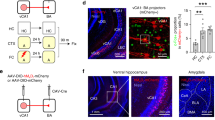
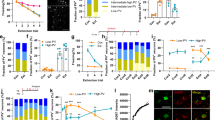
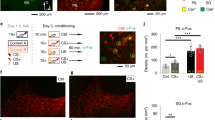
Abstract
Experience-driven synaptic plasticity in the lateral amygdala is thought to underlie the formation of associations between sensory stimuli and an ensuing threat. However, how the central amygdala participates in such a learning process remains unclear. Here we show that PKC-δ-expressing central amygdala neurons are essential for the synaptic plasticity underlying learning in the lateral amygdala, as they convey information about the unconditioned stimulus to lateral amygdala neurons during fear conditioning.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Herry, C. & Johansen, J. P. Encoding of fear learning and memory in distributed neuronal circuits. Nat. Neurosci. 17, 1644–1654 (2014).
Li, H. et al. Experience-dependent modification of a central amygdala fear circuit. Nat. Neurosci. 16, 332–339 (2013).
Penzo, M. A., Robert, V. & Li, B. Fear conditioning potentiates synaptic transmission onto long-range projection neurons in the lateral subdivision of central amygdala. J. Neurosci. 34, 2432–2437 (2014).
Penzo, M. A. et al. The paraventricular thalamus controls a central amygdala fear circuit. Nature 519, 455–459 (2015).
Goosens, K. A. & Maren, S. Pretraining NMDA receptor blockade in the basolateral complex, but not the central nucleus, of the amygdala prevents savings of conditional fear. Behav. Neurosci. 117, 738–750 (2003).
Wilensky, A. E., Schafe, G. E., Kristensen, M. P. & LeDoux, J. E. Rethinking the fear circuit: the central nucleus of the amygdala is required for the acquisition, consolidation, and expression of Pavlovian fear conditioning. J. Neurosci. 26, 12387–12396 (2006).
Yu, K., Garcia da Silva, P., Albeanu, D. F. & Li, B. Central amygdala somatostatin neurons gate passive and active defensive behaviors. J. Neurosci. 36, 6488–6496 (2016).
Ciocchi, S. et al. Encoding of conditioned fear in central amygdala inhibitory circuits. Nature 468, 277–282 (2010).
Roesch, M. R., Esber, G. R., Li, J., Daw, N. D. & Schoenbaum, G. Surprise! Neural correlates of Pearce-Hall and Rescorla-Wagner coexist within the brain. Eur. J. Neurosci 35, 1190–1200 (2012).
Balleine, B. W. & Killcross, S. Parallel incentive processing: an integrated view of amygdala function. Trends Neurosci. 29, 272–279 (2006).
Nabavi, S. et al. Engineering a memory with LTD and LTP. Nature 511, 348–352 (2014).
Haubensak, W. et al. Genetic dissection of an amygdala microcircuit that gates conditioned fear. Nature 468, 270–276 (2010).
Han, S., Soleiman, M. T., Soden, M. E., Zweifel, L. S. & Palmiter, R. D. Elucidating an affective pain circuit that creates a threat memory. Cell 162, 363–374 (2015).
Sato, M. et al. The lateral parabrachial nucleus is actively involved in the acquisition of fear memory in mice. Mol. Brain 8, 22 (2015).
Chen, T. W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Fenno, L. E. et al. Targeting cells with single vectors using multiple-feature Boolean logic. Nat. Methods 11, 763–772 (2014).
Resendez, S. L. et al. Visualization of cortical, subcortical and deep brain neural circuit dynamics during naturalistic mammalian behavior with head-mounted microscopes and chronically implanted lenses. Nat. Protoc. 11, 566–597 (2016).
Johansen, J. P., Tarpley, J. W., LeDoux, J. E. & Blair, H. T. Neural substrates for expectation-modulated fear learning in the amygdala and periaqueductal gray. Nat. Neurosci. 13, 979–986 (2010).
McNally, G. P., Johansen, J. P. & Blair, H. T. Placing prediction into the fear circuit. Trends Neurosci. 34, 283–292 (2011).
Belova, M. A., Paton, J. J., Morrison, S. E. & Salzman, C. D. Expectation modulates neural responses to pleasant and aversive stimuli in primate amygdala. Neuron 55, 970–984 (2007).
Taniguchi, H. et al. A resource of Cre driver lines for genetic targeting of GABAergic neurons in cerebral cortex. Neuron 71, 995–1013 (2011).
Madisen, L. et al. A toolbox of Cre-dependent optogenetic transgenic mice for light-induced activation and silencing. Nat. Neurosci. 15, 793–802 (2012).
He, M. et al. Cell-type-based analysis of microRNA profiles in the mouse brain. Neuron 73, 35–48 (2012).
Murray, A. J. et al. Parvalbumin-positive CA1 interneurons are required for spatial working but not for reference memory. Nat. Neurosci. 14, 297–299 (2011).
Zeng, W. B. et al. Anterograde monosynaptic transneuronal tracers derived from herpes simplex virus 1 strain H129. Mol. Neurodegener 12, 38 (2017).
Bru, T., Salinas, S. & Kremer, E. J. An update on canine adenovirus type 2 and its vectors. Viruses 2, 2134–2153 (2010).
Stephenson-Jones, M. et al. A basal ganglia circuit for evaluating action outcomes. Nature 539, 289–293 (2016).
Beier, K. T. et al. Circuit architecture of VTA dopamine neurons revealed by systematic input-output mapping. Cell 162, 622–634 (2015).
Clem, R. L. & Huganir, R. L. Calcium-permeable AMPA receptor dynamics mediate fear memory erasure. Science 330, 1108–1112 (2010).
Zhang, F., Wang, L. P., Boyden, E. S. & Deisseroth, K. Channelrhodopsin-2 and optical control of excitable cells. Nat. Methods 3, 785–792 (2006).
Chow, B. Y. et al. High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature 463, 98–102 (2010).
Vardy, E. et al. A new DREADD facilitates the multiplexed chemogenetic interrogation of behavior. Neuron 86, 936–946 (2015).
Zhou, P. et al. Efficient and accurate extraction of in vivo calcium signals from microendoscopic video data. Preprint at https://arxiv.org/abs/1605.07266 (2016).
Pnevmatikakis, E. A. et al. Simultaneous denoising, deconvolution, and demixing of calcium imaging data. Neuron 89, 285–299 (2016).
Ragan, T. et al. Serial two-photon tomography for automated ex vivo mouse brain imaging. Nat. Methods 9, 255–258 (2012).
Kim, Y. et al. Whole-brain mapping of neuronal activity in the learned helplessness model of depression. Front. Neural Circuits 10, 3 (2016).
Acknowledgements
We thank G.D. Stuber for helping with the in vivo imaging experiments. We thank G.D. Stuber and J. Johansen for critically reading an earlier version of the manuscript, G.-R. Hwang for technical assistance and members of the Li laboratory for helpful discussions. This work was supported by grants from the National Institutes of Health (NIH) (R01MH101214, B.L.), Human Frontier Science Program (RGP0015/2016, B.L.), NARSAD (23169, B.L., and 21227, S.A.), National Natural Science Foundation of China (81428010, B.L. and M.H., and 91432106, M.H.), China Postdoctoral Science Foundation (2016M590316, L.G.), Louis Feil Trust (B.L.), the Stanley Family Foundation (B.L.), Simons Foundation (344904, B.L.) and Wodecroft Foundation (to B.L.).
Author information
Authors and Affiliations
Contributions
K.Y. and B.L. conceived and designed the study. K.Y., S.A., X.Z. and H.S. conducted experiments (X.Z. performed the experiments in which LA neurons were imaged; S.A. and H.S. performed the experiments in which synaptic plasticity in LA neurons was examined; K.Y. conducted most of the remaining experiments). K.Y., X.Z., S.A. and H.S. analyzed data. C.R., L.F. and K.D. developed the intersectional viral strategy and provided critical reagents. F.Z. and M.-H.L. developed the H129-G4 viral system and performed the anterograde tracing with it. L.G. and M.H. performed imaging with the STPT. P.Z. and L.P. developed and assisted with the imaging analysis methods (CNMF and CNMF-E). B.L. wrote the paper with inputs from all authors.
Corresponding author
Ethics declarations
Competing financial interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–18
Supplementary Video 1
Extraction of the spatial and temporal components of neuronal activity from GCaMP6 signals in PKC-(+ CeL neurons imaged through GRIN lenses.Shown are videos of GCaMP6 signals from PKC-(+ CeL neurons in a representative mouse undergoing fear conditioning. These videos are short clips of movies stitched together, with each clip representing a trial (the onset of which is denoted by each of the solid black lines in the bottom right panel). These videos are played in synchrony, and represent the following contents: “raw data”, GCaMP6 signals without any processing; “background”, the background components in the raw data approximated with our model; “raw–background”, the remaining signals after subtraction of the background from the raw data; “denoised”, the denoised and deconvolved spatiotemporal activity for each neuron obtained from the “raw–background” signals using the CNMF-E; “residual”, the remaining signals. In the “denoised” video, the contours of 5 representative neurons are traced and numbered. The temporal activities of these 5 neurons are displayed in the bottom right panel, in which the moving line indicates the passage of time synchronized with all the videos; the stationary solid black lines denote the onsets of trials (and thus the junctions between the short movie clips); the dashed blue lines denote the onsets of CS; and the dashed red lines denote the onsets of US. Neurons #1-3 are shock-responsive neurons, whereas neurons #4 & 5 only show spontaneous activities. Of note, these denoised and deconvolved signals from adjacent neurons (e.g., #2 and #4) are easily separable. The scale bar beside each of the videos denotes (F values. See Methods for a more detailed description
Supplementary Video 2
Extraction of the spatial and temporal components of neuronal activity from GCaMP6 signals in LA neurons imaged through GRIN lenses. Description: Shown are videos of GCaMP6 signals from LA neurons in a representative mouse receiving tail shocks. These videos are short clips of movies stitched together, with each clip representing a trial (the onset of which is denoted by each of the solid black lines in the bottom right panel). These videos are played in synchrony, and represent the following contents: “raw data”, GCaMP6 signals without any processing; “background”, the background components in the raw data approximated with our model; “raw–background”, the remaining signals after subtraction of the background from the raw data; “denoised”, the denoised and deconvolved spatiotemporal activity for each neuron obtained from the “raw–background” signals using the CNMF-E; “residual”, the remaining signals. In the “denoised” video, the contours of 3 representative neurons are traced and numbered. The temporal activities of these 3 neurons are displayed in the bottom right panel, in which the moving line indicates the passage of time synchronized with all the videos; the stationary solid black lines denote the onsets of trials (and thus the junctions between the short movie clips); and the dashed red lines denote the onsets of shocks. Neurons #1 & 2 are shock-responsive neurons, whereas neurons #3 only shows spontaneous activities. Of note, these denoised and deconvolved signals from adjacent neurons are easily separable. For example, the signals from neuron #1 & 2 are not contaminated by signals from the adjacent neurons, which have high levels of spontaneous activities. The scale bar beside each of the videos denotes (F values. See Methods for a more detailed description.Shown are videos of GCaMP6 signals from LA neurons in a representative mouse receiving tail shocks. These videos are short clips of movies stitched together, with each clip representing a trial (the onset of which is denoted by each of the solid black lines in the bottom right panel). These videos are played in synchrony, and represent the following contents: “raw data”, GCaMP6 signals without any processing; “background”, the background components in the raw data approximated with our model; “raw–background”, the remaining signals after subtraction of the background from the raw data; “denoised”, the denoised and deconvolved spatiotemporal activity for each neuron obtained from the “raw–background” signals using the CNMF-E; “residual”, the remaining signals. In the “denoised” video, the contours of 3 representative neurons are traced and numbered. The temporal activities of these 3 neurons are displayed in the bottom right panel, in which the moving line indicates the passage of time synchronized with all the videos; the stationary solid black lines denote the onsets of trials (and thus the junctions between the short movie clips); and the dashed red lines denote the onsets of shocks. Neurons #1 & 2 are shock-responsive neurons, whereas neurons #3 only shows spontaneous activities. Of note, these denoised and deconvolved signals from adjacent neurons are easily separable. For example, the signals from neuron #1 & 2 are not contaminated by signals from the adjacent neurons, which have high levels of spontaneous activities. The scale bar beside each of the videos denotes (F values. See Methods for a more detailed description
Rights and permissions
About this article
Cite this article
Yu, K., Ahrens, S., Zhang, X. et al. The central amygdala controls learning in the lateral amygdala. Nat Neurosci 20, 1680–1685 (2017). https://doi.org/10.1038/s41593-017-0009-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-017-0009-9
This article is cited by
-
Social buffering in rats reduces fear by oxytocin triggering sustained changes in central amygdala neuronal activity
Nature Communications (2024)
-
Plastic and stimulus-specific coding of salient events in the central amygdala
Nature (2023)
-
Topographic representation of current and future threats in the mouse nociceptive amygdala
Nature Communications (2023)
-
Insular cortical circuits as an executive gateway to decipher threat or extinction memory via distinct subcortical pathways
Nature Communications (2022)
-
A fluorescent sensor for spatiotemporally resolved imaging of endocannabinoid dynamics in vivo
Nature Biotechnology (2022)