Abstract
The bone marrow supports and regulates hematopoiesis, responding to physiological requirements for blood cell production over ontogeny and during pathological challenges. Interactions between hematopoietic cells and niche components are challenging to study mechanistically in the human context, but are important to delineate in order to explore the pathobiology of blood and bone marrow disorders. Organoids are proving transformative in many research settings, but an accurate human bone marrow model incorporating multiple hematopoietic and stromal elements has been lacking. This protocol describes a method to generate three-dimensional, multilineage bone marrow organoids from human induced pluripotent stem cells (hiPSCs), detailing the steps for the directed differentiation of hiPSCs using a series of cytokine cocktails and hydrogel embedding. Over 18 days of differentiation, hiPSCs yield the key lineages that are present in central myelopoietic bone marrow, organized in a well-vascularized architecture that resembles native hematopoietic tissues. This presents a robust, in vitro system that can model healthy and perturbed hematopoiesis in a scalable three-dimensional microenvironment. Bone marrow organoids also support the growth of immortalized cell lines and primary cells from healthy donors and patients with myeloid and lymphoid cancers, including cell types that are poorly viable in standard culture systems. Moreover, we discuss assays for the characterization of organoids, including interrogation of pathogenic remodeling using recombinant TGF-ß treatment, and methods for organoid engraftment with exogenous cells. This protocol can be readily adapted to specific experimental requirements, can be easily implemented by users with tissue culture experience and does not require access to specialist equipment.
Key points
-
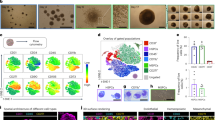
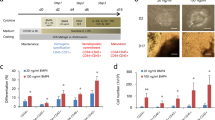
An ex vivo three-dimensional system for modeling human bone marrow is presented. Human induced pluripotent stem cells cultured in a collagen-enriched hydrogel with stimulatory cytokines generate vascularized bone marrow organoids containing mesenchymal stromal cells, fibroblasts, endothelial and hematopoietic cells.
-
The bone marrow organoids can be engrafted with adult donor-derived cells to study the dynamics between these cells and the bone marrow niche, using downstream assays including imaging, genetic characterization and flow cytometry.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The original research relating to this protocol can be accessed in a previous publication22 and via the Github repository (https://github.com/aokhan/BMorganoidV1/). Single-cell RNA sequencing data relevant to the original publication are available at the Gene Expression Omnibus (accession GSE196684). Source data are provided with this paper.
Code availability
Code used to analyze RNA sequencing data relevant to the original publication is available at https://github.com/aokhan/BMorganoidV1/ and https://github.com/supatt-lab/SingCellaR.
References
Tuveson, D. & Clevers, H. Cancer modeling meets human organoid technology. Science 364, 952–955 (2019).
Kim, J., Koo, B.-K. & Knoblich, J. A. Human organoids: model systems for human biology and medicine. Nat. Rev. Mol. Cell Biol. 21, 571–584 (2020).
Veninga, V. & Voest, E. E. Tumor organoids: opportunities and challenges to guide precision medicine. Cancer Cell 39, 1190–1201 (2021).
Li, R. et al. A pro-inflammatory stem cell niche drives myelofibrosis through a targetable galectin 1 axis. Preprint at bioRxiv https://doi.org/10.1101/2023.08.05.550630 (2023).
Méndez-Ferrer, S. et al. Bone marrow niches in haematological malignancies. Nat. Rev. Cancer 20, 285–298 (2020).
Lucas, D. Structural organization of the bone marrow and its role in hematopoiesis. Curr. Opin. Hematol. 28, 36–42 (2020).
Morrison, S. J. & Scadden, D. T. The bone marrow niche for haematopoietic stem cells. Nature 505, 327–334 (2014).
Jardine, L. et al. Blood and immune development in human fetal bone marrow and Down syndrome. Nature 598, 327–331 (2021).
Baryawno, N. et al. A cellular taxonomy of the bone marrow stroma in homeostasis and leukemia. Cell 177, 1915–1932.e16 (2019).
Tikhonova, A. N. et al. The bone marrow microenvironment at single-cell resolution. Nature 569, 222–228 (2019).
Baccin, C. et al. Combined single-cell and spatial transcriptomics reveal the molecular, cellular and spatial bone marrow niche organization. Nat. Cell Biol. 22, 38–48 (2020).
Kent, D., Dykstra, B. & Eaves, C. Isolation and assessment of long‐term reconstituting hematopoietic stem cells from adult mouse bone marrow. Curr. Protoc. Stem Cell Biol. 3, 2A.4.1–2A.4.23 (2007).
Bradley, T. & Metcalf, D. The growth of mouse bone marrow cells in vitro. Aust. J. Exp. Biol. Med. Sci. 44, 287–300 (1966).
de L, M. et al. Cord-blood engraftment with ex vivo mesenchymal-cell coculture. N. Engl. J. Med. 367, 2305–2315 (2012).
Khan, A. O. et al. Post-translational polymodification of β1-tubulin regulates motor protein localisation in platelet production and function. Haematologica 1, 243–260 (2022).
Feng, Q. et al. Scalable generation of universal platelets from human induced pluripotent stem cells. Stem Cell Rep. 3, 817–831 (2014).
Ng, E. S. et al. Differentiation of human embryonic stem cells to HOXA+ hemogenic vasculature that resembles the aorta–gonad–mesonephros. Nat. Biotechnol. 34, 1168–1179 (2016).
Jing, R. et al. EZH1 repression generates mature iPSC-derived CAR T cells with enhanced antitumor activity. Cell Stem Cell 29, 1181–1196.e6 (2022).
Cao, X. et al. Differentiation and functional comparison of monocytes and macrophages from hiPSCs with peripheral blood derivatives. Stem Cell Rep. 12, 1282–1297 (2019).
Ebrahimi, M. et al. Differentiation of human induced pluripotent stem cells into erythroid cells. Stem Cell Res. Ther. 11, 483 (2020).
Moreau, T. et al. Large-scale production of megakaryocytes from human pluripotent stem cells by chemically defined forward programming. Nat. Commun. 7, 11208 (2016).
Khan, A. O. et al. Human bone marrow organoids for disease modelling, discovery and validation of therapeutic targets in hematological malignancies. Cancer Discov. https://doi.org/10.1158/2159-8290.cd-22-0199 (2022).
Zhao, Z. et al. Organoids. Nat. Rev. Methods Prim. 2, 94 (2022).
Wimmer, R. A., Leopoldi, A., Aichinger, M., Kerjaschki, D. & Penninger, J. M. Generation of blood vessel organoids from human pluripotent stem cells. Nat. Protoc. 14, 3082–3100 (2019).
Wimmer, R. A. et al. Human blood vessel organoids as a model of diabetic vasculopathy. Nature 565, 505–510 (2019).
Popescu, D.-M. et al. Decoding human fetal liver haematopoiesis. Nature 574, 365–371 (2019).
Roy, A. et al. Transitions in lineage specification and gene regulatory networks in hematopoietic stem/progenitor cells over human development. Cell Rep. 36, 109698 (2021).
Raic, A., Naolou, T., Mohra, A., Chatterjee, C. & Lee-Thedieck, C. 3D models of the bone marrow in health and disease: yesterday, today, and tomorrow. MRS Commun. 9, 37–52 (2019).
Sharipol, A., Lesch, M. L., Soto, C. A. & Frisch, B. J. Bone marrow microenvironment-on-chip for culture of functional hematopoietic stem cells. Front. Bioeng. Biotechnol. 10, 855777 (2022).
Bessy, T., Itkin, T. & Passaro, D. Bioengineering the bone marrow vascular niche. Front. Cell Dev. Biol. 9, 645496 (2021).
Voeltzel, T. et al. A minimal standardized human bone marrow microphysiological system to assess resident cell behavior during normal and pathological processes. Biomater. Sci. 10, 485–498 (2021).
Giger, S. et al. Microarrayed human bone marrow organoids for modeling blood stem cell dynamics. APL Bioeng. 6, 036101 (2022).
Fairfield, H. et al. Development of a 3D bone marrow adipose tissue model. Bone 118, 77–88 (2019).
Glaser, D. E. et al. Organ-on-a-chip model of vascularized human bone marrow niches. Biomaterials 280, 121245 (2022).
Chou, D. B. et al. On-chip recapitulation of clinical bone-marrow toxicities and patient-specific pathophysiology. Nat. Biomed. Eng. 4, 394–406 (2020).
Zhang, S., Wan, Z. & Kamm, R. D. Vascularized organoids on a chip: strategies for engineering organoids with functional vasculature. Lab Chip 21, 473–488 (2021).
Aazmi, A. et al. Engineered vasculature for organ-on-a-chip systems. Engineering 9, 131–147 (2022).
Marturano-Kruik, A. et al. Human bone perivascular niche-on-a-chip for studying metastatic colonization. Proc. Natl Acad. Sci. USA 115, 1256–1261 (2018).
Cuenca, M. V. et al. Engineered 3D vessel-on-chip using hiPSC-derived endothelial and vascular smooth muscle cells. Stem Cell Rep. 16, 2159–2168 (2021).
Byambaa, B. et al. Bioprinted osteogenic and vasculogenic patterns for engineering 3D bone tissue. Adv. Healthc. Mater. 6, 1700015 (2017).
Simunovic, F. & Finkenzeller, G. Vascularization strategies in bone tissue engineering. Cells 10, 1749 (2021).
Kopp, H.-G., Avecilla, S. T., Hooper, A. T. & Rafii, S. The bone marrow vascular niche: home of HSC differentiation and mobilization. Physiology 20, 349–356 (2005).
Demirci, S., Leonard, A. & Tisdale, J. F. Hematopoietic stem cells from pluripotent stem cells: clinical potential, challenges, and future perspectives. Stem Cells Transl. Med 9, 1549–1557 (2020).
Sugimura, R. et al. Haematopoietic stem and progenitor cells from human pluripotent stem cells. Nature 545, 432–438 (2017).
Sakurai, M. et al. Chemically defined cytokine-free expansion of human haematopoietic stem cells. Nature 615, 127–133 (2023).
Wilkinson, A. C., Ishida, R., Nakauchi, H. & Yamazaki, S. Long-term ex vivo expansion of mouse hematopoietic stem cells. Nat. Protoc. 15, 628–648 (2020).
Wilkinson, A. C. et al. Long-term ex vivo haematopoietic-stem-cell expansion allows nonconditioned transplantation. Nature 571, 117–121 (2019).
Homan, K. A. et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat. Methods 16, 255–262 (2019).
Thamodaran, V., Rani, S. & Velayudhan, S. R. Induced pluripotent stem (iPS) cells, methods and protocols. Methods Mol. Biol. 2454, 755–773 (2021).
Qi, P., Zhou, Y., Wang, D., He, Z. & Li, Z. A new collagen solution with high concentration and collagen native structure perfectly preserved. RSC Adv. 5, 87180–87186 (2015).
Masselink, W. et al. Broad applicability of a streamlined ethyl cinnamate-based clearing procedure. Development 146, dev166884 (2019).
Campbell, T. B., Zhang, S. Y., Valencia, A. & Passegue, E. Bone marrow stromal cell remodeling is a common feature of diverse fibrotic myeloproliferative neoplasm models. Blood 128, 25–25 (2016).
Heaton, H. et al. Souporcell: robust clustering of single-cell RNA-seq data by genotype without reference genotypes. Nat. Methods 17, 615–620 (2020).
Acknowledgements
We thank N. Hayder and S. Reed who helped with sample banking, the University of Birmingham TechHub Imaging Core Facilities, and the Medical Research Council (MRC) Weatherall Institute of Molecular Medicine Flow Cytometry facility and Imaging core. We thank P. Garcia, G. Murphy, V. Steeples, Y. Psaras, and C. Toepffer for the generous provision of the hiPSC lines used (BU3-10, BU8C3, KOLF2). A.O.K. is funded by a Sir Henry Wellcome fellowship (218649/Z/19/Z, 218649/A/19/Z). B.P. receives funding from a Cancer Research UK Advanced Clinician Scientist Fellowship (C67633/A29034), a British Research Council (BRC) Senior Research Fellowship, the Haematology and Stem Cells Theme of the Oxford BRC, a Kay Kendall Leukemia Fund Project Grant and unit funding from the MRC (awarded to the MRC Molecular Haematology Unit, MRC Weatherall Institute of Molecular Medicine). A CC BY or equivalent license is applied to the author accepted manuscript arising from this submission, in accordance with the funders’ open access conditions.
Author information
Authors and Affiliations
Contributions
A.O.K. devised the differentiation protocol and utilization methods, performed cell culture and imaging experiments, co-authored the manuscript and sourced funding for this project. B.P. devised experiments and utilization of the organoids for disease modeling, interpreted data, co-wrote the manuscript and sourced funding for this project. A.-A.O., A.R.-R., Z.C.W. and Y.S. performed flow cytometry experiments, analyzed and interpreted data, and A.-A.O. co-wrote the protocol. J.R. and J.S.R. devised and performed sectioning and related imaging experiments. N.J.J. critically reviewed and edited the manuscript and curated and interpreted data.
Corresponding authors
Ethics declarations
Competing interests
B.P.: Alethiomics (co-founder, equity, consultancy, research funding), Constellation Therapeutics (consultancy), Blueprint Medicines (advisory board), Galecto (research funding), Novartis (paid speaking engagements), GSK (advisory board). A.O.K.: Alethiomics (consultancy). The other authors have no conflicts of interest to declare that are relevant to the content of this article. A patent has been filed by A.O.K. and B.P. relating to work described in this paper (GB2202025.9 and GB221664.47 (WO/2023/156774), PCT/GB2023/050348).
Peer review
Peer review information
Nature Protocols thanks Benjamin Frisch, Veronique Maguer-Satta and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Khan, A. O. et al. Cancer Discov. 13, 364–385 (2022): https://doi.org/10.1158/2159-8290.cd-22-0199
Li, R. et al. Preprint at bioRxiv (2023): https://doi.org/10.1101/2023.08.05.550630
Supplementary Information
Supplementary Information
Supplementary Figs. 1–8.
Source data
Source Data Fig. 3
Statistical source data for the population frequencies in Fig. 3d–i.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Olijnik, AA., Rodriguez-Romera, A., Wong, Z.C. et al. Generating human bone marrow organoids for disease modeling and drug discovery. Nat Protoc (2024). https://doi.org/10.1038/s41596-024-00971-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41596-024-00971-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.