Abstract
Enolase 1 (ENO1) is a glycolytic enzyme that plays essential roles in various pathological activities including cancer development. However, the mechanisms underlying ENO1-contributed tumorigenesis are not well explained. Here, we uncover that ENO1, as an RNA-binding protein, binds to the cytosine-uracil-guanine-rich elements of YAP1 messenger RNA to promote its translation. ENO1 and YAP1 positively regulate alternative arachidonic acid (AA) metabolism by inverse regulation of PLCB1 and HPGD (15-hydroxyprostaglandin dehydrogenase). The YAP1/PLCB1/HPGD axis-mediated activation of AA metabolism and subsequent accumulation of prostaglandin E2 (PGE2) are responsible for ENO1-mediated cancer progression, which can be retarded by aspirin. Finally, aberrant activation of ENO1/YAP1/PLCB1 and decreased HPGD expression in clinical hepatocellular carcinoma samples indicate a potential correlation between ENO1-regulated AA metabolism and cancer development. These findings underline a new function of ENO1 in regulating AA metabolism and tumorigenesis, suggesting a therapeutic potential for aspirin in patients with liver cancer with aberrant expression of ENO1 or YAP1.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
RNA-seq data and RIP-seq data have been deposited in the Gene Expression Omnibus under accession code GSE183703. The KEGG used for gene pathway analysis of RNA-seq data can be accessed at https://www.genome.jp/kegg/. All other data are available in the Source data and Supplementary Information that are provided with this paper.
References
Pavlova, N. N. & Thompson, C. B. The emerging hallmarks of cancer metabolism. Cell Metab. 23, 27–47 (2016).
Zhu, J. & Thompson, C. B. Metabolic regulation of cell growth and proliferation. Nat. Rev. Mol. Cell Biol. 20, 436–450 (2019).
Kim, J. W. & Dang, C. V. Multifaceted roles of glycolytic enzymes. Trends Biochem. Sci. 30, 142–150 (2005).
Yang, W. et al. PKM2 phosphorylates histone H3 and promotes gene transcription and tumorigenesis. Cell 150, 685–696 (2012).
Chang, C. H. et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153, 1239–1251 (2013).
Zhang, C. S. et al. Fructose-1,6-bisphosphate and aldolase mediate glucose sensing by AMPK. Nature 548, 112–116 (2017).
Yang, W. W. et al. Nuclear PKM2 regulates beta-catenin transactivation upon EGFR activation. Nature 480, 118–U289 (2011).
Xu, D. et al. The evolving landscape of noncanonical functions of metabolic enzymes in cancer and other pathologies. Cell Metab. 33, 33–50 (2021).
Schwanhausser, B. et al. Global quantification of mammalian gene expression control. Nature 473, 337–342 (2011).
Hein, M. Y. et al. A human interactome in three quantitative dimensions organized by stoichiometries and abundances. Cell 163, 712–723 (2015).
Itzhak, D. N., Tyanova, S., Cox, J. & Borner, G. H. H. Global, quantitative and dynamic mapping of protein subcellular localization. eLife 5, e16950 (2016).
Diaz-Ramos, A., Roig-Borrellas, A., Garcia-Melero, A. & Lopez-Alemany, R. alpha-Enolase, a multifunctional protein: its role on pathophysiological situations. J. Biomed. Biotechnol. 2012, 156795 (2012).
Pancholi, V. Multifunctional alpha-enolase: its role in diseases. Cell. Mol. Life Sci. 58, 902–920 (2001).
Huppertz, I. et al. Riboregulation of Enolase 1 activity controls glycolysis and embryonic stem cell differentiation. Mol. Cell. 82, 2666–2680.e11 (2022).
Kuhnel, K. & Luisi, B. F. Crystal structure of the Escherichia coli RNA degradosome component enolase. J. Mol. Biol. 313, 583–592 (2001).
Carpousis, A. J. The Escherichia coli RNA degradosome: structure, function and relationship in other ribonucleolytic multienzyme complexes. Biochem. Soc. Trans. 30, 150–155 (2002).
Entelis, N. et al. A glycolytic enzyme, enolase, is recruited as a cofactor of tRNA targeting toward mitochondria in Saccharomyces cerevisiae. Genes Dev. 20, 1609–1620 (2006).
Hernandez-Perez, L. et al. alpha-Enolase binds to RNA. Biochimie 93, 1520–1528 (2011).
Castello, A. et al. Insights into RNA biology from an atlas of mammalian mRNA-binding proteins. Cell 149, 1393–1406 (2012).
Zhang, T. et al. ENO1 suppresses cancer cell ferroptosis by degrading the mRNA of iron regulatory protein 1. Nat. Cancer 3, 75–89 (2022).
Tang, X., Edwards, E. M., Holmes, B. B., Falck, J. R. & Campbell, W. B. Role of phospholipase C and diacylglyceride lipase pathway in arachidonic acid release and acetylcholine-induced vascular relaxation in rabbit aorta. Am. J. Physiol.-Heart C. 290, H37–H45 (2006).
Park, J. B. et al. Phospholipase signalling networks in cancer. Nat. Rev. Cancer 12, 782–792 (2012).
Wang, D. Z. & Dubois, R. N. Eicosanoids and cancer. Nat. Rev. Cancer 10, 181–193 (2010).
Chan, A. T., Ogino, S. & Fuchs, C. S. Aspirin use and survival after diagnosis of colorectal cancer. J. Am. Med Assoc. 302, 649–659 (2009).
Holmes, M. D. et al. Aspirin intake and survival after breast cancer. J. Clin. Oncol. 28, 1467–1472 (2010).
Thun, M. J., Jacobs, E. J. & Patrono, C. The role of aspirin in cancer prevention. Nat. Rev. Clin. Oncol. 9, 259–267 (2012).
Drew, D. A., Cao, Y. & Chan, A. T. Aspirin and colorectal cancer: the promise of precision chemoprevention. Nat. Rev. Cancer 16, 173–186 (2016).
Hogan, D. J., Riordan, D. P., Gerber, A. P., Herschlag, D. & Brown, P. O. Diverse RNA-binding proteins interact with functionally related sets of RNAs, suggesting an extensive regulatory system. PLoS Biol. 6, e255 (2008).
Moroishi, T., Hansen, C. G. & Guan, K. L. The emerging roles of YAP and TAZ in cancer. Nat. Rev. Cancer 15, 73–79 (2015).
Sanchez-Vega, F. et al. Oncogenic signaling pathways in the cancer genome atlas. Cell 173, 321–337 e310 (2018).
Zhao, B. et al. TEAD mediates YAP-dependent gene induction and growth control. Genes Dev. 22, 1962–1971 (2008).
Yu, F. X. et al. Regulation of the Hippo-YAP pathway by G-protein-coupled receptor signaling. Cell 150, 780–791 (2012).
Nguyen, L. T. et al. ERG activates the YAP1 transcriptional program and induces the development of age-related prostate tumors. Cancer Cell 27, 797–808 (2015).
Stein, C. et al. YAP1 exerts its transcriptional control via TEAD-mediated activation of enhancers. PLoS Genet. 11, e1005465 (2015).
Sanchez, M., Galy, B., Muckenthaler, M. U. & Hentze, M. W. Iron-regulatory proteins limit hypoxia-inducible factor-2alpha expression in iron deficiency. Nat. Struct. Mol. Biol. 14, 420–426 (2007).
Wu, N. et al. Translation of yes-associated protein (YAP) was antagonized by its circular RNA via suppressing the assembly of the translation initiation machinery. Cell Death Differ. 26, 2758–2773 (2019).
Brown, S. Z. et al. The RNA-binding protein HuR posttranscriptionally regulates the protumorigenic activator YAP1 in pancreatic ductal adenocarcinoma. Mol. Cell. Biol. 42, e0001822 (2022).
Strnadel, J. et al. eIF5A-PEAK1 signaling regulates YAP1/TAZ protein expression and pancreatic cancer cell growth. Cancer Res. 77, 1997–2007 (2017).
Jin, D. et al. m6A mRNA methylation initiated by METTL3 directly promotes YAP translation and increases YAP activity by regulating the MALAT1-miR-1914-3p-YAP axis to induce NSCLC drug resistance and metastasis. J. Hematol. Oncol. 12, 135 (2019).
Hudson, W. H. & Ortlund, E. A. The structure, function and evolution of proteins that bind DNA and RNA. Nat. Rev. Mol. Cell Bio. 15, 749–760 (2014).
Kang, H. J., Jung, S. K., Kim, S. J. & Chung, S. J. Structure of human alpha-enolase (hENO1), a multifunctional glycolytic enzyme. Acta Crystallogr. D. 64, 651–657 (2008).
Guo, X. C. et al. Single tumor-initiating cells evade immune clearance by recruiting type II macrophages. Genes Dev. 31, 247–259 (2017).
Li, W. et al. YAP transcriptionally regulates COX-2 expression and GCCSysm-4 (G-4), a dual YAP/COX-2 inhibitor, overcomes drug resistance in colorectal cancer. J. Exp. Clin. Cancer Res. 36, 144 (2017).
Kim, H. B. et al. Prostaglandin E2 activates YAP and a positive-signaling loop to promote colon regeneration after colitis but also carcinogenesis in mice. Gastroenterology 152, 616–630 (2017).
Kim, M. K., Jang, J. W. & Bae, S. C. DNA binding partners of YAP/TAZ. BMB Rep. 51, 126–133 (2018).
Kim, M., Kim, T., Johnson, R. L. & Lim, D. S. Transcriptional co-repressor function of the Hippo pathway transducers YAP and TAZ. Cell Rep. 11, 270–282 (2015).
Lacoste, B., Raymond, V. A., Cassim, S., Lapierre, P. & Bilodeau, M. Highly tumorigenic hepatocellular carcinoma cell line with cancer stem cell-like properties. PLoS ONE 12, e0171215 (2017).
Simsek, D. et al. The mammalian ribo-interactome reveals ribosome functional diversity and heterogeneity. Cell 169, 1051–1065 (2017).
Wu, J. et al. Intercellular interaction dictates cancer cell ferroptosis via NF2-YAP signalling. Nature 572, 402–406 (2019).
Yang, W. H. et al. The Hippo pathway effector YAP promotes ferroptosis via the E3 ligase SKP2. Mol. Cancer Res. 19, 1005–1014 (2021).
Zhang, X. et al. Endogenous glutamate determines ferroptosis sensitivity via ADCY10-dependent YAP suppression in lung adenocarcinoma. Theranostics 11, 5650–5674 (2021).
Gao, R. Z. et al. YAP/TAZ and ATF4 drive resistance to Sorafenib in hepatocellular carcinoma by preventing ferroptosis. EMBO Mol. Med. 13, e14351 (2021).
Demetz, E. et al. The arachidonic acid metabolome serves as a conserved regulator of cholesterol metabolism. Cell Metab. 20, 787–798 (2014).
Wang, T. et al. Selective interaction of the C2 domains of phospholipase C-beta1 and -beta2 with activated Galphaq subunits: an alternative function for C2-signaling modules. Proc. Natl Acad. Sci. USA 96, 7843–7846 (1999).
Rhee, S. G. Regulation of phosphoinositide-specific phospholipase C. Annu. Rev. Biochem. 70, 281–312 (2001).
Ha, S. D., Lewin, N., Li, S. S. C. & Kim, S. O. HDAC8 activates AKT through upregulating PLCB1 and suppressing DESC1 expression in MEK1/2 inhibition-resistant cells. Cells 10, 1101 (2021).
Balsinde, J., Winstead, M. V. & Dennis, E. A. Phospholipase A(2) regulation of arachidonic acid mobilization. FEBS Lett. 531, 2–6 (2002).
Norris, P. C., Gosselin, D., Reichart, D., Glass, C. K. & Dennis, E. A. Phospholipase A2 regulates eicosanoid class switching during inflammasome activation. Proc. Natl Acad. Sci. USA 111, 12746–12751 (2014).
Balboa, M. A., Perez, R. & Balsinde, J. Amplification mechanisms of inflammation: paracrine stimulation of arachidonic acid mobilization by secreted phospholipase A(2) is regulated by cytosolic phospholipase A(2)-derived hydroperoxyeicosatetraenoic acid. J. Immunol. 171, 989–994 (2003).
Leslie, C. C. Cytosolic phospholipase A(2): physiological function and role in disease. J. Lipid Res. 56, 1386–1402 (2015).
Kalyvas, A. et al. Differing roles for members of the phospholipase A2 superfamily in experimental autoimmune encephalomyelitis. Brain 132, 1221–1235 (2009).
Simon, T. G. et al. Association between aspirin use and risk of hepatocellular carcinoma. J. Am. Med. Assoc. Oncol. 4, 1683–1690 (2018).
Lee, T. Y. et al. Association of daily aspirin therapy with risk of hepatocellular carcinoma in patients with chronic hepatitis B. J. Am. Med. Assoc. Intern Med 179, 633–640 (2019).
Simon, T. G. et al. Association of aspirin with hepatocellular carcinoma and liver-related mortality. N. Engl. J. Med. 382, 1018–1028 (2020).
Wang, S. J. et al. Association of aspirin therapy with risk of hepatocellular carcinoma: a systematic review and dose-response analysis of cohort studies with 2.5 million participants. Pharmacol. Res. 151, 104585 (2020).
Fox, R. K., Taddei, T. H. & Kaplan, D. E. Aspirin use and risk of hepatocellular carcinoma in hepatitis B. J. Am. Med. Assoc. Intern. Med. 179, 640–641 (2019).
Nair, A. B. & Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 7, 27–31 (2016).
Sun, L. et al. cMyc-mediated activation of serine biosynthesis pathway is critical for cancer progression under nutrient deprivation conditions. Cell Res. 25, 429–444 (2015).
Wu, G. et al. MYC promotes cancer progression by modulating m(6) A modifications to suppress target gene translation. EMBO Rep. 22, e51519 (2021).
Liu, S. et al. METTL13 Methylation of eEF1A increases translational output to promote tumorigenesis. Cell 176, 491–504 e421 (2019).
Xing, S. et al. DIS3L2 promotes progression of hepatocellular carcinoma via hnRNP U-mediated alternative splicing. Cancer Res. 79, 4923–4936 (2019).
Acknowledgements
This work is supported in part by the National Natural Science Foundation of China (grant nos. 91957203 to P.G., 82192893, 81930083 and 81821001 to H.Z., 82130087 to P.G., 82273221 to L.S. and 82203556 to C.S.), the Chinese Academy of Sciences (grant no. XDB39020100 to H.Z.) and the National Key R&D Program of China (grant nos. 2022YFA1304504 to H.Z. and 2018YFA0800300 to P.G.).
Author information
Authors and Affiliations
Contributions
L.S., H.Z. and P.G. conceived the study and supervised the experiments. L.S., C.S., T.Z., S.S., H.Z. and P.G. designed the experiments. L.S. and T.Z. performed RIP and ChIP assays to identify the targets of ENO1 and YAP1, respectively. L.S. and C.S. performed animal studies and analysis of clinical HCC specimens. L.S., C.S. and T.Z. performed immunoblotting, cloning, cell biology and biochemistry experiments. S.S. analyzed RNA-seq and RIP-seq data. X.G., S.Q., P.Z., H.W., W.M., R.Y. and R.C. analyzed the data. W.J. provided clinical specimens. J.C. provided constructive guidance. L.S., C.S., T.Z., H.Z. and P.G. wrote the paper. All the authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
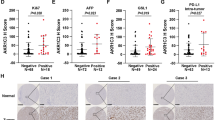
Extended Data Fig. 1 ENO1 regulates the expression of YAP1 target genes.
a, Protein copy numbers of glucose metabolism enzymes in NIH3T3 mouse fibroblast cells (Schwanhausser et al., 2011), and HeLa cells (Hein et al., 2015; Itzhak et al., 2016). b, Venn diagram showing the 21 overlapped target genes enriched by ENO1 IP in our RIP-seq data from HEK293T-Flag-ENO1 cells and the previously published HITS-GBP-CLIP data from HeLa-ENO1-YFP cells (Castello et al., 2012). c, qPCR-based expression analysis of known YAP1 target genes including NPPB, CYR61, CTGF, S1PR1, NEXN, NTF3, KDR, CPA3, PSG1, CCDC80, EGR3, EGR4, DDIT4, and TRAIL in PLC cells expression shRNAs targeting ENO1 or YAP1 (n = 3 independent assays). Error bars denote the mean ± s.e.m. (c).
Extended Data Fig. 2 ENO1 upregulates YAP1 expression by recruiting YTHDF3.
a, Co-IP assay in HEK293T cells transfected with Flag-ENO1 or Hep3B-Flag-ENO1 cells. b, Protein levels of eIF5A, YTHDF3, and YAP1 were measured by immunoblotting analysis in Hep3B cells with eIF5A or YTHDF3 knockdown. Actin served as a loading control. Par: parental; NC: negative control. c, Binding of endogenous YAP1 mRNA transcripts by Flag-ENO1 in Hep3B-Flag-ENO1 cells with or without YTHDF3 knockdown (n = 3 independent assays). Protein levels of ENO1, YTHDF3, and YAP1 were measured. Actin served as a loading control. d, HEK293T cells expressing shRNA targeting endogenous ENO1 were further transfected with plasmids to overexpress Flag-tagged wild-type ENO1, ENO1S40A, or ENO1D245R mutants (n = 1 sample detected at different time points). Then the ENO1 enzyme activity of these cells was measured by the consumption of NADH in an assay buffer that includes pyruvate kinase (PK), lactate dehydrogenase (LDHA), and the required substrates including 2-phospho-D-glycerate (2PG), phosphoenolpyruvate (PEP), and pyruvate. The consumption of NADH is monitored as a decrease of absorbance at 340 nm. e, Flag and YAP1 protein levels were measured in HEK293T cells expressing Flag-tagged wild-type ENO1 and indicated ENO1 mutant variants. Actin served as a negative control. f, g, Photographs showed xenografts described in Fig. 1l (f). ENO1 and YAP1 protein levels were measured (g). Actin served as a negative control. Immunoblots are representative of two independent experiments (a-c,e). Error bars denote the mean ± s.e.m. (c,d). Statistical analyses were performed by two-tailed Student’s t-test (c,d).
Extended Data Fig. 3 ENO1 and YAP1 transcriptionally activate arachidonic acid synthetic enzymes.
a, Venn diagram of the RNA-seq data showing the overlapped genes regulated by ENO1 and YAP1 in PLC cells with ENO1 or YAP1 knockdown. The statistical significance between ENO1-regulated genes and YAP1-regulated genes was determined using Fisher’s exact probability test, P < 0.001. b, KEGG mapper analysis of the pathways in which the co-regulated genes of ENO1 and YAP1 are enriched. 66 genes of metabolic pathways are shown in Supplementary Table 1. c, Gene Ontology (GO) enrichment analysis. The top ten GO terms are listed. Supplementary Table 2 provides a complete list of terms and corresponding gene lists. P-value was computed using the one-sided Fisher’s exact test and corrected for multiple hypothesis testing using false discovery rate (FDR). d, qPCR analysis of ENO1, YAP1, PLCB1, PTGS2 and HPGD expression in PLC cells expressing shRNAs targeting ENO1 or YAP1 (n = 3 independent assays). e, Immunoblotting analysis of ENO1, YAP1, and HPGD expression in PLC cells expressing shRNAs targeting ENO1 or YAP1. Actin served as a negative control. f, Protein levels of ENO1, YAP1, PLCB1, and HPGD were measured in Hepa 1–6 cells expressing shRNAs targeting ENO1 or YAP1. Actin served as a negative control. g, h, Protein levels of ENO1, YAP1, PLCB1, COX2, and HPGD were measured in PLC cells (g) or Hep3B cells (h) expressing ENO1 or YAP1. Actin served as a negative control. i, Protein levels of YAP1, PLCB1, COX2, and HPGD were measured in HEK293T cells expressing wild-type YAP1 or YAP-5SA mutant. Actin served as a negative control. Immunoblots are representative of three independent experiments. Error bars denote the mean ± s.e.m. (d). Statistical analyses were performed by two-tailed Student’s t-test (d).
Extended Data Fig. 4 The NuRD complex is required for ENO1/YAP1-mediated HPGD gene repression.
a, b, qPCR (a) and immunoblotting (b) analysis of HPGD expression levels in PLC cells treated with 1 μM TSA, or expressing shRNAs targeting MTA2, HDAC1, or HDAC2 (mRNA data: n = 3 independent assays). Actin served as a negative control. c, PLC cells expressing Flag-tagged YAP1 or ENO1 were further infected with shRNA viruses targeting HDAC1. Protein levels of YAP1, ENO1, Flag, HDAC1, and HPGD were measured by immunoblotting analysis. Actin served as a negative control. d, A schematic representation of the ENO1-mediated activation of YAP1 transcriptional regulation of the PLCB1 and HPGD loci. Illustration created with Biorender.com. Immunoblots are representative of three independent experiments. Error bars denote the mean ± s.e.m. (a). Statistical analyses were performed by two-tailed Student’s t-test (a).
Extended Data Fig. 5 ENO1 and YAP1 activate AA metabolism in a PLCB1- and HPGD-dependent manner.
a, AA and PGE2 contents were measured in PLC cells expressing ENO1 or YAP1 by ELISA (n = 3 independent assays). b, c, Immunoblotting and qPCR analysis of HPGD expression in PLC cells expressing shRNAs targeting HPGD (b), followed by the detection of PGE2 content and cell growth status (c) (n = 3 independent assays). Actin served as a negative control. d, Protein levels of ENO1 or YAP1, and HPGD (lower panel), and PGE2 content (upper panel) were measured in ENO1 or YAP1-overexpressing PLC cells expressing HPGD (n = 3 independent assays). Actin served as a negative control. e, AA content was measured in ENO1- or YAP1-overexpressing Hep3B cells infected with shRNA viruses targeting PLCB1 (n = 3 independent assays). f, Protein levels of HPGD were measured by immunoblotting in endogenous ENO1-knockdown PLC cells expressing wild-type ENO1, ENO1S40A, and ENO1D245R. AA and PGE2 contents in these cell lines were measured by ELISA (n = 3 independent assays). Actin served as a negative control. Immunoblots are representative of three independent experiments. Error bars denote the mean ± s.e.m. (a-f) or mean ± s.d. (c, lower panel). Statistical analyses were performed by two-tailed Student’s t-test (a-f), or two-way ANOVA (c, lower panel).
Extended Data Fig. 6 The accelerated tumor progression promoted by ENO1/YAP1 depends on PLCB1 expression.
a, Cell growth was measured in ENO1 or YAP1-overexpressing Hep3B cells infected with shRNA viruses targeting PLCB1 (n = 4 independent assays). b, Photographs show xenografts described in Fig. 4a at the end of the experiment. c, Protein levels of Flag, ENO1, YAP1, and PLCB1 in tumor lysates of Extended Data Fig. 6b were measured by immunoblotting analysis. Actin served as a loading control. d, Protein levels of YAP1 and PLCB1 were measured in YAP1-knockdown Hep3B cells further infected with shRNA viruses targeting PLCB1 (upper panel). Actin served as a negative control. Growth curves were measured by trypan blue counting (lower panel, n = 3 independent assays). e, f, Equal numbers of Hep3B cells expressing shYAP1 or/and shPLCB1 were injected subcutaneously into the flanks of BALB/c nude mice (n = 5 mice in each group). Tumor sizes were measured every 3 days using calipers (e). Tumor weights (f) were determined at the end of the experiment (day 30). g, Protein levels of YAP1 and PLCB1 in tumor lysates of Extended Data Fig. 6f were measured by immunoblotting analysis. Actin served as a loading control. Immunoblots are representative of two independent experiments (d). Error bars denote the mean ± s.e.m. (a,d-f). Statistical analyses were performed by two-tailed Student’s t-test (f), or two-way ANOVA with tukey’s multiple comparisons test (a,d,e).
Extended Data Fig. 7 The accelerated tumor progression promoted by ENO1/YAP1 depends on HPGD expression.
a, Cell growth was measured in ENO1 or YAP1-knockdown PLC cells infected with shRNA viruses targeting HPGD (n = 3 independent assays). b, Cell growth was measured in ENO1 or YAP1-overxpressing PLC cells expressing HPGD (n = 3 independent assays). c, Photographs show xenografts described in Fig. 4e at the end of the experiment. d, Protein levels of ENO1, YAP1, and HPGD in tumor lysates of Extended Data Fig. 7c were measured by immunoblotting analysis. Actin served as a loading control. e, PLC cells expressing shRNA targeting endogenous ENO1 were further transfected with plasmids to overexpress Flag-tagged wild-type ENO1, ENO1S40A, ENO1D245R, or ENO1M4mutants. Protein levels of ENO1 and Flag were measured by immunoblotting analysis. Actin served as a loading control. f, ENO1 enzyme activity of cells in Extended Data Fig. 7e was measured (n = 3 independent assays). g, Growth curves were measured by trypan blue counting in PLC cells expressing the EV, wild-type ENO1, or indicated ENO1 mutants with endogenous ENO1 knockdown (n = 3 independent assays). h, Photographs show xenografts described in Fig. 4k at the end of the experiment. i, Protein levels of Flag, ENO1, YAP1, and HPGD were detected in the tumor lysates described in Extended Data Fig. 7h. Actin served as a loading control. Immunoblots are representative of two independent experiments (e). Error bars denote the mean ± s.d. (a,b,g) or mean ± s.e.m. (f). Statistical analyses were performed by two-tailed Student’s t-test (f), two-way ANOVA (g), or two-way ANOVA with tukey’s multiple comparisons test (a,b).
Extended Data Fig. 8 COX2 is responsible for ENO1/YAP1-mediated tumor growth.
a, b, Cell growth was measured in ENO1- or YAP1-overexpressing PLC cells (a) or Hep3B cells (b) infected with shRNA viruses targeting COX2 (n = 3 independent assays). Protein levels of ENO1, YAP1, and COX2 were measured by immunoblotting analysis. Actin served as a loading control. c-f, Equal numbers of Hep3B cells expressing ENO or YAP1 that further knockdown COX2 were injected subcutaneously into the flanks of BALB/c nude mice (n = 5 mice in each group). Tumor sizes were measured every 3 days using calipers (c, d). Tumor weights (e, f) were determined at the end of the experiment (day 29). g, h, Protein levels of ENO1, YAP1, and COX2 in tumor lysates of Extended Data Fig. 8e, f were measured by immunoblotting analysis. Actin served as a loading control. Immunoblots are representative of three independent experiments (a,b). Error bars denote the mean ± s.d. (a,b) or mean ± s.e.m. (c-f). Statistical analyses were performed by two-tailed Student’s t-test (e,f), or two-way ANOVA with tukey’s multiple comparisons test (a-d).
Extended Data Fig. 9 Aspirin inhibits ENO1- and YAP1-induced liver cancer in vivo.
a, Protein levels of COX2 were measured in PLC cells treated with different doses of aspirin. Actin served as a negative control. b, COX2 enzyme activity was measured in the tumor lysates of Fig. 5b using ELISA (n = 10). c, Protein levels of ENO1 and YAP1 were measured in the tumor lysates of Fig. 5b. Actin served as a negative control. d, Protein levels of mus-ENO1 or mus-YAP1 were measured in Hepa 1–6 cells stably infected with viruses expressing mus-ENO1 or mus-YAP1. e, COX2 enzyme activity was measured in the tumor lysates of Fig. 5e, g using ELISA (n = 5). f, g, Protein levels of ENO1 (Flag), YAP1, PLCB1, and HPGD were measured in the tumor lysates of Fig. 5e, g. Calnexin served as a loading control. h, COX2 enzyme activity was measured in the tumor lysates of Fig. 5i using ELISA (n = 5). i, j, protein (i) and mRNA (j) levels of YAP1, PTGS2 (COX2), PLCB1, and HPGD were detected in the tumor lysates of Fig. 5i. Calnexin served as a loading control. Immunoblots are representative of three independent experiments (a,d). Error bars denote the mean ± s.e.m. (b,e,h,j). Statistical analyses were performed by two-tailed Student’s t-test (b,e,h,j).
Extended Data Fig. 10 ENO1 promotes liver cancer progression by regulating PLCB1 and HPGD-mediated arachidonic acid metabolism through YAP1.
a, Summary: ENO1, as an RNA-binding protein, activates YAP1 translation by binding to the (CUG)n triplet rich elements within the 3’UTR of YAP1 mRNA. YAP1 content increases and translocates into the nucleus to promote PLCB1 expression, but directly represses HPGD expression at transcriptional levels, which is responsible for the ENO1-mediated activation of the alterative arachidonic acid pathway, PGE2 accumulation, cell proliferation, and cancer progression. Aspirin, a blocker of the AA pathway by inhibiting COX2 activity and PGE2 production, could inhibit liver cancer progression, especially in liver cancers with elevated expression of ENO1 and YAP1. Illustration created with Biorender.com.
Supplementary information
Supplementary Information
Supplementary Tables 1–9.
Source data
Source Data Fig. 1
Unprocessed western blots.
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 2
Unprocessed western blots.
Source Data Extended Data Fig. 3
Unprocessed western blots.
Source Data Extended Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 5
Unprocessed western blots.
Source Data Extended Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 7
Unprocessed western blots.
Source Data Extended Data Fig. 8
Unprocessed western blots.
Source Data Extended Data Fig. 9
Unprocessed western blots.
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, L., Suo, C., Zhang, T. et al. ENO1 promotes liver carcinogenesis through YAP1-dependent arachidonic acid metabolism. Nat Chem Biol 19, 1492–1503 (2023). https://doi.org/10.1038/s41589-023-01391-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-023-01391-6