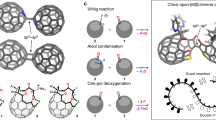
Abstract
Cyclic nanometre-scale sandwich complexes assembled from individual building blocks were synthesized. Sandwich complexes, in which a metal ion is π-coordinated by two planar aromatic organic rings belong to the foundations of organometallic chemistry. They have been successfully used in a wide variety of applications ranging from catalysis, synthesis and electrochemistry to nanotechnology, materials science and medicine1,2. Extending the sandwich structural motif leads to linear multidecker compounds, in which aromatic organic rings and metal atoms are arranged in an alternating fashion. However, the extension to a cyclic multidecker scaffold is unprecedented. Here we show the design, synthesis and characterization of an isomorphous series of circular sandwich compounds, for which the term ‘cyclocenes’ is suggested. These cyclocenes consist of 18 repeating units, forming almost ideally circular, closed rings in the solid state, that can be described by the general formula [cyclo-MII(μ-η8:η8-CotTIPS)]18 (M = Sr, Sm, Eu; CotTIPS = 1,4-(iPr3Si)2C8H62−). Quantum chemical calculations lead to the conclusion that a unique interplay between the ionic metal-to-ligand bonds, the bulkiness of the ligand system and the energy gain on ring closure, which is crucially influenced by dispersion interactions, facilitate the formation of these cyclic systems. Up to now, only linear one-dimensional multidecker sandwich compounds have been investigated for possible applications such as nanowires3,4,5,6,7,8,9,10. This textbook example of cyclic sandwich compounds is expected to open the door for further innovations towards new functional organometallic materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available in the main text or in the supplementary material. Correspondence and request for materials should be addressed to P.W.R.
Code availability
The TURBOMOLE quantum chemistry program suite is available from https://www.turbomole.org. Calculations were done on a cluster consisting of 8 PCs with 24 Intel(R) Xeon(R) Gold 6212U CPUs each.
References
Togni, A. Metallocenes: Synthesis, Reactivity, Applications (Wiley-VCH, 1998).
Štěpnička, P. Ferrocenes: Ligands, Materials and Biomolecules (John Wiley & Sons, Ltd, 2008).
Miyajima, K., Knickelbein, M. B. & Nakajima, A. Stern−Gerlach study of multidecker lanthanide–cyclooctatetraene sandwich clusters. J. Phys. Chem. A. 112, 366–375 (2008).
Hosoya, N. et al. Lanthanide organometallic sandwich nanowires: formation mechanism. J. Phys. Chem. A. 109, 9–12 (2005).
Kurikawa, T. et al. Multiple-decker sandwich complexes of lanthanide-1,3,5,7-cyclooctatetraene [Lnn(C8H8)m] (Ln = Ce, Nd, Eu, Ho, and Yb); localized ionic bonding structure. J. Am. Chem. Soc. 120, 11766–11772 (1998).
Tsuji, T. et al. Liquid-phase synthesis of multidecker organoeuropium sandwich complexes and their physical properties. J. Phys. Chem. C. 118, 5896–5907 (2014).
Huttmann, F., Schleheck, N., Atodiresei, N. & Michely, T. On-surface synthesis of sandwich molecular nanowires on graphene. J. Am. Chem. Soc. 139, 9895–9900 (2017).
Hosoya, N. et al. Formation and electronic structures of organoeuropium sandwich nanowires. J. Phys. Chem. A 118, 8298–8308 (2014).
Miyajima, K., Nakajima, A., Yabushita, S., Knickelbein, M. B. & Kaya, K. Ferromagnetism in one-dimensional vanadium–benzene sandwich clusters. J. Am. Chem. Soc. 126, 13202–13203 (2004).
Xiang, H., Yang, J., Hou, J. G. & Zhu, Q. One-dimensional transition metal–benzene sandwich polymers: possible ideal conductors for spin transport. J. Am. Chem. Soc. 128, 2310–2314 (2006).
Kealy, T. J. & Pauson, P. L. A new type of organo-iron compound. Nature 168, 1039–1040 (1951).
Fischer, E. O. & Pfab, W. Cyclopentadien-Metallkomplexe, ein neuer Typ metallorganischer Verbindungen. Z. Naturforsch., B: Chem. Sci. 7, 377–379 (1952).
Wilkinson, G., Rosenblum, M., Whiting, M. C. & Woodward, R. B. The structure of iron bis-cyclopentadienyl. J. Am. Chem. Soc. 74, 2125–2126 (1952).
Werner, H. & Salzer, A. Die Synthese Eines Ersten Doppel-Sandwich-Komplexes: Das Dinickeltricyclopentadienyl-Kation. Synth. React. Inorg. Met.-Org. Chem. 2, 239–248 (1972).
Elschenbroich, C. Organometallics (Wiley-VCH, 2008).
Zhuo, H.-C., Long, J. R. & Yaghi, O. M. Introduction to metal–organic frameworks. Chem. Rev. 112, 673–674 (2012).
Kreno, L. E. et al. Metal–organic framework materials as chemical sensors. Chem. Rev. 112, 1105–1125 (2012).
Edelmann, F. T. Multiple-decker sandwich complexes of f-elements. New J. Chem. 35, 517–528 (2011).
Grossmann, B. et al. Seven doubly bridged ferrocene units in a cycle. Angew. Chem. Int. Ed. 36, 387–389 (1997).
Herbert, D. E. et al. Redox-active metallomacrocycles and cyclic metallopolymers: photocontrolled ring-opening oligomerization and polymerization of silicon-bridged [1]ferrocenophanes using substitutionally-labile Lewis bases as initiators. J. Am. Chem. Soc. 131, 14958–14968 (2009).
Chan, W. Y., Lough, A. J. & Manners, I. Organometallic macrocycles and cyclic polymers by the bipyridine-initiated photolytic ring opening of a silicon-bridged [1]ferrocenophane. Angew. Chem. Int. Ed. 46, 9069–9072 (2007).
Watts, W. E. The [1,1]ferrocenophane system 1. J. Am. Chem. Soc. 88, 855–856 (1966).
Katz, T. J., Acton, N. & Martin, G. [1n]Ferrocenophanes. J. Am. Chem. Soc. 91, 2804–2805 (1969).
Mueller-Westerhoff, U. T. & Swiegers, G. F. A synthesis of the cyclic ferrocene tetramer [1]4ferrocenophane. Chem. Lett. 23, 67–68 (1994).
Inkpen, M. S. et al. Oligomeric ferrocene rings. Nat. Chem. 8, 825–830 (2016).
Wayda, A. L., Mukerji, I., Dye, J. L. & Rogers, R. D. Divalent lanthanoid synthesis in liquid ammonia. 2. The synthesis and X-ray crystal structure of (C8H8)Yb(C5H5N)3.1/2C5H5N. Organometallics 6, 1328–1332 (1987).
Hayes, R. G. & Thomas, J. L. Synthesis of cyclooctatetraenyleuropium and cyclooctatetraenylytterbium. J. Am. Chem. Soc. 91, 6876–6876 (1969).
Münzfeld, L., Hauser, A., Hädinger, P., Weigend, F. & Roesky, P. W. The archetypal homoleptic lanthanide quadruple-decker—synthesis, mechanistic studies, and quantum chemical investigations. Angew. Chem. Int. Ed. 60, 24493–24499 (2021).
Overby, J. S., Hanusa, T. P. & Young, V. G. Redetermination of the zigzag modification of plumbocene at 173 K. Inorg. Chem. 37, 163–165 (1998).
Morrison, C. A., Wright, D. S. & Layfield, R. A. Interpreting molecular crystal disorder in plumbocene, Pb(C5H5)2: insight from theory. J. Am. Chem. Soc. 124, 6775–6780 (2002).
Suta, M., Kühling, M., Liebing, P., Edelmann, F. T. & Wickleder, C. Photoluminescence properties of the ‘bent sandwich-like’ compounds [Eu(TpiPr2)2] and [Yb(TpiPr2)2] – intermediates between nitride-based phosphors and metallocenes. J. Lumin. 187, 62–68 (2017).
Sztainbuch, I. W., Soos, Z. G. & Spiro, T. G. Herzberg–Teller coupling and configuration interaction in a metalloporphyrin model: 1,3,5,7‐tetramethylcyclo‐octatetraene dianion. J. Chem. Phys. 101, 4644–4648 (1994).
Dorenbos, P. Anomalous luminescence of Eu2+ and Yb2+ in inorganic compounds. J. Phys. Condens. Matter 15, 2645–2665 (2003).
TURBOMOLE v.7.6 (University of Karlsruhe and Forschungszentrum Karlsruhe, 1989–2007).
Balasubramani, S. G. et al. TURBOMOLE: modular program suite for ab initio quantum-chemical and condensed-matter simulations. J. Chem. Phys. 152, 184107 (2020).
Foster, J. M. & Boys, S. F. Canonical configurational interaction procedure. Rev. Mod. Phys. 32, 300–302 (1960).
Reed, A. E., Weinstock, R. B. & Weinhold, F. Natural population analysis. J. Chem. Phys. 83, 735–746 (1985).
Schneider, E. K., Weis, P., Münzfeld, L., Roesky, P. W. & Kappes, M. M. Anionic stacks of alkali-interlinked yttrium and dysprosium bicyclooctatetraenes in isolation. J. Am. Soc. Mass. Spectrom. 33, 695–703 (2022).
Shannon, R. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr., Sect. A. 32, 751–767 (1976).
Acknowledgements
C. Anson, M. Bodensteiner and F. Meurer are acknowledged for discussions about single-crystal X-ray diffraction data refinement. M. Dahlen is acknowledged for supporting PL measurements. Funding was provided by Fonds der Chemischen Industrie, Kekulé fellowship (grant no. 110160). We acknowledge support from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) through the Collaborative Research Centre ‘4f for Future’ (CRC 1573 project no. 471424360, projects C1 and Q).
Author information
Authors and Affiliations
Contributions
Experimental work was carried out by L.M. with support from A.H., P.H. and S.G. PL measurements were carried out by S.L. Single-crystal X-ray diffraction experiments and refinement were done by M.T.G. and L.M. DOSY-NMR was done by N.D.K. and C.Z. Density functional theory calculations were performed by S.G. and F.W. Project administration was done by P.W.R. Supervision was the responsibility of F.W., M.M.K. and P.W.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Conrad Goodwin, Andre Schafer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Münzfeld, L., Gillhuber, S., Hauser, A. et al. Synthesis and properties of cyclic sandwich compounds. Nature 620, 92–96 (2023). https://doi.org/10.1038/s41586-023-06192-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-06192-4
This article is cited by
-
Tunable valence tautomerism in lanthanide–organic alloys
Nature Chemistry (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.