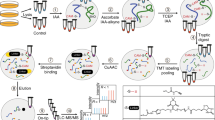
Abstract
Cysteine is unique among all protein-coding amino acids, owing to its intrinsically high nucleophilicity. The cysteinyl thiol group can be covalently modified by a broad range of redox mechanisms or by various electrophiles derived from exogenous or endogenous sources. Measuring the response of protein cysteines to redox perturbation or electrophiles is critical for understanding the underlying mechanisms involved. Activity-based protein profiling based on thiol-reactive probes has been the method of choice for such analyses. We therefore adapted this approach and developed a new chemoproteomic platform, termed ‘QTRP’ (quantitative thiol reactivity profiling), that relies on the ability of a commercially available thiol-reactive probe IPM (2-iodo-N-(prop-2-yn-1-yl)acetamide) to covalently label, enrich and quantify the reactive cysteinome in cells and tissues. Here, we provide a detailed and updated workflow of QTRP that includes procedures for (i) labeling of the reactive cysteinome from cell or tissue samples (e.g., control versus treatment) with IPM, (ii) processing the protein samples into tryptic peptides and tagging the probe-modified peptides with isotopically labeled azido-biotin reagents containing a photo-cleavable linker via click chemistry reaction, (iii) capturing biotin-conjugated peptides with streptavidin beads, (iv) identifying and quantifying the photo-released peptides by mass spectrometry (MS)-based shotgun proteomics and (v) interpreting MS data by a streamlined informatic pipeline using a proteomics software, pFind 3, and an automatic post-processing algorithm. We also exemplified here how to use QTRP for mining H2O2-sensitive cysteines and for determining the intrinsic reactivity of cysteines in a complex proteome. We anticipate that this protocol should find broad applications in redox biology, chemical biology and the pharmaceutical industry. The protocol for sample preparation takes 3 d, whereas MS measurements and data analyses require 75 min and <30 min, respectively, per sample.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE71 partner repository with the dataset identifier PXD016048. Software availability: post-processing algorithm (https://github.com/morpheusliu/Post-processing-program-for-pFind3-results).
References
Paulsen, C. E. & Carroll, K. S. Cysteine-mediated redox signaling: chemistry, biology, and tools for discovery. Chem. Rev. 113, 4633–4679 (2013).
Chouchani, E. T. et al. Mitochondrial ROS regulate thermogenic energy expenditure and sulfenylation of UCP1. Nature 532, 112–116 (2016).
O’Neill, J. S. & Reddy, A. B. Circadian clocks in human red blood cells. Nature 469, 498–503 (2011).
Edgar, R. S. et al. Peroxiredoxins are conserved markers of circadian rhythms. Nature 485, 459–464 (2012).
Yoo, S. K., Starnes, T. W., Deng, Q. & Huttenlocher, A. Lyn is a redox sensor that mediates leukocyte wound attraction in vivo. Nature 480, 109–112 (2011).
Ruppersberg, J. P. et al. Regulation of fast inactivation of cloned mammalian IK(A) channels by cysteine oxidation. Nature 352, 711–714 (1991).
Guo, Z., Kozlov, S., Lavin, M. F., Person, M. D. & Paull, T. T. ATM activation by oxidative stress. Science 330, 517–521 (2010).
Rudolph, T. K. & Freeman, B. A. Transduction of redox signaling by electrophile-protein reactions. Sci. Signal. 2, re7 (2009).
Qin, W. et al. S-glycosylation-based cysteine profiling reveals regulation of glycolysis by itaconate. Nat. Chem. Biol. 15, 983–991 (2019).
Kulkarni, R. A. et al. A chemoproteomic portrait of the oncometabolite fumarate. Nat. Chem. Biol. 15, 391–400 (2019).
Gersch, M., Kreuzer, J. & Sieber, S. A. Electrophilic natural products and their biological targets. Nat. Prod. Rep. 29, 659–682 (2012).
Singh, J., Petter, R. C., Baillie, T. A. & Whitty, A. The resurgence of covalent drugs. Nat. Rev. Drug Discov. 10, 307–317 (2011).
Yang, J., Carroll, K. S. & Liebler, D. C. The expanding landscape of the thiol redox proteome. Mol. Cell. Proteom. 15, 1–11 (2016).
Lee, S. R., Kwon, K. S., Kim, S. R. & Rhee, S. G. Reversible inactivation of protein-tyrosine phosphatase 1B in A431 cells stimulated with epidermal growth factor. J. Biol. Chem. 273, 15366–15372 (1998).
Shiio, Y. & Aebersold, R. Quantitative proteome analysis using isotope-coded affinity tags and mass spectrometry. Nat. Protoc. 1, 139–145 (2006).
Thompson, A. et al. Tandem mass tags: a novel quantification strategy for comparative analysis of complex protein mixtures by MS/MS. Anal. Chem. 75, 1895–1904 (2003).
Vajrychova, M. et al. Quantification of cellular protein and redox imbalance using SILAC-iodoTMT methodology. Redox Biol. 24, 101227 (2019).
Sethuraman, M., McComb, M. E., Heibeck, T., Costello, C. E. & Cohen, R. A. Isotope-coded affinity tag approach to identify and quantify oxidant-sensitive protein thiols. Mol. Cell. Proteom. 3, 273–278 (2004).
Fu, C. et al. Quantitative analysis of redox-sensitive proteome with DIGE and ICAT. J. Proteome Res. 7, 3789–3802 (2008).
Leichert, L. I. et al. Quantifying changes in the thiol redox proteome upon oxidative stress in vivo. Proc. Natl Acad. Sci. USA 105, 8197–8202 (2008).
Topf, U. et al. Quantitative proteomics identifies redox switches for global translation modulation by mitochondrially produced reactive oxygen species. Nat. Commun. 9, 324 (2018).
Weerapana, E. et al. Quantitative reactivity profiling predicts functional cysteines in proteomes. Nature 468, 790–795 (2010).
Weerapana, E., Speers, A. E. & Cravatt, B. F. Tandem orthogonal proteolysis-activity-based protein profiling (TOP-ABPP)—a general method for mapping sites of probe modification in proteomes. Nat. Protoc. 2, 1414–1425 (2007).
Backus, K. M. et al. Proteome-wide covalent ligand discovery in native biological systems. Nature 534, 570–574 (2016).
Bar-Peled, L. et al. Chemical proteomics identifies druggable vulnerabilities in a genetically defined cancer. Cell 171, 696–709.e23 (2017).
van Esbroeck, A. C. M. et al. Activity-based protein profiling reveals off-target proteins of the FAAH inhibitor BIA 10-2474. Science 356, 1084–1087 (2017).
Wang, C., Weerapana, E., Blewett, M. M. & Cravatt, B. F. A chemoproteomic platform to quantitatively map targets of lipid-derived electrophiles. Nat. Methods 11, 79–85 (2014).
Deng, X. et al. Proteome-wide quantification and characterization of oxidation-sensitive cysteines in pathogenic bacteria. Cell Host Microbe 13, 358–370 (2013).
Medina-Cleghorn, D. et al. Mapping proteome-wide targets of environmental chemicals using reactivity-based chemoproteomic platforms. Chem. Biol. 22, 1394–1405 (2015).
Huang, Z. et al. Global portrait of protein targets of metabolites of the neurotoxic compound BIA 10-2474. ACS Chem. Biol. 14, 192–197 (2019).
Maurais, A. J. & Weerapana, E. Reactive-cysteine profiling for drug discovery. Curr. Opin. Chem. Biol. 50, 29–36 (2019).
Senkane, K. et al. The proteome-wide potential for reversible covalency at cysteine. Angew. Chem. Int. Ed. Engl. 58, 11385–11389 (2019).
Fu, L. et al. Systematic and quantitative assessment of hydrogen peroxide reactivity with cysteines across human proteomes. Mol. Cell. Proteom. 16, 1815–1828 (2017).
Guo, J. et al. Resin-assisted enrichment of thiols as a general strategy for proteomic profiling of cysteine-based reversible modifications. Nat. Protoc. 9, 64–75 (2014).
Garcia-Santamarina, S. et al. Monitoring in vivo reversible cysteine oxidation in proteins using ICAT and mass spectrometry. Nat. Protoc. 9, 1131–1145 (2014).
Xiao, H. et al. A quantitative tissue-specific landscape of protein redox regulation during aging. Cell 180, 968–983.e24 (2020).
Paulsen, C. E. et al. Peroxide-dependent sulfenylation of the EGFR catalytic site enhances kinase activity. Nat. Chem. Biol. 8, 57–64 (2011).
Gupta, V., Yang, J., Liebler, D. C. & Carroll, K. S. Diverse redoxome reactivity profiles of carbon nucleophiles. J. Am. Chem. Soc. 139, 5588–5595 (2017).
Akter, S. et al. Chemical proteomics reveals new targets of cysteine sulfinic acid reductase. Nat. Chem. Biol. 14, 995–1004 (2018).
Yang, J., Gupta, V., Carroll, K. S. & Liebler, D. C. Site-specific mapping and quantification of protein S-sulphenylation in cells. Nat. Commun. 5, 4776 (2014).
Yang, J. et al. Global, in situ, site-specific analysis of protein S-sulfenylation. Nat. Protoc. 10, 1022–1037 (2015).
Huang, J. et al. Mining for protein S-sulfenylation in Arabidopsis uncovers redox-sensitive sites. Proc. Natl Acad. Sci. USA 116, 21256–21261 (2019).
Fu, L., Liu, K., Ferreira, R. B., Carroll, K. S. & Yang, J. Proteome-wide analysis of cysteine S-sulfenylation using a benzothiazine-based probe. Curr. Protoc. Protein Sci. 95, e76 (2019).
Fu, L. et al. Direct proteomic mapping of cysteine persulfidation. Antioxid. Redox Signal. Available at https://doi.org/10.1089/ars.2019.7777 (2019).
Petrova, B. et al. Dynamic redox balance directs the oocyte-to-embryo transition via developmentally controlled reactive cysteine changes. Proc. Natl Acad. Sci. USA 115, E7978–E7986 (2018).
Tian, C. et al. Multiplexed thiol reactivity profiling for target discovery of electrophilic natural products. Cell Chem. Biol. 24, 1416–1427.e15 (2017).
Wang, W. et al. An Arabidopsis secondary metabolite directly targets expression of the bacterial type III secretion system to inhibit bacterial virulence. Cell Host Microbe 27, 601–613 (2020).
Sun, R. et al. A chemoproteomic platform to assess bioactivation potential of drugs. Chem. Res. Toxicol. 30, 1797–1803 (2017).
Pei, J.-F. et al. Diurnal oscillations of endogenous H2O2 sustained by p66Shc regulate circadian clocks. Nat. Cell Biol. 21, 1553–1564 (2019).
Martell, J. et al. Global cysteine-reactivity profiling during impaired insulin/IGF-1 signaling in C. elegans identifies uncharacterized mediators of longevity. Cell Chem. Biol. 23, 955–966 (2016).
Chen, X. et al. Discovery of heteroaromatic sulfones as a new class of biologically compatible thiol-selective reagents. ACS Chem. Biol. 12, 2201–2208 (2017).
Reisz, J. A., Bechtold, E., King, S. B., Poole, L. B. & Furdui, C. M. Thiol-blocking electrophiles interfere with labeling and detection of protein sulfenic acids. FEBS J. 280, 6150–6161 (2013).
Cuevasanta, E. et al. Reaction of hydrogen sulfide with disulfide and sulfenic acid to form the strongly nucleophilic persulfide. J. Biol. Chem. 290, 26866–26880 (2015).
Yang, J. Sweetly profiling the cysteinome. Nat. Chem. Biol. 15, 935–936 (2019).
Yang, J., Tallman, K. A., Porter, N. A. & Liebler, D. C. Quantitative chemoproteomics for site-specific analysis of protein alkylation by 4-hydroxy-2-nonenal in cells. Anal. Chem. 87, 2535–2541 (2015).
Zhang, B. et al. Proteogenomic characterization of human colon and rectal cancer. Nature 513, 382–387 (2014).
Chambers, M. C. et al. A cross-platform toolkit for mass spectrometry and proteomics. Nat. Biotechnol. 30, 918–920 (2012).
Kim, S. & Pevzner, P. A. MS-GF+ makes progress towards a universal database search tool for proteomics. Nat. Commun. 5, 5277 (2014).
Ma, Z. Q. et al. IDPicker 2.0: improved protein assembly with high discrimination peptide identification filtering. J. Proteome Res. 8, 3872–3881 (2009).
Pino, L. K. et al. The Skyline ecosystem: informatics for quantitative mass spectrometry proteomics. Mass Spectrom. Rev. 39, 229–244 (2017).
Chi, H. et al. Comprehensive identification of peptides in tandem mass spectra using an efficient open search engine. Nat. Biotechnol. 36, 1059–1061 (2018).
Liu, C. et al. pQuant improves quantitation by keeping out interfering signals and evaluating the accuracy of calculated ratios. Anal. Chem. 86, 5286–5294 (2014).
Tyanova, S., Temu, T. & Cox, J. The MaxQuant computational platform for mass spectrometry-based shotgun proteomics. Nat. Protoc. 11, 2301–2319 (2016).
Tran, N. H. et al. Deep learning enables de novo peptide sequencing from data-independent-acquisition mass spectrometry. Nat. Methods 16, 63–66 (2019).
Luo, Q., Tao, Y., Sheng, W., Lu, J. & Wang, H. Dinitroimidazoles as bifunctional bioconjugation reagents for protein functionalization and peptide macrocyclization. Nat. Commun. 10, 142 (2019).
Yu, J., Yang, X., Sun, Y. & Yin, Z. Highly reactive and tracelessly cleavable cysteine-specific modification of proteins via 4-substituted cyclopentenone. Angew. Chem. Int. Ed. Engl. 57, 11598–11602 (2018).
Abegg, D. et al. Proteome-wide profiling of targets of cysteine reactive small molecules by using ethynyl benziodoxolone reagents. Angew. Chem. Int. Ed. Engl. 54, 10852–10857 (2015).
Uttamapinant, C., Sanchez, M. I., Liu, D. S., Yao, J. Z. & Ting, A. Y. Site-specific protein labeling using PRIME and chelation-assisted click chemistry. Nat. Protoc. 8, 1620–1634 (2013).
Rappsilber, J., Mann, M. & Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2, 1896–1906 (2007).
Chick, J. M. et al. A mass-tolerant database search identifies a large proportion of unassigned spectra in shotgun proteomics as modified peptides. Nat. Biotechnol. 33, 743–749 (2015).
Perez-Riverol, Y. et al. The PRIDE database and related tools and resources in 2019: improving support for quantification data. Nucleic Acids Res. 47, D442–D450 (2019).
Acknowledgements
We thank M. Albertolle of The Scripps Research Institute and A. Foti of the Max Planck Institute for Infection Biology Cellular Microbiology for insightful comments. We thank K. S. Carroll of The Scripps Research Institute for kindly providing the S-sulfenic acid specific probe BTD. The work was supported by grants from the National Key R&D Program of China (2016YFA0501303 to J.Y.), the Natural Science Foundation of China (21922702, 81973279 and 31770885 to J.Y. and 31800036 to L.F.) and the State Key Laboratory of Proteomics (SKLP-K201703 and SKLP-K201804 to J.Y.). We also thank B. Fu and B. Zhong from the MS facility of the National Center for Protein Sciences • Beijing for their help and technical support.
Author information
Authors and Affiliations
Contributions
L.F. and J.Y. developed the QTRP protocol and designed the studies. L.F., Z.L. C.T., Jixiang He and Jingyang He performed the experiments and analyzed the data. K.L. developed the post-processing algorithm. F.H., P.X. and J.Y. supervised the project. J.Y. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks Wei-Jun Qian, Johannes Herrmann and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Fu, L. et al. Mol. Cell. Proteomics 16, 1815–1828 (2017): https://www.mcponline.org/content/16/10/1815.long
Sun, R. et al. Chem. Res. Toxicol. 30, 1797–1803 (2017): https://pubs.acs.org/doi/10.1021/acs.chemrestox.7b00183
Petrova, B. et al. Proc. Natl Acad. Sci. USA 115, E7978–E7986 (2018): http://www.pnas.org/content/115/34/E7978
Pei, J.-F. et al. Nat. Cell Biol. 21, 1553–1564 (2019): https://www.nature.com/articles/s41556-019-0420-4
Wang, W. et al. Cell Host Microbe 27, 601–613 (2020): https://www.sciencedirect.com/science/article/abs/pii/S1931312820301682
Supplementary information
Supplementary Information
Supplementary Figs. 1–13.
Supplementary Data 1
Proteome-wide mapping of oxidation-sensitive cysteines in HEK293T cells by QTRP. HEK293T cell lysates stimulated with or without H2O2 (1 mM, 15 min, 37 °C) and labeled with 100 mM IPM. The IPM-labeled proteome was then processed into tryptic peptides. The resulting probe-labeled peptides were conjugated with both L and H azido-UV-cleavable-biotin (Az-UV-biotin) reagents (1:1) via CuAAC. The L and H ‘Click’ reaction mixtures were cleaned with or without SCX. The biotinylated peptides were captured with streptavidin and photoreleased for LC-MS/MS–based identification and quantification. High RH/L values are indicative of cysteines with less free thiol being available after H2O2 treatment, suggesting potential redox-sensitive targets. A schematic workflow is shown in Fig. 2a.
Supplementary Data 2
Quantitative profiling of the intrinsic reactivity of proteomic cysteines. HEK293T proteomes were labeled with low (10 µM) and high (100 µM) levels of IPM. The probe-labeled proteins were processed into tryptic peptides. The resulting probe-labeled peptides were conjugated with both L and H azido-UV-cleavable-biotin (Az-UV-biotin) reagents (1:1) via CuAAC. The light and heavy ‘Click’ reaction mixtures were cleaned with or without SCX. The biotinylated peptides were captured with streptavidin and photoreleased for LC-MS/MS–based identification and quantification. Those with a calculated ratio of H to L (100 µM versus 10 µM, R10:1) <2.0 were defined as hyperreactive. A schematic workflow is shown in Fig. 2b.
Rights and permissions
About this article
Cite this article
Fu, L., Li, Z., Liu, K. et al. A quantitative thiol reactivity profiling platform to analyze redox and electrophile reactive cysteine proteomes. Nat Protoc 15, 2891–2919 (2020). https://doi.org/10.1038/s41596-020-0352-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-020-0352-2
This article is cited by
-
Catalyst-free late-stage functionalization to assemble α-acyloxyenamide electrophiles for selectively profiling conserved lysine residues
Communications Chemistry (2024)
-
Nucleophilic covalent ligand discovery for the cysteine redoxome
Nature Chemical Biology (2023)
-
Reaction-based fluorogenic probes for detecting protein cysteine oxidation in living cells
Nature Communications (2022)
-
The cytosolic thiol peroxidase PRXIIB is an intracellular sensor for H2O2 that regulates plant immunity through a redox relay
Nature Plants (2022)
-
A modification-centric assessment tool for the performance of chemoproteomic probes
Nature Chemical Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.