Abstract
Toxoplasma gondii is a common protozoan parasite that infects up to one third of the world’s population. Notably, very little is known about innate immune sensing mechanisms for this obligate intracellular parasite by human cells. Here, by applying an unbiased biochemical screening approach, we show that human monocytes recognized the presence of T. gondii infection by detecting the alarmin S100A11 protein, which is released from parasite-infected cells via caspase-1-dependent mechanisms. S100A11 induced a potent chemokine response to T. gondii by engaging its receptor RAGE, and regulated monocyte recruitment in vivo by inducing expression of the chemokine CCL2. Our experiments reveal a sensing system for T. gondii by human cells that is based on the detection of infection-mediated release of S100A11 and RAGE-dependent induction of CCL2, a crucial chemokine required for host resistance to the parasite.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The materials, data, and any associated protocols that support the findings of this study are available from the authors upon reasonable request. All RNA-seq data generated in this study have been deposited in the Gene Expression Omnibus (GEO) under accession code GSE119835.
References
Iwasaki, A. & Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 16, 343–353 (2015).
Man, S. M., Karki, R. & Kanneganti, T. D. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol. Rev. 277, 61–75 (2017).
Kawai, T. & Akira, S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat. Immunol. 11, 373–384 (2010).
Yarovinsky, F. Innate immunity to Toxoplasma gondii infection. Nat. Rev. Immunol. 14, 109–121 (2014).
Pifer, R. & Yarovinsky, F. Innate responses to Toxoplasma gondii in mice and humans. Trends. Parasitol. 27, 388–393 (2011).
Yarovinsky, F. et al. TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science 308, 1626–1629 (2005).
Plattner, F. et al. Toxoplasma profilin is essential for host cell invasion and TLR11-dependent induction of an interleukin-12 response. Cell Host Microbe 3, 77–87 (2008).
Raetz, M. et al. Cooperation of TLR12 and TLR11 in the IRF8-dependent IL-12 response to Toxoplasma gondii profilin. J. Immunol. 191, 4818–4827 (2013).
Koblansky, A. A. et al. Recognition of profilin by Toll-like receptor 12 is critical for host resistance to Toxoplasma gondii. Immunity 38, 119–130 (2013).
Andrade, W. A. et al. Combined action of nucleic acid-sensing Toll-like receptors and TLR11/TLR12 heterodimers imparts resistance to Toxoplasma gondii in mice. Cell Host Microbe 13, 42–53 (2013).
Neal, L. M. & Knoll, L. J. Toxoplasma gondii profilin promotes recruitment of Ly6Chi CCR2+ inflammatory monocytes that can confer resistance to bacterial infection. PLoS Pathog. 10, e1004203 (2014).
Dupont, C. D., Christian, D. A. & Hunter, C. A. Immune response and immunopathology during toxoplasmosis. Semin. Immunopathol. 34, 793–813 (2012).
Roach, J. C. et al. The evolution of vertebrate Toll-like receptors. Proc. Natl Acad. Sci. USA 102, 9577–9582 (2005).
Muller, U. B. & Howard, J. C. The impact of Toxoplasma gondii on the mammalian genome. Curr. Opin. Microbiol. 32, 19–25 (2016).
Debierre-Grockiego, F. et al. Activation of TLR2 and TLR4 by glycosylphosphatidylinositols derived from Toxoplasma gondii. J. Immunol. 179, 1129–1137 (2007).
Ewald, S. E., Chavarria-Smith, J. & Boothroyd, J. C. NLRP1 is an inflammasome sensor for Toxoplasma gondii. Infect. Immun. 82, 460–468 (2014).
Clay, G. M., Sutterwala, F. S. & Wilson, M. E. NLR proteins and parasitic disease. Immunol. Res. 59, 142–152 (2014).
Black, M. W. & Boothroyd, J. C. Lytic cycle of Toxoplasma gondii. Microbiol. Mol. Biol. Rev. 64, 607–623 (2000).
Denkers, E. Y., Schneider, A. G., Cohen, S. B. & Butcher, B. A. Phagocyte responses to protozoan infection and how Toxoplasma gondii meets the challenge. PLoS Pathog. 8, e1002794 (2012).
Hakimi, M. A., Olias, P. & Sibley, L. D. Toxoplasma effectors targeting host signaling and transcription. Clin. Microbiol. Rev. 30, 615–645 (2017).
Gay, G. et al. Toxoplasma gondii TgIST co-opts host chromatin repressors dampening STAT1-dependent gene regulation and IFN-γ-mediated host defenses. J. Exp. Med. 213, 1779–1798 (2016).
Olias, P., Etheridge, R. D., Zhang, Y., Holtzman, M. J. & Sibley, L. D. Toxoplasma effector recruits the Mi-2/NuRD complex to repress STAT1 transcription and block IFN-γ-dependent gene expression. Cell Host Microbe 20, 72–82 (2016).
Naor, A. et al. MYR1-dependent effectors are the major drivers of a host cell’s early response to Toxoplasma, including counteracting MYR1-independent effects. MBio 9, e02401-17 (2018).
Koshy, A. A. et al. Toxoplasma co-opts host cells it does not invade. PLoS Pathog. 8, e1002825 (2012).
Tosh, K. W. et al. The IL-12 response of primary human dendritic cells and monocytes to Toxoplasma gondii is stimulated by phagocytosis of live parasites rather than host cell invasion. J. Immunol. 196, 345–356 (2016).
Christian, D. A. et al. Use of transgenic parasites and host reporters to dissect events that promote interleukin-12 production during toxoplasmosis. Infect. Immun. 82, 4056–4067 (2014).
Melo, M. B., Jensen, K. D. & Saeij, J. P. Toxoplasma gondii effectors are master regulators of the inflammatory response. Trends. Parasitol. 27, 487–495 (2011).
Donato, R. S100: a multigenic family of calcium-modulated proteins of the EF-hand type with intracellular and extracellular functional roles. Int. J. Biochem. Cell. Biol. 33, 637–668 (2001).
Donato, R. et al. Functions of S100 proteins. Curr. Mol. Med. 13, 24–57 (2013).
Ulas, T. et al. S100-alarmin-induced innate immune programming protects newborn infants from sepsis. Nat. Immunol. 18, 622–632 (2017).
Gross, S. R., Sin, C. G., Barraclough, R. & Rudland, P. S. Joining S100 proteins and migration: for better or for worse, in sickness and in health. Cell. Mol. Life Sci. 71, 1551–1579 (2014).
Kierdorf, K. & Fritz, G. RAGE regulation and signaling in inflammation and beyond. J. Leukoc. Biol. 94, 55–68 (2013).
Leclerc, E., Fritz, G., Vetter, S. W. & Heizmann, C. W. Binding of S100 proteins to RAGE: an update. Biochim. Biophys. Acta 1793, 993–1007 (2009).
Koch, M. et al. Structural basis for ligand recognition and activation of RAGE. Structure 18, 1342–1352 (2010).
Penumutchu, S. R., Chou, R. H. & Yu, C. Structural insights into calcium-bound S100P and the V domain of the RAGE complex. PLoS One 9, e103947 (2014).
Hori, M. et al. Mycalolide-B, a novel and specific inhibitor of actomyosin ATPase isolated from marine sponge. FEBS Lett. 322, 151–154 (1993).
Lavine, M. D. & Arrizabalaga, G. Exit from host cells by the pathogenic parasite Toxoplasma gondii does not require motility. Eukaryot. Cell 7, 131–140 (2008).
Dunay, I. R. et al. Gr1(+) inflammatory monocytes are required for mucosal resistance to the pathogen Toxoplasma gondii. Immunity 29, 306–317 (2008).
Serbina, N. V., Jia, T., Hohl, T. M. & Pamer, E. G. Monocyte-mediated defense against microbial pathogens. Annu. Rev. Immunol. 26, 421–452 (2008).
Shi, C. & Pamer, E. G. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 11, 762–774 (2011).
Kim, Y. G. et al. The Nod2 sensor promotes intestinal pathogen eradication via the chemokine CCL2-dependent recruitment of inflammatory monocytes. Immunity 34, 769–780 (2011).
Gov, L., Schneider, C. A., Lima, T. S., Pandori, W. & Lodoen, M. B. NLRP3 and potassium efflux drive rapid IL-1β release from primary human monocytes during Toxoplasma gondii infection. J. Immunol. 199, 2855–2864 (2017).
Robben, P. M., LaRegina, M., Kuziel, W. A. & Sibley, L. D. Recruitment of Gr-1+ monocytes is essential for control of acute toxoplasmosis. J. Exp. Med. 201, 1761–1769 (2005).
Pifer, R., Benson, A., Sturge, C. R. & Yarovinsky, F. UNC93B1 is essential for TLR11 activation and IL-12-dependent host resistance to Toxoplasma gondii. J. Biol. Chem. 286, 3307–3314 (2011).
Saeij, J. P. et al. Toxoplasma co-opts host gene expression by injection of a polymorphic kinase homologue. Nature 445, 324–327 (2007).
Reese, M. L., Zeiner, G. M., Saeij, J. P., Boothroyd, J. C. & Boyle, J. P. Polymorphic family of injected pseudokinases is paramount in Toxoplasma virulence. Proc. Natl Acad. Sci. USA 108, 9625–9630 (2011).
Butcher, B. A. et al. Toxoplasma gondii rhoptry kinase ROP16 activates STAT3 and STAT6 resulting in cytokine inhibition and arginase-1-dependent growth control. PLoS Pathog. 7, e1002236 (2011).
Burger, E. et al. Loss of Paneth cell autophagy causes acute susceptibility to Toxoplasma gondii–mediated inflammation. Cell Host Microbe 23, 177–190.e4 (2018).
Lopez-Yglesias, A. H., Burger, E., Araujo, A., Martin, A. T. & Yarovinsky, F. T-bet-independent Th1 response induces intestinal immunopathology during Toxoplasma gondii infection. Mucosal Immunol. 11, 921–931 (2018).
Raetz, M. et al. Parasite-induced TH1 cells and intestinal dysbiosis cooperate in IFN-γ-dependent elimination of Paneth cells. Nat. Immunol. 14, 136–142 (2013).
Acknowledgements
This work was supported by National Institute of Allergy and Infectious Diseases grants R01AI136538 and R01AI121090 and by the Burroughs Wellcome Foundation.
Author information
Authors and Affiliations
Contributions
A.S. and F.Y. conceived of the study, interpreted data and wrote the manuscript; A.S. performed and analyzed all experiments, except those in Fig. 6 and Supplementary Fig. 8 (performed by A.A. and E.T.C.). E.T.C. contributed to Supplementary Fig. 7. T.J.M. and M.R.E. contributed to Supplementary Fig. 3. D.P.B. contributed to Fig. 1a,b and Supplementary Fig. 1.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
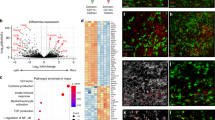
Supplementary Figure 1 Global gene transcriptome analysis of human PBMCs infected with T. gondii.
a, List of the most significant differentially expressed genes in human PBMCs infected with the Pru strain of T. gondii (MOI 3:1) for 12 h. b, Heat map of differentially expressed genes based on RNA-seq results of PBMCs infected with the RH strain of T. gondii (MOI 3:1) for 12 h.
Supplementary Figure 2 Identification of S100A11.
a, Amino acid peptides detected by mass spectrometry. The red boxes denote the most frequently detected peptides by mass spectrometry. The data shown are representative of three independent experiments. b, Complete gel images from Figs. 4c and 5. Dotted markings indicate the parts used for the figures.
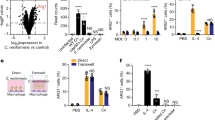
Supplementary Figure 3 S100A11 participates in induction of CCL2.
a–d, Knockdown of S100A11 in THP-1 cells was performed by siRNA targeting S100A11 or irrelevant targets (siRNA-GFP and ‘scrambled siRNA’). The efficiency of S100A11 knockdown was verified by immunoblot (a), qRT–PCR (b), and hS100A11-specific ELISA (c). THP-1 cells with the reduced S100A11 expression produced less CCL2 when infected with RH88 (c) or Pru (d) strains of T. gondii. The data shown (mean ± s.d.) are representative of three independent experiments. Each symbol represents an individual experimental sample.
Supplementary Figure 4 Gene ontology analysis identifies the RAGE pathway as activated in PBMCs infected by T. gondii (Pru strain).
Differentially expressed genes (fold change > 2, P < 0.001) in PBMCs (n = 5) identified by Ingenuity Pathway Analysis (IPA) software in response to the T. gondii Pru strain.
Supplementary Figure 5 Gene ontology analysis identifies the RAGE pathway as activated in PBMCs infected by T. gondii (RH88 strain).
Differentially expressed genes (fold change > 2, P < 0.001) in PBMCs (n = 5) identified by Ingenuity Pathway Analysis (IPA) software in response to T. gondii RH88 strain.
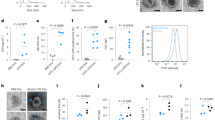
Supplementary Figure 6 Parasite invasion is required for induction of CCL2 responses.
a, Parental (TATi) and inducible profilin knockout parasites (ΔPRFe/PRFi) were grown for 4 d ± anhydrotetracycline (ATc), harvested and incubated with THP-1 cells at a 3:1 ratio for 16 h in triplicates. CCL2 expression was then measured by RT–PCR. The experiment shown is representative of five independent experiments performed. b, MYB- (3 μM) or DMSO-pretreated T. gondii parasites were added to THP-1 cells (MOI 3:1) for 16 h, and CCL2 expression was analyzed by RT–PCR. Each symbol represents an individual experimental sample. The data shown represent the mean ± s.d.
Supplementary Figure 7 Generation of S100a11 KO mice.
a, A schematic diagram of the CRISPR/Cas9 strategy used to generate S100a11-deficient mice and primer design used in the study. Exons 2 and 3 of the S100a11 gene were targeted by two sgRNAs depicted as sg#9 and sg#3. b, Representative genotyping of the targeted alleles with a set of primers S100A11 wtF and S100A11 wtR that result in PCR products of 2.7 kb for WT mice and 557 bp for the S100a11 KO allele, as a result of deletion of exons 2 and 3.
Supplementary Figure 8 S100A11 regulates monocyte recruitment during mucosal response to T. gondii.
a,b, WT and S100a11 KO mice (n = 5) were infected orally with T. gondii, and the presence of monocytes and neutrophils in small intestinal lamina propria (n = 3) was analyzed (a) and quantified by flow cytometry on day 7 after infection (b). c, CCL2 and IFN-γ secretion in small intestine. d, Parasite burden in small intestine (n = 5) was measured by qRT–PCR. e, Histological analysis of the small intestines (n = 5) of infected WT and S100a11 KO mice with 20 cysts of ME49 T. gondii on day 7 after infection. Image fields are representative of pathology in multiple tissue sections, and chosen sections were selected by blinded observation. f, Histological changes in the small intestine were analyzed on day 7 after infection based on an additive scoring system. The data shown (mean ± s.d.) are representative of three independent experiments. Each symbol represents an individual experimental sample, and unpaired two-tailed Student’s t test was used for statistical analysis; ns, not significant.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8
Rights and permissions
About this article
Cite this article
Safronova, A., Araujo, A., Camanzo, E.T. et al. Alarmin S100A11 initiates a chemokine response to the human pathogen Toxoplasma gondii. Nat Immunol 20, 64–72 (2019). https://doi.org/10.1038/s41590-018-0250-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-018-0250-8
This article is cited by
-
Role of the S100 protein family in rheumatoid arthritis
Arthritis Research & Therapy (2022)
-
Itaconate inhibits TET DNA dioxygenases to dampen inflammatory responses
Nature Cell Biology (2022)
-
Toxoplasma gondii infection and its implications within the central nervous system
Nature Reviews Microbiology (2021)
-
Essential roles of S100A10 in Toll-like receptor signaling and immunity to infection
Cellular & Molecular Immunology (2020)
-
Single-cell RNA-seq reveals CD16- monocytes as key regulators of human monocyte transcriptional response to Toxoplasma
Scientific Reports (2020)