Abstract
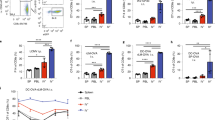
Increasing evidence indicates close interaction between immune cells and the brain, revising the traditional view of the immune privilege of the brain. However, the specific mechanisms by which immune cells promote normal neural function are not entirely understood. Mucosal-associated invariant T cells (MAIT cells) are a unique type of innate-like T cell with molecular and functional properties that remain to be better characterized. In the present study, we report that MAIT cells are present in the meninges and express high levels of antioxidant molecules. MAIT cell deficiency in mice results in the accumulation of reactive oxidative species in the meninges, leading to reduced expression of junctional protein and meningeal barrier leakage. The presence of MAIT cells restricts neuroinflammation in the brain and preserves learning and memory. Together, our work reveals a new functional role for MAIT cells in the meninges and suggests that meningeal immune cells can help maintain normal neural function by preserving meningeal barrier homeostasis and integrity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
ScRNA-seq data have been deposited in the National Center for Biotechnology Information’s Gene Expression Omnibus under accession nos. GSE189656 and GSE189661. Source data are provided with this paper.
References
Galea, I., Bechmann, I. & Perry, V. H. What is immune privilege (not)? Trends Immunol. 28, 12–18 (2007).
Buckley, M. W. & McGavern, D. B. Immune dynamics in the CNS and its barriers during homeostasis and disease. Immunol. Rev. 306, 58–75 (2022).
Alves de Lima, K. et al. Meningeal gammadelta T cells regulate anxiety-like behavior via IL-17a signaling in neurons. Nat. Immunol. 21, 1421–1429 (2020).
Brombacher, T. M. et al. IL-13-mediated regulation of learning and memory. J. Immunol. 198, 2681–2688 (2017).
Derecki, N. C. et al. Regulation of learning and memory by meningeal immunity: a key role for IL-4. J. Exp. Med. 207, 1067–1080 (2010).
Fung, I. T. H. et al. Activation of group 2 innate lymphoid cells alleviates aging-associated cognitive decline. J. Exp. Med. 217, e20190915 (2020).
Herz, J. et al. GABAergic neuronal IL-4R mediates T cell effect on memory. Neuron 109, 3609–3618.e3609 (2021).
Ribeiro, M. et al. Meningeal gammadelta T cell-derived IL-17 controls synaptic plasticity and short-term memory. Sci. Immunol. 4, eaay5199 (2019).
Derk, J., Jones, H. E., Como, C., Pawlikowski, B. & Siegenthaler, J. A. Living on the edge of the CNS: meninges cell diversity in health and disease. Front. Cell. Neurosci. 15, 703944 (2021).
Horng, S. et al. Astrocytic tight junctions control inflammatory CNS lesion pathogenesis. J. Clin. Invest. 127, 3136–3151 (2017).
Roth, T. L. et al. Transcranial amelioration of inflammation and cell death after brain injury. Nature 505, 223–228 (2014).
Hinks, T. S. C. & Zhang, X. W. MAIT cell activation and functions. Front. Immunol. 11, 1014 (2020).
Kjer-Nielsen, L. et al. MR1 presents microbial vitamin B metabolites to MAIT cells. Nature 491, 717–723 (2012).
Treiner, E. et al. Selection of evolutionarily conserved mucosal-associated invariant T cells by MR1. Nature 422, 164–169 (2003).
Legoux, F. et al. Microbial metabolites control the thymic development of mucosal-associated invariant T cells. Science 366, 494–499 (2019).
Constantinides, M. G. et al. MAIT cells are imprinted by the microbiota in early life and promote tissue repair. Science 366, eaax6624 (2019).
Rudak, P. T., Choi, J. & Haeryfar, S. M. M. MAIT cell-mediated cytotoxicity: roles in host defense and therapeutic potentials in infectious diseases and cancer. J. Leukoc. Biol. 104, 473–486 (2018).
Leng, T. et al. TCR and inflammatory signals tune human MAIT cells to exert specific tissue repair and effector functions. Cell Rep. 28, 3077–3091.e3075 (2019).
Lamichhane, R. et al. TCR- or cytokine-activated CD8+ mucosal-associated invariant T cells are rapid polyfunctional effectors that can coordinate immune responses. Cell Rep. 28, 3061–3076.e3065 (2019).
Hinks, T. S. C. et al. Activation and in vivo evolution of the MAIT cell transcriptome in mice and humans reveals tissue repair functionality. Cell Rep. 28, 3249–3262 e3245 (2019).
Legoux, F., Salou, M. & Lantz, O. MAIT cell development and functions: the microbial connection. Immunity 53, 710–723 (2020).
Ye, L. et al. Mucosal-associated invariant T cells restrict allergic airway inflammation. J. Allergy Clin. Immunol. 145, 1469–1473.e1464 (2020).
Pomaznoy, M. et al. Quantitative and qualitative perturbations of CD8+ MAITs in healthy Mycobacterium tuberculosis-infected individuals. Immunohorizons 4, 292–307 (2020).
Steinbrenner, H., Speckmann, B. & Klotz, L. O. Selenoproteins: antioxidant selenoenzymes and beyond. Arch. Biochem. Biophys. 595, 113–119 (2016).
Mukherjee, C. et al. Oligodendrocytes provide antioxidant defense function for neurons by secreting ferritin heavy chain. Cell Metab. 32, 259–272.e210 (2020).
Kumar, S. & Gupta, S. Thymosin beta 4 prevents oxidative stress by targeting antioxidant and anti-apoptotic genes in cardiac fibroblasts. PLoS ONE 6, e26912 (2011).
Li, X., Wang, L. & Chen, C. Effects of exogenous thymosin beta4 on carbon tetrachloride-induced liver injury and fibrosis. Sci. Rep. 7, 5872 (2017).
Shah, R. et al. Thymosin beta4 prevents oxidative stress, inflammation, and fibrosis in ethanol- and LPS-induced liver injury in mice. Oxid. Med. Cell. Longev. 2018, 9630175 (2018).
Yaman, O. M. et al. Protective effect of thymosin beta4 against abdominal aortic ischemia–reperfusion-induced acute lung injury in rats. Medicine 55, 187 (2019).
Kashiba, M. et al. Prosaposin regulates coenzyme Q10 levels in HepG2 cells, especially those in mitochondria. J. Clin. Biochem. Nutr. 55, 85–89 (2014).
Kashiba, M., Terashima, M., Sagawa, T., Yoshimura, S. & Yamamoto, Y. Prosaposin knockdown in Caco-2 cells decreases cellular levels of coenzyme Q10 and ATP, and results in the loss of tight junction barriers. J. Clin. Biochem. Nutr. 60, 81–85 (2017).
Tian, R. et al. Genome-wide CRISPRi/a screens in human neurons link lysosomal failure to ferroptosis. Nat. Neurosci. 24, 1020–1034 (2021).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Jones, H. E., Abrams, K. A. & Siegenthaler, J. A. Techniques for visualizing fibroblast-vessel interactions in the developing and adult CNS. Neurophotonics 9, 021911 (2022).
Banks, W. A. et al. Lipopolysaccharide-induced blood-brain barrier disruption: roles of cyclooxygenase, oxidative stress, neuroinflammation, and elements of the neurovascular unit. J. Neuroinflam. 12, 223 (2015).
Chen, Y. & Colonna, M. Microglia in Alzheimer’s disease at single-cell level. Are there common patterns in humans and mice? J. Exp. Med. 218, e20202717 (2021).
Ocanas, S.R. et al. Minimizing the ex vivo confounds of cell-isolation techniques on transcriptomic and translatomic profiles of purified microglia. eNeuro https://doi.org/10.1523/ENEURO.0348-21.2022 (2022).
Marsh, S. E. et al. Dissection of artifactual and confounding glial signatures by single-cell sequencing of mouse and human brain. Nat. Neurosci. 25, 306–316 (2022).
Ajami, B., Bennett, J. L., Krieger, C., McNagny, K. M. & Rossi, F. M. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nat. Neurosci. 14, 1142–1149 (2011).
Varvel, N. H. et al. Infiltrating monocytes promote brain inflammation and exacerbate neuronal damage after status epilepticus. Proc. Natl Acad. Sci. USA 113, E5665–E5674 (2016).
Filiano, A. J. et al. Unexpected role of interferon-gamma in regulating neuronal connectivity and social behaviour. Nature 535, 425–429 (2016).
Koay, H. F. et al. A divergent transcriptional landscape underpins the development and functional branching of MAIT cells. Sci. Immunol. 4, eaay6039 (2019).
Voet, S., Prinz, M. & van Loo, G. Microglia in central nervous system inflammation and multiple sclerosis pathology. Trends Mol. Med. 25, 112–123 (2019).
Augusto-Oliveira, M. et al. What do microglia really do in healthy adult brain? Cells 8, 1293 (2019).
Bachiller, S. et al. Microglia in neurological diseases: a road map to brain-disease dependent-inflammatory response. Front. Cell. Neurosci. 12, 488 (2018).
Corbett, A. J. et al. T-cell activation by transitory neo-antigens derived from distinct microbial pathways. Nature 509, 361–365 (2014).
Mastorakos, P. et al. Temporally distinct myeloid cell responses mediate damage and repair after cerebrovascular injury. Nat. Neurosci. 24, 245–258 (2021).
Acknowledgements
We thank A. Rabson, H. Ching Lin, P. Jiang and Z. Pang for critical discussion and technical help. This work was supported by the US NIH (grant nos. R01HL137813, R01AG057782 and R01HL155021 to Q.Y.), the NIH Intramural Research Programs (to D.B.M., Y.B. and A.B.) and support to the Child Health Institute of New Jersey from the Robert Wood Johnson Foundation (grant no. 74260).
Author information
Authors and Affiliations
Contributions
Y.Z., D.B.M. and Q.Y. conceived the ideas and designed the experiments. Y.Z. and Q.Y. performed the experiments, with the help of J.T.B., K.S., E.X., A.H., J.C., W.S., D.B.S.’A. and D.B.M. M.L. and A.B. provided Fig. 2b and Table 1. V.M.L. and Y.B. provided Fig. 2f,g,h. S.D.’S. developed and optimized in vitro MAIT cell culture methods for Fig. 3. Y.Z. and Q.Y. wrote the manuscript with the help of D.B.M. All authors reviewed, edited and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks Robyn Klein, Paul Klenerman and Julie Siegenthaler for their contribution to the peer review of this work. L. A. Dempsey was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Expression of transcription factors by meningeal MAIT cells.
Feature plots depicting expression for the indicated genes by scRNA-seq analysis with meningeal MAIT cells and CD4 and CD8 T cells in 7-month-old Mr1+/+ and Mr1−/− mice. Data are from 6 mice pooled per group.
Extended Data Fig. 2 Validation of adoptive transfer of MAIT cells.
a, Representative flow cytometry profiles depict gating strategies to sort donor MAIT cells for adoptive transfer experiments. Meningeal tissue was pooled, with tissue from 5 mice in 1 ml of Liberase digestion buffer. Digested tissue from up to 60 mice was pooled per sample, followed by staining with MR1 tetramers and surface antibodies to identify MAIT cells. Events of around 8% of the sample (equivalent to around 5 mice) were collected in the representative flow cytometry profiles. CD3 and TCRβ antibodies were not included for purification of MAIT cells for adoptive transfer. Donor MAIT cells were identified as CD45+ Thy1.2hiIL-18RhiMR1-tetramer+cells. b, Representative profiles of MAIT cells in the meninges of 7-month-old Mr1−/− mice that received adoptive transfer of PBS or MAIT cells. Plots were pre-gated on CD45+CD11b−B220−NK1.1- Thy1.2+ cells. c, Numbers of meningeal MAIT cells per mouse in the meninges and in the small intestinal laminal propria (SILP) of recipient mice at the indicated time points after adoptive transfer. d, Percentages of MAIT cells in the total T cell subset in the dura/arachnoid meningeal tissue obtained from inner calvaria and in the leptomeninges of recipient mice at 6 months post adoptive transfer. Error bars = Mean ± SE. Data are from 3 independent experiments, 2-5 recipient mice pooled in one sample for each experiment (b–d). Each data point indicates one independent experiment (c, d).
Extended Data Fig. 3 MAIT cells repress ROS accumulation and preserve expression of junctional molecules in dura/arachnoid meningeal tissue isolated from inner calvaria.
a, Representative profile of Reactive Oxygen Species (ROS) in the dura/arachnoid meningeal tissue obtained from the inner calvaria of 7-month-old Mr1+/+ and Mr1−/− mice with intra-cisterna magna administration of CellROX Green Reagent. b, Numbers of ROS positive cells in the meningeal tissue. c, Representative imaging profile of ROS in the dura/arachnoid meningeal tissue of 7-month-old Mr1−/− mice that received adoptive transfer of PBS or MAIT cells. d, Numbers of ROS + cells in the meningeal tissue in Mr1−/− mice that received adoptive transfer of PBS or MAIT cells. e, Representative imaging profile depicting expression of CD31 and E-cadherin in dura/arachnoid meningeal tissue in 7-month-old Mr1+/+ and Mr1−/− mice, with i.c.m. injection of fluorescence conjugated CD31 and E-cadherin antibodies. f, Fluorescence intensity of E-cadherin in the meninges of Mr1+/+ and Mr1−/− mice. g, Representative imaging profile depicting expression of CD31 and E-cadherin in dura/arachnoid meningeal tissue in 7-month-old Mr1−/− mice that received adoptive transfer of PBS or MAIT cells. h, Fluorescence intensity of E-cadherin in the meninges of Mr1−/− mice that received adoptive transfer of PBS or MAIT cells. i, Representative profile of ROS detection in meninges of Mr1−/− mice treated with Glutathione or PBS control. j, Numbers of ROS positive cells in 7-month-old Mr1−/− mice treated with Glutathione or PBS control. k, Representative imaging profile depicting expression of CD31 and E-cadherin in dura/arachnoid meningeal tissue in 7-month-old Mr1−/− mice treated with Glutathione or PBS control. l, Fluorescence intensity of E-cadherin in dura/arachnoid meningeal tissue in Mr1−/− mice treated with Glutathione or PBS control. Error bars = Mean ± SE. Data are from 6 mice per group, representative of 2 independent experiments. **P < 0.01 using two-sided Student’s t-test; exact P values are provided in the source data.
Extended Data Fig. 4 Mr1−/− mice do not exhibit significant defects in BBB integrity.
Na-fluorescein concentrations in the serum and brain of 7-month-old mice with intraperitoneal administration of Na-fluorescein. Error bars = Mean ± SE; Data are from 6 mice per group, 2 independent experiments. **P < 0.01, n.s = not statistically significant (P > 0.05) using two-sided ANOVA with Dunnett’s correction; exact P values are provided in the source data.
Extended Data Fig. 5 Myeloid cells and astrocytes in the hippocampus of Mr1−/− mice.
a, UMAP analysis for the entire population of live cells isolated from the hippocampus of 7-month-old Mr1+/+ and Mr1−/− mice. b, Representative flow cytometry profiles for monocytes and neutrophils in the hippocampus of 7-month-old Mr1+/+ and Mr1−/− mice. Lungs from wild-type mice were used as a positive control for gating monocytes and neutrophils. c, Numbers of monocytes and neutrophils in the hippocampus of 7-month-old Mr1+/+ and Mr1−/− mice. d, Representative profile of immunofluorescence staining of GFAP in the HP DG region of 7-month-old Mr1+/+ and Mr1−/− mice. e, Numbers of astrocytes (GFAP+ cells) in the HP DG regions. f, UMAP analysis of astrocytes in the hippocampus of 7-month-old Mr1+/+ and Mr1−/− mice. scRNA-seq was performed with FACS-sorted live cells in the hippocampus. Gating strategy to sort live cells for scRNA-seq is shown in Supplementary Fig. 2. UMAP profiles for total live cells in the hippocampus are provide in a. The data subset for microglia was created using the ‘subset’ function of Seurat. g, Feature plots depict expression of Il33 and Gfap in astrocytes. h, Genes highly expressed by each astrocyte subset. i. Proportions of each astrocyte subset in Mr1+/+ and Mr1−/− mice. j, Differentially expressed genes (DEG) in the total astrocyte population and in each astrocyte subset, between Mr1+/+ and Mr1−/− mice. k, Feature plots depict expression of Uba52 in astrocytes. Error bars = Mean ± SE. Data are from 6 mice per group, pooled from two independent experiments (b-e), or are from 6 mice pooled per group (a, f-k). *P < 0.05, **P < 0.01, n.s = not statistically significant (P > 0.05) using two-sided Student’s t-test (c, e), or two-sided Wilcoxon rank sum test (k); exact P values are provided in the source data (c, e) or Supplementary Table 11.
Extended Data Fig. 6 Behavior tests results of Mr1−/− mice and control wild-type mice.
a, Total distance travelled, and percentages of time spent in the central zone, the corner, and the peripheral zone in Open Field test, by 7-month-old Mr1+/+ and Mr1−/− mice. b, Percentages of time spent in the closed arm in Elevated Plus Maze test, by 7-month-old Mr1+/+ and Mr1−/− mice. c, Percentages of time spent in the novel arms in Y-maze test, by 7-month-old Mr1−/− mice that received adoptive transfer of control CD4/CD8 T cells or PBS control. d, Escape latency in the 4 day training period of Water Maze test, by 7-month-old Mr1−/− mice that received adoptive transfer of control CD4/CD8 T cells or PBS control. e, Entries to the target zone, latency to the target zone, and Percentage of time spent in the target quadrant, in day 5 probe trial of Water Maze Test, by 7-month-old Mr1−/− mice that received adoptive transfer of control CD4/CD8 T cells or PBS control. f, Fluorescence intensities of E-cadherin in the leptomeninges of 7-month-old Mr1−/− mice that received adoptive transfer of control CD4/CD8 T cells or PBS control. g, Fluorescence intensities of E-cadherin in the leptomeninges of 7-month-old Mr1−/− mice that received adoptive transfer of MAIT T cells or PBS control. h, Percentages of time spent in the novel (N) and familiar (F) arms in Y-maze test, by young 5−week-old Mr1+/+ and Mr1−/− mice. i, Escape latency in Water Maze 4-day training, by 5-week-old Mr1+/+ and Mr1−/− mice. Error bars = Mean ± SE. Data are from 9 mice per group (a–e) or 5 mice per group (f, g), or 10 mice per group (h, i), representative of 2 independent experiments. *P < 0.05, n.s = not statistically significant (P > 0.05) using two-sided Student’s t-test; exact P values are provided in the source data.
Supplementary information
Supplementary Information
Supplementary Figs. 1 and 2.
Supplementary Table
Supplementary Tables 1–13.
Source data
Source Data Fig. 1
Numerical data used for plots.
Source Data Fig. 3
Numerical data used for plots.
Source Data Fig. 4
Numerical data used for plots.
Source Data Fig. 5
Numerical data used for plots.
Source Data Fig. 6
Numerical data used for plots.
Source Data Fig. 7
Numerical data used for plots.
Source Data Extended Data Fig. 2
Numerical data used for plots.
Source Data Extended Data Fig. 3
Numerical data used for plots.
Source Data Extended Data Fig. 4
Numerical data used for plots.
Source Data Extended Data Fig. 5
Numerical data used for plots.
Source Data Extended Data Fig. 6
Numerical data used for plots.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Bailey, J.T., Xu, E. et al. Mucosal-associated invariant T cells restrict reactive oxidative damage and preserve meningeal barrier integrity and cognitive function. Nat Immunol 23, 1714–1725 (2022). https://doi.org/10.1038/s41590-022-01349-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-022-01349-1
This article is cited by
-
Role of meningeal immunity in brain function and protection against pathogens
Journal of Inflammation (2024)
-
Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases
Signal Transduction and Targeted Therapy (2024)
-
Meningeal interleukin-17-producing T cells mediate cognitive impairment in a mouse model of salt-sensitive hypertension
Nature Neuroscience (2024)
-
MR1 antigen presentation to MAIT cells and other MR1-restricted T cells
Nature Reviews Immunology (2024)
-
Same yet different — how lymph node heterogeneity affects immune responses
Nature Reviews Immunology (2024)