Abstract
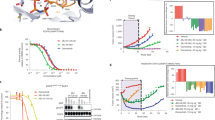
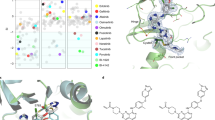
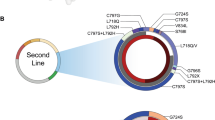
Although most activating mutations of epidermal growth factor receptor (EGFR)-mutant non–small cell lung cancers (NSCLCs) are sensitive to available EGFR tyrosine kinase inhibitors (TKIs), a subset with alterations in exon 20 of EGFR and HER2 are intrinsically resistant and lack an effective therapy. We used in silico, in vitro, and in vivo testing to model structural alterations induced by exon 20 mutations and to identify effective inhibitors. 3D modeling indicated alterations restricted the size of the drug-binding pocket, limiting the binding of large, rigid inhibitors. We found that poziotinib, owing to its small size and flexibility, can circumvent these steric changes and is a potent inhibitor of the most common EGFR and HER2 exon 20 mutants. Poziotinib demonstrated greater activity than approved EGFR TKIs in vitro and in patient-derived xenograft models of EGFR or HER2 exon 20 mutant NSCLC and in genetically engineered mouse models of NSCLC. In a phase 2 trial, the first 11 patients with NSCLC with EGFR exon 20 mutations receiving poziotinib had a confirmed objective response rate of 64%. These data identify poziotinib as a potent, clinically active inhibitor of EGFR and HER2 exon 20 mutations and illuminate the molecular features of TKIs that may circumvent steric changes induced by these mutations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bezjak, A. et al. Symptom improvement in lung cancer patients treated with erlotinib: quality of life analysis of the National Cancer Institute of Canada Clinical Trials Group Study BR.21. J. Clin. Oncol. 24, 3831–3837 (2006).
Rosell, R. et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 13, 239–246 (2012).
Wang, Y., Schmid-Bindert, G. & Zhou, C. Erlotinib in the treatment of advanced non-small cell lung cancer: an update for clinicians. Ther. Adv. Med. Oncol. 4, 19–29 (2012).
Wheatley-Price, P., Ding, K., Seymour, L., Clark, G. M. & Shepherd, F. A. Erlotinib for advanced non-small-cell lung cancer in the elderly: an analysis of the National Cancer Institute of Canada Clinical Trials Group Study BR.21. J. Clin. Oncol. 26, 2350–2357 (2008).
Zhou, C. et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 12, 735–742 (2011).
Mitsudomi, T. & Yatabe, Y. Mutations of the epidermal growth factor receptor gene and related genes as determinants of epidermal growth factor receptor tyrosine kinase inhibitors sensitivity in lung cancer. Cancer Sci. 98, 1817–1824 (2007).
Yu, H. A. et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin. Cancer Res. 19, 2240–2247 (2013).
Pao, W. et al. EGF receptor gene mutations are common in lung cancers from “never smokers” and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc. Natl Acad. Sci. USA 101, 13306–13311 (2004).
Paez, J. G. et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304, 1497–1500 (2004).
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–2139 (2004).
Cappuzzo, F. et al. Epidermal growth factor receptor gene and protein and gefitinib sensitivity in non-small-cell lung cancer. J. Natl Cancer Inst. 97, 643–655 (2005).
Costa, D. B., Kobayashi, S., Tenen, D. G. & Huberman, M. S. Pooled analysis of the prospective trials of gefitinib monotherapy for EGFR-mutant non-small cell lung cancers. Lung Cancer 58, 95–103 (2007).
Sequist, L. V. et al. First-line gefitinib in patients with advanced non-small-cell lung cancer harboring somatic EGFR mutations. J. Clin. Oncol. 26, 2442–2449 (2008).
Maemondo, M. et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N. Engl. J. Med. 362, 2380–2388 (2010).
Oxnard, G. R. et al. Natural history and molecular characteristics of lung cancers harboring EGFR exon 20 insertions. J. Thorac. Oncol. 8, 179–184 (2013).
Yasuda, H. et al. Structural, biochemical, and clinical characterization of epidermal growth factor receptor (EGFR) exon 20 insertion mutations in lung cancer. Sci. Transl. Med. 5, 216ra177 (2013).
Arcila, M. E. et al. EGFR exon 20 insertion mutations in lung adenocarcinomas: prevalence, molecular heterogeneity, and clinicopathologic characteristics. Mol. Cancer Ther. 12, 220–229 (2013).
Yasuda, H., Kobayashi, S. & Costa, D. B. EGFR exon 20 insertion mutations in non-small-cell lung cancer: preclinical data and clinical implications. Lancet Oncol. 13, e23–e31 (2012).
Arcila, M. E. et al. Prevalence, clinicopathologic associations, and molecular spectrum of ERBB2 (HER2) tyrosine kinase mutations in lung adenocarcinomas. Clin. Cancer Res. 18, 4910–4918 (2012).
Mazières, J. et al. Lung cancer that harbors an HER2 mutation: epidemiologic characteristics and therapeutic perspectives. J. Clin. Oncol. 31, 1997–2003 (2013).
Costa, D. B. et al. Pulse afatinib for ERBB2 exon 20 insertion-mutated lung adenocarcinomas. J. Thorac. Oncol. 11, 918–923 (2016).
De Grève, J. et al. Clinical activity of afatinib (BIBW 2992) in patients with lung adenocarcinoma with mutations in the kinase domain of HER2/neu. Lung Cancer 76, 123–127 (2012).
Mazières, J. et al. Lung cancer patients with HER2 mutations treated with chemotherapy and HER2 targeted drugs: results from the EUHER2 cohort study. Ann. Oncol. 27, 281–286 (2016).
Kosaka, T. et al. Response heterogeneity of EGFR and HER2 exon 20 insertions to covalent EGFR and HER2 inhibitors. Cancer Res. 77, 2712–2721 (2017).
Hyman, D. M. et al. HER kinase inhibition in patients with HER2- and HER3-mutant cancers. Nature 554, 189–194 (2018).
Perera, S. A. et al. HER2YVMA drives rapid development of adenosquamous lung tumors in mice that are sensitive to BIBW2992 and rapamycin combination therapy. Proc. Natl Acad. Sci. USA 106, 474–479 (2009).
Wang, S. E. et al. HER2 kinase domain mutation results in constitutive phosphorylation and activation of HER2 and EGFR and resistance to EGFR tyrosine kinase inhibitors. Cancer Cell 10, 25–38 (2006).
Yang, M. et al. NSCLC harboring EGFR exon-20 insertions after the regulatory C-helix of kinase domain responds poorly to known EGFR inhibitors. Int. J. Cancer 139, 171–176 (2016).
Cha, M. Y. Antitumor activity of HM781-36B, a highly effective pan-HER inhibitor in erlotinib-resistant NSCLC and other EGFR-dependent cancer models. Int. J. Cancer 10, 2445–2454 (2012).
Cho, J. et al. Cetuximab response of lung cancer-derived EGF receptor mutants is associated with asymmetric dimerization. Cancer Res. 73, 6770–6779 (2013).
Wind, S., Schmid, M., Erhardt, J., Goeldner, R. G. & Stopfer, P. Pharmacokinetics of afatinib, a selective irreversible ErbB family blocker, in patients with advanced solid tumours. Clin. Pharmacokinet. 52, 1101–1109 (2013).
Li, D. et al. BIBW2992, an irreversible EGFR/HER2 inhibitor highly effective in preclinical lung cancer models. Oncogene 27, 4702–4711 (2008).
Thress, K. S. et al. Acquired EGFR C797S mutation mediates resistance to AZD9291 in non-small cell lung cancer harboring EGFR T790M. Nat. Med. 21, 560–562 (2015).
Chabon, J. J. et al. Circulating tumour DNA profiling reveals heterogeneity of EGFR inhibitor resistance mechanisms in lung cancer patients. Nat. Commun. 7, 11815 (2016).
Nilsson, M. B. Stress hormones promote EGFR inhibitor resistance in NSCLC: implications for combinations with beta blockers. Sci. Transl. Med. 9, eaao4307 (2017).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2017. CA Cancer J. Clin. 67,7–30 (2017).
Kim D.-W. et al. Phase I studies of HM781-36B, an irreversible pan-HER tyrosine kinase inhibitor (TKI) in patients with advanced solid tumor and the therapeutic potential in patients with advanced non-small cell lung cancer (NSCLC). J. Thorac. Oncol. 8, S607 (2013).
Kim, T. M. et al. A phase I study of HM781–36B, a novel pan-HER inhibitor, in patients (pts) with advanced solid tumors. J. Clin. Oncol. 30, 3076 (2012).
Noh, Y. H. et al. Population pharmacokinetics of HM781-36 (poziotinib), pan-human EGF receptor (HER) inhibitor, and its two metabolites in patients with advanced solid malignancies. Cancer Chemother. Pharmacol. 75, 97–109 (2015).
Han, J. Y. et al. A phase II study of poziotinib in patients with epidermal growth factor receptor (EGFR)-mutant lung adenocarcinoma who have acquired resistance to egfr-tyrosine kinase inhibitors. Cancer Res. Treat. 49, 10–19 (2017).
Planchard, D. et al. Osimertinib Western and Asian clinical pharmacokinetics in patients and healthy volunteers: implications for formulation, dose, and dosing frequency in pivotal clinical studies. Cancer Chemother. Pharmacol. 77, 767–776 (2016).
Jänne, P. A. et al. Phase I dose-escalation study of the pan-HER inhibitor, PF299804, in patients with advanced malignant solid tumors. Clin. Cancer Res. 17, 1131–1139 (2011).
Kourie, H. R., Chaix, M., Gombos, A., Aftimos, P. & Awada, A. Pharmacodynamics, pharmacokinetics and clinical efficacy of neratinib in HER2-positive breast cancer and breast cancer with HER2 mutations. Expert Opin. Drug Metab. Toxicol. 12, 947–957 (2016).
Ito, Y. et al. Safety, efficacy and pharmacokinetics of neratinib (HKI-272) in Japanese patients with advanced solid tumors: a Phase 1 dose-escalation study. Jpn. J. Clin. Oncol. 42, 278–286 (2012).
Sequist, L. V. et al. Neratinib, an irreversible pan-ErbB receptor tyrosine kinase inhibitor: results of a phase II trial in patients with advanced non-small-cell lung cancer. J. Clin. Oncol. 28, 3076–3083 (2010).
Yang, J. C. et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 16, 830–838 (2015).
Kris, M. G. et al. Targeting HER2 aberrations as actionable drivers in lung cancers: phase II trial of the pan-HER tyrosine kinase inhibitor dacomitinib in patients with HER2-mutant or amplified tumors. Ann. Oncol. 26, 1421–1427 (2015).
Tsigelny, I. F. et al. Molecular determinants of drug-specific sensitivity for epidermal growth factor receptor (EGFR) exon 19 and 20 mutants in non-small cell lung cancer. Oncotarget 6, 6029–6039 (2015).
Wheler, J. et al. Revisiting clinical trials using EGFR inhibitor-based regimens in patients with advanced non-small cell lung cancer: a retrospective analysis of an MD Anderson Cancer Center phase I population. Oncotarget 4, 772–784 (2013).
Wheler, J. J. et al. Combining erlotinib and cetuximab is associated with activity in patients with non-small cell lung cancer (including squamous cell carcinomas) and wild-type EGFR or resistant mutations. Mol. Cancer Ther. 12, 2167–2175 (2013).
Shen, X. et al. A systematic analysis of the resistance and sensitivity of HER2YVMA receptor tyrosine kinase mutant to tyrosine kinase inhibitors in HER2-positive lung cancer. J. Recept. Signal Transduct. Res. 36, 89–97 (2016).
Davies, K. D. et al. Resistance to ROS1 inhibition mediated by EGFR pathway activation in non-small cell lung cancer. PLoS One. 8, e82236 (2013).
Hong, J. et al. Anthrax edema toxin inhibits endothelial cell chemotaxis via Epac and Rap1. J. Biol. Chem. 282, 19781–19787 (2007).
Acknowledgements
The authors wish to thank the patients and their families for participation in this study and Spectrum Pharmaceuticals for providing the drug used in the compassionate-use protocol. The authors also wish to thank J.S. (a patient) for efforts in initiating the poziotinib trial. This work was supported by the Lung SPORE grant P50 CA070907 and P50 CA196530 (J.V.H.); the Lung Cancer Research Fund (Y.Y.E.); generous philanthropic contributions to the University of Texas MD Anderson Lung Cancer Moon Shots Program (J.V.H.); NIH R01 CA190628 (J.V.H.), NIH Cancer Center Support Grant P30 CA016672 (J.V.H.), the Rexanna Foundation for Fighting Lung Cancer (J.V.H.), the Exon 20 Group (J.V.H.), the University of Texas MD Anderson Cancer Center Bruton Endowed Chair in Tumor Biology (J.V.H.), the Stading Fund for EGFR inhibitor resistance (J.V.H.), the Hallman fund (J.V.H.), and the Fox Lung EGFR Inhibitor Fund (J.V.H.). Additional support was provided from the Christine J. Burge Endowment for Lung Cancer Research at the University of Colorado Cancer Center, the Burge family, the Miramont Cares foundation (R.C.D.), a fellowship from the Italian Association for Cancer Research (A.T.), and Cancer Prevention Research Institute of Texas DP150086, National Science Foundation CHE-1411859, and National Institute of General Medical Sciences GM070737 (S.Z.). The clinical study was supported by Spectrum Pharmaceuticals.
Author information
Authors and Affiliations
Contributions
J.P.R., Y.Y.E., and J.V.H. coordinated the study. J.P.R., Y.Y.E., Z.T., A.P., H.S., S.Z., S. Liu. S. Li, T.C., A.E-B., A.T.L., and J.V.H. designed and/or performed experiments. J.P.R., Y.Y.E., Z.T., B.W.C., S.Z., E.R., A.T., K.P., K.-K.W, R.C.D., and J.V.H. interpreted data. A.T., S.B.G., and K.P. validated and provided the YUL0019 cell line. J.R.B. referred two patients described in the study. V.P., C.L., M.A., Y.Y.E., and J.V.H. treated patients on the clinical study. J.P.R., M.B.N, Y.Y.E., and J.V.H wrote the manuscript. All authors edited and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
J.P.R., M.B.N., and J.V.H. have filed patent applications under the Patent Cooperation Treaty and in Taiwan. J.V.H. has had grant or research support from AstraZeneca, Bayer, and GlaxoSmithKline and has served on advisory committees for AstraZeneca, Boehringer Ingelheim, Exelixis, Genentech, GSK, Lilly, Novartis, Spectrum, and Synta. R.C.D. has licensing fees, honorarium, and travel expenses from Ariad Pharmaceuticals, has a Sponsored Research Agreement from Threshold Pharmaceuticals, and has served as an advisory Board member for AstraZeneca.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Figures and Tables
Supplementary Figures 1-10 and Supplementary Tables 1-4
Rights and permissions
About this article
Cite this article
Robichaux, J.P., Elamin, Y.Y., Tan, Z. et al. Mechanisms and clinical activity of an EGFR and HER2 exon 20–selective kinase inhibitor in non–small cell lung cancer. Nat Med 24, 638–646 (2018). https://doi.org/10.1038/s41591-018-0007-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-018-0007-9
This article is cited by
-
Oncogenic alterations in advanced NSCLC: a molecular super-highway
Biomarker Research (2024)
-
Novel therapeutic strategies for rare mutations in non-small cell lung cancer
Scientific Reports (2024)
-
EGFR and ERBB2 Exon 20 Insertion Mutations in Chinese Non-small Cell Lung Cancer Patients: Pathological and Molecular Characterization, and First-Line Systemic Treatment Evaluation
Targeted Oncology (2024)
-
The development of amivantamab for the treatment of non-small cell lung cancer
Respiratory Research (2023)
-
A real-world study of the efficacy and safety of furmonertinib for patients with non-small cell lung cancer with EGFR exon 20 insertion mutations
Journal of Cancer Research and Clinical Oncology (2023)