Abstract
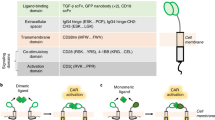
Chimeric antigen receptors (CARs) are synthetic receptors that target and reprogram T cells to acquire augmented antitumor properties1. CD19-specific CARs that comprise CD28 and CD3ζ signaling motifs2 have induced remarkable responses in patients with refractory leukemia3,4,5 and lymphoma6 and were recently approved by the US Food and Drug Administration7. These CARs program highly performing effector functions that mediate potent tumor elimination4,8 despite the limited persistence they confer on T cells3,4,5,6,8. Extending their functional persistence without compromising their potency should improve current CAR therapies. Strong T cell activation drives exhaustion9,10, which may be accentuated by the redundancy of CD28 and CD3ζ signaling11,12 as well as the spatiotemporal constraints imparted by the structure of second-generation CARs2. Thus, we hypothesized that calibrating the activation potential of CD28-based CARs would differentially reprogram T cell function and differentiation. Here, we show that CARs encoding a single immunoreceptor tyrosine-based activation motif direct T cells to different fates by balancing effector and memory programs, thereby yielding CAR designs with enhanced therapeutic profiles.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The RNA-seq data have been deposited in the Gene Expression Omnibus and are available under accession number GSE121226. Raw data for the figures in the manuscript will be made available upon request to the corresponding author.
Change history
28 January 2019
The original Nature Research Reporting Summary that was published with this article was an outdated version. The correct version is now available online.
28 January 2019
In the version of this article originally published, there was an error in the legend for Extended Data Fig. 7. The legend for panel f was originally: “f, FACS analysis of IL7R–, CD62L– and CD45RA– expression on TRAC-1928ζ and TRAC-1XX CAR T cells at day 63 post CAR infusion (representative for at least n = 3 mice per group in one independent experiment).” The legend should have been: “f, FACS analysis of IL7R+, CD62L+ and CD45RA+ expression on TRAC-1928ζ and TRAC-1XX CAR T cells at day 63 post CAR infusion (representative for at least n = 3 mice per group in one independent experiment).” The error has been corrected in the HTML and PDF versions of this article.
References
Sadelain, M., Rivière, I. & Riddell, S. Therapeutic T cell engineering. Nature 545, 423–431 (2017).
Maher, J., Brentjens, R. J., Gunset, G., Rivière, I. & Sadelain, M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta/CD28 receptor. Nat. Biotechnol. 20, 70–75 (2002).
Brentjens, R. J. et al. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci. Transl. Med. 5, 177ra38 (2013).
Lee, D. W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528 (2015).
Park, J. H. et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 378, 449–459 (2018).
Neelapu, S. S. et al. Axicabtagene Ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Sadelain, M. CD19 CAR T cells. Cell 171, 1471 (2017).
Zhao, Z. et al. Structural design of engineered costimulation determines tumor rejection kinetics and persistence of CAR T cells. Cancer Cell 28, 415–428 (2015).
Youngblood, B., Davis, C. W. & Ahmed, R. Making memories that last a lifetime: heritable functions of self-renewing memory CD8 T cells. Int. Immunol. 22, 797–803 (2010).
Wherry, E. J. & Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 15, 486–499 (2015).
Acuto, O. & Michel, F. CD28-mediated co-stimulation: a quantitative support for TCR signalling. Nat. Rev. Immunol. 3, 939–951 (2003).
Smith-Garvin, J. E., Koretzky, G. A. & Jordan, M. S. T cell activation. Annu. Rev. Immunol. 27, 591–619 (2009).
Love, P. E. & Hayes, S. M. ITAM-mediated signaling by the T-cell antigen receptor. Cold Spring Harb. Perspect. Biol. 2, a002485 (2010).
Kersh, E. N., Shaw, A. S. & Allen, P. M. Fidelity of T cell activation through multistep T cell receptor zeta phosphorylation. Science 281, 572–575 (1998).
Isakov, N. et al. ZAP-70 binding specificity to T cell receptor tyrosine-based activation motifs: the tandem SH2 domains of ZAP-70 bind distinct tyrosine-based activation motifs with varying affinity. J. Exp. Med. 181, 375–380 (1995).
van Oers, N. S. et al. The 21- and 23-kD forms of TCR zeta are generated by specific ITAM phosphorylations. Nat. Immunol. 1, 322–328 (2000).
Chae, W. J. et al. Qualitatively differential regulation of T cell activation and apoptosis by T cell receptor zeta chain ITAMs and their tyrosine residues. Int. Immunol. 16, 1225–1236 (2004).
Mukhopadhyay, H., Cordoba, S. P., Maini, P. K., van der Merwe, P. A. & Dushek, O. Systems model of T cell receptor proximal signaling reveals emergent ultrasensitivity. PLoS Comput. Biol. 9, e1003004 (2013).
Eyquem, J. et al. Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 543, 113–117 (2017).
Brentjens, R. J. et al. Eradication of systemic B-cell tumors by genetically targeted human T lymphocytes co-stimulated by CD80 and interleukin-15. Nat. Med. 9, 279–286 (2003).
Gattinoni, L. et al. A human memory T cell subset with stem cell-like properties. Nat. Med. 17, 1290–1297 (2011).
Chang, J. T., Wherry, E. J. & Goldrath, A. W. Molecular regulation of effector and memory T cell differentiation. Nat. Immunol. 15, 1104–1115 (2014).
Yu, B. et al. Epigenetic landscapes reveal transcription factors that regulate CD8+ T cell differentiation. Nat. Immunol. 18, 573–582 (2017).
Kaech, S. M. & Cui, W. Transcriptional control of effector and memory CD8+ T cell differentiation. Nat. Rev. Immunol. 12, 749–761 (2012).
Ichii, H. et al. Role for Bcl-6 in the generation and maintenance of memory CD8+T cells. Nat. Immunol. 3, 558–563 (2002).
Zhou, X. & Xue, H. H. Cutting edge: generation of memory precursors and functional memory CD8+ T cells depends on T cell factor-1 and lymphoid enhancer-binding factor-1. J. Immunol. 189, 2722–2726 (2012).
Carlson, C. M. et al. Kruppel-like factor 2 regulates thymocyte and T-cell migration. Nature 442, 299–302 (2006).
Bai, A., Hu, H., Yeung, M. & Chen, J. Kruppel-like factor 2 controls T cell trafficking by activating L-selectin (CD62L) and sphingosine-1-phosphate receptor 1 transcription. J. Immunol. 178, 7632–7639 (2007).
Daniels, M. A. & Teixeiro, E. TCR signaling in T cell memory. Front. Immunol. 6, 617 (2015).
Fraietta, J. A. et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat. Med. 24, 563–571 (2018).
Sommermeyer, D. et al. Chimeric antigen receptor-modified T cells derived from defined CD8+ and CD4+ subsets confer superior antitumor reactivity in vivo. Leukemia 30, 492–500 (2016).
Zhao, Y. et al. A herceptin-based chimeric antigen receptor with modified signaling domains leads to enhanced survival of transduced T lymphocytes and antitumor activity. J. Immunol. 183, 5563–5574 (2009).
James, J. R. Tuning ITAM multiplicity on T cell receptors can control potency and selectivity to ligand density. Sci. Signal. 11, eaan1088 (2018).
Kochenderfer, J. N., Yu, Z., Frasheri, D., Restifo, N. P. & Rosenberg, S. A. Adoptive transfer of syngeneic T cells transduced with a chimeric antigen receptor that recognizes murine CD19 can eradicate lymphoma and normal B cells. Blood 116, 3875–3886 (2010).
Rivière, I., Brose, K. & Mulligan, R. C. Effects of retroviral vector design on expression of human adenosine deaminase in murine bone marrow transplant recipients engrafted with genetically modified cells. Proc. Natl Acad. Sci. USA 92, 6733–6737 (1995).
Gong, M. C. et al. Cancer patient T cells genetically targeted to prostate-specific membrane antigen specifically lyse prostate cancer cells and release cytokines in response to prostate-specific membrane antigen. Neoplasia 1, 123–127 (1999).
Brentjens, R. J. et al. Genetically targeted T cells eradicate systemic acute lymphoblastic leukemia xenografts. Clin. Cancer Res. 13, 5426–5435 (2007).
Ghosh, A. et al. Adoptively transferred TRAIL+ T cells suppress GVHD and augment antitumor activity. J. Clin. Invest. 123, 2654–2662 (2013).
Gade, T. P. et al. Targeted elimination of prostate cancer by genetically directed human T lymphocytes. Cancer Res. 65, 9080–9088 (2005).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Acknowledgements
We thank J. Mansilla-Soto, S. van der Stegen, F. Perna, and other Sadelain laboratory members (Memorial Sloan Kettering Cancer Center) for helpful and critical discussions and G. Gunset for excellent technical assistance. We thank the following Memorial Sloan Kettering Cancer Center (MSKCC) core facilities for the excellent support: SKI Flow Cytometry core facility; animal facility; bioinformatics core; and integrated genomics operation core, funded by the NCI Cancer Center Support Grant (P30 CA08748), Cycle for Survival, and the Marie-Josée and Henry R. Kravis Center for Molecular Oncology. We thank the Care-for-Rare Foundation (J.F.), the German Research Foundation (DFG) (J.F.), the Edythe Griffinger Fellowship (J.S.), and the Louis V. Gerstner Jr. Graduate School of Biomedical Sciences (A.D.) for their support. This work was in part supported by the Lake Road Foundation, the Mr. William H. and Mrs. Alice Goodwin and the Commonwealth Foundation for Cancer Research, the Lymphoma and Leukemia Society, and the MSKCC Support Grant/Core Grant (P30 CA008748).
Author information
Authors and Affiliations
Contributions
J.E. and Y.-J.H. share second authorship. J.F. and J.S. designed the study, performed the experiments, analyzed and interpreted the data, and wrote the manuscript. J.E. and A.D. performed CRISPR–Cas9-mediated gene editing and J.E. performed in vivo experiments. Y.-J.H. analyzed the RNA-seq data. Z.Z. and J.L. performed in vivo experiments. A.C. and M.H. contributed to vector construction. M.S. designed the study, analyzed and interpreted the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
A patent application has been submitted based in part on results presented in this manuscript. J.F., J.S., M.H., and M.S. are listed as the inventors. The Memorial Sloan Kettering Cancer Center has licensed this intellectual property, has received license fees, and has the potential to receive royalties under the license.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
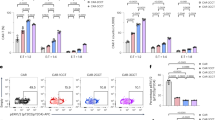
Extended Data Fig. 1 Impact of ITAM-mutated 1928ζ CARs on T cell function in vitro, T cell differentiation and antitumor activity in vivo.
a, Cytotoxic activity as determined by 4-h 51Cr release assay 1 week after expansion of effector cells on irradiated 3T3-CD19 (data are shown as means of n = 2 independent experiments performed in triplicates). b, Cumulative cell counts of indicated CAR T cells upon weekly stimulation with CD19+ target cells (n = 3 independent experiments). All data are means ± s.e.m. P values were calculated with two-tailed paired Student’s t-test. c, NALM6-bearing mice were treated with 5 × 104 CAR+ T cells. Kaplan–Meier analysis of survival comparing the in vivo efficacy of wild-type 1928ζ or indicated 1928ζ mutants (n = 10 mice, pooled data from two independent experiments). Control (Ctl) refers to untreated mice (n = 6). P value was determined by a one-sided log-rank Mantel–Cox test. d, Phenotype of CAR T cells as demonstrated by percentage of central memory (CD62L+CD45RA−) and effector memory (CD62L−CD45RA−) CD4+CAR T cells 48 h upon second stimulation with CD19+ target cells. Two-tailed paired Student’s t-test was performed, data represent means ± s.e.m. of n = 4 independent experiments. e, NALM6-bearing mice were treated with 5 × 104 CAR T cells and euthanized at day 10 after infusion; bone marrow CAR T cells were analyzed by FACS. Representative flow cytometric analysis of phenotype for indicated CAR T cells as determined by CD62L/CD45RA expression, gated on CAR+CD4+ T cells. Representative of 5 mice per group in at least n = 2 independent experiments with similar results.
Extended Data Fig. 2 Analysis of effector function in 1928ζ mutants compared to wild-type 1928ζ.
a, Cytotoxic activity of 1928ζ mutants compared to wild-type 1928ζ using an 18-h bioluminescence assay with FFL-expressing NALM6 cells as targets. Experiments were performed 1 week after expansion of effector cells on CD19+ target cells. Data are means ± s.e.m. (n = 4 independent experiments performed in triplicates). *P < 0.05 (21: P = 0.0273, 2-1: P = 0.0387, 2-2: P = 0.0125), **P = 0.0018 as calculated by two-tailed paired Student’s t-test of average of triplicates. b,c, Granzyme B (GrB) expression (n = 4 independent experiments) on CD8+ CAR T cells (b) and cytokine secretion (c) of CD4+ and CD8+ CAR T cells upon 2nd stimulation with CD19-expressing target cells. All data are means ± s.e.m. (IFNγ and IL2, n = 4; TNFα, n = 5 independent experiments). Unstimulated (Unstim.) wild-type 1928ζ cells were used as control. Significant differences compared to 1928ζ were determined by two-tailed paired Student’s t-test.
Extended Data Fig. 3 Impact of ITAM location within 1928ζ CARs on T cell function and therapeutic potency.
a, Cytotoxic activity as determined by 4-h 51Cr release assay 1 week after expansion of effector cells on irradiated 3T3-CD19 (data are means of n = 2 independent experiments performed in triplicates). b, Cumulative cell counts of indicated CAR T cells upon weekly stimulation with CD19+ target cells (n = 3 independent experiments). All data are means ± s.e.m.; P values were calculated with two-tailed paired Student’s t-test. c, Cytotoxic activity of D12 and D23 compared to wild-type 1928ζ as determined by 18-h bioluminescence assay with FFL-expressing NALM6 cells as targets. Experiments were performed 1 week after expansion of effector cells on CD19+ target cells. Data are means ± s.e.m. (n = 4 independent experiments performed in triplicates). P value was calculated by two-tailed paired Student’s t-test of average of triplicates and showed no significant difference (P > 0.05) between D12/D23 and wild-type 1928ζ for all E/T ratios. d, NALM6-bearing mice were treated with 5 × 104 CAR T cells. Kaplan–Meier analysis of survival of mice treated with wild-type 1928ζ or indicated 1928ζ mutants (n = 10 mice per group). Control refers to untreated mice (n = 6). P value was calculated by a one-sided log-rank Mantel–Cox test.
Extended Data Fig. 4 Influence of ITAM location within 1928ζ CARs on effector function in vitro.
a, Granzyme B (GrB) expression on CD8+ CAR T cells (n = 5 independent experiments). b, Cytokine secretion of CD4+ and CD8+ CAR T cells upon second stimulation with CD19-expressing target cells. Unstimulated wild-type 1928ζ cells were used as control. All data are means ± s.e.m. (IFNγ and IL2, n = 4; TNFα, n = 5 independent experiments). Each individual symbol indicates one sample. Significant differences compared to 1928ζ were determined by two-tailed paired Student’s t-test.
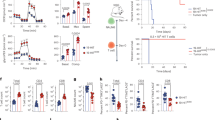
Extended Data Fig. 5 T cell differentiation and effector function of TRAC-encoded 1928ζ mutants.
NALM6-bearing mice were treated with 1 × 105 CAR T cells and euthanized at day 17 after infusion. Bone marrow and spleen CAR T cells were analyzed and counted by FACS. a, Histogram and flow cytometric analysis of CAR expression 4 d after CAR gene integration into the TRAC locus. Representative of four independent experiments with similar results. b, Cell numbers of CD4+ and CD8+ CAR T cells, c, Percentage of CD8+ TCM (CD62L+CD45RA−) and flow cytometric analysis of CD62L/CD45RA expression on bone marrow CD8+CAR T cells (representative of n = 5 mice per group in one independent experiment). d, Ratio of CAR+IL7R+ to tumor cells and exemplary flow cytometric analysis of IL7R+ CAR T cells in the bone marrow of mice. e, Enumeration of CAR T cells in the spleen of mice. In b, c, d and e all data are means ± s.e.m., two-tailed Mann–Whitney analysis was performed, n = 5 mice per group. f, Cytotoxic activity of TRAC-1XX, TRAC-XX3 and wild-type TRAC-1928ζ (18-h bioluminescence assay with FFL-expressing NALM6 as targets). Experiments were performed 4 d post transduction, 1 week and 3 weeks after expansion with weekly CD19 antigen stimulations. Symbols demonstrate means of triplicates (one representative donor).
Extended Data Fig. 6 In vivo T cell exhaustion of TRAC-1928ζ mutants compared to wild-type TRAC-1928ζ.
a, NALM6-bearing mice were treated with 1 × 105 CAR T cells and euthanized at day 17 after infusion. FACS analysis of expression of exhaustion markers on CAR+ T cells, representative of n = 5 mice per group in one independent experiment. b–g, NALM6-bearing mice were treated with 1 × 105 TRAC-edited naive T cells. 16 (b–c) and 36 (e,g) d after CAR administration, TRAC-1928ζ and TRAC-1XX cells from bone marrow and spleen were exposed to ex vivo stimulation with NALM6 or PMA/Ionomycin (Iono). Cytokine and granzyme B (GrB)/ CD107a expression on CAR T cells as demonstrated by percentage of expression and flow cytometric analyses, representative for n = 3 mice in two independent experiments (b) and for n = 3 replicates (g). Expression of exhaustion markers PD1+LAG3+ on CAR T cells (d) and cytotoxic activity (f) of TRAC-1XX (day 36) after 10 h of co-culture with NALM6. All data are means ± s.e.m., n = 3 mice per group
Extended Data Fig. 7 T cell memory formation in TRAC-1XX compared to wild-type TRAC-1928ζ.
NALM6-bearing mice were treated with 1 × 105 or 5 × 105 TRAC-edited naive T cells. a–c, 16 and 36 d after administration of 1 × 105 TRAC-1928ζ and TRAC-1XX, CARs were isolated from bone marrow and spleen. a–b, Cell number of CAR T cells (a), central memory (TCM: CD62L+CD45RA−), effector (TEFF: CD62L−CD45RA+) and IL7R-expressing bone marrow CAR T cells (b). All data are means ± s.e.m., n = 3 mice per group. c, Representative flow cytometric analysis of CD62L/CD45RA expression on TRAC-1928ζ and TRAC-1XX bone marrow CAR T cells at day 36 in one independent experiment (n = 3 mice per group). d–g, NALM6-bearing mice were treated with 5 × 105 TRAC-edited naive T cells and were either rechallenged with NALM6 cells (n = 5 mice per group) or no further rechallenge with tumor was performed (TRAC-1928ζ, n = 6 mice; TRAC-1XX, n = 7 mice). d,e, Cell number of total CAR T cells (d), TCM, TEFF and IL7R+ CAR T cells (e) in the spleen of treated mice 63 d post CAR administration (rechallenge: TRAC-1928ζ, n = 4 mice; TRAC-1XX, n = 5 mice. No rechallenge, n = 5 mice per group). All data are means ± s.e.m.; a two-tailed unpaired Student's t-test was used for statistical analysis. f, FACS analysis of IL7R+, CD62L+ and CD45RA+ expression on TRAC-1928ζ and TRAC-1XX CAR T cells at day 63 post CAR infusion (representative for at least n = 3 mice per group in one independent experiment). g, Expression of exhaustion markers PD1+TIM3+LAG3+ on CAR T cells derived from the spleen (rechallenge: TRAC-1928ζ, n = 4 mice; TRAC-1XX: n = 5 mice. No rechallenge, n = 5 mice per group). All data are means ± s.e.m.; P value was determined by a two-tailed unpaired Student's t-test.
Extended Data Fig. 8 Transcriptional profiles of TRAC-encoded 1928ζ mutants and sorted control T cells.
a, Principal component analysis (PCA) of global transcriptional profiles of CD8+ TRAC-1XX, TRAC-XX3 and TRAC-1928ζ after stimulation with CD19 target cells (left) and of sorted control T cell subsets (right): TN, TSCM and TEFF. Experiment was performed in technical triplicates for each CAR construct and in six replicates for each control subset. b, Representative GSEA enrichment plot (GSE10239), demonstrating downregulation of memory- relative to effector-related genes and naive- relative to effector-related genes in 1928ζ versus 1XX and in 1928ζ versus XX3 (n = 3 mice per group). c, Heat map of 900 differentially expressed genes among CD8+ T cell subsets as described by Gattinoni et al21 (left) compared to differential gene expression of sorted control T cell subsets (TEFF, TSCM and TN)21.
Extended Data Fig. 9 Impact of CD3ζ ITAM mutations in TRAC-1928ζ on T cell differentiation state and effector profile.
a, GSEA of a signature of the top 200 genes upregulated in exhausted CD8 T cells relative to naive or memory CD8 T cells as derived from GSE41867, demonstrating enrichment of exhaustion signature in TRAC-1928ζ versus TRAC-1XX or TRAC-XX3 and in the sorted control TEFF versus TN and TSCM. Experiment was performed in technical triplicates for each CAR construct and in six replicates for each control subset. b, Gene ontology analysis demonstrating significantly enriched gene sets associated with inflammation, cytokine and chemokine signaling in 1928ζ versus XX3, 1XX versus XX3 and 1928ζ versus 1XX (n = 3). Transcriptional analysis was performed after CAR gene integration into the TRAC locus of naive T cells and stimulation with CD19+ target cells. Results are shown in order of significance as –log10 (corrected P value). P values were determined by a one-tailed Fisher’s exact test and the Benjamini–Hochberg method was used to correct for multiple hypotheses testing. c, Heat map of selected differentially expressed genes between CAR constructs related to inflammation, cytokine and chemokine activity. d, Flow cytometric analysis of T cell differentiation state on CD8+CAR T cells after stimulation with CD19 antigen (representative for n = 2 independent experiments with similar results).
Extended Data Fig. 10 Gating strategy to analyze CAR T cells obtained from bone marrow of treated mice.
a,b Representative flow cytometric analysis of TRAC-1928ζ (a) compared to TRAC-1XX (b) on day 17 post CAR infusion. Placement of gating was based on FMO controls.
Supplementary information
Rights and permissions
About this article
Cite this article
Feucht, J., Sun, J., Eyquem, J. et al. Calibration of CAR activation potential directs alternative T cell fates and therapeutic potency. Nat Med 25, 82–88 (2019). https://doi.org/10.1038/s41591-018-0290-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0290-5
This article is cited by
-
Challenges and strategies associated with CAR-T cell therapy in blood malignancies
Experimental Hematology & Oncology (2024)
-
Base-editing mutagenesis maps alleles to tune human T cell functions
Nature (2024)
-
Prophylactic and long-lasting efficacy of senolytic CAR T cells against age-related metabolic dysfunction
Nature Aging (2024)
-
CAR-T cell therapy: a game-changer in cancer treatment and beyond
Clinical and Translational Oncology (2024)
-
Leveraging CD16 fusion receptors to remodel the immune response for enhancing anti-tumor immunotherapy in iPSC-derived NK cells
Journal of Hematology & Oncology (2023)