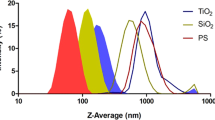
Abstract
Engineered nanomaterials (NMs) are often compositionally indistinguishable from their natural counterparts, and thus their tracking in the environment or within the biota requires the development of appropriate labeling tools. Stable isotope labeling has become a well-established such tool, developed to assign ‘ownership’ or a ‘source’ to engineered NMs, enabling their tracing and quantification, especially in complex environments. A particular methodological challenge for stable isotope labeling is to ensure that the label is traceable in a range of environmental or biological scenarios but does not induce modification of the properties of the NM or lose its signal, thus retaining realism and relevance. This protocol describes a strategy for stable isotope labeling of several widely used metal and metal oxide NMs, namely ZnO, CuO, Ag, and TiO2, using isotopically enriched precursors, namely 67Zn or 68Zn metal, 65CuCl2, 107Ag or 109Ag metal, and 47TiO2 powder. A complete synthesis requires 1–8 d, depending on the type of NM, the precursors used, and the synthesis methods adopted. The physicochemical properties of the labeled particles are determined by optical, diffraction, and spectroscopic techniques for quality control. The procedures for tracing the labels in aquatic (snail and mussel) and terrestrial (earthworm) organisms and for monitoring the environmental transformation of labeled silver (Ag) NMs are also described. We envision that this labeling strategy will be adopted by industry to facilitate applications such as nanosafety assessments before NMs enter the market and environment, as well as for product authentication and tracking.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper are available from the corresponding author upon reasonable request.
References
Keller, A. A. & Lazareva, A. Predicted releases of engineered nanomaterials: from global to regional to local. Environ. Sci. Technol. Lett 1, 65–70 (2013).
Valsami-Jones, E. & Lynch, I. How safe are nanomaterials? Science 350, 388–389 (2015).
Holden, P. A. et al. Considerations of environmentally relevant test conditions for improved evaluation of ecological hazards of engineered nanomaterials. Environ. Sci. Technol. 50, 6124–6145 (2016).
Larner, F. & Rehkämper, M. Evaluation of stable isotope tracing for ZnO nanomaterials—new constraints from high precision isotope analyses and modeling. Environ. Sci. Technol. 46, 4149–4158 (2012).
Leavitt, S., Dueser, R. & Goodell, H. Plant regulation of essential and non-essential heavy metals. J. Appl. Ecol. 16, 203–212 (1979).
Sridhar, B. B. M., Han, F. X., Diehl, S. V., Monts, D. L. & Su, Y. Effects of Zn and Cd accumulation on structural and physiological characteristics of barley plants. Braz. J. Plant Physiol. 19, 15–22 (2007).
Hu, C. W. et al. Toxicological effects of TiO2 and ZnO nanoparticles in soil on earthworm Eisenia fetida. Soil Biol. Biochem. 42, 586–591 (2010).
Kool, P. L., Ortiz, M. D. & van Gestel, G. A. M. Chronic toxicity of ZnO nanoparticles, non-nano ZnO and ZnCl2 to Folsomia candida (Collembola) in relation to bioavailability in soil. Environ. Pollut. 159, 2713–2719 (2011).
Gottschalk, F., Sun, T. & Nowack, B. Environmental concentrations of engineered nanomaterials: review of modeling and analytical studies. Environ. Pollut. 181, 287–300 (2013).
Lowry, G. V., Gregory, K. B., Apte, S. C. & Lead, J. R. Transformations of nanomaterials in the environment. Environ. Sci. Technol. 46, 6893–6899 (2012).
Zhang, P. et al. Biotransformation of ceria nanoparticles in cucumber plants. ACS Nano 6, 9943–9950 (2012).
Santra, S. et al. TAT conjugated, FITC doped silica nanoparticles for bioimaging applications. Chem. Commun. 24, 2810–2811 (2004).
Zhi, L., Ren, M., Qu, M., Zhang, H. & Wang, D. Wnt ligands differentially regulate toxicity and translocation of graphene oxide through different mechanisms in Caenorhabditis elegans. Sci. Rep. 6, 39261 (2016).
Zhang, Z. et al. Uptake and distribution of ceria nanoparticles in cucumber plants. Metallomics 3, 816–822 (2011).
Freund, B. et al. A simple and widely applicable method to 59Fe-radiolabel monodisperse superparamagnetic iron oxide nanoparticles for in vivo quantification studies. ACS Nano 6, 7318–7325 (2012).
Kannan, R. et al. Functionalized radioactive gold nanoparticles in tumor therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 4, 42–51 (2012).
Yang, K., Feng, L., Hong, H., Cai, W. & Liu, Z. Preparation and functionalization of graphene nanocomposites for biomedical applications. Nat. Protoc. 8, 2392–2403 (2013).
Wiederhold, J. G. Metal stable isotope signatures as tracers in environmental geochemistry. Environ. Sci. Technol. 49, 2606–2624 (2015).
Simon, J. et al. Calcium and titanium isotope fractionation in refractory inclusions: tracers of condensation and inheritance in the early solar protoplanetary disk. Earth. Planet. Sci. Lett. 472, 277–288 (2017).
Šillerová, H. et al. Stable isotope tracing of Ni and cu pollution in north-East Norway: potentials and drawbacks. Environ. Pollut. 228, 149–157 (2017).
Chokkathukalam, A., Kim, D. H., Barrett, M. P., Breitling, R. & Creek, D. J. Stable isotope-labeling studies in metabolomics: new insights into structure and dynamics of metabolic networks. Bioanalysis 6, 511–524 (2014).
Chahrour, O., Cobice, D. & Malone, J. Stable isotope labelling methods in mass spectrometry-based quantitative proteomics. J. Pharm. Biomed. Anal. 113, 2–20 (2015).
Yuan, J., Bennett, B. D. & Rabinowitz, J. D. Kinetic flux profiling for quantitation of cellular metabolic fluxes. Nat. Protoc. 3, 1328 (2008).
Mugoni, V., Medana, C. & Santoro, M. M. 13C-isotope-based protocol for prenyl lipid metabolic analysis in zebrafish embryos. Nat. Protoc. 8, 2337 (2013).
Sharp, Z. Principles of Stable Isotope Geochemistry 2nd edn. Pearson Education (2017).
Dybowska, A., Misra, S. K. & Valsami-Jones, E. Labelling nanoparticles with non-radioactive isotopes. in Isotopes in Nanoparticles—Fundamentals and Applications (eds Llop, J., Gomez-Vallejo, V. & Gibson, N.) 455–485 (Pan Stanford Publishing Singapore, 2016).
Becker, J. S., Matusch, A. & Wu, B. Bioimaging mass spectrometry of trace elements–recent advance and applications of LA-ICP-MS: a review. Anal. Chim. Acta 835, 1–18 (2014).
Proetto, M. T. et al. Cellular delivery of nanoparticles revealed with combined optical and isotopic nanoscopy. ACS Nano 10, 4046–4054 (2016).
Lee, P. L. et al. Development and validation of TOF-SIMS and CLSM imaging method for cytotoxicity study of ZnO nanoparticles in HaCaT cells. J. Hazard. Mater. 277, 3–12 (2014).
Yu, S., Yin, Y., Zhou, X., Dong, L. & Liu, J. Transformation kinetics of silver nanoparticles and silver ions in aquatic environments revealed by double stable isotope labeling. Environ. Sci. Nano 3, 883–893 (2016).
Dybowska, A. D. et al. Synthesis of isotopically modified ZnO nanoparticles and their potential as nanotoxicity tracers. Environ. Pollut. 159, 266–273 (2011).
Laycock, A. et al. Novel multi-isotope tracer approach to test ZnO nanoparticle and soluble Zn bioavailability in joint soil exposures. Environ. Sci. Technol. 51, 12756–12763 (2017).
Misra, S. K. et al. Isotopically modified nanoparticles for enhanced detection in bioaccumulation studies. Environ. Sci. Technol. 46, 1216–1222 (2012).
Croteau, M.-Nl., Misra, S. K., Luoma, S. N. & Valsami-Jones, E. Bioaccumulation and toxicity of CuO nanoparticles by a freshwater invertebrate after waterborne and dietborne exposures. Environ. Sci. Technol. 48, 10929–10937 (2014).
Laycock, A. et al. Synthesis and characterization of isotopically labeled silver nanoparticles for tracing studies. Environ. Sci. Nano 1, 271–283 (2014).
Croteau, M.-N., Dybowska, A. D., Luoma, S. N., Misra, S. K. & Valsami-Jones, E. Isotopically modified silver nanoparticles to assess nanosilver bioavailability and toxicity at environmentally relevant exposures. Environ. Chem. 11, 247–256, https://doi.org/10.1071/EN13141 (2014).
Bourgeault, A. et al. The challenge of studying TiO2 nanoparticle bioaccumulation at environmental concentrations: crucial use of a stable isotope tracer. Environ. Sci. Technol. 49, 2451–2459 (2015).
Meermann, B., Wichmann, K., Lauer, F., Vanhaecke, F. & Ternes, T. A. Application of stable isotopes and AF4/ICP-SFMS for simultaneous tracing and quantification of iron oxide nanoparticles in a sediment–slurry matrix. J. Anal. At. Spectrom 31, 890–901 (2016).
Meuller, B. O. et al. Review of spark discharge generators for production of nanoparticle aerosols. Aerosol Sci. Technol. 46, 1256–1270 (2012).
Croteau, M. N., Dybowska, A. D., Luoma, S. N. & Valsami-Jones, E. A novel approach reveals that zinc oxide nanoparticles are bioavailable and toxic after dietary exposures. Nanotoxicology 5, 79–90 (2011).
Laycock, A. et al. Earthworm uptake routes and rates of ionic Zn and ZnO nanoparticles at realistic concentrations, traced using stable isotope labeling. Environ. Sci. Technol. 50, 412–419 (2015).
Pick, D., Leiterer, M. & Einax, J. W. Reduction of polyatomic interferences in biological material using dynamic reaction cell ICP-MS. Microchem. J. 95, 315–319 (2010).
Khosravi, K., Hoque, M. E., Dimock, B., Hintelmann, H. & Metcalfe, C. D. A novel approach for determining total titanium from titanium dioxide nanoparticles suspended in water and biosolids by digestion with ammonium persulfate. Anal. Chim. Acta 713, 86–91 (2012).
Kay, A. R. Detecting and minimizing zinc contamination in physiological solutions. BMC Physiol. 4, 1–9 (2004).
Vanhaecke, F., Vanhoe, H., Dams, R. & Vandecasteele, C. The use of internal standards in ICP-MS. Talanta 39, 737–742 (1992).
Liu, J. Y. & Robert, H. H. Ion release kinetics and particle persistence in aqueous nano-silver colloids. Environ. Sci. Technol. 44, 2169–2175 (2010).
US Environmental Protection Agency. Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms, EPA-821-R-02-012. 5th edn (US Environmental Protection Agency Office of Water, Cincinnati, OH, 2002).
Eisler, R. Silver hazards to fish, wildlife and invertebrates: a synoptic review in Contaminant Hazard Reviews Biological Science Report 32 (US Department of the Interior, National Biological Service, 1981).
Heggelund, L. R. et al. Soil pH effects on the comparative toxicity of dissolved zinc, non-nano and nano ZnO to the earthworm Eisenia fetida. Nanotoxicology 8, 559–572 (2014).
Lu, D. et al. Stable silver isotope fractionationin the natural transformation process of silver nanoparticles. Nat. Nanotechnol. 11, 682–686 (2016).
Acknowledgements
This work was supported by Marie Skłodowska-Curie Individual Fellowships (NanoLabels 750455 to P.Z.; NanoBBB 798505 to Z.G.) under the European Union’s Horizon 2020 research program. Financial support from MHRD-IMPRINT funds is appreciated. Partial funding from EU H2020 project ACEnano (grant agreement no. 2016-720952) is also acknowledged.
Author information
Authors and Affiliations
Contributions
P.Z., S.M., and Z.G. wrote the paper. E.V.-J. and M.R. designed part of the experiments and revised the paper. S.M. performed part of the experiments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Dybowska, A.D. et al. Environ. Pollut. 159, 266–273 (2011): https://doi.org/10.1016/j.envpol.2010.08.032
Yu, S., Yin, Y., Zhou, X., Dong, L. & Liu, J. Environ. Sci. Nano 3, 883–893 (2016): https://doi.org/10.1039/c6en00104a
Misra, S.K. et al. Environ. Sci. Technol. 46, 1216–1222 (2012): https://doi.org/10.1021/es2039757
Laycock, A. et al. Environ. Sci. Nano 1, 271–283 (2014): https://doi.org/10.1039/c3en00100h
Bourgeault, A. et al. Environ. Sci. Technol. 49, 2451–2459 (2015): https://doi.org/10.1021/es504638f
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, P., Misra, S., Guo, Z. et al. Stable isotope labeling of metal/metal oxide nanomaterials for environmental and biological tracing. Nat Protoc 14, 2878–2899 (2019). https://doi.org/10.1038/s41596-019-0205-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-019-0205-z
This article is cited by
-
Characterization and environmental applications of soil biofilms: a review
Environmental Chemistry Letters (2024)
-
An analytical workflow for dynamic characterization and quantification of metal-bearing nanomaterials in biological matrices
Nature Protocols (2022)
-
Nanocontaminants in soil: emerging concerns and risks
International Journal of Environmental Science and Technology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.