigma-Aldrich, St. Louis, MO); antibodies against dsRNA (Scicons, city?, Hungary); antibodies against NeuN, DENV NS1, and NS4B (GeneTex, San Antonio, TX); Pitstop 2 and rabbit anti-mouse immunoglobulin G (IgG) conjugated with horseradish peroxidase (HRP; Abcam, Cambridge, MA); and Alexa Fluor 488- and Alexa Fluor 594-conjugated goat anti-mouse and goat anti-rabbit [?]antibodies (Invitrogen, Carlsbad, CA).
Isolation of neuronal cells
Hippocampal neurons were dissected from E16.5 C57BL/6 mouse embryos (Jackson Laboratory, Bar Harbor, MA) and suspended in 0.02% trypsin-EDTA (Invitrogen) at 37 °C for 10 min according to a previous study45.
DENV infection in vitro and in vivo
The in vitro and in vivo infectious procedures were carried out according to our previous studies16, 46. Seven-day-old ICR strain suckling mice were inoculated intracerebrally with 2.5 × 105 plaque-forming units (PFU) and intraperitoneally with 7.5 × 105 PFU of DENV2 (PL046), which was combined with or without BafA1 (1 mg/kg) treatment.
Immunostaining
Procedures were carried out according to our previous studies16. For actin staining, Alexa Fluor 594 phalloidin (Thermo Fisher Scientific, Pittsburgh, PA) was used. Cells were visualized under a fluorescent microscope (BX51; Olympus, Tokyo, Japan) or a laser-scanning confocal microscope (SPII; Leica Mikrosysteme Vertrieb, Bensheim, Germany). Cells were analyzed using flow cytometry (FACSCalibur; BD Biosciences, where?).
TdT-mediated dUTP nick end labeling (TUNEL) assay
Apoptotic cells were assessed by TUNEL staining using an ApoAlert DNA fragmentation assay kit (Clontech, Mountain View, CA) according to the manufacturer’s instructions.
Fluorescent DENV
Fluorescent DENV was prepared by labeling with Alexa Fluor 594 succinimidyl ester (AF594SE, Molecular Probes, Invitrogen) according to a method described in a previous study47. Labeled viruses were purified using Amicon Ultra-15 PLTK Ultracel-PL Membrane (30 kDa) centrifugal filter units (Millipore, where?) to remove excess dye.
Cell viability and cytotoxicity
Cell viability and cytotoxicity were respectively assessed using a colorimetric Cell Counting Kit-8 (Dojindo Molecular Technologies, Kumamoto, Japan) and Cytotoxicity Detection kit assays (Roche Diagnostics, Lewes, UK), according to the manufacturer’s instructions.
Western blotting
The general protocol for Western blotting was performed according to previous studies16, 46.
Reverse-transcription (RT)-polymerase chain reaction (PCR) and quantitative (q)PCR
Total RNA was extracted using the TRIZol (Invitrogen) RNA extraction reagent. Complementary (c)DNA was synthesized with an RT reaction using a PrimeScriptTM RT reagent kit (Takara, Tokyo, Japan). The qPCR was conducted using KAPA SYBR FAST qPCR Master Mix (Life Technologies and Kapa Biosystems, Woburn, MA). The PCR was performed using a StepOnePlusTM real-time PCR system (Applied Biosystems, Foster City, CA) with the following pair of specific primers: primer sequences for NS1 (forward): 5′-ATGGATCCGATAGTGGTTGCGTTGTGA-3′ and NS1 (reverse): 5′-ATCTCGAGGGCTGTGACCAAGGAGTT-3′.
Plaque assay
Detecting viral replication was performed with a plaque assay according to previous studies16, 46.
Reporter assay
BHK-21 cells harboring the luciferase-expressing DENV replicon (BHK-D2-Fluc-SGR-Neo-1) were constructed and maintained according to a previous study48.
Statistical analysis
Data are presented as the mean ± standard deviation (SD). Data were analyzed by an unpaired Student’s t-test or by one-way analysis of variance (ANOVA) with Tukey’s multiple-comparison test. Statistical significance was defined as p < 0.05.
References
Diaz-Quijano, F. A. Dengue. The New England journal of medicine 367, 180; author reply 181, doi:10.1056/NEJMc1205584#SA1 (2012).
Yacoub, S., Mongkolsapaya, J. & Screaton, G. Recent advances in understanding dengue. F1000Research 5, doi:10.12688/f1000research.6233.1 (2016).
Cruz-Oliveira, C. et al. Receptors and routes of dengue virus entry into the host cells. FEMS Microbiol Rev 39, 155–170, doi:10.1093/femsre/fuu004 (2015).
Acosta, E. G., Castilla, V. & Damonte, E. B. Alternative infectious entry pathways for dengue virus serotypes into mammalian cells. Cellular microbiology 11, 1533–1549, doi:10.1111/j.1462-5822.2009.01345.x (2009).
De La Guardia, C. & Lleonart, R. Progress in the identification of dengue virus entry/fusion inhibitors. Biomed Res Int 2014, 825039, doi:10.1155/2014/825039 (2014).
Kaptein, S. J. & Neyts, J. Towards antiviral therapies for treating dengue virus infections. Curr Opin Pharmacol 30, 1–7, doi:10.1016/j.coph.2016.06.002 (2016).
Solomon, T. Flavivirus encephalitis. The New England journal of medicine 351, 370–378, doi:10.1056/NEJMra030476 (2004).
Mailles, A., Stahl, J. P. & Bloch, K. C. Update and new insights in encephalitis. Clinical microbiology and infection: the official publication of the European Society of Clinical Microbiology and Infectious Diseases, doi:10.1016/j.cmi.2017.05.002 (2017).
Acevedo, N. et al. Zika Virus, Chikungunya Virus, and Dengue Virus in Cerebrospinal Fluid from Adults with Neurological Manifestations, Guayaquil, Ecuador. Frontiers in microbiology 8, 42, doi:10.3389/fmicb.2017.00042 (2017).
Carod-Artal, F. J., Wichmann, O., Farrar, J. & Gascon, J. Neurological complications of dengue virus infection. The Lancet. Neurology 12, 906–919, doi:10.1016/S1474-4422(13)70150-9 (2013).
Solomon, T. et al. Neurological manifestations of dengue infection. Lancet 355, 1053–1059, doi:10.1016/S0140-6736(00)02036-5 (2000).
Verma, R., Sahu, R. & Holla, V. Neurological manifestations of dengue infection: a review. Journal of the neurological sciences 346, 26–34, doi:10.1016/j.jns.2014.08.044 (2014).
Verma, R. et al. Neurological complications of dengue fever: Experience from a tertiary center of north India. Annals of Indian Academy of Neurology 14, 272–278, doi:10.4103/0972-2327.91946 (2011).
de Miranda, A. S. et al. Dengue-3 encephalitis promotes anxiety-like behavior in mice. Behavioural brain research 230, 237–242, doi:10.1016/j.bbr.2012.02.020 (2012).
Amaral, D. C. et al. Intracerebral infection with dengue-3 virus induces meningoencephalitis and behavioral changes that precede lethality in mice. Journal of neuroinflammation 8, 23, doi:10.1186/1742-2094-8-23 (2011).
Tsai, T. T. et al. Microglia retard dengue virus-induced acute viral encephalitis. Scientific reports 6, 27670, doi:10.1038/srep27670 (2016).
Meador-Woodruff, J. H. et al. Dopamine receptor gene expression in the human medial temporal lobe. Neuropsychopharmacology 10, 239–248, doi:10.1038/npp.1994.27 (1994).
Smith, J. L. et al. Inhibition of dengue virus replication by a class of small-molecule compounds that antagonize dopamine receptor d4 and downstream mitogen-activated protein kinase signaling. J Virol 88, 5533–5542, doi:10.1128/JVI.00365-14 (2014).
Simanjuntak, Y., Liang, J. J., Lee, Y. L. & Lin, Y. L. Repurposing of prochlorperazine for use against dengue virus infection. J Infect Dis 211, 394–404, doi:10.1093/infdis/jiu377 (2015).
Despres, P., Flamand, M., Ceccaldi, P. E. & Deubel, V. Human isolates of dengue type 1 virus induce apoptosis in mouse neuroblastoma cells. J Virol 70, 4090–4096 (1996).
Su, H. L. et al. The effect of human bcl-2 and bcl-X genes on dengue virus-induced apoptosis in cultured cells. Virology 282, 141–153, doi:10.1006/viro.2000.0820 (2001).
Jan, J. T. et al. Potential dengue virus-triggered apoptotic pathway in human neuroblastoma cells: arachidonic acid, superoxide anion, and NF-kappaB are sequentially involved. J Virol 74, 8680–8691 (2000).
Castellanos, J. E., Neissa, J. I. & Camacho, S. J. Dengue virus induces apoptosis in SH-SY5Y human neuroblastoma cells. Biomedica 36, 156–158, doi:10.7705/biomedica.v36i0.2984 (2016).
Despres, P., Frenkiel, M. P., Ceccaldi, P. E., Duarte Dos Santos, C. & Deubel, V. Apoptosis in the mouse central nervous system in response to infection with mouse-neurovirulent dengue viruses. J Virol 72, 823–829 (1998).
Rodenhuis-Zybert, I. A., Wilschut, J. & Smit, J. M. Dengue virus life cycle: viral and host factors modulating infectivity. Cellular and molecular life sciences: CMLS 67, 2773–2786, doi:10.1007/s00018-010-0357-z (2010).
van der Schaar, H. M. et al. Dissecting the cell entry pathway of dengue virus by single-particle tracking in living cells. PLoS pathogens 4, e1000244, doi:10.1371/journal.ppat.1000244 (2008).
Duan, X., Lu, X., Li, J. & Liu, Y. Novel binding between pre-membrane protein and vacuolar ATPase is required for efficient dengue virus secretion. Biochem Biophys Res Commun 373, 319–324, doi:10.1016/j.bbrc.2008.06.041 (2008).
Cotter, K., Stransky, L., McGuire, C. & Forgac, M. Recent Insights into the Structure, Regulation, and Function of the V-ATPases. Trends Biochem Sci 40, 611–622, doi:10.1016/j.tibs.2015.08.005 (2015).
Harrold, M. W. et al. Charged analogues of chlorpromazine as dopamine antagonists. J Med Chem 30, 1631–1635 (1987).
Farias, K. J., Machado, P. R., de Almeida Junior, R. F., de Aquino, A. A. & da Fonseca, B. A. Chloroquine interferes with dengue-2 virus replication in U937 cells. Microbiol Immunol 58, 318–326, doi:10.1111/1348-0421.12154 (2014).
Farias, K. J., Machado, P. R., Muniz, J. A., Imbeloni, A. A. & da Fonseca, B. A. Antiviral activity of chloroquine against dengue virus type 2 replication in Aotus monkeys. Viral Immunol 28, 161–169, doi:10.1089/vim.2014.0090 (2015).
Akpovwa, H. Chloroquine could be used for the treatment of filoviral infections and other viral infections that emerge or emerged from viruses requiring an acidic pH for infectivity. Cell Biochem Funct 34, 191–196, doi:10.1002/cbf.3182 (2016).
Savarino, A., Boelaert, J. R., Cassone, A., Majori, G. & Cauda, R. Effects of chloroquine on viral infections: an old drug against today’s diseases? Lancet Infect Dis 3, 722–727 (2003).
Tricou, V. et al. A randomized controlled trial of chloroquine for the treatment of dengue in Vietnamese adults. PLoS Negl Trop Dis 4, e785, doi:10.1371/journal.pntd.0000785 (2010).
Ang, F., Wong, A. P., Ng, M. M. & Chu, J. J. Small interference RNA profiling reveals the essential role of human membrane trafficking genes in mediating the infectious entry of dengue virus. Virol J 7, 24, doi:10.1186/1743-422X-7-24 (2010).
Sessions, O. M. et al. Discovery of insect and human dengue virus host factors. Nature 458, 1047–1050, doi:10.1038/nature07967 (2009).
Kang, S., Shields, A. R., Jupatanakul, N. & Dimopoulos, G. Suppressing dengue-2 infection by chemical inhibition of Aedes aegypti host factors. PLoS Negl Trop Dis 8, e3084, doi:10.1371/journal.pntd.0003084 (2014).
Mosso, C., Galvan-Mendoza, I. J., Ludert, J. E. & del Angel, R. M. Endocytic pathway followed by dengue virus to infect the mosquito cell line C6/36 HT. Virology 378, 193–199, doi:10.1016/j.virol.2008.05.012 (2008).
Perez, L. & Carrasco, L. Involvement of the vacuolar H(+)-ATPase in animal virus entry. J Gen Virol 75(Pt 10), 2595–2606, doi:10.1099/0022-1317-75-10-2595 (1994).
Mateo, R. et al. Inhibition of cellular autophagy deranges dengue virion maturation. J Virol 87, 1312–1321, doi:10.1128/JVI.02177-12 (2013).
Jain, B., Chaturvedi, U. C. & Jain, A. Role of intracellular events in the pathogenesis of dengue; an overview. Microb Pathog 69–70, 45–52, doi:10.1016/j.micpath.2014.03.004 (2014).
Miagostovich, M. P. et al. Retrospective study on dengue fatal cases. Clin Neuropathol 16, 204–208 (1997).
Meertens, L. et al. Axl Mediates ZIKA Virus Entry in Human Glial Cells and Modulates Innate Immune Responses. Cell reports 18, 324–333, doi:10.1016/j.celrep.2016.12.045 (2017).
King, N. J. et al. Immunopathology of flavivirus infections. Immunology and cell biology 85, 33–42, doi:10.1038/sj.icb.7100012 (2007).
Pao, P. C. et al. A novel RING finger protein, Znf179, modulates cell cycle exit and neuronal differentiation of P19 embryonal carcinoma cells. Cell death and differentiation 18, 1791–1804, doi:10.1038/cdd.2011.52 (2011).
Cheng, Y. L. et al. Activation of Nrf2 by the dengue virus causes an increase in CLEC5A, which enhances TNF-alpha production by mononuclear phagocytes. Scientific reports 6, 32000, doi:10.1038/srep32000 (2016).
Zhang, S. L., Tan, H. C., Hanson, B. J. & Ooi, E. E. A simple method for Alexa Fluor dye labelling of dengue virus. J Virol Methods 167, 172–177, doi:10.1016/j.jviromet.2010.04.001 (2010).
Su, C. I., Tseng, C. H., Yu, C. Y. & Lai, M. M. SUMO Modification Stabilizes Dengue Virus Nonstructural Protein 5 To Support Virus Replication. J Virol 90, 4308–4319, doi:10.1128/JVI.00223-16 (2016).
Acknowledgements
We thank the Core Facility Center of Taipei Medical University (TMU) for providing the FACSCanto II flow cytometer (BD Biosciences) and TCS SP5 Confocal Spectral Microscope Imaging System (Leica). This study was supported by grants from the Ministry of Science and Technology (MOST102-2628-B-038-011-MY3, 105-2321-B-038-002, and 106-2321-B-038-002) and the intramural funding 105TMU-CIT-01-2, Taipei, Taiwan.
Author information
Authors and Affiliations
Contributions
M.R.H., T.T.T., M.K.J., and C.C.T. conducted the experiments; C.L.C., C.C.T., and C.F.L. designed the experiments; Y.C.L. and C.H.C. provided materials; M.R.H., T.T.T., and C.F.L. wrote the paper; and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ho, MR., Tsai, TT., Chen, CL. et al. Blockade of dengue virus infection and viral cytotoxicity in neuronal cells in vitro and in vivo by targeting endocytic pathways. Sci Rep 7, 6910 (2017). https://doi.org/10.1038/s41598-017-07023-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-07023-z
This article is cited by
-
Quantification of the interaction forces between dengue virus and dopamine type-2 receptor using optical tweezers
Virology Journal (2024)
-
In vitro synergistic antiviral activity of repurposed drugs against enterovirus 71
Archives of Virology (2024)
-
Role of angiotensin II in cellular entry and replication of dengue virus
Archives of Virology (2024)
-
Signaling events evoked by domain III of envelop glycoprotein of tick-borne encephalitis virus and West Nile virus in human brain microvascular endothelial cells
Scientific Reports (2022)
-
Inhibition of dengue virus infection by trifluoperazine
Archives of Virology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.