Abstract
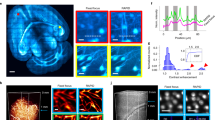
The microscopic visualization of large-scale three-dimensional (3D) samples by optical microscopy requires overcoming challenges in imaging quality and speed and in big data acquisition and management. We report a line-illumination modulation (LiMo) technique for imaging thick tissues with high throughput and low background. Combining LiMo with thin tissue sectioning, we further develop a high-definition fluorescent micro-optical sectioning tomography (HD-fMOST) method that features an average signal-to-noise ratio of 110, leading to substantial improvement in neuronal morphology reconstruction. We achieve a >30-fold lossless data compression at a voxel resolution of 0.32 × 0.32 × 1.00 μm3, enabling online data storage to a USB drive or in the cloud, and high-precision (95% accuracy) brain-wide 3D cell counting in real time. These results highlight the potential of HD-fMOST to facilitate large-scale acquisition and analysis of whole-brain high-resolution datasets.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The imaging data reported in the paper are available at http://atlas.brainsmatics.org/a/zhong2019. Source data are provided with this paper.
Code availability
The code for data acquisition in this paper is available at http://atlas.brainsmatics.org/a/zhong2019.
References
Oron, D., Tal, E. & Silberberg, Y. Scanningless depth-resolved microscopy. Opt. Express 13, 1468–1476 (2005).
Dana, H. & Shoham, S. Numerical evaluation of temporal focusing characteristics in transparent and scattering media. Opt. Express 19, 4937–4948 (2011).
Neil, M. A. A., Juskaitis, R. & Wilson, T. Method of obtaining optical sectioning by using structured light in a conventional microscope. Opt. Lett. 22, 1905–1907 (1997).
Lim, D., Chu, K. K. & Mertz, J. Wide-field fluorescence sectioning with hybrid speckle and uniform-illumination microscopy. Opt. Lett. 33, 1819–1821 (2008).
Ford, T. N., Lim, D. & Mertz, J. Fast optically sectioned fluorescence HiLo endomicroscopy. J. Biomed. Opt. 17, 021105 (2012).
Abeytunge, S., Li, Y., Larson, B., Toledo-Crow, R. & Rajadhyaksha, M. Rapid confocal imaging of large areas of excised tissue with strip mosaicing. J. Biomed. Opt. 16, 050504–050504 (2011).
Seiriki, K. et al. High-speed and scalable whole-brain imaging in rodents and primates. Neuron 94, 1085–1100 (2017).
Gong, H. et al. High-throughput dual-colour precision imaging for brain-wide connectome with cytoarchitectonic landmarks at the cellular level. Nat. Commun. 7, 12142 (2016).
Economo, M. N. et al. A platform for brain-wide imaging and reconstruction of individual neurons. eLife 5, e10566 (2016).
Gong, H. et al. Continuously tracing brain-wide long-distance axonal projections in mice at a one-micron voxel resolution. Neuroimage 74, 87–98 (2013).
Li, A. et al. Micro-optical sectioning tomography to obtain a high-resolution atlas of the mouse brain. Science 330, 1404–1408 (2010).
Sun, Q. et al. A whole-brain map of long-range inputs to GABAergic interneurons in the mouse medial prefrontal cortex. Nat. Neurosci. 22, 1357–1370 (2019).
Parekh, R. & Ascoli, G. A. Neuronal morphology goes digital: a research hub for cellular and system neuroscience. Neuron 77, 1017–1038 (2013).
Meijering, E., Carpenter, A. E., Peng, H., Hamprecht, F. A. & Olivo-Marin, J.-C. Imagining the future of bioimage analysis. Nat. Biotechnol. 34, 1250–1255 (2016).
Jiang, T. et al. A platform for efficient identification of molecular phenotypes of brain-wide neural circuits. Sci. Rep. 7, 13891 (2017).
Sun, P. et al. Highly efficient and super-bright neurocircuit tracing using vector mixing-based virus cocktail. Preprint at bioRxiv https://doi.org/10.1101/705772 (2020).
Kita, T. & Kita, H. The subthalamic nucleus is one of multiple innervation sites for long-range corticofugal axons: a single-axon tracing study in the rat. J. Neurosci. 32, 5990–5999 (2012).
Winnubst, J. et al. Reconstruction of 1,000 projection neurons reveals new cell types and organization of long-range connectivity in the mouse brain. Cell 179, 268–281.e213 (2019).
Zhou, H. et al. GTree: an open-source tool for dense reconstruction of brain-wide neuronal population. Neuroinform https://doi.org/10.1007/s12021-020-09484-6 (2020).
Do, J. P. et al. Cell type-specific long-range connections of basal forebrain circuit. eLife 5, e13214 (2016).
Yang, Z., Hu, B., Zhang, Y., Luo, Q. & Gong, H. Development of a plastic embedding method for large-volume and fluorescent-protein-expressing tissues. PLoS ONE 8, e60877 (2013).
Stokseth, P. A. Properties of a defocused optical system. J. Opt. Soc. Am. 59, 1314–1321 (1969).
Quan, T. et al. NeuroGPS: automated localization of neurons for brain circuits using L1 minimization model. Sci. Rep. 3, 1414 (2013).
Dong, H. W. The Allen Reference Atlas: A Digital Color Brain Atlas of the C57BL/6J Male Mouse (John Wiley & Sons, 2008).
Avants, B. B. et al. A reproducible evaluation of ANTs similarity metric performance in brain image registration. Neuroimage 54, 2033–2044 (2011).
Li, Y. et al. TDat: an efficient platform for processing petabyte-scale whole-brain volumetric images. Front. Neural Circuits 11, 51 (2017).
Ni, H. et al. A robust image registration interface for large volume brain atlas. Sci. Rep. 10, 2139 (2020).
Acknowledgements
We thank the MOST group members of the Britton Chance Center for Biomedical Photonics for assistance with experiments and comments on the manuscript. We are grateful to X. Yang and J. Chen for experimental help, T. Quan, M. Liao, K. Ning, N. Wang, J. Zhou, S. Bao, M. Tian and B. Lu for morphology reconstruction and analysis, and H. Li, H. Dong, Z. J. Huang, M. Luo, T. Xu, Y. Han, X. Chen and Y. Sun for constructive comments. This work was supported by National Key Research and Development Program of China (grant no. 2017YFA0700402 to J.Y.), Science Fund for Creative Research Group of China (grant no. 61721092 to Q.L.), National Natural Science Foundation of China (grant nos. 81827901 to Q.L. and A.L., 91749209 to Q.L., 61890950 to H.G., J.Y. and A.L., 61890953 to H.G., J.Y. and X.L., 61890954 to A.L.), and Fundamental Research Funds for the Central Universities (HUST: grant nos. 2019kfyXMBZ011, 2019kfyXMBZ039, 2018KFYXMPT018, 2018KFYXKJC030 to Q.L., H.G. and Z.Z.). We also thank Analytical and Testing Center (HUST) and Optical Bioimaging Core Facility of WNLO-HUST for the support in data acquisition, and the director fund of the WNLO.
Author information
Authors and Affiliations
Contributions
J.Y., Q.L., Z.Q. and H.G. conceived and designed the study and wrote the manuscript. Q.Z., R.J., D.Z., C.J., Z.D. and J.Y. constructed the microscopes and performed data acquisition. X.L., P.L., C.Z., S.J., Z.Z. and H.G. designed the tissue preparation and built the virus tracing samples. J.Y., A.L., Q.Z., X.J., Z.F., H.G. and Q.L. performed image processing and visualization and analyzed the data. J.Y., Q.Z., A.L., H.G. and Q.L. modified the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Q.L., J.Y., Q.Z., R.J. and H.G. have filed patent applications based on this work.
Additional information
Peer review information Nature Methods thanks Lamiae Abdeladim and the other, anonymous, reviewer for their contribution to the peer review of this work. Nina Vogt was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1 and 2, Figs. 1–20 and Tables 1–7.
Supplementary Video 1
Background inhibition using LiMo, point-confocal, 2PM and line-confocal methods.
Supplementary Video 2
Whole-brain MIP of the sparsely labeled mouse brain.
Supplementary Video 3
3D rendering of the injection site of the same dataset with Supplementary Video 2.
Supplementary Video 4
3D rendering of the reconstructed neurons.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Rights and permissions
About this article
Cite this article
Zhong, Q., Li, A., Jin, R. et al. High-definition imaging using line-illumination modulation microscopy. Nat Methods 18, 309–315 (2021). https://doi.org/10.1038/s41592-021-01074-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-021-01074-x
This article is cited by
-
Multimodal Fusion of Brain Imaging Data: Methods and Applications
Machine Intelligence Research (2024)
-
TSA-PACT: a method for tissue clearing and immunofluorescence staining on zebrafish brain with improved sensitivity, specificity and stability
Cell & Bioscience (2023)
-
Upper brainstem cholinergic neurons project to ascending and descending circuits
BMC Biology (2023)
-
D-LMBmap: a fully automated deep-learning pipeline for whole-brain profiling of neural circuitry
Nature Methods (2023)
-
Three-dimensional mapping in multi-samples with large-scale imaging and multiplexed post staining
Communications Biology (2023)