Abstract
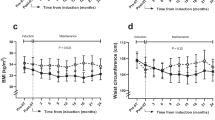
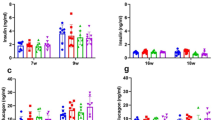
Pancreatic beta cell loss is a key factor in the pathogenesis of type 1 diabetes (T1D), but therapies to halt this process are lacking. We previously reported that the approved antihypertensive calcium-channel blocker verapamil, by decreasing the expression of thioredoxin-interacting protein, promotes the survival of insulin-producing beta cells and reverses diabetes in mouse models1. To translate these findings into humans, we conducted a randomized double-blind placebo-controlled phase 2 clinical trial (NCT02372253) to assess the efficacy and safety of oral verapamil added for 12 months to a standard insulin regimen in adult subjects with recent-onset T1D. Verapamil treatment, compared with placebo was well tolerated and associated with an improved mixed-meal-stimulated C-peptide area under the curve, a measure of endogenous beta cell function, at 3 and 12 months (prespecified primary endpoint), as well as with a lower increase in insulin requirements, fewer hypoglycemic events and on-target glycemic control (secondary endpoints). Thus, addition of once-daily oral verapamil may be a safe and effective novel approach to promote endogenous beta cell function and reduce insulin requirements and hypoglycemic episodes in adult individuals with recent-onset T1D.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Xu, G., Chen, J., Jing, G. & Shalev, A. Preventing beta-cell loss and diabetes with calcium channel blockers. Diabetes 61, 848–856 (2012).
Davis, A. K. et al. Prevalence of detectable C-peptide according to age at diagnosis and duration of type 1 diabetes. Diabetes Care 38, 476–481 (2015).
Liu, E. H. et al. Pancreatic beta cell function persists in many patients with chronic type 1 diabetes, but is not dramatically improved by prolonged immunosuppression and euglycaemia from a beta cell allograft. Diabetologia 52, 1369–1380 (2009).
The Diabetes Control and Complications Trial Research Group. Effect of intensive therapy on residual beta-cell function in patients with type 1 diabetes in the diabetes control and complications trial: a randomized, controlled trial. Ann. Intern. Med. 128, 517–523 (1998).
Shalev, A. et al. Oligonucleotide microarray analysis of intact human pancreatic islets: identification of glucose-responsive genes and a highly regulated TGFbeta signaling pathway. Endocrinology 143, 3695–3698 (2002).
Chen, J. et al. Thioredoxin-interacting protein deficiency induces Akt/Bcl-xL signaling and pancreatic beta cell mass and protects against diabetes. FASEB J. 22, 3581–3594 (2008).
Chen, J., Saxena, G., Mungrue, I. N., Lusis, A. J. & Shalev, A. Thioredoxin-interacting protein: a critical link between glucose toxicity and beta cell apoptosis. Diabetes 57, 938–944 (2008).
Minn, A. H., Hafele, C. & Shalev, A. Thioredoxin-interacting protein is stimulated by glucose through a carbohydrate response element and induces beta-cell apoptosis. Endocrinology 146, 2397–2405 (2005).
Minn, A. H. et al. Gene expression profiling in INS-1 cells overexpressing thioredoxin-interacting protein. Biochem. Biophys. Res. Commun. 336, 770–778 (2005).
Chen, J., Cha-Molstad, H., Szabo, A. & Shalev, A. Diabetes induces and calcium channel blockers prevent cardiac expression of pro-apoptotic thioredoxin-interacting protein. Am. J. Physiol. Endocrinol. Metab. 296, 1133–1139 (2009).
Afzal, N. et al. Beneficial effects of verapamil in diabetic cardiomyopathy. Diabetes 37, 936–942 (1988).
Cohn, R. D. et al. Prevention of cardiomyopathy in mouse models lacking the smooth muscle sarcoglycan-sarcospan complex. J. Clin. Invest. 107, R1–R7 (2001).
Xu, G., Chen, J., Jing, G. & Shalev, A. Thioredoxin-interacting protein regulates insulin transcription through microRNA-204. Nat. Med. 19, 1141–1146 (2013).
Jo, S. et al. miR-204 controls glucagon-like peptide 1 receptor expression and agonist function. Diabetes 67, 256–264 (2018).
Zhou, R., Tardivel, A., Thorens, B., Choi, I. & Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 11, 136–140 (2010).
Yin, T., Kuo, S. C., Chang, Y. Y., Chen, Y. T. & Wang, K. K. Verapamil use is associated with reduction of newly diagnosed diabetes mellitus. J. Clin. Endocrinol. Metab. 102, 2604–2610 (2017).
Cooper-Dehoff, R. et al. Predictors of development of diabetes mellitus in patients with coronary artery disease taking antihypertensive medications (findings from the INternational VErapamil SR-Trandolapril STudy [INVEST]). Am. J. Cardiol. 98, 890–894 (2006).
Cooper-DeHoff, R. M. et al. Blood pressure control and cardiovascular outcomes in high-risk Hispanic patients: findings from the International Verapamil SR/Trandolapril Study (INVEST). Am. Heart J. 151, 1072–1079 (2006).
Busch Sorensen, M. et al. Influence of short term verapamil treatment on glucose metabolism in patients with non-insulin dependent diabetes mellitus. Eur. J. Clin. Pharmacol. 41, 401–404 (1991).
Khodneva, Y., Shalev, A., Frank, S. J., Carson, A. P. & Safford, M. M. Calcium channel blocker use is associated with lower fasting serum glucose among adults with diabetes from the REGARDS study. Diabetes Res. Clin. Pract. 115, 115–121 (2016).
Nambam, B., Bratina, N. & Schatz, D. Immune interventions for type 1 diabetes mellitus. Diabetes Technol. & Ther. 19, S74–S81 (2017).
Alhadj Ali, M.et al. Metabolic and immune effects of immunotherapy with proinsulin peptide in human new-onset type 1 diabetes. Sci. Transl. Med. 9, eaaf7779 (2017).
Herold, K. C. et al. Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. New Engl. J. Med. 346, 1692–1698 (2002).
Aronson, R. et al. Low-dose otelixizumab anti-CD3 monoclonal antibody DEFEND-1 study: results of the randomized phase III study in recent-onset human type 1 diabetes. Diabetes Care 37, 2746–2754 (2014).
Pescovitz, M. D. et al. Rituximab, B-lymphocyte depletion, and preservation of beta-cell function. New Engl. J. Med. 361, 2143–2152 (2009).
Pozzilli, P., Maddaloni, E. & Buzzetti, R. Combination immunotherapies for type 1 diabetes mellitus. Nat. Rev. Endocrinol. 11, 289–297 (2015).
Greenbaum, C. J. et al. Fall in C-peptide during first 2 years from diagnosis: evidence of at least two distinct phases from composite Type 1 Diabetes TrialNet data. Diabetes 61, 2066–2073 (2012).
Hao, W. et al. Fall in C-peptide during first 4 years from diagnosis of type 1 diabetes: variable relation to age, HbA1c, and insulin dose. Diabetes Care 39, 1664–1670 (2016).
Greenbaum, C. J. et al. Mixed-meal tolerance test versus glucagon stimulation test for the assessment of beta-cell function in therapeutic trials in type 1 diabetes. Diabetes Care 31, 1966–1971 (2008).
Palmer, J. P. et al. C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve beta-cell function: report of an ADA workshop, 21-22 October 2001. Diabetes 53, 250–264 (2004).
Lachin, J. M. et al. Sample size requirements for studies of treatment effects on beta-cell function in newly diagnosed type 1 diabetes. PLoS One 6, e26471 (2011).
Moore, C. G., Carter, R. E., Nietert, P. J. & Stewart, P. W. Recommendations for planning pilot studies in clinical and translational research. Clin. Transl. Sci. 4, 332–337 (2011).
Li, P., Stuart, E. A. & Allison, D. B. Multiple imputation: a flexible tool for handling missing data. JAMA 314, 1966–1967 (2015).
Acknowledgements
The work was supported by JDRF grant 3-SRA-2014-302-M-R to A.S. The UAB Physiology Core was supported by DRC P30DK079626, and the UAB Center for Clinical and Translational Science was supported by UL1TR001417. We thank M. Preuss for excellent administrative support during the study.
Author information
Authors and Affiliations
Contributions
F.O., T.G. and A.J.P. were responsible for patient care, MMTTs, and sample and data collection. G.X., T.B.G. and L.A.T. helped with sample preparation. P.L. provided statistical advice. F.O. and A.S. designed the studies and analyzed the results. A.S. wrote the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–4
Rights and permissions
About this article
Cite this article
Ovalle, F., Grimes, T., Xu, G. et al. Verapamil and beta cell function in adults with recent-onset type 1 diabetes. Nat Med 24, 1108–1112 (2018). https://doi.org/10.1038/s41591-018-0089-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0089-4
This article is cited by
-
Systematic immune cell dysregulation and molecular subtypes revealed by single-cell RNA-seq of subjects with type 1 diabetes
Genome Medicine (2024)
-
Regulation of autophagy by perilysosomal calcium: a new player in β-cell lipotoxicity
Experimental & Molecular Medicine (2024)
-
Phosphoinositides and intracellular calcium signaling: novel insights into phosphoinositides and calcium coupling as negative regulators of cellular signaling
Experimental & Molecular Medicine (2023)
-
Candesartan, an angiotensin-II receptor blocker, ameliorates insulin resistance and hepatosteatosis by reducing intracellular calcium overload and lipid accumulation
Experimental & Molecular Medicine (2023)
-
TXNIP: A key protein in the cellular stress response pathway and a potential therapeutic target
Experimental & Molecular Medicine (2023)