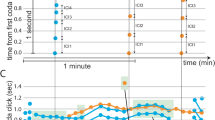
Abstract
Investigation of the neural mechanisms underlying social hierarchy requires a reliable and effective behavioral test. The tube test is a simple and robust behavioral assay that we recently validated as a reliable measure of social hierarchy in mice. The test was demonstrated to produce results largely consistent with the results seen when using other dominance measures, including the warm spot test, territory urine marking or the courtship ultrasound vocalization test. Here, we describe a step-by-step procedure to use the tube test to measure dominance within a cage of four male C57/BL6 mice as an example application. The procedure comprises three stages: habituation, training to pass through the tube, and the tube test itself. The social rank of each mouse is determined by the number of wins it gains when competing against the other three cagemates. A stable rank is derived when all mice maintain the same ranking for 4 consecutive days. The time required to acquire a stable rank usually varies from 4 to 14 d. An additional 5 d is required for habituation and training.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Change history
11 April 2019
In the version of this paper originally published, an affiliation for Zhengxiao Fan was omitted. In addition, the Reporting Summary incorrectly indicated that human research participants had been used in the study, instead of animal subjects. These errors have been corrected in the PDF and HTML versions of the protocol.
References
Cole, B. J. Dominance hierarchies in leptothorax ants. Science 212, 83–84 (1981).
Grosenick, L., Clement, T. S. & Fernald, R. D. Fish can infer social rank by observation alone. Nature 445, 429–432 (2007).
Dunbar, R. I. & Dunbar, E. P. Dominance and reproductive success among female gelada baboons. Nature 266, 351–352 (1977).
Qu, C., Ligneul, R., Van der Henst, J. B. & Dreher, J. C. An integrative interdisciplinary perspective on social dominance hierarchies. Trends Cogn. Sci. 21, 893–908 (2017).
Bercovitch, F. B. & Clarke, A. S. Dominance rank, cortisol concentrations, and reproductive maturation in male rhesus macaques. Phys. Behav. 58, 215–221 (1995).
Bernstein, I. S. Dominance—the baby and the bathwater. Behav. Brain Sci. 4, 419–429 (1981).
Sapolsky, R. M. The influence of social hierarchy on primate health. Science 308, 648–652 (2005).
Yeh, S. R., Fricke, R. A. & Edwards, D. H. The effect of social experience on serotonergic modulation of the escape circuit of crayfish. Science 271, 366–369 (1996).
Russell, J. A. & Mehrabian, A. Evidence for a three-factory theory of emotions. J. Res. Pers. 11, 273–294 (1977).
Bakker, I., van der Voordt, T., Vink, P. & de Boon, J. Pleasure, arousal, dominance: Mehrabian and Russell revisited. Curr. Psychol. 33, 405–421 (2014).
Mehrabian, A. Pleasure-arousal-dominance: a general framework for describing and measuring individual differences in temperament. Curr. Psychol. 14, 261–292 (1966).
Drews, C. The concept and definition of dominance in animal behaviour. Behaviour 125, 283–313 (1993).
Hand, J. L. Resolution of social conflicts—dominance, egalitarianism, spheres of dominance, and game theory. Quart. Rev. Biol. 61, 201–220 (1986).
Wang, F., Kessels, H. W. & Hu, H. The mouse that roared: neural mechanisms of social hierarchy. Trends Neurosci. 2014, 1–9 (2014).
Chou, M. Y. et al. Social conflict resolution regulated by two dorsal habenular subregions in zebrafish. Science 352, 87–90 (2016).
Lindzey, G., Winston, H. & Manosevitz, M. Social dominance in inbred mouse strains. Nature 191, 474–476 (1961).
Wang, F. et al. Bidirectional control of social hierarchy by synaptic efficacy in medial prefrontal cortex. Science 334, 693–697 (2011).
Merlot, E., Moze, E., Bartolomucci, A., Dantzer, R. & Neveu, P. J. The rank assessed in a food competition test influences subsequent reactivity to immune and social challenges in mice. Brain Behav. Immun. 18, 468–475 (2004).
Cordero, M. I. & Sandi, C. Stress amplifies memory for social hierarchy. Front. Neurosci. 1, 175–184 (2007).
Lee, Y. P., Craig, J. V. & Dayton, A. D. The Social Rank Index as a measure of social status and its association with egg production in White Leghorn pullets. Appl. Anim. Ethol. 8, 377–390 (1982).
Hessing, M. J. C. & Tielen, M. J. M. The effect of climatic environment and relocating and mixing on health status and productivity of pigs. Anim. Prod. 59, 131–139 (1994).
Jupp, B. et al. Social dominance in rats: effects on cocaine self-administration, novelty reactivity and dopamine receptor binding and content in the striatum. Psychopharmacology 233, 579–589 (2016).
Timmer, M. & Sandi, C. A role for glucocorticoids in the long-term establishment of a social hierarchy. Psychoneuroendocrinology 35, 1543–1552 (2010).
Ujita, W., Kohyama-Koganeya, A., Endo, N., Saito, T. & Oyama, H. Mice lacking a functional NMDA receptor exhibit social subordination in a group-housed environment. FEBS J. 285, 188–196 (2018).
Lucion, A. & Vogel, W. Effects of stress on defensive aggression and dominance in a water competition test. Integr. Physiol. Behav. Sci. 29, 415–422 (1994).
Zhou, T. et al. History of winning remodels thalamo-PFC circuit to reinforce social dominance. Science 357, 162–168 (2017).
Zhu, H. & Hu, H. L. Brain’s neural switch for social dominance in animals. Sci. China Life Sci. 61, 113–114 (2018).
Ralls, K. Mammalian scent marking. Science 171, 443–449 (1971).
Desjardins, C., Maruniak, J. A. & Bronson, F. H. Social rank in house mice—differentiation revealed by ultraviolet visualization of urinary marking patterns. Science 182, 939–941 (1973).
Long, S. Y. Hair-nibbling and whisker-trimming as indicators of social hierarchy in mice. Anim. Behav. 20, 10–12 (1972).
Kalueff, A. V., Minasyan, A., Keisala, T., Shah, Z. H. & Tuohimaa, P. Hair barbering in mice: implications for neurobehavioural research. Behav. Processes 71, 8–15 (2006).
Strozik, E. & Festing, M. F. W. Whisker trimming in mice. Lab Anim. 15, 309–312 (1981).
Hauschka, T. S. Whisker-eating mice. J. Hered. 43, 77–80 (1952).
Dizinno, G., Whitney, G. & Nyby, J. Ultrasonic vocalizations by male mice (Mus musculus) to female sex-pheromone—experiential determinants. Behav. Biol. 22, 104–113 (1978).
Nyby, J., Dizinno, G. A. & Whitney, G. Social status and ultrasonic vocalizations of male mice. Behav. Biol. 18, 285–289 (1976).
Damato, F. R. Courtship ultrasonic vocalizations and social status in mice. Anim. Behav. 41, 875–885 (1991).
Williamson, C. M., Romeo, R. D. & Curley, J. P. Dynamic changes in social dominance and mPOA GnRH expression in male mice following social opportunity. Hormones Behav. 87, 80–88 (2017).
Crawley, J. N. Behavioral phenotyping of transgenic and knockout mice: experimental design and evaluation of general health, sensory functions, motor abilities, and specific behavioral tests. Brain Res. 835, 18–26 (1999).
Garfield, A. S. et al. Distinct physiological and behavioural functions for parental alleles of imprinted Grb10. Nature 469, 534–538 (2011).
Zhou, Y. et al. Mice with Shank3 mutations associated with ASD and schizophrenia display both shared and distinct defects. Neuron 89, 147–162 (2016).
Hou, X. H. et al. Central control circuit for context-dependent micturition. Cell 167, 73–86.e12 (2016).
Zhou, T., Sandi, C. & Hu, H. Advances in understanding neural mechanisms of social dominance. Curr. Opin. Neurobiol. 49, 99–107 (2018).
Larrieu, T. et al. Hierarchical status predicts behavioral vulnerability and nucleus accumbens metabolic profile following chronic social defeat stress. Curr. Biol. 27, 2202–2210.e04 (2017).
Park, M. J., Seo, B. A., Lee, B., Shin, H. S. & Kang, M. G. Stress-induced changes in social dominance are scaled by AMPA-type glutamate receptor phosphorylation in the medial prefrontal cortex. Sci. Rep. 8, 15008 (2018).
lindzey, G., Manoseitz, M. & Winston, H. Social dominance in the mouse. Psychon. Sci. 5, 451–452 (1966).
Baenninger, L. Social dominance orders in the rat: “spontaneous,” food, and water competition. J. Comp. Physiol. Psych. 71, 202–209 (1970).
Miczek, K. A. & Barry, H.3rd. What does the tube test measure?. Behav. Biol. 13, 537–539 (1975).
Miczek, K. A. & Barry, H. Delta-9-tetrahydrocannabinol and aggressive behavior in rats. Behav. Biol. 11, 261–267 (1974).
Stagkourakis, S. et al. A neural network for intermale aggression to establish social hierarchy. Nat. Neurosci. 21, 834–842 (2018).
Zhong, J. et al. Cyclic ADP-ribose and heat regulate oxytocin release via CD38 and TRPM2 in the hypothalamus during social or psychological stress in mice. Front. Neurosci. 10, 304 (2016).
van den Berg, W. E., Lamballais, S. & Kushner, S. A. Sex-specific mechanism of social hierarchy in mice. Neuropsychopharmacology 40, 1364–1372 (2015).
Anacker, A. M. J., Smith, M. L. & Ryabinin, A. E. Establishment of stable dominance interactions in prairie vole peers: relationships with alcohol drinking and activation of the paraventricular nucleus of the hypothalamus. Soc. Neurosci. 9, 484–494 (2014).
Matthews, G. A. et al. Dorsal raphe dopamine neurons represent the experience of social isolation. Cell 164, 617–631 (2016).
Kunkel, T. & Wang, H. B. Socially dominant mice in C57BL6 background show increased social motivation. Behav. Brain Res. 336, 173–176 (2018).
Saxena, K. et al. Experiential contributions to social dominance in a rat model of fragile-X syndrome. Proc. Biol. Sci. 285, 20180294 (2018).
Yang, C. R. et al. Enhanced aggressive behaviour in a mouse model of depression. Neurotox. Res. 27, 129–142 (2015).
Huang, W. H. et al. Early adolescent Rai1 reactivation reverses transcriptional and social interaction deficits in a mouse model of Smith-Magenis syndrome. Proc. Natl. Acad. Sci. USA 115, 10744–10749 (2018).
Cao, W. Y. et al. Role of early environmental enrichment on the social dominance tube test at adulthood in the rat. Psychopharmacology 234, 3321–3334 (2017).
Tada, H. et al. Neonatal isolation augments social dominance by altering actin dynamics in the medial prefrontal cortex. Proc. Natl. Acad. Sci. USA 113, E7097–E7105 (2016).
Arrant, A. E., Filiano, A. J., Warmus, B. A., Hall, A. M. & Roberson, E. D. Progranulin haploinsufficiency causes biphasic social dominance abnormalities in the tube test. Genes Brain Behav. 15, 588–603 (2016).
Friard, O. & Gamba, M. BORIS: a free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol. Evol. 7, 1325–1330 (2016).
Acknowledgements
We thank the Hu laboratory members for valuable discussions and advice. This study was supported by grants from the National Natural Science Foundation of China (31830032, 81527901, 91432108 and 31225010), the National Key R&D Program of China (2016YFA0501000) to H.H., the Non-profit Central Research Institute Fund of the Chinese Academy of Medical Sciences (2017PT31038 and 2018PT31041) and the 111 Project (B13026) to H.H.
Author information
Authors and Affiliations
Contributions
T.Z., H.Z. and H.H. designed the experimental strategy. Z.F., T.Z. and H.Z. optimized experimental procedures. Z.F., S.W., Y.W. and H.H. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information: Nature Protocols thanks Christian Broberger, Carmen Sandi and other (anonymous) reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Wang, F. et al. Science 334, 693–697 (2011): https://doi.org/10.1126/science.1209951
Zhou, T. et al. Science 357, 162–168 (2017): https://doi.org/10.1126/science.aak9726
Supplementary information
Supplementary Video 1
Training stage of the tube test.
Supplementary Video 2
Testing stage of the tube test.
Supplementary Video 3
Annotation of different behavior epochs during a tube test trial. Adapted with permission from Zhou et al.26.
Supplementary Video 4
Optogenetics in tube test. A tube with a 15-mm slit is used for optogenetics. After a stable tube test rank is determined, acute dmPFC photostimulation of a previously rank 3 mouse induces winning against a rank 1 mouse. Adapted with permission from Zhou et al.26.
Rights and permissions
About this article
Cite this article
Fan, Z., Zhu, H., Zhou, T. et al. Using the tube test to measure social hierarchy in mice. Nat Protoc 14, 819–831 (2019). https://doi.org/10.1038/s41596-018-0116-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0116-4
This article is cited by
-
Acetylcholine muscarinic M1 receptors in the rodent prefrontal cortex modulate cognitive abilities to establish social hierarchy
Neuropsychopharmacology (2024)
-
Single cell transcriptomic representation of social dominance in prefrontal cortex and the influence of preweaning maternal and postweaning social environment
Scientific Reports (2024)
-
Prefrontocortical Astrocytes Regulate Dominance Hierarchy in Male Mice
Neuroscience Bulletin (2024)
-
Sustained overexpression of spliced X-box-binding protein-1 in neurons leads to spontaneous seizures and sudden death in mice
Communications Biology (2023)
-
Cortical astrocytes modulate dominance behavior in male mice by regulating synaptic excitatory and inhibitory balance
Nature Neuroscience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.