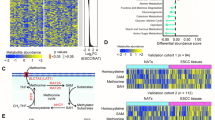
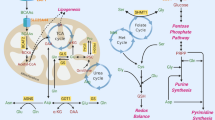
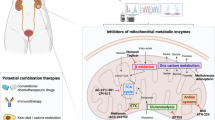
Abstract
Understanding cellular metabolism holds immense potential for developing new classes of therapeutics that target metabolic pathways in cancer. Metabolic pathways are altered in bulk neoplastic cells in comparison to normal tissues. However, carcinoma cells within tumors are heterogeneous, and tumor-initiating cells (TICs) are important therapeutic targets that have remained metabolically uncharacterized. To understand their metabolic alterations, we performed metabolomics and metabolite tracing analyses, which revealed that TICs have highly elevated methionine cycle activity and transmethylation rates that are driven by MAT2A. High methionine cycle activity causes methionine consumption to far outstrip its regeneration, leading to addiction to exogenous methionine. Pharmacological inhibition of the methionine cycle, even transiently, is sufficient to cripple the tumor-initiating capability of these cells. Methionine cycle flux specifically influences the epigenetic state of cancer cells and drives tumor initiation. Methionine cycle enzymes are also enriched in other tumor types, and MAT2A expression impinges upon the sensitivity of certain cancer cells to therapeutic inhibition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The metabolomics datasets generated or analyzed during this study are included in this published article in Supplementary Tables 2 and 3. Additional datasets are also available from the corresponding author upon reasonable request. Source data are available online for Figs. 1–5 and Extended Data Figs. 1 and 3–5.
Change history
21 May 2019
In the version of this article originally published, there is an error in Fig. 5a. Originally, ‘MAT2A’ appeared between ‘Methionine’ and ‘Homocysteine’. ‘MAT2A’ should have been ‘MTR’. The error has been corrected in the PDF and HTML versions of this article.
References
Chaneton, B. et al. Serine is a natural ligand and allosteric activator of pyruvate kinase M2. Nature 491, 458–462 (2012).
DeBerardinis, R. J., Lum, J. J., Hatzivassiliou, G. & Thompson, C. B. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 7, 11–20 (2008).
Jain, M. et al. Metabolite profiling identifies a key role for glycine in rapid cancer cell proliferation. Science 336, 1040–1044 (2012).
Maddocks, O. D. et al. Serine starvation induces stress and p53-dependent metabolic remodelling in cancer cells. Nature 493, 542–546 (2013).
Mayers, J. R. & Vander Heiden, M. G. Famine versus feast: understanding the metabolism of tumors in vivo. Trends Biochem. Sci. 40, 130–140 (2015).
Vander Heiden, M. G. et al. Metabolic pathway alterations that support cell proliferation. Cold Spring Harb. Symp. Quant. Biol. 76, 325–334 (2011).
Wise, D. R. & Thompson, C. B. Glutamine addiction: a new therapeutic target in cancer. Trends Biochem. Sci. 35, 427–433 (2010).
Anastasiou, D. et al. Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to cellular antioxidant responses. Science 334, 1278–1283 (2011).
Kim, D. et al. SHMT2 drives glioma cell survival in ischaemia but imposes a dependence on glycine clearance. Nature 520, 363–367 (2015).
Son, J. et al. Glutamine supports pancreatic cancer growth through a KRAS-regulated metabolic pathway. Nature 496, 101–105 (2013).
Locasale, J. W. et al. Phosphoglycerate dehydrogenase diverts glycolytic flux and contributes to oncogenesis. Nat. Genet. 43, 869–874 (2011).
Possemato, R. et al. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature 476, 346–350 (2011).
Kaelin, W. G. Jr & McKnight, S. L. Influence of metabolism on epigenetics and disease. Cell 153, 56–69 (2013).
Kaelin, W. G. Jr Cancer and altered metabolism: potential importance of hypoxia-inducible factor and 2-oxoglutarate-dependent dioxygenases. Cold Spring Harb. Symp. Quant. Biol. 76, 335–345 (2011).
Lu, C. et al. IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature 483, 474–478 (2012).
Letouze, E. et al. SDH mutations establish a hypermethylator phenotype in paraganglioma. Cancer Cell 23, 739–752 (2013).
Xiao, M. et al. Inhibition of α-KG-dependent histone and DNA demethylases by fumarate and succinate that are accumulated in mutations of FH and SDH tumor suppressors. Genes Dev. 26, 1326–1338 (2012).
Kreso, A. & Dick, J. E. Evolution of the cancer stem cell model. Cell Stem Cell 14, 275–291 (2014).
Meacham, C. E. & Morrison, S. J. Tumour heterogeneity and cancer cell plasticity. Nature 501, 328–337 (2013).
O’Brien, C. A., Kreso, A. & Dick, J. E. Cancer stem cells in solid tumors: an overview. Semin. Radiat. Oncol. 19, 71–77 (2009).
Oskarsson, T., Batlle, E. & Massague, J. Metastatic stem cells: sources, niches, and vital pathways. Cell Stem Cell 14, 306–321 (2014).
Tan, W. L. et al. Novel therapeutic targets on the horizon for lung cancer. Lancet Oncol. 17, e347–e362 (2016).
Lee, Y. A. et al. Identification of tumor initiating cells with a small-molecule fluorescent probe by using vimentin as a biomarker. Angew. Chem. Int. Ed. Engl. 57, 2851–2854 (2018).
Zhang, W. C. et al. Glycine decarboxylase activity drives non-small cell lung cancer tumor-initiating cells and tumorigenesis. Cell 148, 259–272 (2012).
Shiraki, N. et al. Methionine metabolism regulates maintenance and differentiation of human pluripotent stem cells. Cell Metab. 19, 780–794 (2014).
Tsuyama, T., Shiraki, N. & Kume, S. Definitive endoderm differentiation of human embryonic stem cells combined with selective elimination of undifferentiated cells by methionine deprivation. Methods Mol. Biol. 1341, 173–180 (2016).
Shyh-Chang, N. et al. Influence of threonine metabolism on S-adenosylmethionine and histone methylation. Science 339, 222–226 (2013).
Maddocks, O. D., Labuschagne, C. F., Adams, P. D. & Vousden, K. H. Serine metabolism supports the methionine cycle and DNA/RNA methylation through de novo ATP synthesis in cancer cells. Mol. Cell 61, 210–221 (2016).
Labuschagne, C. F., van den Broek, N. J., Mackay, G. M., Vousden, K. H. & Maddocks, O. D. Serine, but not glycine, supports one-carbon metabolism and proliferation of cancer cells. Cell Rep. 7, 1248–1258 (2014).
Martinez-Chantar, M. L. et al. l-Methionine availability regulates expression of the methionine adenosyltransferase 2A gene in human hepatocarcinoma cells: role of S-adenosylmethionine. J. Biol. Chem. 278, 19885–19890 (2003).
Pendleton, K. E. et al. The U6 snRNA m6A methyltransferase METTL16 regulates SAM synthetase intron retention. Cell 169, 824–835 (2017).
Quinlan, C. L. et al. Targeting S-adenosylmethionine biosynthesis with a novel allosteric inhibitor of Mat2A. Nat. Chem. Biol. 13, 785–792 (2017).
Eagle, H. The specific amino acid requirements of a human carcinoma cell (Stain HeLa) in tissue culture. J. Exp. Med. 102, 37–48 (1955).
Eagle, H., Oyama, V. I. & Levy, M. Amino acid requirements of normal and malignant human cells in tissue culture. Arch. Biochem. Biophys. 67, 432–446 (1957).
Carey, B. W., Finley, L. W. S., Cross, J. R., Allis, C. D. & Thompson, C. B. Intracellular α-ketoglutarate maintains the pluripotency of embryonic stem cells. Nature 518, 413–416 (2015).
Guo, H. Y., Herrera, H., Groce, A. & Hoffman, R. M. Expression of the biochemical defect of methionine dependence in fresh patient tumors in primary histoculture. Cancer Res. 53, 2479–2483 (1993).
Yano, S. et al. Selective methioninase-induced trap of cancer cells in S/G2 phase visualized by FUCCI imaging confers chemosensitivity. Oncotarget 5, 8729–8736 (2014).
De La Haba, G. & Cantoni, G. L. The enzymatic synthesis of S-adenosyl-l-homocysteine from adenosine and homocysteine. J. Biol. Chem. 234, 603–608 (1959).
Zhang, W. et al. Fluorinated N,N-dialkylaminostilbenes repress colon cancer by targeting methionine S-adenosyltransferase 2A. ACS Chem. Biol. 8, 796–803 (2013).
Jiang, X. et al. Functional characterization of D9, a novel deazaneplanocin A (DZNep) analog, in targeting acute myeloid leukemia (AML). PloS One 10, e0122983 (2015).
Miranda, T. B. et al. DZNep is a global histone methylation inhibitor that reactivates developmental genes not silenced by DNA methylation. Mol. Cancer Ther. 8, 1579–1588 (2009).
Tan, J. et al. Pharmacologic disruption of Polycomb-repressive complex 2-mediated gene repression selectively induces apoptosis in cancer cells. Genes Dev. 21, 1050–1063 (2007).
Cantor, J. R. & Sabatini, D. M. Cancer cell metabolism: one hallmark, many faces. Cancer Discov. 2, 881–898 (2012).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Ward, P. S. & Thompson, C. B. Metabolic reprogramming: a cancer hallmark even Warburg did not anticipate. Cancer Cell 21, 297–308 (2012).
Halpern, B. C., Clark, B. R., Hardy, D. N., Halpern, R. M. & Smith, R. A. The effect of replacement of methionine by homocystine on survival of malignant and normal adult mammalian cells in culture. Proc. Natl Acad. Sci. USA 71, 1133–1136 (1974).
Hoffman, R. M. & Erbe, R. W. High in vivo rates of methionine biosynthesis in transformed human and malignant rat cells auxotrophic for methionine. Proc. Natl Acad. Sci. USA 73, 1523–1527 (1976).
Stern, P. H., Wallace, C. D. & Hoffman, R. M. Altered methionine metabolism occurs in all members of a set of diverse human tumor cell lines. J. Cell. Physiol. 119, 29–34 (1984).
Batlle, E. & Clevers, H. Cancer stem cells revisited. Nat. Med. 23, 1124–1134 (2017).
Lyssiotis, C. A. & Kimmelman, A. C. Metabolic interactions in the tumor microenvironment. Trends Cell Biol. 27, 863–875 (2017).
Mayers, J. R. et al. Tissue of origin dictates branched-chain amino acid metabolism in mutant Kras-driven cancers. Science 353, 1161–1165 (2016).
Davidson, S. M. et al. Environment impacts the metabolic dependencies of Ras-driven non-small cell lung cancer. Cell Metab. 23, 517–528 (2016).
Guo, H. et al. Therapeutic tumor-specific cell cycle block induced by methionine starvation in vivo. Cancer Res. 53, 5676–5679 (1993).
Ikeda, S., Kawahara-Miki, R., Iwata, H., Sugimoto, M. & Kume, S. Role of methionine adenosyltransferase 2A in bovine preimplantation development and its associated genomic regions. Sci. Rep. 7, 3800 (2017).
Katoh, Y. et al. Methionine adenosyltransferase II serves as a transcriptional corepressor of Maf oncoprotein. Mol. Cell 41, 554–566 (2011).
Kera, Y. et al. Methionine adenosyltransferase II-dependent histone H3K9 methylation at the COX-2 gene locus. J. Biol. Chem. 288, 13592–13601 (2013).
Marjon, K. et al. MTAP deletions in cancer create vulnerability to targeting of the MAT2A/PRMT5/RIOK1 axis. Cell Rep. 15, 574–587 (2016).
McDonald, O. G., Wu, H., Timp, W., Doi, A. & Feinberg, A. P. Genome-scale epigenetic reprogramming during epithelial-to-mesenchymal transition. Nat. Struct. Mol. Biol. 18, 867–874 (2011).
Feinberg, A. P., Ohlsson, R. & Henikoff, S. The epigenetic progenitor origin of human cancer. Nat. Rev. Genet. 7, 21–33 (2006).
Hansen, K. D. et al. Increased methylation variation in epigenetic domains across cancer types. Nat. Genet. 43, 768–775 (2011).
Kahlert, C. et al. Increased expression of ALCAM/CD166 in pancreatic cancer is an independent prognostic marker for poor survival and early tumour relapse. Br. J. Cancer 101, 457–464 (2009).
Papal, S., Monti, C. E., Tennison, M. E. & Swaroop, A. Molecular dissection of cone photoreceptor-enriched genes encoding transmembrane and secretory proteins. J. Neurosci. Res. 97, 16–28 (2019).
Wu, M. et al. Multiparameter metabolic analysis reveals a close link between attenuated mitochondrial bioenergetic function and enhanced glycolysis dependency in human tumor cells. Am. J. Physiol Cell Physiol. 292, C125–C136 (2007).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Smith, C. A., Want, E. J., O’Maille, G., Abagyan, R. & Siuzdak, G. XCMS: processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching and identification. Anal. Chem. 78, 779–787 (2006).
Alves, T. C. et al. Integrated, step-wise, mass-isotopomeric flux analysis of the TCA cycle. Cell Metab. 22, 936–947 (2015).
Davis, M. A. et al. Calpain drives pyroptotic vimentin cleavage, intermediate filament loss, and cell rupture that mediates immunostimulation. Proc. Natl Acad. Sci. USA 116, 5061–5070 (2019).
Zizza, P. et al. TRF2 positively regulates SULF2 expression increasing VEGF-A release and activity in tumor microenvironment. Nucleic Acids Res. https://doi.org/10.1093/nar/gkz041(2019).
Garcia-Lopez, S. et al. Deregulation of the imprinted DLK1-DIO3 locus ncRNAs is associated with replicative senescence of human adipose-derived stem cells. PloS One 13, e0206534 (2018).
Wang, L. et al. H3K36 trimethylation mediated by SETD2 regulates the fate of bone marrow mesenchymal stem cells. PLoS Biol. 16, e2006522 (2018).
Fofou-Caillierez, M. B. et al. Interaction between methionine synthase isoforms and MMACHC: characterization in cblG-variant, cblG and cblC inherited causes of megaloblastic anaemia. Hum. Mol. Genet. 22, 4591–4601 (2013).
Zhu, B. et al. The protective role of DOT1L in UV-induced melanomagenesis. Nat. Commun. 9, 259 (2018).
Limm, K., Dettmer, K., Reinders, J., Oefner, P. J. & Bosserhoff, A. K. Characterization of the methylthioadenosine phosphorylase polymorphism rs7023954—incidence and effects on enzymatic function in malignant melanoma. PloS One 11, e0160348 (2016).
Cooper, S. et al. Jarid2 binds mono-ubiquitylated H2A lysine 119 to mediate crosstalk between Polycomb complexes PRC1 and PRC2. Nat. Commun. 7, 13661 (2016).
Chen, X., Zhi, X., Wang, J. & Su, J. RANKL signaling in bone marrow mesenchymal stem cells negatively regulates osteoblastic bone formation. Bone Res. 6, 34 (2018).
Li, Y. et al. Genome-wide analyses reveal a role of Polycomb in promoting hypomethylation of DNA methylation valleys. Genome Biol. 19, 18 (2018).
Fang, D., Gan, H., Wang, H., Zhou, H. & Zhang, Z. Probe the function of histone lysine 36 methylation using histone H3 lysine 36 to methionine mutant transgene in mammalian cells. Cell Cycle 16, 1781–1789 (2017).
Zhang, X. et al. H3 ubiquitination by NEDD4 regulates H3 acetylation and tumorigenesis. Nat. Commun. 8, 14799 (2017).
Freeman, T. J. et al. Inhibition of pannexin 1 reduces the tumorigenic properties of human melanoma cells. Cancers 11, E102 (2019).
Gerhart, S. V. et al. Activation of the p53–MDM4 regulatory axis defines the anti-tumour response to PRMT5 inhibition through its role in regulating cellular splicing. Sci. Rep. 8, 9711 (2018).
Sengupta, S. et al. Genome-wide expression profiling reveals EBV-associated inhibition of MHC class I expression in nasopharyngeal carcinoma. Cancer Res. 66, 7999–8006 (2006).
Haslinger, C. et al. Microarray gene expression profiling of B-cell chronic lymphocytic leukemia subgroups defined by genomic aberrations and VH mutation status. J. Clin. Oncol. 22, 3937–3949 (2004).
Zhang, J. et al. The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature 481, 157–163 (2012).
Piccaluga, P. P. et al. Gene expression analysis of peripheral T cell lymphoma, unspecified, reveals distinct profiles and new potential therapeutic targets. J. Clin. Invest. 117, 823–834 (2007).
Bonome, T. et al. A gene signature predicting for survival in suboptimally debulked patients with ovarian cancer. Cancer Res. 68, 5478–5486 (2008).
Talantov, D. et al. Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin. Cancer Res. 11, 7234–7242 (2005).
Wallace, T. A. et al. Tumor immunobiological differences in prostate cancer between African-American and European-American men. Cancer Res. 68, 927–936 (2008).
Zhao, H. et al. Different gene expression patterns in invasive lobular and ductal carcinomas of the breast. Mol. Biol. Cell 15, 2523–2536 (2004).
Acknowledgements
We thank SingHealth Advanced Molecular Pathology Laboratory for their assistance in generating tumor microarray data, M.Y. Lee, K.H.E. Lim, X.H. Yeo, G.S. Tan, M. Nichane, L. Chen, W.A. Zaw, S.L. Khaw, M.S. Noghabi and R. Ettikan for technical assistance, and S.-C. Ng for critical comments. This research is supported by the National Research Foundation, Singapore (NRF-NRFF2015-04), the National Medical Research Council, Singapore (LCG17MAY004; NMRC/OFIRG/0064/2017; NMRC/TCR/007-NCC/2013; OFYIRG16nov013), the Agency for Science, Research and Technology, Singapore (1331AEG071; 334I00053; SPF 2012/001), and the Singapore Ministry of Education under its Research Centers of Excellence initiative. Z. Wang dedicates this manuscript to the memory of Joseph P. Calarco, a wonderful friend and scientist.
Author information
Authors and Affiliations
Contributions
L.Y.Y., P.K.W.C., C.C.T., K.L.E.P., N.B. and Y.S.H. performed metabolomic studies. Z. Wang, J.H.J.L., H.Y.-K.A., L.S.K.C., H.Y.C., X.J. and Z. Wu performed molecular, cell-based and mouse xenograft experiments. A.T., A.M.H., Q.Y., E.H.T., W.T.L., T.K.H.L., J.Y., S.M. and D.S.W.T. provided key reagents and interpreted the data. Z. Wang, B.L. and W.L.T. designed the research and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Metabolomic characterization of lung tumor-initiating cells and differentiated cells.
a, Mean tumor mass of implanted cells. Error bars, s.d.; n = 6 tumors. b, Left, mean percentage of CD166+ cells. Error bars, s.d.; n = 4 biologically independent experiments. Right, representative flow cytometry plots, independently repeated four times. Unstained cells are in blue. c, Proliferation curves generated from mean cell viability. Error bars, s.d.; n = 6 biologically independent experiments. d, Mean ECAR. Error bars, s.d.; n = 6 biologically independent experiments. e,f, Top, immunoblots of the indicated cells, independently repeated three times. Bottom, mean ATP levels. Formate was supplemented at 0.5 mM. Error bars, s.d.; n = 6 (e) and n = 5 (f) biologically independent experiments. g, Top, mean tumor mass of implanted cells. Error bars, s.d.; n = 5 tumors. Bottom, mean tumor volume. Error bars, s.e.m.; n = 4 tumors. Mean tumor mass and volume for Control-knockdown (a and Fig. 1c) are included. h, Left, mean percentage of CD166+ cells. Error bars, s.d.; n = 4 biologically independent experiments. Right, representative flow cytometry plots, independently repeated four times. Unstained cells are in blue. i, Mean tumor mass and volume of implanted cells. Error bars, s.d. (left) and s.e.m. (right); n = 4 tumors. Mean tumor mass and volume for TS and Adh cells (a and Fig. 1c) are included. j, Immunoblots of cells grown in the indicated conditions. Independent blots were repeated at least three times with similar results. Histone H3 is used as a loading control. k, Mean abundance of metabolites in cells sorted by CD166 expression from three different tumors. Error bars, s.d. *P < 0.05, one-sided multiple t test corrected for multiple comparisons by the Holm–Sidak method. Exact P values (vs. CD166– cells) are as follows: methionine, 0.0102522; SAM, 0.01934. l, Immunoblots of sorted cells. Independent blots were repeated at least three times with similar results. GAPDH and total histone H3 were used as loading controls.
Extended Data Fig. 2 The metabolic requirements of lung tumor-initiating cells.
a, Left, mean number of colonies. Error bars, s.d.; n = 4 biologically independent experiments. Right, mean tumor mass. Error bars, s.d.; n = 9 (no methionine, no serine/glycine), n = 6 (no glutamine) tumors. Mean TS tumor masses (Extended Data Fig. 1a) were included. b, Imaging experiments were independently repeated three times. Top, lesion from TS-implanted lung (l) and normal bronchiole (r). Black bars, 50 μm. Middle, GFP-positive lesion (l) and normal bronchiole (r). Scale bars, 40 μm. Bottom, mean number of GFP-positive lesions. Error bars, s.d.; n = 5 lungs. ****P < 0.0001 by unpaired two-sided Student’s t test. c, Mean tumor mass. Error bars, s.d.; n = 5 tumors. d, Top, mean percentage of CD166+ cells. n = 4 biologically independent experiments. Error bars, s.d. Bottom, representative flow cytometry plots independently repeated four times. Unstained cells are in blue. e, Proliferation curves generated from mean cell viability. Error bars, s.d.; n = 6 biologically independent experiments. f, Left, mean number of colonies. Error bars, s.d.; n = 5 (no methionine + homocysteine), n = 3 (no methionine + SAM), n = 6 (48/48) biologically independent replicates. Right, mean tumor mass. Error bars, s.d.; n = 9 (no methionine + SAM, 48/48), n = 5 (no methionine + homocysteine) tumors. Tumor masses for no methionine (a) and TS cells (Extended Data Fig. 1a) are included. ****P < 0.0001, two-sided Student’s t test with Welch’s correction. P values (vs. no methionine): no methionine + homocysteine, 0.0505; no methionine + SAM, <0.0001; 48/48, <0.0001. g, Mean α-ketoglutarate/succinate ratios. Error bars, s.d.; n = 4 biologically independent experiments. h, Proliferation curves generated from mean cell viability. Error bars, s.d.; n = 6 biologically independent experiments. i, Top, representative flow cytometry plots independently repeated four times. Left, Complete condition. Right, Thymidine treated positive control. Bottom, mean percentage of cells in G2/M. Error bars, s.d.; n = 4 biologically independent experiments. j, Mean percentage of Annexin V+ cells. Error bars, s.d.; n = 4 biologically independent experiments. k, Proliferation curves generated from mean cell viability. Error bars, s.d.; n = 6 biologically independent experiments.
Extended Data Fig. 3 Metabolic labeling and tracking of methionine cycle flux.
a, Changes to [13C]methionine through the methionine cycle. Red circles, 13C; blue triangle, ATP; +, positive charge; black circle, 12C; black letters, enzymes. b, Top, cells were starved of methionine (16 h; l) or not starved (r) before [13C]methionine pulse–chase. Bottom, metabolite species detected are indicated on the right, and proportional abundance (% APE) is indicated on the left. Data represent the mean ± s.e.m.; n = 3 technical replicates. Technical replicates are shown to demonstrate technical consistency. n = 2 (bottom left) and n = 3 (bottom right) biologically independent experiments. c,d, Immunoblots of the indicated cells. β-actin (c) and total histone H3 (d) were used as loading controls. Independent blots were repeated at least three times with similar results. e, Mean tumor mass of implanted cells. Tumor masses from control- and GLDC-knockdown cells (Extended Data Fig. 1a) are included. Error bars, s.d.; n = 6 tumors. f, Immunoblots of the indicated lines. β-actin was used as a loading control. Independent blots were repeated at least three times with similar results. g, Percent APE of metabolite species derived from deuterated homocysteine. Data represent the mean ± s.e.m.; n = 3 technical replicates. Technical replicates are shown to demonstrate technical consistency. n = 3 biologically independent experiments. h, Mean tumor mass. Tumor masses from control- and GLDC-knockdown cells (Extended Data Fig. 1a) are included. Error bars, s.d.; n = 7 tumors. i, Mean ATP levels in the indicated cells supplemented with formate (0.5 mM), methyl-THF (20 μM) or adenosine (200 μM). Error bars, s.d.; n = 6 biologically independent experiments; **P < 0.005, Student’s two-sided t test with Welch’s correction. Exact P values (vs. GLDC shRNA) are as follows: GLDC shRNA + methyl-THF, 0.7947; GLDC shRNA + adenosine, 0.0011; GLDC shRNA + formate, 0.0010.
Extended Data Fig. 4 Functional and clinical relevance of methionine cycle enzymes in NSCLC.
a, Immunoblots of the indicated enzymes. β-actin was used as a loading control. Independent blots were repeated at least three times with similar results. b, MTHFR immunohistochemistry (performed once) of a tumor microarray (n = 47) containing paired tumor and normal sections. Top, representative staining intensity. White bar, 20 µm. Bottom, box-and-whisker plots comparing the intensity of tumor and normal sections. Intensity was defined as the product of the maximum immunostaining intensity and the percentage of tumor cells stained per section. Box, twenty-fifth to seventy-fifth percentile; the median value coincides with the seventy-fifth percentile; whiskers indicate the minima and maxima. **P = 0.0005, paired Student’s two-sided t test. t = 3.776, d.f. = 46. c, MTHFR immunohistochemistry (performed once) of an NSCLC tumor microarray (n = 153). Top, representative staining intensity. White bar, 200 µm. Bottom, contingency table correlating staining intensity with NSCLC grade. Chi-squared test P value (P = 0.2297) is indicated at the bottom right. χ2 = 8.116, d.f. = 6. d,e, Immunoblots of MAT2A in the indicated cells or tumors. GAPDH was used as a loading control. Independent blots were repeated at least three times with similar results. f, Proliferation curves generated from mean cell viability of the indicated lines. Error bars, s.d.; n = 10 biologically independent experiments.
Extended Data Fig. 5 Small-molecule inhibition of the methionine cycle disrupts the tumorigenicity of lung tumor-initiating cells.
a, Mean tumor mass of implanted cells. Error bars, s.d.; n = 6 (D9, DMSO), n = 9 (FIDAS, FIDAS + SAM) tumors. b, Left, mean percentage of CD166+ cells. Error bars, s.d.; n = 4 biologically independent experiments. Right, representative flow cytometry plots independently repeated four times. Unstained cells are in blue. c,d, Immunoblots of the indicated cells. β-catenin was used as a loading control. Independent blots were repeated at least three times with similar results. e, Proliferation curves generated from mean cell viability. Error bars, s.d.; n = 10 biologically independent experiments. f, Mean percentage of Annexin V+ cells. Error bars, s.d.; n = 4 (DMSO, D9), n = 3 (FIDAS) biologically independent experiments. g,h, Proliferation curves generated from mean cell viability. Error bars, s.d.; n = 10 biologically independent experiments. i, Mean tumor mass of implanted cells. Error bars, s.d.; n = 7 (control), n = 6 (FIDAS) and n = 9 (cisplatin) tumors. j, Mean number of GFP+ lesions. Error bars, s.d.; n = 5 lungs. ****P < 0.0001, two-sided unpaired Student’s t test. k, Individual weight plots of nine mice undergoing the indicated treatment. l, Mean MAT2A mRNA levels in normal versus tumor tissue, including glioblastoma (TCGA), colorectal cancer (TCGA), nasopharyngeal carcinoma81, leukemia82,83, lymphoma84, ovarian carcinoma85, melanoma86, prostate adenocarcinoma87 and breast cancer88. ****P < 0.0001, ***P < 0.001, **P ≤ 0.01, *P ≤ 0.05, Student’s unpaired two-sided t test with Welch’s correction. P values and numbers of normal and tumor samples are as follows: brain: P < 0.0001; n = 10 and n = 547; nasopharynx, P = 0.0005; n = 10 and n = 31; skin: P = 0.0015; n = 7 and n = 63; lymphatic system, P < 0.0001; n = 20 and n = 40; bone marrow (childhood acute lymphatic leukemia): P = 0.0492; n = 8 and n = 566; bone marrow (chronic lymphatic leukemia): P = 0.0003; n = 11 and n = 100; ovary: P < 0.0001; n = 10 and n = 185; breast, P = 0.0215; n = 5 and n = 59; prostate: P = 0.0010; n = 20 and n = 69; colon: P < 0.0001; n = 22 and n = 215. m, Immunoblots of the indicated cells. Independent blots were repeated at least three times with similar results.
Supplementary information
Supplementary Information
Supplementary Tables 1–7 and Figures 1 and 2
Source data
Source Data Fig. 1
Unprocessed Western Blots
Source Data Fig. 2
Unprocessed Western Blots
Source Data Fig. 3
Unprocessed Western Blots
Source Data Fig. 4
Unprocessed Western Blots
Source Data Fig. 5
Unprocessed Western Blots
Source Data Extended Data Fig. 1
Unprocessed Western Blots
Source Data Extended Data Fig. 3
Unprocessed Western Blots
Source Data Extended Data Fig. 4
Unprocessed Western Blots
Source Data Extended Data Fig. 5
Unprocessed Western Blots
Rights and permissions
About this article
Cite this article
Wang, Z., Yip, L.Y., Lee, J.H.J. et al. Methionine is a metabolic dependency of tumor-initiating cells. Nat Med 25, 825–837 (2019). https://doi.org/10.1038/s41591-019-0423-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-019-0423-5
This article is cited by
-
Blocking methionine catabolism induces senescence and confers vulnerability to GSK3 inhibition in liver cancer
Nature Cancer (2024)
-
Cellular metabolism regulates the differentiation and function of T-cell subsets
Cellular & Molecular Immunology (2024)
-
Unveiling the methionine cycle: a key metabolic signature and NR4A2 as a methionine-responsive oncogene in esophageal squamous cell carcinoma
Cell Death & Differentiation (2024)
-
8-Cl-Ado and 8-NH2-Ado synergize with venetoclax to target the methionine-MAT2A-SAM axis in acute myeloid leukemia
Leukemia (2024)
-
Immune, metabolic landscapes of prognostic signatures for lung adenocarcinoma based on a novel deep learning framework
Scientific Reports (2024)