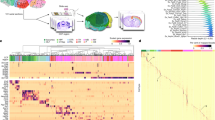
Abstract
The nucleus accumbens (NAc) plays an important role in regulating multiple behaviors, and its dysfunction has been linked to many neural disorders. However, the molecular, cellular and anatomic heterogeneity underlying its functional diversity remains incompletely understood. In this study, we generated a cell census of the mouse NAc using single-cell RNA sequencing and multiplexed error-robust fluorescence in situ hybridization, revealing a high level of cell heterogeneity in this brain region. Here we show that the transcriptional and spatial diversity of neuron subtypes underlie the NAc’s anatomic and functional heterogeneity. These findings explain how the seemingly simple neuronal composition of the NAc achieves its highly heterogenous structure and diverse functions. Collectively, our study generates a spatially resolved cell taxonomy for understanding the structure and function of the NAc, which demonstrates the importance of combining molecular and spatial information in revealing the fundamental features of the nervous system.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The scRNA-seq data have been deposited at the Gene Expression Omnibus with accession number GSE118020. The MERFISH data are available at the Brain Image Library (https://download.brainimagelibrary.org/fc/4c/fc4c2570c3711952/).
Code availability
Code for MERFISH image acquisition is available at https://github.com/ZhuangLab. Code for MERFISH image analysis is available at https://github.com/ZhuangLab/MERlin. Code for MERFISH and scRNA-seq integration is available at https://github.com/YiZhang-lab/NAcMERFISHscRNAseqAnalysis.
References
Floresco, S. B. The nucleus accumbens: an interface between cognition, emotion, and action. Annu. Rev. Psychol. 66, 25–52 (2015).
O’Connor, E. C. et al. Accumbal D1R neurons projecting to lateral hypothalamus authorize feeding. Neuron 88, 553–564 (2015).
Dolen, G., Darvishzadeh, A., Huang, K. W. & Malenka, R. C. Social reward requires coordinated activity of nucleus accumbens oxytocin and serotonin. Nature 501, 179–184 (2013).
Smith-Roe, S. L. & Kelley, A. E. Coincident activation of NMDA and dopamine D1 receptors within the nucleus accumbens core is required for appetitive instrumental learning.J. Neurosci. 20, 7737–7742 (2000).
Kravitz, A. V., Tye, L. D. & Kreitzer, A. C. Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nat. Neurosci. 15, 816–818 (2012).
Berridge, K. C. & Kringelbach, M. L. Pleasure systems in the brain. Neuron 86, 646–664 (2015).
Pascoli, V., Turiault, M. & Luscher, C. Reversal of cocaine-evoked synaptic potentiation resets drug-induced adaptive behaviour. Nature 481, 71–75 (2012).
Krishnan, V. et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 131, 391–404 (2007).
Bewernick, B. H. et al. Nucleus accumbens deep brain stimulation decreases ratings of depression and anxiety in treatment-resistant depression. Biol. Psychiatry 67, 110–116 (2010).
McCollum, L. A., Walker, C. K., Roche, J. K. & Roberts, R. C. Elevated excitatory input to the nucleus accumbens in schizophrenia: a postmortem ultrastructural study. Schizophr. Bull. 41, 1123–1132 (2015).
Oh, S. W. et al. A mesoscale connectome of the mouse brain. Nature 508, 207–214 (2014).
Groenewegen, H. J., Wright, C. I., Beijer, A. V. & Voorn, P. Convergence and segregation of ventral striatal inputs and outputs. Ann. N. Y. Acad. Sci. 877, 49–63 (1999).
Salgado, S. & Kaplitt, M. G. The nucleus accumbens: a comprehensive review. Stereotact. Funct. Neurosurg. 93, 75–93 (2015).
Britt, J. P. et al. Synaptic and behavioral profile of multiple glutamatergic inputs to the nucleus accumbens. Neuron 76, 790–803 (2012).
Pascoli, V. et al. Contrasting forms of cocaine-evoked plasticity control components of relapse. Nature 509, 459–464 (2014).
Yang, H. et al. Nucleus accumbens subnuclei regulate motivated behavior via direct inhibition and disinhibition of VTA dopamine subpopulations. Neuron 97, 434–449 (2018).
de Jong, J. W. et al. A neural circuit mechanism for encoding aversive stimuli in the mesolimbic dopamine system. Neuron 101, 133–151 (2019).
Lammel, S. et al. Input-specific control of reward and aversion in the ventral tegmental area. Nature 491, 212–217 (2012).
Tepper, J. M., Tecuapetla, F., Koos, T. & Ibanez-Sandoval, O. Heterogeneity and diversity of striatal GABAergic interneurons. Front. Neuroanat. 4, 150 (2010).
Gerfen, C. R. & Surmeier, D. J. Modulation of striatal projection systems by dopamine. Annu. Rev. Neurosci. 34, 441–466 (2011).
Kreitzer, A. C. & Malenka, R. C. Striatal plasticity and basal ganglia circuit function. Neuron 60, 543–554 (2008).
Gerfen, C. R. et al. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 250, 1429–1432 (1990).
Yawata, S., Yamaguchi, T., Danjo, T., Hikida, T. & Nakanishi, S. Pathway-specific control of reward learning and its flexibility via selective dopamine receptors in the nucleus accumbens. Proc. Natl Acad. Sci. USA 109, 12764–12769 (2012).
Voorn, P., Gerfen, C. R. & Groenewegen, H. J. Compartmental organization of the ventral striatum of the rat: immunohistochemical distribution of enkephalin, substance P, dopamine, and calcium-binding protein. J. Comp. Neurol. 289, 189–201 (1989).
Brimblecombe, K. R. & Cragg, S. J. The striosome and matrix compartments of the striatum: a path through the labyrinth from neurochemistry toward function. ACS Chem. Neurosci. 8, 235–242 (2017).
Kupchik, Y. M. et al. Coding the direct/indirect pathways by D1 and D2 receptors is not valid for accumbens projections. Nat. Neurosci. 18, 1230–1232 (2015).
Al-Hasani, R. et al. Distinct subpopulations of nucleus accumbens dynorphin neurons drive aversion and reward. Neuron 87, 1063–1077 (2015).
Gibson, G. D. et al. Distinct accumbens shell output pathways promote versus prevent relapse to alcohol seeking. Neuron 98, 512–520 (2018).
Gokce, O. et al. Cellular taxonomy of the mouse striatum as revealed by single-cell RNA-seq. Cell Rep. 16, 1126–1137 (2016).
Stanley, G., Gokce, O., Malenka, R. C., Sudhof, T. C. & Quake, S. R. Continuous and discrete neuron types of the adult murine striatum. Neuron 105, 688–699 (2020).
Saunders, A. et al. Molecular diversity and specializations among the cells of the adult mouse brain. Cell 174, 1015–1030 (2018).
Chen, K. H., Boettiger, A. N., Moffitt, J. R., Wang, S. & Zhuang, X. RNA imaging. Spatially resolved, highly multiplexed RNA profiling in single cells. Science 348, aaa6090 (2015).
Moffitt, J. R. et al. Molecular, spatial, and functional single-cell profiling of the hypothalamic preoptic region. Science 362, eaau5324 (2018).
Chen, R., Wu, X., Jiang, L. & Zhang, Y. Single-cell RNA-seq reveals hypothalamic cell diversity. Cell Rep. 18, 3227–3241 (2017).
Zeisel, A. et al. Brain structure. Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science 347, 1138–1142 (2015).
Zeisel, A. et al. Molecular architecture of the mouse nervous system. Cell 174, 999–1014 (2018).
Ibanez-Sandoval, O. et al. A novel functionally distinct subtype of striatal neuropeptide Y interneuron. J. Neurosci. 31, 16757–16769 (2011).
Munoz-Manchado, A. B. et al. Diversity of interneurons in the dorsal striatum revealed by single-cell RNA sequencing and PatchSeq. Cell Rep. 24, 2179–2190 (2018).
Walaas, S. I., Aswad, D. W. & Greengard, P. A dopamine- and cyclic AMP-regulated phosphoprotein enriched in dopamine-innervated brain regions. Nature 301, 69–71 (1983).
Hintiryan, H. et al. The mouse cortico-striatal projectome. Nat. Neurosci. 19, 1100–1114 (2016).
Lee, J., Wang, W. & Sabatini, B. L. Anatomically segregated basal ganglia pathways allow parallel behavioral modulation. Nat. Neurosci. 23, 1388–1398 (2020).
Xiao, X. et al. A genetically defined compartmentalized striatal direct pathway for negative reinforcement. Cell 183, 211–227 (2020).
Castro, D. C. & Bruchas, M. R. A motivational and neuropeptidergic hub: anatomical and functional diversity within the nucleus accumbens shell. Neuron 102, 529–552 (2019).
Chaudhri, N., Sahuque, L. L., Schairer, W. W. & Janak, P. H. Separable roles of the nucleus accumbens core and shell in context- and cue-induced alcohol-seeking. Neuropsychopharmacology 35, 783–791 (2010).
Di Chiara, G. Nucleus accumbens shell and core dopamine: differential role in behavior and addiction. Behav. Brain Res. 137, 75–114 (2002).
Ito, R., Robbins, T. W. & Everitt, B. J. Differential control over cocaine-seeking behavior by nucleus accumbens core and shell. Nat. Neurosci. 7, 389–397 (2004).
Castro, D. C. & Berridge, K. C. Opioid hedonic hotspot in nucleus accumbens shell: mu, delta, and kappa maps for enhancement of sweetness ‘liking’ and ‘wanting’. J. Neurosci. 34, 4239–4250 (2014).
Puga, L., Alcantara-Alonso, V., Coffeen, U., Jaimes, O. & de Gortari, P. TRH injected into the nucleus accumbens shell releases dopamine and reduces feeding motivation in rats. Behav. Brain Res. 306, 128–136 (2016).
Darcq, E. & Kieffer, B. L. Opioid receptors: drivers to addiction? Nat. Rev. Neurosci. 19, 499–514 (2018).
Le Merrer, J., Becker, J. A., Befort, K. & Kieffer, B. L. Reward processing by the opioid system in the brain. Physiol. Rev. 89, 1379–1412 (2009).
Banghart, M. R., Neufeld, S. Q., Wong, N. C. & Sabatini, B. L. Enkephalin disinhibits mu opioid receptor-rich striatal patches via delta opioid receptors. Neuron 88, 1227–1239 (2015).
Tejeda, H. A. et al. Pathway- and cell-specific kappa-opioid receptor modulation of excitation-inhibition balance differentially gates D1 and D2 accumbens neuron activity. Neuron 93, 147–163 (2017).
Eng, C. L. et al. Transcriptome-scale super-resolved imaging in tissues by RNA seqFISH. Nature 568, 235–239 (2019).
Francis, T. C., Yano, H., Demarest, T. G., Shen, H. & Bonci, A. High-frequency activation of nucleus accumbens D1-MSNs drives excitatory potentiation on D2-MSNs. Neuron 103, 432–444 (2019).
Lechan, R. M. & Fekete, C. The TRH neuron: a hypothalamic integrator of energy metabolism. Prog. Brain Res. 153, 209–235 (2006).
Zhang, M. et al. Molecular, spatial and projection diversity of neurons in primary motor cortex revealed by in situ single-cell transcriptomics. Preprint at https://www.biorxiv.org/content/10.1101/2020.06.04.105700v1 (2020).
Brewer, G. J. & Torricelli, J. R. Isolation and culture of adult neurons and neurospheres. Nat. Protoc. 2, 1490–1498 (2007).
Zheng, G. X. et al. Massively parallel digital transcriptional profiling of single cells. Nat. Commun. 8, 14049 (2017).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Kuhn, M. Building predictive models in R using the caret package. J. Stat. Softw. 1, 2008 (2008).
Mayer, C. et al. Developmental diversification of cortical inhibitory interneurons. Nature 555, 457–462 (2018).
Shekhar, K. et al. Comprehensive classification of retinal bipolar neurons by single-cell transcriptomics. Cell 166, 1308–1323 (2016).
Wolf, F. A., Angerer, P. & Theis, F. J. SCANPY: large-scale single-cell gene expression data analysis. Genome Biol. 19, 15 (2018).
Satija, R., Farrell, J. A., Gennert, D., Schier, A. F. & Regev, A. Spatial reconstruction of single-cell gene expression data. Nat. Biotechnol. 33, 495–502 (2015).
Blondel, V. D., Guillaume, J.-L., Lambiotte, R. & Lefebvre, E. Fast unfolding of communities in large networks. J. Stat. Mech. 2008, P10008 (2008).
Moffitt, J. R. et al. High-throughput single-cell gene-expression profiling with multiplexed error-robust fluorescence in situ hybridization. Proc. Natl Acad. Sci. USA 113, 11046–11051 (2016).
Xia, C., Fan, J., Emanuel, G., Hao, J. & Zhuang, X. Spatial transcriptome profiling by MERFISH reveals subcellular RNA compartmentalization and cell cycle-dependent gene expression. Proc. Natl Acad. Sci. USA 116, 19490–19499 (2019).
Traag, V. A., Waltman, L. & van Eck, N. J. From Louvain to Leiden: guaranteeing well-connected communities. Sci. Rep. 9, 5233 (2019).
Korsunsky, I. et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 16, 1289–1296 (2019).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Gu, Z., Eils, R. & Schlesner, M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics 32, 2847–2849 (2016).
Kolde, R. Pheatmap: pretty heatmaps [Software] (2015).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, 2016).
Tasic, B. et al. Shared and distinct transcriptomic cell types across neocortical areas. Nature 563, 72–78 (2018).
Acknowledgements
We thank Z. Chen and Q. Yin for their help with sequencing and B. L. Sabatini for critical reading of the manuscript. We acknowledge the support of the HMS Neurobiology Imaging Facility (supported by NINDS P30 Core Center Grant no. NS072030) and its staff, R. Carelli and M. Ocana, and the Mouse Behavior Core of Harvard Medical School and its director, B. Caldarone. This project was supported by the National Institutes of Health (NIDA R01DA042283 to Y.Z. and NIMH U19MH114821 to X.Z.) and the Howard Hughes Medical Institute. Y.Z. and X.Z. are investigators of the Howard Hughes Medical Institute. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.Z. conceived the project. Y.Z. and X.Z. supervised the project. R.C, T.R.B., Y.Z. and X.Z. designed the experiments. R.C. performed the scRNA-seq experiments. T.R.B. and J.H. performed the MERFISH experiments. M.N.D analyzed the scRNA-seq data. T.R.B. and M.N.D. analyzed the MERFISH data. A.B. and R.C. performed the RNAscope experiments. W.C. and L.M.T. helped with preparing scRNA-seq samples. R.C, T.R.B., Y.Z. and X.Z. interpreted the data. R.C., T.R.B., M.N.D, Y.Z. and X.Z. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
X.Z. is a co-founder and consultant of Vizgen. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Charles Gerfen, Arpiar Saunders, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Identification of the transcriptionally distinct cell types in mouse NAc.
a, Schematic diagram showing the nucleus accumbens region used for single-cell RNA sequencing. Adult mouse brain was first cut into 0.5mm-thick serial coronal sections and then NAc tissues (shown in purple contours) were dissected from successive slices along the rostral-caudal axis. The brain pictures were taken from Allen Mouse Brain Atlas. b, Bargraphs showing the distribution of UMI and gene number detected in each cell across the 11 samples. Different samples are color-coded. The black central line is the median, the box limits indicate the upper and lower quartiles, the whiskers indicate the 1.5 interquartile range and dots represent outliers. c, Histograms showing the percentage of cells from each sample that contribute to the major cell clusters. Different samples are color-coded. d, Bargraphs showing the distribution of UMI and gene number detected in each cell across the 9 major cell clusters. Different cell clusters are color-coded. The black central line is the median, the box limits indicate the upper and lower quartiles, the whiskers indicate the 1.5 interquartile range and dots represent outliers. Astro, astrocyte; D1, medial spiny neuron, D1-receoptor subtype; D2, medial spiny neuron, D2-receptor subtype; Endo, endothelial cell; IN, interneuron; Micro, microglia; NB, neural stem cells and neuroblast; Oligo, oligodendrocyte; OPC, oligodendrocyte progenitor cell. e, ROC curves showing the high accuracy of the cell identity predictor, especially for non-neuronal cells. The curves of different cell types are represented by different colors. f, Heatmap showing the results of predicted identity of cells with 800 to 1,500 genes detected. The prediction probability is color-coded. g, In situ hybridization of the cell-cycle gene, Top2a, showing the distribution of neural stem cells and neuroblasts in the ventral wall of the lateral ventricle. The purple contour indicates the NAc, and the boxed region in the left panel is enlarged and shown on the right. The ISH data was obtained from Allen Mouse Brain Atlas. Scale bar, 100 µm.
Extended Data Fig. 2 Gene expression and spatial pattern of NAc interneuron subtypes.
a, Hierarchical relationship of the 13 NAc interneuron subtypes identified from scRNA-seq. The dendrogram indicate the relatedness among interneuron subtypes based on their gene expression. The markers and the number of cells of each subtypes are shown. b, Heatmap showing the expression pattern of interneuron markers across different subtypes. Each column represents a single cell. The gene expression level is color coded. The 13 interneuron subtypes are indicated by different colors on top. c, ISH showing the expression of selected interneuron subtype markers identified by scRNA-seq in NAc. The regions around the anterior commissure (AC) were shown except for Pvalb, which was more enriched in lateral part of NAc. The images were obtained from the Allen Mouse Brain Atlas. d, tSNE plots showing the expression of interneuron subtype markers across NAc interneuron subtypes. The gene expression level is color-coded. e, FISH showing the overlap of selected interneuron markers in NAc. Arrow heads indicate the cells co-express the two genes. Arrows indicate cells expressing one marker gene. Three independent experiments were performed with similar results. Scale bar, 50 µm.
Extended Data Fig. 3 Molecularly defined MSN subtypes exhibit distinct spatial distribution in NAc.
a, Histograms showing the percentage of cells from each of the 11 samples across D1 and D2 MSN subtypes. Different samples are represented in different colors. b, tSNE plots showing the expression pattern of Lrpprc, Foxp2, Foxp1 and Isl1 across MSN populations. The expression level is color-coded. c, ISH of Tshz1 showing the distribution of Tshz1+ cells in NAc. Sagittal section of mouse brain including NAc is shown. The boxed region is enlarged and shown on the right. Data are obtained from Allen Mouse Brain Atlas. Scale bars, 500 µm. d, e, Ddit4l (d) and Trhr (e) are enriched in subpopulation of MSNs. Left panel, tSNE plots showing the expression pattern of Ddit4l (d) and Trhr (e) across MSN populations. The expression level is color-coded. The MSN subtypes labeled by Ddit4l or Trhr were indicated. Right panels, ISH image showing the distribution of Ddit4l+ (d) and Trhr+ (e) cells in NAc. The boxed regions are enlarged and shown on the right. Data is obtained from Allen Mouse Brain Atlas. Scale bar, 500 µm. f, Heatmaps showing the gene expression correlation of differentially expresses genes in D1 MSNs (upper panel) and D2 MSNs (lower panel). The correlation is color-coded, and genes were sorted into groups with higher intra-group correlation. g, h, Violin plots showing the expression of selected markers across high-resolution D1 (g) and D2 (h) MSN subtypes. Different MSN subtypes are color-coded. The mRNA level is presented on a log scale and adjusted for different genes.
Extended Data Fig. 4 MERFISH revealed major cell types in striatum.
a, Schematic diagram showing the striatal slices used for MERFISH. Adult mouse brain was first cut into 10 µm -thick serial coronal sections. Twelve brain slices with 100 μm interval between adjacent slices were used for MERFISH experiment. The brain pictures were taken from Allen Mouse Brain Atlas, note that only 6 slices were shown. b, One example image showing the maximum projection of images taken in one representative field-of-view (FOV) during MERFISH. The boxed region was enlarged and shown on the right. Individual RNA molecules were detected as single dots. c, DAPI (left) and poly(A) RNA (right) images were used to define the boundaries of each cell in white. The mRNA molecules detected were assigned to different cells based on the cell boundaries. d, Scatterplot showing the average counts of each genes per cell detected by MERFISH in the two biological replicates. e, Scatterplot showing the average copy number of each genes per cell detected by MERFISH and bulk RNA-seq. The Pearson correlation coefficient is 0.72. f, Heatmap showing the expression pattern of selected genes in striatum as determined by MERFISH (upper panels), which are highly similar to the patterns determined by conventional ISH (lower panels). The ISH data are obtained from Allen Mouse Brain Atlas. Scale bars, 500 µm. g, the Harmony algorithm69 based UMAP plots showing the cells from the two replicates of MERFISH experiments with good overlap. h, Bar graph showing the proportion of major cell types from the two batches of MERFISH experiments after the Harmony integration. i, Spatial pattern of interneuron, astrocyte, microglia, endothelial cell and OPC in coronal brain sections at different anterior-posterior positions. Three of the twelve slices from a male mouse were shown. Colored dots were cells belong to the specified cell populations, while gray dots indicate all other cells. The 200, 500 and 800 μm labels indicate the distance from the anterior position (Bregma 1.94 mm). The dorsal-ventral (DV) and medial-lateral (ML) axes are indicated.
Extended Data Fig. 5 MERFISH identifies molecularly distinct D1 and D2 MSN subtypes in striatum.
a, b, The Harmony algorithm69 based UMAP plots showing the D1 MSNs (a) and D2 MSNs (b) from the two replicates of MERFISH experiments. c, Bar graph showing the proportion of different D1 and D2 MSN subtypes from the two replicates of MERFISH experiments. d, e, Heatmaps showing the expression pattern of differentially expressed genes detected by MERFISH across D1 (d) and D2 (e) MSN subtypes. The expression level is color coded. The width of each column represents the abundance of each MSN subtype. f, Integrative analysis (Using the Harmony algorithm) of D2 MSNs from MERFISH and scRNA-seq experiments. The D2 MSNs from MERFISH and scRNA-seq experiments were integrated into the same UMAP space. The initial identity of each cell was color coded and shown in the left and middle panels. Based on the nearest neighbors from the MERFISH experiments, the cells from scRNA-seq were assigned to one of the MERFISH D2 MSN subtypes shown on the right panel. g, Pie charts showing the percentage of D1 (upper) and D2 (lower) MSNs from scRNA-seq experiments which could or could not be assigned to a certain MEFFISH identify due to insufficient MERFISH k-NN belonging to the same cluster. h, The Harmony algorithm based UMAP showing the D1 MSNs (upper panels) and D2 MSNs (lower panels) from scRNA-seq and MERFISH experiments. The cells from different experiments were integrated into the same UMAP spaces. The arrows indicated UMAP spaces that are mainly occupied by cells from non-NAc region, thus the scRNA-seq cells were depleted comparing to MERFISH cells. i, Dotplots showing the expression of selected MSN subtype markers in D1 (left) and D2 (ringht) MSN subtypes defined by MERFISH and scRNA-seq. The expression level is color-coded (MERFISH and scRNA-seq data are normalized and scaled separately). Dot size represents the fraction of cells expressing the gene in each subtype. Corresponding MERFISH and scRNA-seq clusters are grouped together. The names and marker genes of MSN subtypes identified by MERFISH but without corresponding scRNA-seq subtypes are marked in blue.
Extended Data Fig. 6 Molecular and spatial features of MSN subtypes underlie anatomic organization of striatum.
a, Spatial patterns of different MSNs groups in striatal region. The MSN subtype groups are the same as Fig. 6e. Different MSN subtypes are color coded. The D1 and D2 MSNs are shown in upper and lower panels, respectively. b, Heatmap showing the patterns of differentially expressed genes among D1 (left panel) and D2 (right panel) MSN subtype groups located in major striatal divisions. The MSN groups corresponding to different anatomic regions are the same as Fig. 6e, but the two NAc groups are combined. Reln, Nnat and Rasgrf2 are enriched in DS, NAc and OT, respectively, and the patterns are shared in D1 and D2 MSN subtypes. c, The spatial heatmaps showing the expression pattern of Nrgn, Reln, Wfs1, Crym, Rasgrf2, Rprm in coronal sections, which are enriched in different striatal divisions. The expression level is color coded. d, Spatial and gene expression features of D1 MSN subtypes representing striosome (D1_DS2) and matrix (D1_DS1, D1_DS3 and D1_DS4) structure in DS. The upper and lower panels show D1 subtypes representing striosome and matrix, respectively. The left panels show spatial pattern of D1 subtypes corresponding to striosome and matrix. The middle and right panels are heatmaps showing the expression of striosome enriched gene Oprm1 and matrix enriched gene Calb1 in these D1 MSN subtypes. e, Dotplot showing the expression pattern of selected genes in D1 and D2 MSN subtypes representing striosome and matrix. D1 Matrix: D1_DS1, D1_DS3 and D1_DS4; D1 Striosome: D1_DS4; D2 Matrix: D2_DS1 and D2_DS3; D2 Striosome: D2_DS2. The expression level is color coded. Dot size represents the fraction of cells in the subtype. f, Heatmap showing the expression of selected marker genes that distinguish atypical D1 and D2 MSN subtypes from other D1 and D2 MSN subtypes. The expression level is color coded. g, Heatmap showing the pattern of differentially expressed genes among the three atypical D1 MSN subtypes. Selected marker genes enriched in different atypical D1 MSN subtypes are labeled.
Extended Data Fig. 7 Molecular and spatial features of MSN subtypes underlie anatomic heterogeneity of NAc.
a, Spatial patterns of selected D2 MSN subtypes in coronal sections at different anterior-posterior positions. The subtypes enriched in anterior and posterior NAc are shown in upper and lower panels, respectively. Different subtypes are represented by different colors. The 100, 300, 500, 700, 900 and 1100 μm labels indicate the distance from the anterior position (Bregma 1.94 mm). The dorsal-ventral (DV) and medial-lateral (ML) axes are indicated. b, The distribution of the three D2 MSN subtypes shown in (a) along the AP axis. c, The spatial pattern of MSN subtypes enriched in NAc core (left panel) or shell (right panel). Different subtypes are indicated with different colors. The core region is indicated by dashed line. d, Heatmap showing the expression of core enriched gene Calb1 and shell enriched genes Gucy1a3 in coronal brain sections. The expression level was color coded and the core region was indicated with dashed line. e, Density maps showing the enrichment of different D1 and D2 MSN subtypes in the dorsolateral and ventromedial part of NAc core. The same region as Fig. 7f are shown. The NAc core and AC structure are labeled with dashed lines. The dorsal-ventral (DV) and medial-lateral (ML) axes are indicated. f, Heatmaps showing the differentially expressed genes between D1 (upper panel) and D2 (lower panel) MSN subtypes enriched in different subregions of NAc core. The expression level is color coded. g, Heatmap showing the differential expression of Rgs9, Ppp3ca, Peg10 and Dlk1 between the dorsolateral and ventromedial part of NAc core. The same region as Fig. 7f is presented. The NAc core and AC structure are labeled with dashed lines. The gene expression level is color coded. h, MERFISH and ISH detection of Drd2 expression in striatum. The dorsal part of NAc lateral shell with low Drd2 expression is labeled with dashed line. The boxed region in the ISH image is enlarged and shown on the right. The ISH data is from Allen Brain Atlas. i, Density maps showing the enrichment of different groups of MSN subtypes in distinct subregions in NAc medial shell.
Supplementary information
Supplementary Tables
List of Supplementary Tables. Table S1: Summary of the scRNA-seq experiments. Table S2: Major cell populations identified by scRNA-seq. Table S3: Markers of scRNA-seq D1 and D2 MSN subtypes. Table S4: List of MERFISH target genes. Table S5: Markers of MERFISH D1 and D2 MSN subtypes. Table S6: DEGs in major striatal subregions. Table S7: DEGs in different atypical MSN subtypes. Table S8: DEGs among NAc core subregions.
Rights and permissions
About this article
Cite this article
Chen, R., Blosser, T.R., Djekidel, M.N. et al. Decoding molecular and cellular heterogeneity of mouse nucleus accumbens. Nat Neurosci 24, 1757–1771 (2021). https://doi.org/10.1038/s41593-021-00938-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-021-00938-x
This article is cited by
-
Gene panel selection for targeted spatial transcriptomics
Genome Biology (2024)
-
MENDER: fast and scalable tissue structure identification in spatial omics data
Nature Communications (2024)
-
Updating the striatal–pallidal wiring diagram
Nature Neuroscience (2024)
-
Spatial transcriptomics reveal neuron–astrocyte synergy in long-term memory
Nature (2024)
-
A spatially-resolved transcriptional atlas of the murine dorsal pons at single-cell resolution
Nature Communications (2024)