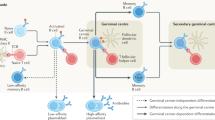
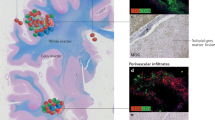
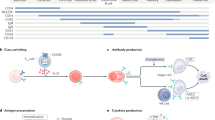
Abstract
There is growing recognition that B cell contributions to normal immune responses extend well beyond their potential to become antibody-producing cells, including roles at the innate–adaptive interface and their potential to modulate the responses of other immune cells such as T cells and myeloid cells. These B cell functions can have both pathogenic and protective effects in the context of central nervous system (CNS) inflammation. Here, we review recent advances in the field of multiple sclerosis (MS), which has traditionally been viewed as primarily a T cell–mediated disease, and we consider antibody-dependent and, particularly, emerging antibody-independent functions of B cells that may be relevant in both the peripheral and CNS disease compartments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Dutta, R. & Trapp, B. D. Relapsing and progressive forms of multiple sclerosis: insights from pathology. Curr. Opin. Neurol. 27, 271–278 (2014).
Lassmann, H. Multiple sclerosis pathology. Cold Spring Harb. Perspect. Med. 8, a028936 (2018).
Klineova, S. & Lublin, F.D. Clinical course of multiple sclerosis. Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a028928 (2018).
Baranzini, S. E. & Oksenberg, J. R. The genetics of multiple sclerosis: from 0 to 200 in 50 years. Trends Genet. 33, 960–970 (2017).
Olsson, T., Barcellos, L. F. & Alfredsson, L. Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nat. Rev. Neurol. 13, 25–36 (2017).
Glatigny, S. & Bettelli, E. Experimental autoimmune encephalomyelitis (EAE) as animal models of multiple sclerosis (MS). Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a028977 (2018).
Dendrou, C. A., Fugger, L. & Friese, M. A. Immunopathology of multiple sclerosis. Nat. Rev. Immunol. 15, 545–558 (2015).
Kaskow, B. J. & Baecher-Allan, C. Effector T cells in multiple sclerosis. Cold Spring Harb. Perspect. Med. 8, a029025 (2018).
Kitz, A., Singer, E., Hafler, D. & Regulatory, T. Regulatory T cells: from discovery to autoimmunity. Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a029041 (2018).
Bar-Or, A. et al. Rituximab in relapsing-remitting multiple sclerosis: a 72-week, open-label, phase I trial. Ann. Neurol. 63, 395–400 (2008).
Hauser, S. L. et al. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N. Engl. J. Med. 358, 676–688 (2008).
Kappos, L. et al. Ocrelizumab in relapsing-remitting multiple sclerosis: a phase 2, randomised, placebo-controlled, multicentre trial. Lancet 378, 1779–1787 (2011).
Sorensen, P. S. et al. Safety and efficacy of ofatumumab in relapsing-remitting multiple sclerosis: a phase 2 study. Neurology 82, 573–581 (2014).
Hauser, S. L. et al. Ocrelizumab versus Interferon beta-1a in relapsing multiple sclerosis. N. Engl. J. Med. 376, 221–234 (2017).
Bar-Or, A. et al. Subcutaneous ofatumumab in relapsing-remitting multiple sclerosis patients: the MIRROR study. Neurology doi:https://doi.org/10.1212/WNL.0000000000005516 (2018).
Lucchinetti, C. F. et al. Inflammatory cortical demyelination in early multiple sclerosis. N. Engl. J. Med. 365, 2188–2197 (2011).
Michel, L. et al. B cells in the multiple sclerosis central nervous system: trafficking and contribution to CNS-compartmentalized inflammation. Front. Immunol. 6, 636 (2015).
Magliozzi, R. et al. A gradient of neuronal loss and meningeal inflammation in multiple sclerosis. Ann. Neurol. 68, 477–493 (2010). This paper provides the first description of links between meningeal inflammation and both neuronal loss and microglia activation.
Ransohoff, R. M. Animal models of multiple sclerosis: the good, the bad and the bottom line. Nat. Neurosci. 15, 1074–1077 (2012).
Magliozzi, R. et al. Meningeal B-cell follicles in secondary progressive multiple sclerosis associate with early onset of disease and severe cortical pathology. Brain 130, 1089–1104 (2007).
Nikolakopoulou, A. M., Dutta, R., Chen, Z., Miller, R. H. & Trapp, B. D. Activated microglia enhance neurogenesis via trypsinogen secretion. Proc. Natl. Acad. Sci. USA 110, 8714–8719 (2013).
Mahad, D. H., Trapp, B. D. & Lassmann, H. Pathological mechanisms in progressive multiple sclerosis. Lancet Neurol. 14, 183–193 (2015).
Zrzavy, T. et al. Loss of ‘homeostatic’ microglia and patterns of their activation in active multiple sclerosis. Brain 140, 1900–1913 (2017).
McHeyzer-Williams, M. G. & Nossal, G. J. Clonal analysis of autoantibody-producing cell precursors in the preimmune B cell repertoire. J. Immunol. 141, 4118–4123 (1988).
Wardemann, H. et al. Predominant autoantibody production by early human B cell precursors. Science 301, 1374–1377 (2003).
Shlomchik, M. J. Sites and stages of autoreactive B cell activation and regulation. Immunity 28, 18–28 (2008).
Meffre, E. The establishment of early B cell tolerance in humans: lessons from primary immunodeficiency diseases. Ann. NY Acad. Sci. 1246, 1–10 (2011).
Hervé, M. et al. CD40 ligand and MHC class II expression are essential for human peripheral B cell tolerance. J. Exp. Med. 204, 1583–1593 (2007).
Kinnunen, T. et al. Accumulation of peripheral autoreactive B cells in the absence of functional human regulatory T cells. Blood 121, 1595–1603 (2013).
Samuels, J., Ng, Y. S., Coupillaud, C., Paget, D. & Meffre, E. Impaired early B cell tolerance in patients with rheumatoid arthritis. J. Exp. Med. 201, 1659–1667 (2005).
Yurasov, S. et al. Defective B cell tolerance checkpoints in systemic lupus erythematosus. J. Exp. Med. 201, 703–711 (2005).
Kinnunen, T. et al. Specific peripheral B cell tolerance defects in patients with multiple sclerosis. J. Clin. Invest. 123, 2737–2741 (2013).
Dhaeze, T. et al. Circulating follicular regulatory T cells are defective in multiple sclerosis. J. Immunol. 195, 832–840 (2015).
Viglietta, V., Baecher-Allan, C., Weiner, H. L. & Hafler, D. A. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J. Exp. Med. 199, 971–979 (2004).
Coles, A. J. et al. Alemtuzumab for patients with relapsing multiple sclerosis after disease-modifying therapy: a randomised controlled phase 3 trial. Lancet 380, 1829–1839 (2012).
Prineas, J. W. & Graham, J. S. Multiple sclerosis: capping of surface immunoglobulin G on macrophages engaged in myelin breakdown. Ann. Neurol. 10, 149–158 (1981).
Genain, C. P., Cannella, B., Hauser, S. L. & Raine, C. S. Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat. Med. 5, 170–175 (1999).
Villar, L. M. et al. Early differential diagnosis of multiple sclerosis using a new oligoclonal band test. Arch. Neurol. 62, 574–577 (2005).
Villar, L. M. et al. Intrathecal synthesis of oligoclonal IgM against myelin lipids predicts an aggressive disease course in MS. J. Clin. Invest. 115, 187–194 (2005).
Villar, L. M. et al. Immunoglobulin M oligoclonal bands: biomarker of targetable inflammation in primary progressive multiple sclerosis. Ann. Neurol. 76, 231–240 (2014). This paper indicates that IgM OCB is an important biomarker for anti-CD20 treatment in patients with PPMS.
Qin, Y. et al. Clonal expansion and somatic hypermutation of V(H) genes of B cells from cerebrospinal fluid in multiple sclerosis. J. Clin. Invest. 102, 1045–1050 (1998).
Baranzini, S. E. et al. B cell repertoire diversity and clonal expansion in multiple sclerosis brain lesions. J. Immunol. 163, 5133–5144 (1999).
Colombo, M. et al. Accumulation of clonally related B lymphocytes in the cerebrospinal fluid of multiple sclerosis patients. J. Immunol. 164, 2782–2789 (2000).
Qin, Y. et al. Intrathecal B-cell clonal expansion, an early sign of humoral immunity, in the cerebrospinal fluid of patients with clinically isolated syndrome suggestive of multiple sclerosis. Lab. Invest. 83, 1081–1088 (2003).
Von Büdingen, H. C. et al. Clonally expanded plasma cells in the cerebrospinal fluid of patients with central nervous system autoimmune demyelination produce “oligoclonal bands”. J. Neuroimmunol. 218, 134–139 (2010).
Colombo, M. et al. Maintenance of B lymphocyte-related clones in the cerebrospinal fluid of multiple sclerosis patients. Eur. J. Immunol. 33, 3433–3438 (2003).
Lovato, L. et al. Related B cell clones populate the meninges and parenchyma of patients with multiple sclerosis. Brain 134, 534–541 (2011).
Von Büdingen, H. C. et al. B cell exchange across the blood-brain barrier in multiple sclerosis. J. Clin. Invest. 122, 4533–4543 (2012).
Palanichamy, A. et al. Immunoglobulin class-switched B cells form an active immune axis between CNS and periphery in multiple sclerosis. Sci. Transl. Med. 6, 248ra106 (2014).
Stern, J. N. H. et al. B cells populating the multiple sclerosis brain mature in the draining cervical lymph nodes. Sci. Transl. Med. 6, 248ra107 (2014). Refs. 48–50 together demonstrate the dynamic changes in B cells across the blood–brain barrier.
Eggers, E. L. et al. Clonal relationships of CSF B cells in treatment-naive multiple sclerosis patients. JCI Insight 2, 92724 (2017).
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Aspelund, A. et al. A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J. Exp. Med. 212, 991–999 (2015).
Absinta, M. et al. Human and nonhuman primate meninges harbor lymphatic vessels that can be visualized noninvasively by MRI. eLife 6, e29738 (2017).
Bar-Or, A. et al. Analyses of all matrix metalloproteinase members in leukocytes emphasize monocytes as major inflammatory mediators in multiple sclerosis. Brain 126, 2738–2749 (2003).
Alter, A. et al. Determinants of human B cell migration across brain endothelial cells. J. Immunol. 170, 4497–4505 (2003).
Blauth, K., Owens, G. P. & Bennett, J. L. The ins and outs of B cells in multiple sclerosis. Front. Immunol. 6, 565 (2015).
Alvarez, E. et al. Predicting optimal response to B-cell depletion with rituximab in multiple sclerosis using CXCL13 index, magnetic resonance imaging and clinical measures. Mult. Scler. J. Exp. Transl. Clin. 1, 2055217315623800 (2015).
Hohlfeld, R., Dornmair, K., Meinl, E. & Wekerle, H. The search for the target antigens of multiple sclerosis, part 2: CD8+ T cells, B cells, and antibodies in the focus of reverse-translational research. Lancet Neurol. 15, 317–331 (2016).
Brändle, S. M. et al. Distinct oligoclonal band antibodies in multiple sclerosis recognize ubiquitous self-proteins. Proc. Natl. Acad. Sci. USA 113, 7864–7869 (2016).
Winger, R. C. & Zamvil, S. S. Antibodies in multiple sclerosis oligoclonal bands target debris. Proc. Natl. Acad. Sci. USA 113, 7696–7698 (2016). This study identifies antibodies, found in the CSF in patients with MS, that recognize intracellular antigens.
Kuhle, J. et al. Lack of association between antimyelin antibodies and progression to multiple sclerosis. N. Engl. J. Med. 356, 371–378 (2007).
Zhou, D. et al. Identification of a pathogenic antibody response to native myelin oligodendrocyte glycoprotein in multiple sclerosis. Proc. Natl. Acad. Sci. USA 103, 19057–19062 (2006).
Ketelslegers, I. A. et al. Anti-MOG antibodies plead against MS diagnosis in an acquired demyelinating syndromes cohort. Mult. Scler. 21, 1513–1520 (2015).
Srivastava, R. et al. Potassium channel KIR4.1 as an immune target in multiple sclerosis. N. Engl. J. Med. 367, 115–123 (2012).
Kraus, V. et al. Potassium channel KIR4.1-specific antibodies in children with acquired demyelinating CNS disease. Neurology 82, 470–473 (2014).
Brickshawana, A. et al. Investigation of the KIR4.1 potassium channel as a putative antigen in patients with multiple sclerosis: a comparative study. Lancet Neurol. 13, 795–806 (2014).
Nerrant, E. et al. Lack of confirmation of anti-inward rectifying potassium channel 4.1 antibodies as reliable markers of multiple sclerosis. Mult. Scler. 20, 1699–1703 (2014).
Waters, P. J. et al. Serologic diagnosis of NMO: a multicenter comparison of aquaporin-4-IgG assays. Neurology 78, 665–671 (2012).
Pitzalis, C., Jones, G. W., Bombardieri, M. & Jones, S. A. Ectopic lymphoid-like structures in infection, cancer and autoimmunity. Nat. Rev. Immunol. 14, 447–462 (2014).
Lisak, R. P. et al. Secretory products of multiple sclerosis B cells are cytotoxic to oligodendroglia in vitro. J. Neuroimmunol. 246, 85–95 (2012).
Lisak, R. P. et al. B cells from patients with multiple sclerosis induce cell death via apoptosis in neurons in vitro. J. Neuroimmunol. 309, 88–99 (2017).
Krumbholz, M. et al. BAFF is produced by astrocytes and up-regulated in multiple sclerosis lesions and primary central nervous system lymphoma. J. Exp. Med. 201, 195–200 (2005).
Krumbholz, M., Derfuss, T., Hohlfeld, R. & Meinl, E. B cells and antibodies in multiple sclerosis pathogenesis and therapy. Nat. Rev. Neurol. 8, 613–623 (2012).
Touil, H. et al. Human central nervous system astrocytes support survival and activation of B cells: implications for MS pathogenesis. J. Neuroinflammation 15, 114 (2018).
Montalban, X. et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N. Engl. J. Med. 376, 209–220 (2017).
Cross, A. H., Stark, J. L., Lauber, J., Ramsbottom, M. J. & Lyons, J. A. Rituximab reduces B cells and T cells in cerebrospinal fluid of multiple sclerosis patients. J. Neuroimmunol. 180, 63–70 (2006).
Monson, N. L., Cravens, P. D., Frohman, E. M., Hawker, K. & Racke, M. K. Effect of rituximab on the peripheral blood and cerebrospinal fluid B cells in patients with primary progressive multiple sclerosis. Arch. Neurol. 62, 258–264 (2005).
Rodríguez-Pinto, D. B cells as antigen presenting cells. Cell. Immunol. 238, 67–75 (2005).
Pierce, S. K. et al. Antigen-presenting function of B lymphocytes. Immunol. Rev. 106, 149–180 (1988).
Rivera, A., Chen, C. C., Ron, N., Dougherty, J. P. & Ron, Y. Role of B cells as antigen-presenting cells in vivo revisited: antigen-specific B cells are essential for T cell expansion in lymph nodes and for systemic T cell responses to low antigen concentrations. Int. Immunol. 13, 1583–1593 (2001).
Molnarfi, N. et al. MHC class II-dependent B cell APC function is required for induction of CNS autoimmunity independent of myelin-specific antibodies. J. Exp. Med. 210, 2921–2937 (2013).
Barnett, L. G. et al. B cell antigen presentation in the initiation of follicular helper T cell and germinal center differentiation. J. Immunol. 192, 3607–3617 (2014).
Walters, S. N., Webster, K. E., Daley, S. & Grey, S. T. A role for intrathymic B cells in the generation of natural regulatory T cells. J. Immunol. 193, 170–176 (2014).
Milich, D. R. et al. Role of B cells in antigen presentation of the hepatitis B core. Proc. Natl. Acad. Sci. USA 94, 14648–14653 (1997).
Zeng, Q. et al. B cells mediate chronic allograft rejection independently of antibody production. J. Clin. Invest. 124, 1052–1056 (2014).
Serreze, D. V. et al. B lymphocytes are critical antigen-presenting cells for the initiation of T cell-mediated autoimmune diabetes in nonobese diabetic mice. J. Immunol. 161, 3912–3918 (1998).
Li, R. et al. Antibody-independent function of human B cells contributes to antifungal T cell responses. J. Immunol. 198, 3245–3254 (2017).
Weber, M. S. et al. B-cell activation influences T-cell polarization and outcome of anti-CD20 B-cell depletion in central nervous system autoimmunity. Ann. Neurol. 68, 369–383 (2010).
Chen, L. & Flies, D. B. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat. Rev. Immunol. 13, 227–242 (2013).
Bar-Or, A. et al. Immunological memory: contribution of memory B cells expressing costimulatory molecules in the resting state. J. Immunol. 167, 5669–5677 (2001).
Henn, A. D. et al. Functionally distinct subpopulations of CpG-activated memory B cells. Sci. Rep. 2, 345 (2012).
O’Neill, S. K. et al. Expression of CD80/86 on B cells is essential for autoreactive T cell activation and the development of arthritis. J. Immunol. 179, 5109–5116 (2007).
Gimmi, C. D. et al. B-cell surface antigen B7 provides a costimulatory signal that induces T cells to proliferate and secrete interleukin 2. Proc. Natl. Acad. Sci. USA 88, 6575–6579 (1991).
Genç, K., Dona, D. L. & Reder, A. T. Increased CD80+ B cells in active multiple sclerosis and reversal by interferon beta-1b therapy. J. Clin. Invest. 99, 2664–2671 (1997).
Bodhankar, S., Galipeau, D., Vandenbark, A. A. & Offner, H. PD-1 interaction with PD-L1 but not PD-L2 on B-cells mediates protective effects of estrogen against EAE. J. Clin. Cell. Immunol. 4, 143 (2013).
Ray, A., Basu, S., Williams, C. B., Salzman, N. H. & Dittel, B. N. A novel IL-10-independent regulatory role for B cells in suppressing autoimmunity by maintenance of regulatory T cells via GITR ligand. J. Immunol. 188, 3188–3198 (2012).
Zuccarino-Catania, G. V. et al. CD80 and PD-L2 define functionally distinct memory B cell subsets that are independent of antibody isotype. Nat. Immunol. 15, 631–637 (2014). This study proposes that costimulatory/inhibitory molecules can be used as markers to define functionally distinct B cell subpopulations.
Shen, P. & Fillatreau, S. Antibody-independent functions of B cells: a focus on cytokines. Nat. Rev. Immunol. 15, 441–451 (2015).
Li, R. et al. Cytokine-defined B cell responses as therapeutic targets in multiple sclerosis. Front. Immunol. 6, 626 (2016).
Duddy, M. E., Alter, A. & Bar-Or, A. Distinct profiles of human B cell effector cytokines: a role in immune regulation? J. Immunol. 172, 3422–3427 (2004).
Duddy, M. et al. Distinct effector cytokine profiles of memory and naive human B cell subsets and implication in multiple sclerosis. J. Immunol. 178, 6092–6099 (2007). This is the first study indicating abnormal B cell cytokine responses in MS.
Bar-Or, A. et al. Abnormal B-cell cytokine responses a trigger of T-cell-mediated disease in MS? Ann. Neurol. 67, 452–461 (2010).
Barr, T. A. et al. B cell depletion therapy ameliorates autoimmune disease through ablation of IL-6-producing B cells. J. Exp. Med. 209, 1001–1010 (2012).
Miyazaki, Y. et al. A novel microRNA-132-sirtuin-1 axis underlies aberrant B-cell cytokine regulation in patients with relapsing-remitting multiple sclerosis. PLoS One 9, e105421 (2014).
Li, R. et al. Proinflammatory GM-CSF-producing B cells in multiple sclerosis and B cell depletion therapy. Sci. Transl. Med. 7, 310ra166 (2015). This paper describes novel B cell subpopulations and how abnormal B cell–myeloid cell interactions might play an important role in MS pathogenesis.
Correale, J., Farez, M. & Razzitte, G. Helminth infections associated with multiple sclerosis induce regulatory B cells. Ann. Neurol. 64, 187–199 (2008).
Rauch, P. J. et al. Innate response activator B cells protect against microbial sepsis. Science 335, 597–601 (2012).
Hilgendorf, I. et al. Innate response activator B cells aggravate atherosclerosis by stimulating T helper-1 adaptive immunity. Circulation 129, 1677–1687 (2014).
Weber, G. F. et al. Pleural innate response activator B cells protect against pneumonia via a GM-CSF-IgM axis. J. Exp. Med. 211, 1243–1256 (2014).
Fillatreau, S., Sweenie, C. H., McGeachy, M. J., Gray, D. & Anderton, S. M. B cells regulate autoimmunity by provision of IL-10. Nat. Immunol. 3, 944–950 (2002).
Lampropoulou, V. et al. TLR-activated B cells suppress T cell-mediated autoimmunity. J. Immunol. 180, 4763–4773 (2008).
Matsushita, T., Yanaba, K., Bouaziz, J. D., Fujimoto, M. & Tedder, T. F. Regulatory B cells inhibit EAE initiation in mice while other B cells promote disease progression. J. Clin. Invest. 118, 3420–3430 (2008). This paper raises the possibility that different B cell subsets may play opposing roles in the pathogenesis of neuroinflammatory diseases.
Yoshizaki, A. et al. Regulatory B cells control T-cell autoimmunity through IL-21-dependent cognate interactions. Nature 491, 264–268 (2012).
Matsumoto, M. et al. Interleukin-10-producing plasmablasts exert regulatory function in autoimmune inflammation. Immunity 41, 1040–1051 (2014).
Ochoa-Repáraz, J., Mielcarz, D. W., Haque-Begum, S. & Kasper, L. H. Induction of a regulatory B cell population in experimental allergic encephalomyelitis by alteration of the gut commensal microflora. Gut Microbes 1, 103–108 (2010).
Rosser, E. C. et al. Regulatory B cells are induced by gut microbiota-driven interleukin-1β and interleukin-6 production. Nat. Med. 20, 1334–1339 (2014).
Blair, P. A. et al. CD19+CD24hiCD38hi B cells exhibit regulatory capacity in healthy individuals but are functionally impaired in systemic lupus erythematosus patients. Immunity 32, 129–140 (2010).
Iwata, Y. et al. Characterization of a rare IL-10-competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood 117, 530–541 (2011). Refs. 118,119 show the existence of regulatory B cell subpopulations in humans.
Rieger, A. & Bar-Or, A. B-cell-derived interleukin-10 in autoimmune disease: regulating the regulators. Nat. Rev. Immunol. 8, 486–487 (2008).
Correale, J. & Farez, M. Association between parasite infection and immune responses in multiple sclerosis. Ann. Neurol. 61, 97–108 (2007).
Bjarnadóttir, K. et al. B cell-derived transforming growth factor-β1 expression limits the induction phase of autoimmune neuroinflammation. Sci. Rep. 6, 34594 (2016).
Shen, P. et al. IL-35-producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Nature 507, 366–370 (2014). This is the first study demonstrating antibody-independent roles of plasma cells.
Wang, R. X. et al. Interleukin-35 induces regulatory B cells that suppress autoimmune disease. Nat. Med. 20, 633–641 (2014). Refs. 123,124 identify a novel population of regulatory B cells characterized by IL-35 expression.
Fritz, J. H. et al. Acquisition of a multifunctional IgA+ plasma cell phenotype in the gut. Nature 481, 199–203 (2011).
Patel, D. J. & Wang, Z. Readout of epigenetic modifications. Annu. Rev. Biochem. 82, 81–118 (2013).
Zan, H. & Casali, P. Epigenetics of peripheral B-cell differentiation and the antibody response. Front. Immunol. 6, 631 (2015).
Wu, H. et al. Epigenetic regulation in B-cell maturation and its dysregulation in autoimmunity. Cell. Mol. Immunol. https://doi.org/10.1038/cmi.2017.133 (2018).
Sievers, C. et al. Altered microRNA expression in B lymphocytes in multiple sclerosis: towards a better understanding of treatment effects. Clin. Immunol. 144, 70–79 (2012).
Palanichamy, A. et al. Rituximab efficiently depletes increased CD20-expressing T cells in multiple sclerosis patients. J. Immunol. 193, 580–586 (2014).
Kappos, L. et al. Atacicept in multiple sclerosis (ATAMS): a randomised, placebo-controlled, double-blind, phase 2 trial. Lancet Neurol. 13, 353–363 (2014).
Sergott, R. C. et al. ATON: results from a phase II randomized trial of the B-cell-targeting agent atacicept in patients with optic neuritis. J. Neurol. Sci. 351, 174–178 (2015).
Bossen, C. & Schneider, P. BAFF, APRIL and their receptors: structure, function and signaling. Semin. Immunol. 18, 263–275 (2006).
Steri, M. et al. Overexpression of the cytokine BAFF and autoimmunity risk. N. Engl. J. Med. 376, 1615–1626 (2017).
Baker, D., Marta, M., Pryce, G., Giovannoni, G. & Schmierer, K. Memory B cells are major targets for effective immunotherapy in relapsing multiple sclerosis. EBioMedicine 16, 41–50 (2017).
Miyazaki, Y. et al. Suppressed pro-inflammatory properties of circulating B cells in patients with multiple sclerosis treated with fingolimod, based on altered proportions of B-cell subpopulations. Clin. Immunol. 151, 127–135 (2014).
Nakamura, M. et al. Differential effects of fingolimod on B-cell populations in multiple sclerosis. Mult. Scler. 20, 1371–1380 (2014).
Grützke, B. et al. Fingolimod treatment promotes regulatory phenotype and function of B cells. Ann. Clin. Transl. Neurol. 2, 119–130 (2015).
Lundy, S. K. et al. Dimethyl fumarate treatment of relapsing-remitting multiple sclerosis influences B-cell subsets. Neurol. Neuroimmunol. Neuroinflamm. 3, e211 (2016).
Li, R. et al. Dimethyl fumarate treatment mediates an anti-inflammatory shift in B cell subsets of patients with multiple sclerosis. J. Immunol. 198, 691–698 (2017).
Longbrake, E.E. et al. Dimethyl fumarate induces changes in B- and T-lymphocyte function independent of the effects on absolute lymphocyte count. Mult. Scler. https://doi.org/10.1177/1352458517707069 (2017).
Smith, M. D., Martin, K. A., Calabresi, P. A. & Bhargava, P. Dimethyl fumarate alters B-cell memory and cytokine production in MS patients. Ann. Clin. Transl. Neurol. 4, 351–355 (2017).
Agius, M.A. et al. Safety and tolerability of inebilizumab (MEDI-551), an anti-CD19 monoclonal antibody, in patients with relapsing forms of multiple sclerosis: results from a phase 1 randomised, placebo-controlled, escalating intravenous and subcutaneous dose study. Mult. Scler. https://doi.org/10.1177/1352458517740641 (2017).
Satterthwaite, A. B. Bruton’s tyrosine kinase, a component of B cell signaling pathways, has multiple roles in the pathogenesis of lupus. Front. Immunol. 8, 1986 (2018).
Rizzo, F. et al. Interferon-β therapy specifically reduces pathogenic memory B cells in multiple sclerosis patients by inducing a FAS-mediated apoptosis. Immunol. Cell Biol. 94, 886–894 (2016).
Rudick, R. A. et al. Cerebrospinal fluid abnormalities in a phase III trial of Avonex (IFNbeta-1a) for relapsing multiple sclerosis. J. Neuroimmunol. 93, 8–14 (1999).
Noronha, A., Toscas, A. & Jensen, M. A. Interferon beta decreases T cell activation and interferon gamma production in multiple sclerosis. J. Neuroimmunol. 46, 145–153 (1993).
Rep, M. H. et al. Interferon (IFN)-beta treatment enhances CD95 and interleukin 10 expression but reduces interferon-gamma producing T cells in MS patients. J. Neuroimmunol. 96, 92–100 (1999).
Lucas, M. et al. Regulation by interferon beta-1a of reactive oxygen metabolites production by lymphocytes and monocytes and serum sulfhydryls in relapsing multiple sclerosis patients. Neurochem. Int. 42, 67–71 (2003).
Hussien, Y., Sanna, A., Söderström, M., Link, H. & Huang, Y. M. Multiple sclerosis: expression of CD1a and production of IL-12p70 and IFN-gamma by blood mononuclear cells in patients on combination therapy with IFN-beta and glatiramer acetate compared to monotherapy with IFN-beta. Mult. Scler. 10, 16–25 (2004).
Zafranskaya, M. et al. Interferon-beta therapy reduces CD4+ and CD8+ T-cell reactivity in multiple sclerosis. Immunology 121, 29–39 (2007).
Hamamcioglu, K. & Reder, A. T. Interferon-beta regulates cytokines and BDNF: greater effect in relapsing than in progressive multiple sclerosis. Mult. Scler. 13, 459–470 (2007).
Korporal, M. et al. Interferon beta-induced restoration of regulatory T-cell function in multiple sclerosis is prompted by an increase in newly generated naive regulatory T cells. Arch. Neurol. 65, 1434–1439 (2008).
Comabella, M. et al. Changes in matrix metalloproteinases and their inhibitors during interferon-beta treatment in multiple sclerosis. Clin. Immunol. 130, 145–150 (2009).
Rasouli, J. et al. Expression of GM-CSF in T cells is increased in multiple sclerosis and suppressed by IFN-β therapy. J. Immunol. 194, 5085–5093 (2015).
Trinschek, B., Luessi, F., Gross, C. C., Wiendl, H. & Jonuleit, H. Interferon-beta therapy of multiple sclerosis patients improves the responsiveness of T cells for immune suppression by regulatory T cells. Int. J. Mol. Sci. 16, 16330–16346 (2015).
Duda, P. W., Schmied, M. C., Cook, S. L., Krieger, J. I. & Hafler, D. A. Glatiramer acetate (Copaxone) induces degenerate, Th2-polarized immune responses in patients with multiple sclerosis. J. Clin. Invest. 105, 967–976 (2000).
Karandikar, N. J. et al. Glatiramer acetate (Copaxone) therapy induces CD8+ T cell responses in patients with multiple sclerosis. J. Clin. Invest. 109, 641–649 (2002).
Salama, H. H., Hong, J., Zang, Y. C., El-Mongui, A. & Zhang, J. Blocking effects of serum reactive antibodies induced by glatiramer acetate treatment in multiple sclerosis. Brain 126, 2638–2647 (2003).
Rieks, M. et al. Induction of apoptosis of CD4+ T cells by immunomodulatory therapy of multiple sclerosis with glatiramer acetate. Eur. Neurol. 50, 200–206 (2003).
Putheti, P., Soderstrom, M., Link, H. & Huang, Y. M. Effect of glatiramer acetate (Copaxone) on CD4+CD25high T regulatory cells and their IL-10 production in multiple sclerosis. J. Neuroimmunol. 144, 125–131 (2003).
Dhib-Jalbut, S. et al. Glatiramer acetate-reactive peripheral blood mononuclear cells respond to multiple myelin antigens with a Th2-biased phenotype. J. Neuroimmunol. 140, 163–171 (2003).
Aharoni, R., Kayhan, B., Eilam, R., Sela, M. & Arnon, R. Glatiramer acetate-specific T cells in the brain express T helper 2/3 cytokines and brain-derived neurotrophic factor in situ. Proc. Natl Acad. Sci. USA 100, 14157–14162 (2003).
Weber, M. S. et al. Multiple sclerosis: glatiramer acetate inhibits monocyte reactivity in vitro and in vivo. Brain 127, 1370–1378 (2004).
Kim, H. J. et al. Type 2 monocyte and microglia differentiation mediated by glatiramer acetate therapy in patients with multiple sclerosis. J. Immunol. 172, 7144–7153 (2004).
Tennakoon, D. K. et al. Therapeutic induction of regulatory, cytotoxic CD8+ T cells in multiple sclerosis. J. Immunol. 176, 7119–7129 (2006).
Blanco, Y. et al. Effect of glatiramer acetate (Copaxone) on the immunophenotypic and cytokine profile and BDNF production in multiple sclerosis: a longitudinal study. Neurosci. Lett. 406, 270–275 (2006).
Biegler, B. W. et al. Glatiramer acetate (GA) therapy induces a focused, oligoclonal CD8+ T-cell repertoire in multiple sclerosis. J. Neuroimmunol. 180, 159–171 (2006).
Burger, D. et al. Glatiramer acetate increases IL-1 receptor antagonist but decreases T cell-induced IL-1beta in human monocytes and multiple sclerosis. Proc. Natl Acad. Sci. USA 106, 4355–4359 (2009).
Pul, R. et al. Glatiramer acetate increases phagocytic activity of human monocytes in vitro and in multiple sclerosis patients. PLoS One 7, e51867 (2012).
Sellner, J. et al. Glatiramer acetate attenuates the pro-migratory profile of adhesion molecules on various immune cell subsets in multiple sclerosis. Clin. Exp. Immunol. 173, 381–389 (2013).
Ayers, C. L. et al. Modulation of immune function occurs within hours of therapy initiation for multiple sclerosis. Clin. Immunol. 147, 105–119 (2013).
Ireland, S. J. et al. The effect of glatiramer acetate therapy on functional properties of B cells from patients with relapsing-remitting multiple sclerosis. JAMA Neurol. 71, 1421–1428 (2014).
Ahn, Y. H. et al. Glatiramer acetate attenuates the activation of CD4+ T cells by modulating STAT1 and -3 signaling in glia. Sci. Rep. 7, 40484 (2017).
Watson, C. M., Davison, A. N., Baker, D., O’Neill, J. K. & Turk, J. L. Suppression of demyelination by mitoxantrone. Int. J. Immunopharmacol. 13, 923–930 (1991).
Fox, E. J. Mechanism of action of mitoxantrone. Neurology 63 (Suppl. 6), S15–S18 (2004).
Chan, A., Weilbach, F. X., Toyka, K. V. & Gold, R. Mitoxantrone induces cell death in peripheral blood leucocytes of multiple sclerosis patients. Clin. Exp. Immunol. 139, 152–158 (2005).
Putzki, N. et al. Mitoxantrone does not restore the impaired suppressive function of natural regulatory T cells in patients suffering from multiple sclerosis: a longitudinal ex vivo and in vitro study. Eur. Neurol. 61, 27–32 (2009).
Vogelgesang, A., Rosenberg, S., Skrzipek, S., Bröker, B. M. & Dressel, A. Mitoxantrone treatment in multiple sclerosis induces TH2-type cytokines. Acta Neurol. Scand. 122, 237–243 (2010).
Bar-Or, A. et al. Teriflunomide effect on immune response to influenza vaccine in patients with multiple sclerosis. Neurology 81, 552–558 (2013).
Bar-Or, A., Pachner, A., Menguy-Vacheron, F., Kaplan, J. & Wiendl, H. Teriflunomide and its mechanism of action in multiple sclerosis. Drugs 74, 659–674 (2014).
Bar-Or, A. et al. Randomized study of teriflunomide effects on immune responses to neoantigen and recall antigens. Neurol. Neuroimmunol. Neuroinflamm. 2, e70 (2015).
Gandoglia, I. et al. Teriflunomide treatment reduces B cells in patients with MS. Neurol. Neuroimmunol. Neuroinflamm. 4, e403 (2017).
Li, L. et al. The effects of teriflunomide on lymphocyte subpopulations in human peripheral blood mononuclear cells in vitro. J. Neuroimmunol. 265, 82–90 (2013).
Spencer, C. M., Crabtree-Hartman, E. C., Lehmann-Horn, K., Cree, B. A. & Zamvil, S. S. Reduction of CD8+ T lymphocytes in multiple sclerosis patients treated with dimethyl fumarate. Neurol. Neuroimmunol. Neuroinflamm. 2, e76 (2015).
Berkovich, R. & Weiner, L. P. Effects of dimethyl fumarate on lymphocyte subsets. Mult. Scler. Relat. Disord. 4, 339–341 (2015).
Longbrake, E. E. et al. Dimethyl fumarate selectively reduces memory T cells in multiple sclerosis patients. Mult. Scler. 22, 1061–1070 (2016).
Michell-Robinson, M. A. et al. Effects of fumarates on circulating and CNS myeloid cells in multiple sclerosis. Ann. Clin. Transl. Neurol. 3, 27–41 (2015).
Gross, C. C. et al. Dimethyl fumarate treatment alters circulating T helper cell subsets in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 3, e183 (2015).
Wu, Q. et al. Dimethyl fumarate selectively reduces memory T cells and shifts the balance between Th1/Th17 and Th2 in multiple sclerosis patients. J. Immunol. 198, 3069–3080 (2017).
Fleischer, V. et al. Treatment response to dimethyl fumarate is characterized by disproportionate CD8+ T cell reduction in MS. Mult. Scler. 24, 632–641 (2018).
Ghadiri, M. et al. Dimethyl fumarate-induced lymphopenia in MS due to differential T-cell subset apoptosis. Neurol. Neuroimmunol. Neuroinflamm. 4, e340 (2017).
Diebold, M. et al. Dimethyl fumarate influences innate and adaptive immunity in multiple sclerosis. J. Autoimmun. 86, 39–50 (2018).
Bielekova, B. et al. Regulatory CD56bright natural killer cells mediate immunomodulatory effects of IL-2Ralpha-targeted therapy (daclizumab) in multiple sclerosis. Proc. Natl Acad. Sci. USA 103, 5941–5946 (2006).
Wynn, D. et al. Daclizumab in active relapsing multiple sclerosis (CHOICE study): a phase 2, randomised, double-blind, placebo-controlled, add-on trial with interferon beta. Lancet Neurol. 9, 381–390 (2010).
Lin, Y. C. et al. Daclizumab reverses intrathecal immune cell abnormalities in multiple sclerosis. Ann. Clin. Transl. Neurol. 2, 445–455 (2015).
Lin, Y. C. et al. Patients with MS under daclizumab therapy mount normal immune responses to influenza vaccination. Neurol. Neuroimmunol. Neuroinflamm. 3, e196 (2016).
Niino, M. et al. Natalizumab effects on immune cell responses in multiple sclerosis. Ann. Neurol. 59, 748–754 (2006).
Stüve, O. et al. Immune surveillance in multiple sclerosis patients treated with natalizumab. Ann. Neurol. 59, 743–747 (2006).
Krumbholz, M., Meinl, I., Kümpfel, T., Hohlfeld, R. & Meinl, E. Natalizumab disproportionately increases circulating pre-B and B cells in multiple sclerosis. Neurology 71, 1350–1354 (2008).
Sellebjerg, F. et al. Increased cerebrospinal fluid concentrations of the chemokine CXCL13 in active MS. Neurology 73, 2003–2010 (2009).
Putzki, N., Baranwal, M. K., Tettenborn, B., Limmroth, V. & Kreuzfelder, E. Effects of natalizumab on circulating B cells, T regulatory cells and natural killer cells. Eur. Neurol. 63, 311–317 (2010).
Kowarik, M. C. et al. Differential effects of fingolimod (FTY720) on immune cells in the CSF and blood of patients with MS. Neurology 76, 1214–1221 (2011).
Planas, R., Jelči, I., Schippling, S., Martin, R. & Sospedra, M. Natalizumab treatment perturbs memory- and marginal zone-like B-cell homing in secondary lymphoid organs in multiple sclerosis. Eur. J. Immunol. 42, 790–798 (2012).
Börnsen, L. et al. Effect of natalizumab on circulating CD4+ T-cells in multiple sclerosis. PLoS One 7, e47578 (2012).
Warnke, C. et al. Natalizumab exerts a suppressive effect on surrogates of B cell function in blood and CSF. Mult. Scler. 21, 1036–1044 (2015).
Saraste, M., Penttilä, T. L. & Airas, L. Natalizumab treatment leads to an increase in circulating CXCR3-expressing B cells. Neurol. Neuroimmunol. Neuroinflamm. 3, e292 (2016).
Kimura, K. et al. Disrupted balance of T cells under natalizumab treatment in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 3, e210 (2016).
Mameli, G. et al. Natalizumab therapy modulates miR-155, miR-26a and proinflammatory cytokine expression in MS patients. PLoS One 11, e0157153 (2016).
Serpero, L. D. et al. Fingolimod modulates peripheral effector and regulatory T cells in MS patients. J. Neuroimmune Pharmacol. 8, 1106–1113 (2013).
Claes, N. et al. Compositional changes of B and T cell subtypes during fingolimod treatment in multiple sclerosis patients: a 12-month follow-up study. PLoS One 9, e111115 (2014).
Muls, N., Dang, H. A., Sindic, C. J. & van Pesch, V. Fingolimod increases CD39-expressing regulatory T cells in multiple sclerosis patients. PLoS One 9, e113025 (2014).
Mazzola, M. A. et al. Identification of a novel mechanism of action of fingolimod (FTY720) on human effector T cell function through TCF-1 upregulation. J. Neuroinflammation 12, 245 (2015).
Blumenfeld, S., Staun-Ram, E. & Miller, A. Fingolimod therapy modulates circulating B cell composition, increases B regulatory subsets and production of IL-10 and TGFβ in patients with multiple sclerosis. J. Autoimmun. 70, 40–51 (2016).
Miyazaki, Y. et al. Fingolimod induces BAFF and expands circulating transitional B cells without activating memory B cells and plasma cells in multiple sclerosis. Clin. Immunol. 187, 95–101 (2018).
Ghadiri, M. et al. Reconstitution of the peripheral immune repertoire following withdrawal of fingolimod. Mult. Scler. 23, 1225–1232 (2017).
Jones, J. L. et al. Human autoimmunity after lymphocyte depletion is caused by homeostatic T-cell proliferation. Proc. Natl. Acad. Sci. USA 110, 20200–20205 (2013).
Thomas, K., Eisele, J., Rodriguez-Leal, F. A., Hainke, U. & Ziemssen, T. Acute effects of alemtuzumab infusion in patients with active relapsing-remitting MS. Neurol. Neuroimmunol. Neuroinflamm. 3, e228 (2016).
Gross, C. C. et al. Alemtuzumab treatment alters circulating innate immune cells in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm. 3, e289 (2016).
Cox, A. L. et al. Lymphocyte homeostasis following therapeutic lymphocyte depletion in multiple sclerosis. Eur. J. Immunol. 35, 3332–3342 (2005).
Thompson, S. A., Jones, J. L., Cox, A. L., Compston, D. A. & Coles, A. J. B-cell reconstitution and BAFF after alemtuzumab (Campath-1H) treatment of multiple sclerosis. J. Clin. Immunol. 30, 99–105 (2010).
Hill-Cawthorne, G. A. et al. Long term lymphocyte reconstitution after alemtuzumab treatment of multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 83, 298–304 (2012).
Heidt, S., Hester, J., Shankar, S., Friend, P. J. & Wood, K. J. B cell repopulation after alemtuzumab induction-transient increase in transitional B cells and long-term dominance of naïve B cells. Am. J. Transplant. 12, 1784–1792 (2012).
Zhang, X. et al. Differential reconstitution of T cell subsets following immunodepleting treatment with alemtuzumab (anti-CD52 monoclonal antibody) in patients with relapsing-remitting multiple sclerosis. J. Immunol. 191, 5867–5874 (2013).
Wilk, E. et al. Depletion of functionally active CD20+ T cells by rituximab treatment. Arthritis Rheum. 60, 3563–3571 (2009).
Piccio, L. et al. Changes in B- and T-lymphocyte and chemokine levels with rituximab treatment in multiple sclerosis. Arch. Neurol. 67, 707–714 (2010).
Studer, V., Rossi, S., Motta, C., Buttari, F. & Centonze, D. Peripheral B cell depletion and central proinflammatory cytokine reduction following repeated intrathecal administration of rituximab in progressive Multiple Sclerosis. J. Neuroimmunol. 276, 229–231 (2014).
Dall’Era, M. et al. Reduced B lymphocyte and immunoglobulin levels after atacicept treatment in patients with systemic lupus erythematosus: results of a multicenter, phase Ib, double-blind, placebo-controlled, dose-escalating trial. Arthritis Rheum. 56, 4142–4150 (2007).
van Vollenhoven, R. F., Kinnman, N., Vincent, E., Wax, S. & Bathon, J. Atacicept in patients with rheumatoid arthritis and an inadequate response to methotrexate: results of a phase II, randomized, placebo-controlled trial. Arthritis Rheum. 63, 1782–1792 (2011).
Ma, N. et al. BAFF suppresses IL-15 expression in B cells. J. Immunol. 192, 4192–4201 (2014).
Munafo, A., Priestley, A., Nestorov, I., Visich, J. & Rogge, M. Safety, pharmacokinetics and pharmacodynamics of atacicept in healthy volunteers. Eur. J. Clin. Pharmacol. 63, 647–656 (2007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
A.B.-O. has consulted for multiple entities involved in B cell–targeting therapies and has received consulting fees and/or grant support from Atara Biotherapeutics, Biogen Idec, Celgene/Receptos, Genentech/Roche, GlaxoSmithKline, MAPI, Medimmune, Merck/EMD Serono, Novartis and Sanofi-Genzyme. R.L. and K.R.P. have nothing to disclose.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, R., Patterson, K.R. & Bar-Or, A. Reassessing B cell contributions in multiple sclerosis. Nat Immunol 19, 696–707 (2018). https://doi.org/10.1038/s41590-018-0135-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-018-0135-x