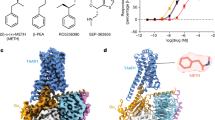
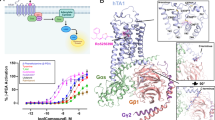
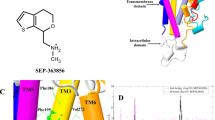
Abstract
Trace-amine-associated receptors (TAARs), a group of biogenic amine receptors, have essential roles in neurological and metabolic homeostasis1. They recognize diverse endogenous trace amines and subsequently activate a range of G-protein-subtype signalling pathways2,3. Notably, TAAR1 has emerged as a promising therapeutic target for treating psychiatric disorders4,5. However, the molecular mechanisms underlying its ability to recognize different ligands remain largely unclear. Here we present nine cryo-electron microscopy structures, with eight showing human and mouse TAAR1 in a complex with an array of ligands, including the endogenous 3-iodothyronamine, two antipsychotic agents, the psychoactive drug amphetamine and two identified catecholamine agonists, and one showing 5-HT1AR in a complex with an antipsychotic agent. These structures reveal a rigid consensus binding motif in TAAR1 that binds to endogenous trace amine stimuli and two extended binding pockets that accommodate diverse chemotypes. Combined with mutational analysis, functional assays and molecular dynamic simulations, we elucidate the structural basis of drug polypharmacology and identify the species-specific differences between human and mouse TAAR1. Our study provides insights into the mechanism of ligand recognition and G-protein selectivity by TAAR1, which may help in the discovery of ligands or therapeutic strategies for neurological and metabolic disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Cryo-EM coordinates and density maps have been deposited at the PDB and the Electron Microscopy Data Bank under accession codes 8JLN, EMD-36401 (T1AM–hTAAR1–Gs complex); 8JLO, EMD-36402 (SEP-363856–hTAAR1–Gs complex); 8JLP, EMD-36403 (RO-6889450–hTAAR1–Gs complex); 8JLQ, EMD-36404 (fenoldopam–hTAAR1–Gs complex); 8JLR, EMD-36405 (A77636–hTAAR1–Gs complex); 8JLJ, EMD-36399 (T1AM–mTAAR1–Gs complex); 8JLK, EMD-36400 (SEP-363856–mTAAR1–Gs complex); 8JSO, EMD-36625 ((S)-AMPH–hTAAR1–Gs complex); and 8JSP, EMD-36626 (SEP-363856–5-HT1AR–Gi complex).
References
Gainetdinov, R. R., Hoener, M. C. & Berry, M. D. Trace amines and their receptors. Pharmacol. Rev. 70, 549–620 (2018).
Underhill, S. M. et al. Amphetamines signal through intracellular TAAR1 receptors coupled to Gα13 and GαS in discrete subcellular domains. Mol. Psychiatry 26, 1208–1223 (2021).
Navarro, H. A., Gilmour, B. P. & Lewin, A. H. A rapid functional assay for the human trace amine-associated receptor 1 based on the mobilization of internal calcium. J. Biomol. Screen. 11, 688–693 (2006).
Halff, E. F., Rutigliano, G., Garcia-Hidalgo, A. & Howes, O. D. Trace amine-associated receptor 1 (TAAR1) agonism as a new treatment strategy for schizophrenia and related disorders. Trends Neurosci. 46, 60–74 (2023).
Raony, I., Domith, I., Lourenco, M. V., Paes-de-Carvalho, R. & Pandolfo, P. Trace amine-associated receptor 1 modulates motor hyperactivity, cognition, and anxiety-like behavior in an animal model of ADHD. Prog. Neuropsychopharmacol. Biol. Psychiatry 117, 110555 (2022).
Lindemann, L. & Hoener, M. C. A renaissance in trace amines inspired by a novel GPCR family. Trends Pharmacol. Sci. 26, 274–281 (2005).
Khan, M. Z. & Nawaz, W. The emerging roles of human trace amines and human trace amine-associated receptors (hTAARs) in central nervous system. Biomed. Pharmacother. 83, 439–449 (2016).
Borowsky, B. et al. Trace amines: identification of a family of mammalian G protein-coupled receptors. Proc. Natl Acad. Sci. USA 98, 8966–8971 (2001).
Bunzow, J. R. et al. Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor. Mol. Pharmacol. 60, 1181–1188 (2001).
Guo, L. et al. Structural basis of amine odorant perception by a mammal olfactory receptor. Nature 618, 193–200 (2023).
Liberles, S. D. & Buck, L. B. A second class of chemosensory receptors in the olfactory epithelium. Nature 442, 645–650 (2006).
Barnes, D. A., Galloway, D. A., Hoener, M. C., Berry, M. D. & Moore, C. S. TAAR1 expression in human macrophages and brain tissue: a potential novel facet of MS neuroinflammation. Int. J. Mol. Sci. 22, 11576 (2021).
Xie, Z. & Miller, G. M. Trace amine-associated receptor 1 as a monoaminergic modulator in brain. Biochem. Pharmacol. 78, 1095–1104 (2009).
Michael, E. S., Covic, L. & Kuliopulos, A. Trace amine-associated receptor 1 (TAAR1) promotes anti-diabetic signaling in insulin-secreting cells. J. Biol. Chem. 294, 4401–4411 (2019).
Koblan, K. S. et al. A non-D2-receptor-binding drug for the treatment of schizophrenia. N. Engl. J. Med. 382, 1497–1506 (2020).
Miller, G. M. The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity. J. Neurochem. 116, 164–176 (2011).
Heal, D. J., Smith, S. L., Gosden, J. & Nutt, D. J. Amphetamine, past and present-a pharmacological and clinical perspective. J. Psychopharmacol. 27, 479–496 (2013).
Faraone, S. V. The pharmacology of amphetamine and methylphenidate: relevance to the neurobiology of attention-deficit/hyperactivity disorder and other psychiatric comorbidities. Neurosci. Biobehav. Rev. 87, 255–270 (2018).
Sharma, A. & Couture, J. A review of the pathophysiology, etiology, and treatment of attention-deficit hyperactivity disorder (ADHD). Ann. Pharmacother. 48, 209–225 (2014).
Shoptaw, S. J., Kao, U. & Ling, W. Treatment for amphetamine psychosis. Cochrane Database Syst. Rev. 2009, CD003026 (2009).
Fulde, G. W. & Forster, S. L. The impact of amphetamine-type stimulants on emergency services. Curr. Opin. Psychiatry 28, 275–279 (2015).
Bramness, J. G. & Rognli, E. B. Psychosis induced by amphetamines. Curr. Opin. Psychiatry 29, 236–241 (2016).
Sulzer, D., Sonders, M. S., Poulsen, N. W. & Galli, A. Mechanisms of neurotransmitter release by amphetamines: a review. Prog. Neurobiol. 75, 406–433 (2005).
Reith, M. E. A. & Gnegy, M. E. Molecular mechanisms of amphetamines. Handb. Exp. Pharmacol. 258, 265–297 (2020).
Zucchi, R., Chiellini, G., Scanlan, T. S. & Grandy, D. K. Trace amine-associated receptors and their ligands. Br. J. Pharmacol. 149, 967–978 (2006).
Berry, M. D., Gainetdinov, R. R., Hoener, M. C. & Shahid, M. Pharmacology of human trace amine-associated receptors: therapeutic opportunities and challenges. Pharmacol. Ther. 180, 161–180 (2017).
Tan, E. S. et al. The molecular basis of species-specific ligand activation of trace amine-associated receptor 1 (TAAR1). ACS Chem. Biol. 4, 209–220 (2009).
Renwick, A. G., Thompson, J. P., O’Shaughnessy, M. & Walter, E. J. The metabolism of cyclamate to cyclohexylamine in humans during long-term administration. Toxicol. Appl. Pharmacol. 196, 367–380 (2004).
Huang, S. et al. GPCRs steer Gi and Gs selectivity via TM5-TM6 switches as revealed by structures of serotonin receptors. Mol. Cell 82, 2681–2695 (2022).
Xu, X. et al. Binding pathway determines norepinephrine selectivity for the human β1AR over β2AR. Cell Res. 31, 569–579 (2021).
Rasmussen, S. G. et al. Crystal structure of the β2 adrenergic receptor–Gs protein complex. Nature 477, 549–555 (2011).
Kohrle, J. & Biebermann, H. 3-Iodothyronamine—a thyroid hormone metabolite with distinct target profiles and mode of action. Endocr. Rev. 40, 602–630 (2019).
Scanlan, T. S. et al. 3-Iodothyronamine is an endogenous and rapid-acting derivative of thyroid hormone. Nat. Med. 10, 638–642 (2004).
Ballesteros, J. A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 25, 366–428 (1995).
Francesconi, V. et al. Novel 1-amidino-4-phenylpiperazines as potent agonists at human TAAR1 receptor: rational design, synthesis, biological evaluation and molecular docking studies. Pharmaceuticals 13, 391 (2020).
Guariento, S. et al. Rational design, chemical synthesis and biological evaluation of novel biguanides exploring species-specificity responsiveness of TAAR1 agonists. Eur. J. Med. Chem. 146, 171–184 (2018).
Bokoch, M. P. et al. Ligand-specific regulation of the extracellular surface of a G-protein-coupled receptor. Nature 463, 108–112 (2010).
Nuber, S. et al. β-Arrestin biosensors reveal a rapid, receptor-dependent activation/deactivation cycle. Nature 531, 661–664 (2016).
Liao, S., Pino, M. J. Jr, Deleon, C., Lindner-Jackson, M. & Wu, C. Interaction analyses of hTAAR1 and mTAAR1 with antagonist EPPTB. Life Sci. 300, 120553 (2022).
Dedic, N. et al. SEP-363856, a novel psychotropic agent with a unique, non-D2 receptor mechanism of action. J. Pharmacol. Exp. Ther. 371, 1–14 (2019).
Kaplan, A. L. et al. Bespoke library docking for 5-HT2A receptor agonists with antidepressant activity. Nature 610, 582–591 (2022).
Xu, P. et al. Structural genomics of the human dopamine receptor system. Cell Res. 33, 604–616 (2023).
Dedic, N., Dworak, H., Zeni, C., Rutigliano, G. & Howes, O. D. Therapeutic potential of TAAR1 agonists in schizophrenia: evidence from preclinical models and clinical studies. Int. J. Mol. Sci. 22, 13185 (2021).
Angrist, B. M., Shopsin, B. & Gershon, S. Comparative psychotomimetic effects of stereoisomers of amphetamine. Nature 234, 152–153 (1971).
Brogden, R. N. & Markham, A. Fenoldopam: a review of its pharmacodynamic and pharmacokinetic properties and intravenous clinical potential in the management of hypertensive urgencies and emergencies. Drugs 54, 634–650 (1997).
Xiao, P. et al. Ligand recognition and allosteric regulation of DRD1–Gs signaling complexes. Cell 184, 943–956 (2021).
Katritch, V. et al. Allosteric sodium in class A GPCR signaling. Trends Biochem. Sci. 39, 233–244 (2014).
Fenalti, G. et al. Molecular control of delta-opioid receptor signalling. Nature 506, 191–196 (2014).
Wolf, G. et al. Could psychedelic drugs have a role in the treatment of schizophrenia? Rationale and strategy for safe implementation. Mol. Psychiatry 28, 44–58 (2023).
Sukhanov, I. et al. Increased context-dependent conditioning to amphetamine in mice lacking TAAR1. Pharmacol. Res. 103, 206–214 (2016).
Achat-Mendes, C., Lynch, L. J., Sullivan, K. A., Vallender, E. J. & Miller, G. M. Augmentation of methamphetamine-induced behaviors in transgenic mice lacking the trace amine-associated receptor 1. Pharmacol. Biochem. Behav. 101, 201–207 (2012).
Qu, Q. et al. Insights into distinct signaling profiles of the μOR activated by diverse agonists. Nat. Chem. Biol. 19, 423–430 (2023).
Congreve, M., de Graaf, C., Swain, N. A. & Tate, C. G. Impact of GPCR structures on drug discovery. Cell 181, 81–91 (2020).
Carpenter, B., Nehme, R., Warne, T., Leslie, A. G. & Tate, C. G. Structure of the adenosine A2A receptor bound to an engineered G protein. Nature 536, 104–107 (2016).
Xiao, P. et al. Tethered peptide activation mechanism of the adhesion GPCRs ADGRG2 and ADGRG4. Nature 604, 771–778 (2022).
Zhao, J. et al. Prospect of acromegaly therapy: molecular mechanism of clinical drugs octreotide and paltusotine. Nat. Commun. 14, 962 (2023).
Mastronarde, D. N. SerialEM: a program for automated tilt series acquisition on Tecnai microscopes using prediction of specimen position. Microsc. Microanal. 9, 1182–1183 (2003).
Scheres, S. H. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M., A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Xu, P. et al. Structural insights into the lipid and ligand regulation of serotonin receptors. Nature 592, 469–473 (2021).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. D 75, 861–877 (2019).
Williams, C. J. et al. MolProbity: more and better reference data for improved all-atom structure validation. Protein Sci. 27, 293–315 (2018).
Pettersen, E. F. et al. UCSF ChimeraX: structure visualization for researchers, educators, and developers. Protein Sci. 30, 70–82 (2021).
Krissinel, E. Enhanced fold recognition using efficient short fragment clustering. J. Mol. Biochem. 1, 76–85 (2012).
Hori, T. et al. Na+-mimicking ligands stabilize the inactive state of leukotriene B4 receptor BLT1. Nat. Chem. Biol. 14, 262–269 (2018).
Wang, C. et al. Structural basis for molecular recognition at serotonin receptors. Science 340, 610–614 (2013).
Peng, Y. et al. 5-HT2C receptor structures reveal the structural basis of GPCR polypharmacology. Cell 172, 719–730 (2018).
Olsson, M. H., Sondergaard, C. R., Rostkowski, M. & Jensen, J. H. PROPKA3: consistent treatment of internal and surface residues in empirical pKa predictions. J. Chem. Theory Comput. 7, 525–537 (2011).
Webb, B. & Sali, A. Comparative protein structure modeling using MODELLER. Curr. Protoc. Bioinform. 54, 5.6.1–5.6.37 (2016).
Lomize, M. A., Pogozheva, I. D., Joo, H., Mosberg, H. I. & Lomize, A. L. OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res. 40, D370–D376 (2012).
Jo, S., Kim, T., Iyer, V. G. & Im, W. CHARMM-GUI: a web-based graphical user interface for CHARMM. J. Comput. Chem. 29, 1859–1865 (2008).
Wu, E. L. et al. CHARMM-GUI Membrane Builder toward realistic biological membrane simulations. J. Comput. Chem. 35, 1997–2004 (2014).
Pall, S. et al. Heterogeneous parallelization and acceleration of molecular dynamics simulations in GROMACS. J. Chem. Phys. 153, 134110 (2020).
Huang, J. et al. CHARMM36m: an improved force field for folded and intrinsically disordered proteins. Nat. Methods 14, 71–73 (2017).
Feng, Y. et al. Mechanism of activation and biased signaling in complement receptor C5aR1. Cell Res. 33, 312–324 (2023).
Olsen, R. H. J. et al. TRUPATH, an open-source biosensor platform for interrogating the GPCR transducerome. Nat. Chem. Biol. 16, 841–849 (2020).
Yang, X. et al. Molecular mechanism of allosteric modulation for the cannabinoid receptor CB1. Nat. Chem. Biol. 18, 831–840 (2022).
Sveidahl Johansen, O. et al. Lipolysis drives expression of the constitutively active receptor GPR3 to induce adipose thermogenesis. Cell 184, 3502–3518 (2021).
Ping, Y. Q. et al. Structures of the glucocorticoid-bound adhesion receptor GPR97–Go complex. Nature 589, 620–626 (2021).
De Lean, A., Stadel, J. M. & Lefkowitz, R. J. A ternary complex model explains the agonist-specific binding properties of the adenylate cyclase-coupled beta-adrenergic receptor. J. Biol. Chem. 255, 7108–7117 (1980).
Strachan, R. T. et al. Divergent transducer-specific molecular efficacies generate biased agonism at a G protein-coupled receptor (GPCR). J. Biol. Chem. 289, 14211–14224 (2014).
Cheng, J. et al. Autonomous sensing of the insulin peptide by an olfactory G protein-coupled receptor modulates glucose metabolism. Cell Metab. 34, 240–255 (2022).
Fu, Y. et al. Cartilage oligomeric matrix protein is an endogenous β-arrestin-2-selective allosteric modulator of AT1 receptor counteracting vascular injury. Cell Res. 31, 773–790 (2021).
Cao, Y. et al. Unraveling allostery within the angiotensin II type 1 receptor for Gαq and β-arrestin coupling. Sci. Signal. 16, eadf2173 (2023).
Acknowledgements
We thank the staff at the Translational Medicine Core Facility of Advanced Medical Research Institute at Shandong University for their technical assistance; the staff of Sichuan University West China Cryo-EM Center and Cryo-EM Center in Southern University of Science and Technology for cryo-EM data collection; and the staff at the Cryo-EM Center in Shandong University for technical support. This work was supported by the National Natural Science Foundation of China (32100988; 32371288 to W.Y., T2221004 to S.Y. and Z.S., 31972916 to Z.S., 81825022 to J.-P.S., 82271190 and 32100965 to L.C.), Science and Technology Department of Sichuan Province (2022ZYD0085 to Z.S.), Frontiers Medical Center, Tianfu Jincheng Laboratory Foundation (TFJC2023010010 to Z.S.), the China Postdoctoral Science Foundation (BX20220219 to Z.X.), 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (ZYYC20023 to Z.S.), National Science Fund for Excellent Young Scholars (82122070 to F.Y.), National Key R&D Program of China (2022YFC2702603 to F.Y.), STI2030-Major Projects (2021ZD0203000 and 2021ZD0203003 to X.W.), the Beijing National Laboratory for Molecular Sciences (BNLMS202108 to X.W.), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0450102 to X.W.) and the Chinese Academy of Sciences (CAS) Pioneer Hundred Talents Program. This work used resources from the Duyu High Performance Computing Center, Sichuan University, and Big Data Platform at West China Hospital of Sichuan University (WCH-BDP).
Author information
Authors and Affiliations
Contributions
Z.S., W.Y., X. Yu and J.-P.S. initiated structural studies of TAAR and their ligands. Z.X., J.Y., S.S. and Y.F. designed the expression constructs, and purified and prepared the hTAAR1–Gs complexes (T1AM–hTAAR1–Gs, SEP-363856–hTAAR1–Gs, (S)-AMPH–hTAAR1–Gs, RO-6889450–hTAAR1–Gs, fenoldopam–hTAAR1–Gs, A77636–hTAAR1–Gs) and SEP-363856–5-HT1AR–Gi complexes with assistance from H.H. and L.S.; L.G. and W.Z. performed purification of the mTAAR1–Gs complexes (SEP-363856–mTAAR1–Gs, T1AM–mTAAR–Gs) under the supervision of X. Yong and F.Y.; Z.X., C.Z. and X.T. performed cryo-EM screening, data collection, model building, refinement and MD simulations in the study. Hongshuang Wang and X.W. prepared the AMPH. H.H. and L.S. prepared plasmids. C.W., Y.D. and K.W. performed functional assays on T1AM, SEP-363856, RO-6889450, fenoldopam and A77636 with the help of P.C., S.G. and Heli Wang. K.W., S.G. and C.W. contributed to the G-protein-dissociation assay. W.Z. performed radioligand-binding assays. X.Z. contributed to the purification of scFv16. Z.X., S.S., X. Yong and C.W. prepared figures with assistance from X.T., C.Z., L.C., J.Z., L.G. and F.Y.; Z.X., S.S. and C.W. planned and coordinated the entire project with the help of Y.X., X. Yong and L.L.; W.Y. and Z.S. supervised the overall project and wrote the manuscript with the help of J.-P.S., X. Yu, S.Y. and L.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Tao Che, Thomas Keck and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Activation of TAAR1 signalling induced by different ligands.
Concentration dependent activation of hTAAR1 (a, c) and mTAAR1 (b, d) in response to different agonists stimulation examined by G-proteins dissociation assays. Data are presented as mean ± SEM of three independent experiments performed in triplicate.
Extended Data Fig. 2 Structural comparison of TAAR1 with other aminergic receptors.
a, Superposed structures of diverse agonist-bound TAAR1 with T1AM-hTAAR1. The root mean square deviation (RMSD) levels were calculated using T1AM-hTAAR1 structure as the reference. b, Structural superposition of T1AM-hTAAR1 structure with DRD1 (PDB:7CKZ), 5-HT4R (PDB:7XTA), β1AR (PDB:7BTS) and β2AR (PDB:3SN6), respectively. c, Structural comparison of T1AM-hTAAR1 with SEP-363856-hTAAR1 and RO-6889450-hTAAR1 (left panel) or T1AM-mTARR1 with SEP-363856-mTARR1 (right panel) revealed a similar ECL2 in TAAR1 receptor. d, Structural superposition of T1AM-hTAAR1 structure with SEP-363856-bound 5-HT1AR, dopamine-bound DRD1 (PDB:7CKZ) and adrenaline-bound β2AR (PDB: 3SN6).
Extended Data Fig. 3 Pharmacological characterization of T1AM at TAAR1.
a, Sequence alignment of residues involved in T1AM binding in hTAAR1 and mTAAR1. The conserved residues are shown as green, residues that specifically interacted with T1AM are coloured as grey. b, Structural comparison of ligand binding mode of T1AM in mTAAR1 with dopamine in DRD1. c, Heatmap visualizes the effect of mutations in the ligand binding pocket of mTAAR1 in response to T1AM, as determined by the CAMYEL assay. The heatmap is coloured according to the value of ΔpEC50 (ΔpEC50 = pEC50 of mutant - pEC50 of WT) and Emax (relative WT%). ND, not detected or cannot be established over the tested concentration range. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type. d, e, Representative curve of key residues involved in activation of hTAAR1 (d) or mTAAR1 (e) by CAMYEL assay. Data represent mean ± SEM from three independent experiments performed in triplicate. WT, wild type. f, Competitive binding curves of T1AM to wild type hTAAR1 or its mutants in pocket 1 and pocket 2. [3H]-tyramine was used in our study. The Ki values of wild type hTAAR1 for T1AM are 23.59 ± 0.62 nM. Data are presented as mean ± SEM of three independent experiments performed in triplicate. g, Representative curve for effects of mutations in pocket 1 (left panel) and pocket 2 (right panel) from hTAAR1 in response to the T1AM stimulation by FlAsH-BRET assay. Values are means ± SEM from three independent experiments performed in triplicate. h, Structural comparison of the hTAAR1-T1AM (sea green) with mTAAR1-T1AM (light salmon) reveals notable differences (hThr/mAla5.42) in ligand binding pocket. Residue are shown as tan sticks in hTAAR1 and medium slate blue sticks in mTAAR1.
Extended Data Fig. 4 Sequence alignment of key residues of TAARs in the ligand binding pocket.
The key residues in pocket 1, pocket 2, pocket 3 and pocket 4 of TAAR1 are sequentially aligned among members of TAAR subfamily.
Extended Data Fig. 5 Pharmacological characterization of SEP-363856 at TAAR1 and 5-HT1AR.
a, 2D representation of interaction of SEP-363856 with h/mTAAR1. b, The superimposed structures of SEP-363856-bound hTAAR1 (green/blue) with SEP-363856-bound mTAAR1 (yellow/cyan) reveal nearly identical binding pose of ligand. c, d, Comparison of the binding mode of SEP-363856 and T1AM in hTAAR1 (c) and mTAAR1 (d). e-g, Glosensor assay of receptors WT and mutations in hTAAR1 (e), mTAAR1 (f) and 5-HT1AR (g) on SEP-363856-induced Gs signalling. Data represent mean ± SEM from three independent experiments performed in triplicate. WT, wild type. h, Competitive binding curves of SEP-363856 to wild type hTAAR1 or its mutants in pocket 1 and pocket 2. [3H]-tyramine was used in our study. The Ki values of wild type hTAAR1 for SEP-363856 are 22.59 ± 0.60 nM. Data are presented as mean ± SEM of three independent experiments performed in triplicate. i, j, Representative curve for effects of the Phe186ECL2 mutations in hTAAR1 (i) or Phe185ECL2 mutations in mTAAR1 (j) on SEP-363856-induced Gs signalling detected by Glosensor assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type. k, The residues in ECL2 of TAAR1 are sequentially aligned among aminergic receptors. * Representing the corresponding residues in aminergic receptors that relatively to Phe186ECL2 of hTAAR1. l, Structural comparison of the hTAAR1-SEP363856 with 5-HT1AR-SEP363856 reveals that the residues Phe186ECL2 in hTAAR1 and Lys191ECL2 in 5-HT1AR are different. Residues are shown as dodger blue sticks in hTAAR1 and pale violet red sticks in 5-HT1AR, respectively. m, Representative curve for effects of the Lys191ECL2A mutation in 5-HT1AR on SEP-363856-induced Gi signalling detected by Glosensor assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type.
Extended Data Fig. 6 Structural features of RO-6889450 and (S)-AMPH bound.
a, 2D representation of interaction of RO-6889450 with hTAAR1. b, Representative dose response curves of the RO-6889450-induced FlAsH-BRET ratio for the mutants of residues Phe1544.56 and Ser1945.42 in pocket 3. Values are means ± SEM from three independent experiments performed in triplicate. c, Mutational effects of Phe1544.56A and Ser1945.42A on hTAAR1-Gs (left panel), hTAAR1-Gi (middle panel) or hTAAR1-Gq (right panel) signalling in response to RO-6889450 stimulation by Gαs-γ1, Gαi1-γ2 or Gαq-γ9 dissociation assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type. d, Structural comparison of (S)-AMPH with SEP-363856-bound in hTAAR1 at pocket 1 and pocket 2. e, Conformational changes of key residues from pocket 2 in TAAR1 binds to (S)-AMPH or T1AM. f, The orientation of (S)-AMPH in 500 ns molecular dynamic (MD) simulation course. g, Heatmap was visualized the effects of mutations of key residues from hTAAR1 in response to (S)-AMPH. The heatmap is coloured according to the value of ΔpEC50 (ΔpEC50 = pEC50 of mutant - pEC50 of WT) and Emax (relative WT%). ND, not detected or cannot be established over the tested concentration range. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type. h, Representative curve for effects of mutations in pocket 1 (upper panel) and pocket 2 (lower panel) on (S)-AMPH-induced Gs signalling using Gαs-γ1 dissociation assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type.
Extended Data Fig. 7 Structural features of fenoldopam- or A77636-bound hTAAR1.
a, Screening of 22 synthetic molecules for hTAAR1 activation, as assessed using the Glosensor assay. At the administration of 10 μM, two of the 22 compounds showed >50% of activity of agonist β-PEA and SEP-363856. Data shown as mean ± standard error of the mean (SEM) (n = 4 biologically independent samples). ***P < 0.001. Comparison between buffer and several ligands. All the data were determined by one-way of variance ANOVA with Dunnett’s test (From left to right, P = < 0.001, <0.001, <0.001, <0.001, 0.999, 0.265, > 0.999, > 0.999, 0.988, > 0.999, 0.999, 0.995, 0.999, 0.971, 0.969, > 0.999, 0.994, 0.994, > 0.999, > 0.999, > 0.999, 0.988, 0.948, > 0.999). b, Representative dose-response curves for hTAAR1-Gs signalling simulated by the endogenous agonist β-PEA, as well as SEP-363856, fenoldopam, and A77636, as determined by the CAMYEL assay. All curves are mean ± SEM of three independent experiments performed in triplicate (n = 3). c, d, Activation potency and efficacy of hTAAR1 and mTAAR1 induced by β-PEA, fenoldopam and A77636 using CAMYEL assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. e, f, Structural details of fenoldopam (magenta, stick) (e) and A77636 (orange, stick) (f) bound to hTAAR1. Schematic representation of residues contacts with fenoldopam and A77636 in hTAAR1. Residues in pocket 1, 2 and 4 are shown in pink boxes, green boxes and medium slate blue boxes, respectively. Residues located concurrently in pocket 2 and pocket 4 are placed in a green/medium slate blue mixed box. g, Different binding pose of fenoldopam in the structure between DRD1 and hTAAR1. h, Structural comparison of fenoldopam-DRD1 with A77636-hTAAR1.
Extended Data Fig. 8 The role of pocket 4 in TAAR1.
a-h, Structural representations of the polar interaction network of the residues at position 2.61, 2.64, 3.32 and 7.43, for the hTAAR1 with SEP-363856 (a), T1AM (b), (S)-AMPH (c), RO-6889450 (d), fenoldopam (g) and A77636 (h), or mTAAR1 with SEP-363856 (e), T1AM (f), respectively. The estimated hydrogen bonds are represented with red dash lines. i, Representative curve for effects of the S802.61A, R832.64A and H993.28A mutations in hTAAR1 on ligands (fenoldopam, A77636, T1AM, SEP-363856) induced Gs signalling using CAMYEL assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type. j, Representative curve for effects of the S802.61A, R832.64A and H993.28A mutations in hTAAR1 on ligands (fenoldopam, A77636, T1AM, SEP-363856) binding using FlAsH-BRET assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate.
Extended Data Fig. 9 Activation of TAAR1 by diverse agonists.
a, Cut-away view of the ligand-binding pocket in the hTAAR1 complexes: T1AM, SEP-363856, (S)-AMPH, RO-6889450, fenoldopam and A77636. b, Cut-away view of the ligand-binding pocket in the mTAAR1 complexes: T1AM, SEP-363856. c, The residues of the core ligand binding pocket are resembled in the SEP-363856-, RO-6889450- and fenoldopam-bound structures. The hydrogen bonds shown as red dashed lines. d, e, Structural representation of the detailed interactions of SEP-363856 and RO-6889450 with the key residues in pocket 3 (e), and SEP-363856, A77636 and fenoldopam with the key residues in pocket 4 (f). The hydrogen bonding is shown as red dashed lines. The red arrows indicate direction of movement. f, Structural superimposition of A77636-bound hTAAR1 with SEP-33856-bound mTAAR1 revealed that Pro183ECL2 and Tyr2877.39 in mTAAR1 are different from the allelic residue in hTAAR1. g, Representative curve for effects of the V184ECL2A and V184ECL2P mutations in hTAAR1 on A77636-induced Gs signalling using CAMYEL assay. Data are presented as mean ± SEM of three independent experiments performed in triplicate. WT, wild type. h, j, Schematic diagram representing the key residues that contribute to the Gs (h) or Gi (j) proteins coupling with TAAR1. hTAAR1 is depicted as a green cartoon. Residues crucial for Gs or Gi activation are denoted with blue circle (h) or pink circle (j), respectively. i, k, Heatmap illustrating the effect of mutations on the signalling pathways of hTAAR1-Gs (i) and hTAAR1-Gi (k) in response to (S)-AMPH, as evaluated by the Gαs-γ1 and Gαi1-γ2 dissociation assays. Data are presented as mean ± SEM of three independent experiments performed in double.
Supplementary information
Supplementary Information
Supplementary Figs. 1–19, Supplementary Tables 1–5 and Supplementary Methods.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Z., Guo, L., Yu, J. et al. Ligand recognition and G-protein coupling of trace amine receptor TAAR1. Nature 624, 672–681 (2023). https://doi.org/10.1038/s41586-023-06804-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-06804-z
This article is cited by
-
Dopamine D1 receptor in medial prefrontal cortex mediates the effects of TAAR1 activation on chronic stress-induced cognitive and social deficits
Neuropsychopharmacology (2024)
-
Newly identified structures of trace-amine associated receptor-1 (TAAR1) will aid discovery of next generation neuropsychiatric drugs
Molecular Psychiatry (2024)
-
Structures of the amphetamine-binding receptor will aid drug discovery
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.