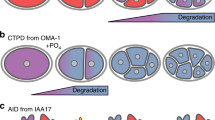
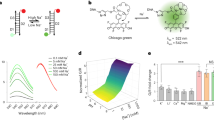
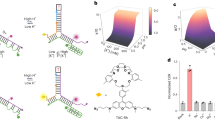
Abstract
It is extremely challenging to quantitate lumenal Ca2+ in acidic Ca2+ stores of the cell because all Ca2+ indicators are pH sensitive, and Ca2+ transport is coupled to pH in acidic organelles. We have developed a fluorescent DNA-based reporter, CalipHluor, that is targetable to specific organelles. By ratiometrically reporting lumenal pH and Ca2+ simultaneously, CalipHluor functions as a pH-correctable Ca2+ reporter. By targeting CalipHluor to the endolysosomal pathway, we mapped lumenal Ca2+ changes during endosomal maturation and found a surge in lumenal Ca2+ specifically in lysosomes. Using lysosomal proteomics and genetic analysis, we found that catp-6, a Caenorhabditis elegans homolog of ATP13A2, was responsible for lysosomal Ca2+ accumulation—an example of a lysosome-specific Ca2+ importer in animals. By enabling the facile quantification of compartmentalized Ca2+, CalipHluor can expand the understanding of subcellular Ca2+ importers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and the findings of this study are available from the corresponding author upon reasonable request.
Change history
14 January 2019
The originally published paper has been updated to include the following new reference, added as ref. 18: Albrecht, T., Zhao, Y., Nguyen, T. H., Campbell, R. E. & Johnson, J. D. Fluorescent biosensors illuminate calcium levels within defined beta-cell endosome subpopulations. Cell Calcium 57, 263–274 (2015). Subsequent references have been renumbered in the reference list and throughout the text. Minor text changes were made in the sentence in which this new reference is first cited: “Previous attempts used endocytic tracers bearing either pH- or Ca2+-sensitive dyes to serially measure population-averaged pH and apparent Ca2+ in different batches of cells, thus scrambling information from individual endosomes13–17” in the original introduction was changed to “Previous attempts used endocytic tracers bearing either pH- or Ca2+-sensitive dyes13–17 or fluorescent-protein-based sensors18 to serially measure population-averaged pH and apparent Ca2+ in different batches of cells, thus scrambling information from individual endosomes.” These changes have been made in the HTML and PDF versions of the article.
References
Clapham, D. E. Calcium signaling. Cell 131, 1047–1058 (2007).
Bagur, R. & Hajnóczky, G. Intracellular Ca2+ sensing: its role in calcium homeostasis and signaling. Mol. Cell 66, 780–788 (2017).
Yang, J., Zhao, Z., Gu, M., Feng, X. & Xu, H. Release and uptake mechanisms of vesicular Ca2+ stores. Protein Cell https://doi.org/10.1007/s13238-018-0523-x (2018).
Parenti, G., Andria, G. & Ballabio, A. Lysosomal storage diseases: from pathophysiology to therapy. Annu. Rev. Med. 66, 471–486 (2015).
Plotegher, N. & Duchen, M. R. Crosstalk between lysosomes and mitochondria in Parkinson’s disease. Front. Cell Dev. Biol. 5, 110 (2017).
Xu, H., Martinoia, E. & Szabo, I. Organellar channels and transporters. Cell Calcium 58, 1–10 (2015).
Calcraft, P. J. et al. NAADP mobilizes calcium from acidic organelles through two-pore channels. Nature 459, 596–600 (2009).
Huang, P. et al. P2X4 forms functional ATP-activated cation channels on lysosomal membranes regulated by luminal pH. J. Biol. Chem. 289, 17658–17667 (2014).
Kiselyov, K. et al. TRPML: transporters of metals in lysosomes essential for cell survival? Cell Calcium 50, 288–294 (2011).
Lloyd-Evans, E. On the move, lysosomal CAX drives Ca2+ transport and motility. J. Cell Biol. 212, 755–757 (2016).
Melchionda, M., Pittman, J. K., Mayor, R. & Patel, S. Ca2+/H+ exchange by acidic organelles regulates cell migration in vivo. J. Cell Biol. 212, 803–813 (2016).
Morgan, A. J., Davis, L. C. & Galione, A. Imaging approaches to measuring lysosomal calcium. Methods Cell Biol. 126, 159–195 (2015).
Christensen, K. A., Myers, J. T. & Swanson, J. A. pH-dependent regulation of lysosomal calcium in macrophages. J. Cell Sci. 115, 599–607 (2002).
Lloyd-Evans, E. et al. Niemann–Pick disease type C1 is a sphingosine storage disease that causes deregulation of lysosomal calcium. Nat. Med. 14, 1247–1255 (2008).
Garrity, A. G. et al. The endoplasmic reticulum, not the pH gradient, drives calcium refilling of lysosomes. eLife 5, e15887 (2016).
Sherwood, M. W. et al. Activation of trypsinogen in large endocytic vacuoles of pancreatic acinar cells. Proc. Natl Acad. Sci. USA 104, 5674–5679 (2007).
Gerasimenko, J. V., Tepikin, A. V., Petersen, O. H. & Gerasimenko, O. V. Calcium uptake via endocytosis with rapid release from acidifying endosomes. Curr. Biol. 8, 1335–1338 (1998).
Albrecht, T., Zhao, Y., Nguyen, T. H., Campbell, R. E. & Johnson, J. D. Fluorescent biosensors illuminate calcium levels within defined beta-cell endosome subpopulations. Cell Calcium 57, 263–274 (2015).
Johnson, D. E., Ostrowski, P., Jaumouillé, V. & Grinstein, S. The position of lysosomes within the cell determines their luminal pH. J. Cell Biol. 212, 677–692 (2016).
Chakraborty, K., Veetil, A. T., Jaffrey, S. R. & Krishnan, Y. Nucleic acid-based nanodevices in biological imaging. Annu. Rev. Biochem. 85, 349–373 (2016).
Modi, S. et al. A DNA nanomachine that maps spatial and temporal pH changes inside living cells. Nat. Nanotechnol. 4, 325–330 (2009).
Surana, S., Bhat, J. M., Koushika, S. P. & Krishnan, Y. An autonomous DNA nanomachine maps spatiotemporal pH changes in a multicellular living organism. Nat. Commun. 2, 340 (2011).
Saha, S., Prakash, V., Halder, S., Chakraborty, K. & Krishnan, Y. A pH-independent DNA nanodevice for quantifying chloride transport in organelles of living cells. Nat. Nanotechnol. 10, 645–651 (2015).
Modi, S., Nizak, C., Surana, S., Halder, S. & Krishnan, Y. Two DNA nanomachines map pH changes along intersecting endocytic pathways inside the same cell. Nat. Nanotechnol. 8, 459–467 (2013).
Chakraborty, K., Leung, K. & Krishnan, Y. High lumenal chloride in the lysosome is critical for lysosome function. eLife 6, e28862 (2017).
Toyoshima, C. & Inesi, G. Structural basis of ion pumping by Ca2+-ATPase of the sarcoplasmic reticulum. Annu. Rev. Biochem. 73, 269–292 (2004).
Schmidt, K., Wolfe, D. M., Stiller, B. & Pearce, D. A. Cd2+, Mn2+, Ni2+ and Se2+ toxicity to Saccharomyces cerevisiae lacking YPK9p the orthologue of human ATP13A2. Biochem. Biophys. Res. Commun. 383, 198–202 (2009).
Ramonet, D. et al. PARK9-associated ATP13A2 localizes to intracellular acidic vesicles and regulates cation homeostasis and neuronal integrity. Hum. Mol. Genet. 21, 1725–1743 (2012).
Fares, H. & Greenwald, I. Regulation of endocytosis by CUP-5, the Caenorhabditis elegans mucolipin-1 homolog. Nat. Genet. 28, 64–68 (2001).
Salgado, E. N., Garcia Rodriguez, B., Narayanaswamy, N., Krishnan, Y. & Harrison, S. C. Visualization of Ca2+ loss from rotavirus during cell entry. J. Virol. https://doi.org/10.1128/JVI.01327–18 (2018).
Jewett, J. C., Sletten, E. M. & Bertozzi, C. R. Rapid Cu-free click chemistry with readily synthesized biarylazacyclooctynones. J. Am. Chem. Soc. 132, 3688–3690 (2010).
Huotari, J. & Helenius, A. Endosome maturation. EMBO J. 30, 3481–3500 (2011).
Hu, Y.-B., Dammer, E. B., Ren, R.-J. & Wang, G. The endosomal-lysosomal system: from acidification and cargo sorting to neurodegeneration. Transl. Neurodegener. 4, 18 (2015).
Vandecaetsbeek, I., Vangheluwe, P., Raeymaekers, L., Wuytack, F. & Vanoevelen, J. The Ca2+ pumps of the endoplasmic reticulum and Golgi apparatus. Cold Spring Harb. Perspect. Biol. 3, a004184 (2011).
Tharkeshwar, A. K. et al. A novel approach to analyze lysosomal dysfunctions through subcellular proteomics and lipidomics: the case of NPC1 deficiency. Sci. Rep. 7, 41408 (2017).
Chapel, A. et al. An extended proteome map of the lysosomal membrane reveals novel potential transporters. Mol. Cell. Proteomics 12, 1572–1588 (2013).
Lübke, T., Lobel, P. & Sleat, D. E. Proteomics of the lysosome. Biochim. Biophys. Acta 1793, 625–635 (2009).
Brozzi, A., Urbanelli, L., Germain, P. L., Magini, A. & Emiliani, C. hLGDB: a database of human lysosomal genes and their regulation. Database (Oxford) 2013, bat024 (2013).
Schröder, B. A., Wrocklage, C., Hasilik, A. & Saftig, P. The proteome of lysosomes. Proteomics 10, 4053–4076 (2010).
Cao, Q., Yang, Y., Zhong, X. Z. & Dong, X.-P. The lysosomal Ca2+ release channel TRPML1 regulates lysosome size by activating calmodulin. J. Biol. Chem. 292, 8424–8435 (2017).
Sahoo, N. et al. Gastric acid secretion from parietal cells is mediated by a Ca2+ efflux channel in the tubulovesicle. Dev. Cell 41, 262–273 (2017).
Bargal, R. et al. Identification of the gene causing mucolipidosis type IV. Nat. Genet. 26, 118–123 (2000).
Schaheen, L., Dang, H. & Fares, H. Basis of lethality in C. elegans lacking CUP-5, the mucolipidosis type IV orthologue. Dev. Biol. 293, 382–391 (2006).
C. elegans Deletion Mutant Consortium. Large-scale screening for targeted knockouts in the Caenorhabditis elegans genome. G3 (Bethesda) 2, 1415–1425 (2012).
Schaheen, L., Patton, G. & Fares, H. Suppression of the cup-5 mucolipidosis type IV-related lysosomal dysfunction by the inactivation of an ABC transporter in C. elegans. Development 133, 3939–3948 (2006).
Fares, H. & Greenwald, I. Genetic analysis of endocytosis in Caenorhabditis elegans: coelomocyte uptake defective mutants. Genetics 159, 133–145 (2001).
van Veen, S. et al. Cellular function and pathological role of ATP13A2 and related P-type transport ATPases in Parkinson’s disease and other neurological disorders. Front. Mol. Neurosci. 7, 48 (2014).
Schöndorf, D. C. et al. iPSC-derived neurons from GBA1-associated Parkinson’s disease patients show autophagic defects and impaired calcium homeostasis. Nat. Commun. 5, 4028 (2014).
Usenovic, M., Tresse, E., Mazzulli, J. R., Taylor, J. P. & Krainc, D. Deficiency of ATP13A2 leads to lysosomal dysfunction, α-synuclein accumulation, and neurotoxicity. J. Neurosci. 32, 4240–4246 (2012).
Bras, J., Verloes, A., Schneider, S. A., Mole, S. E. & Guerreiro, R. J. Mutation of the parkinsonism gene ATP13A2 causes neuronal ceroid-lipofuscinosis. Hum. Mol. Genet. 21, 2646–2650 (2012).
Estrada-Cuzcano, A. et al. Loss-of-function mutations in the ATP13A2/PARK9 gene cause complicated hereditary spastic paraplegia (SPG78). Brain 140, 287–305 (2017).
Grynkiewicz, G., Poenie, M. & Tsien, R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 260, 3440–3450 (1985).
Collot, M. et al. CaRuby-Nano: a novel high affinity calcium probe for dual color imaging. eLife 4, e05808 (2015).
Moore, D. & Dowhan, D. Purification and concentration of DNA from aqueous solutions. Curr. Protoc. Mol. Biol. 59, 2.1.1–2.1.10 (2002).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Kamath, R. S. & Ahringer, J. Genome-wide RNAi screening in Caenorhabditis elegans. Methods 30, 313–321 (2003).
Evangelidis, G. D. & Psarakis, E. Z. Parametric image alignment using enhanced correlation coefficient maximization. IEEE Trans. Pattern Anal. Mach. Intell. 30, 1858–1865 (2008).
Sauvola, J. & Pietikäinen, M. Adaptive document image binarization. Pattern Recognit. 33, 225–236 (2000).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Engelstein, M. et al. An efficient, automatable template preparation for high throughput sequencing. Microb. Comp. Genomics 3, 237–241 (1998).
Vandeventer, P. E. et al. Multiphasic DNA adsorption to silica surfaces under varying buffer, pH, and ionic strength conditions. J. Phys. Chem. B 116, 5661–5670 (2012).
Acknowledgements
We thank J. Kuriyan and M. Zajac for valuable comments. We thank the Integrated Light Microscopy facility at the University of Chicago, the Caenorhabditis Genetic Center for strains, and Ausubel Lab for Arhinger Library RNAi clones. We thank the Krainc lab at Northwestern University (Chicago, IL, USA) for the fibroblast cells harboring mutations in ATP13A2 (L6025). HDF cells were a kind gift from the lab of J. Rowley (University of Chicago, Chicago, IL, USA). This work was supported by the University of Chicago Women’s Board; a Pilot and Feasibility award from an NIDDK center grant no. P30DK42086 to the University of Chicago Digestive Diseases Research Core Center; MRSEC grant no. DMR-1420709; the National Center for Advancing Translational Sciences of the National Institutes of Health through grant no. 1UL1TR002389-01 that funds the Institute for Translational Medicine, Chicago Biomedical Consortium, with support from the Searle Funds at The Chicago Community Trust, C-084; and University of Chicago start-up funds to Y.K. Y.K. is a Brain Research Foundation Fellow.
Author information
Authors and Affiliations
Contributions
K.C. and Y.K. designed the project. N.N. synthesized and designed the calcium dye. N.N., K.C., A.S., E.Z., and K.L. performed experiments. J.D. provided key resources. N.N., K.C., A.S., and Y.K. analyzed the data. K.C. and Y.K wrote the paper. All authors discussed the results and gave inputs on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 In vitro characterization of Rhod-5F.
(a) Chemical structure of Rhod-5F. (b) Fluorescence emission spectra of Rhod-5F(green) and Alexa Fluor 647 (red) with increasing [Ca2+] upon exciting Rhod-5F and Alexa Fluor 647 at 560 nm and 650 nm, respectively. (c) Normalized O/R ratio of Rhod-5F/Alexa Fluor 647 with increasing [Ca2+] at pH 7.2 and 5.5. Error bar represents mean + s.e.m. of three independent experiments.
Supplementary Figure 2 Characterization of CalipHluorLy and CalipHluor.
(a) Gel showing the conjugation of Rhod-5F to D2-DBCO strand. Gels were visualized in EtBr and TMR channels. (b) Native PAGE showing formation of CalipHluorLy. Gels were visualized in Alexa Fluor 488, TMR and Alexa Fluor 647 channels. (c) Schematic of working principle of CalipHluor. pH-induced FRET changes between Alexa Fluor 488 (donor, green sphere) and Alexa Fluor 647 (acceptor, red star) is used to report pH ratiometrically. A Ca2+ sensitive fluorophore (Rhod-5F, yellow diamond) and Alexa Fluor 647 report Ca2+ (at a given pH) ratiometrically by direct excitation of each dye. (d) Gel showing the conjugation of Rhod-5F to O3-DBCO strand. Gels were visualized in EtBr and TMR channels. (e) Native PAGE showing formation of CalipHluor. Gels were visualized in Alexa Fluor 488, TMR and Alexa Fluor 647 channels. (f) Emission spectra of CalipHluor at pH values ranging from 7.5 to 5.0 upon excitation at 488 nm. (g) Normalized ratio of fluorescence intensity of donor to that of acceptor (D/A) of CalipHluor as a function of pH. (D λex = 495 nm, λem = 520 nm; A λex = 495 nm, λem = 665 nm). Gels were performed twice independently. Error bar represents mean + S.E.M of three independent experiments.
Supplementary Figure 3 In vivo performance of CalipHluorLy.
Representative pseudo color images of coelomocytes labeled with CalipHluorLy and clamped at the indicated (a) pH and (b) free [Ca2+] at pH 5.5. Scale bar 5 μm. Experiments were repeated three times independently with similar results.
Supplementary Figure 4 Comparison of in vitro and in vivo pH and Ca2+ calibration profile of CalipHluorLy.
(a-d) D/A ratios of CalipHluorLy as a function of pH clamped at different amounts of added [Ca2+]. (e-h) Normalized O/R ratios of CalipHluorLy as a function of free [Ca2+] clamped at different pH points. For in vivo n = 10 worms; 15 cells and 50 endosomes were quantified; in vitro n = 2. Error bar represents mean + s.e.m.
Supplementary Figure 5 Endocytic trafficking of CalipHluorA647 in coelomocytes.
(a-c) Representative confocal images taken 5 min, 17 min, and 60 min following injection of CalipHluorA647 in worms expressing GFP::RAB-5, GFP::RAB-7 and LMP-1::GFP. Scale bar 5 μm. Experiment was performed once. n = 10 worms.
Supplementary Figure 6 Lethality rescue and RNAi controls.
a) catp-6 rescues lethality of cup-5 +/−. Representative images showing the number of progeny of cup-5 +/− worms in plates containing RNAi bacteria of mrp-4 (positive control), clh-6, catp-6, catp-5 and e.v. (control). Experiments were repeated twice independently with similar results. b) RT-PCR analysis of total RNA isolated from C. elegans pre- and post-RNAi. Lanes correspond to PCR-amplified cDNA of the indicated gene product isolated from wild type without RNAi treatment (denoted by gene name) and the corresponding dsRNA-fed worms (denoted as. ‘— gene name) c) Representative images of worms expressing LMP-1::GFP (green) in the background of various indicated RNAi, which were injected with CalipHluorLyA647 (red) and imaged 60 mins post-injection. Scale bar: 5 μm. d) Quantification of colocalization between the CalipHluorLyA647 and GFP in LMP-1::GFP worms. n = 10 cells; error bars represent mean + s.e.m.
Supplementary Figure 7 Characterization of CalipHluormLy.
a) Schematic of the working principle of CalipHluormLy. An Oregon Green based pH sensor (green sphere), an ion insensitive Alexa Fluor 647 (red star) and a Ca2+ sensitive fluorophore (Rhod-5F, yellow diamond). Calibration curves comparing in vitro (red) and on beads (orange) calibration at; b) pH 4.6 and c) pH 5.1. Comparison of d) Kd and e) Fold change (FC) in O/R of CalipHluorLy (pink) and CalipHluormLy (black). f) Representative images Comparison of g) Fold change (FC) in O/R and h) Kd of CalipHluormLy in vitro (pink), on beads (orange) and in cellulo (gray). (n = 5 cells; 30 endosomes; n = 60 beads). Experiments were performed three times independently. *Error is obtained from Hill equation fit. Error bars represent mean + s.e.m. Scale bar: 10 µm.
Supplementary Figure 8 Uptake of CalipHluormLy in fibroblast cells.
a)-b) CalipHluormLy internalization by primary human skin fibroblasts is competed out by excess maleylated BSA (mBSA, 10 μM), revealing that uptake is mediated by scavenger receptors. Cells are imaged in Alexa Fluor 647 channel. AF: autofluorescence. Scale bar: 10 µm. Experiments were performed in triplicate. Error bars indicate the mean of three independent experiments +/− s.e.m. (n = 25 cells).
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–8, Supplementary Note 1 and Supplementary Tables 1–3
Rights and permissions
About this article
Cite this article
Narayanaswamy, N., Chakraborty, K., Saminathan, A. et al. A pH-correctable, DNA-based fluorescent reporter for organellar calcium. Nat Methods 16, 95–102 (2019). https://doi.org/10.1038/s41592-018-0232-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-018-0232-7