Abstract
Phytoplankton blooms in coastal oceans can be beneficial to coastal fisheries production and ecosystem function, but can also cause major environmental problems1,2—yet detailed characterizations of bloom incidence and distribution are not available worldwide. Here we map daily marine coastal algal blooms between 2003 and 2020 using global satellite observations at 1-km spatial resolution. We found that algal blooms occurred in 126 out of the 153 coastal countries examined. Globally, the spatial extent (+13.2%) and frequency (+59.2%) of blooms increased significantly (P < 0.05) over the study period, whereas blooms weakened in tropical and subtropical areas of the Northern Hemisphere. We documented the relationship between the bloom trends and ocean circulation, and identified the stimulatory effects of recent increases in sea surface temperature. Our compilation of daily mapped coastal phytoplankton blooms provides the basis for global assessments of bloom risks and benefits, and for the formulation or evaluation of management or policy actions.
Similar content being viewed by others
Main
Phytoplankton blooms are accumulations of microscopic algae in the surface layer of fresh and marine water bodies. Although many blooms can occur naturally, nutrients linked to anthropogenic eutrophication are expected to intensify their frequency globally2,3,4. Many algal blooms are beneficial, fixing carbon at the base of the food chain and supporting fisheries and ecosystems worldwide. However, proliferations of algae that cause harm (termed harmful algal blooms (HABs)) have become a major environmental problem worldwide5,6,7. For instance, the toxins produced by some algal species can accumulate in the food web, causing closures of fisheries as well as illness or mortality of marine species and humans8,9,10. In other cases, the decay of a dense algal bloom can deplete oxygen in bottom waters, forming anoxic ‘dead zones’ that can cause fish and invertebrate die-offs and ecosystem restructuring, with serious consequences for the well-being of coastal communities1,11. Unfortunately, algal bloom frequency and distribution are projected to increase with future climate change12,13, with some changes causing adverse effects on aquatic ecosystems, fisheries and coastal resources.
Owing to substantial heterogeneity in space and time, algal blooms are challenging to characterize on a large scale5,14, and thus present knowledge does not allow us to answer one of the most fundamental questions: whether algal blooms have changed in recent decades on a global basis6,15,16. For example, although HAB events have been compiled into the UNESCO (United Nations Educational, Scientific, and Cultural Organization) Intergovernmental Oceanographic Commission Harmful Algae Event Database (HAEDAT) globally since 1985, bloom trends are difficult to resolve, owing to inconsistent sampling efforts and the diversity of the eco-environmental or socio-economic effects6. Alternatively, satellite data have been used to monitor the ocean surface continuously since 1997 and have enabled bloom detection in many coastal regions17,18,19. However, the technical difficulties in dealing with complex optical features across different types of coastal waters have so far prohibited their application globally20. To fill this knowledge gap, we developed a method to map global coastal algal blooms and used this tool to examine satellite images between 2003 and 2020, addressing three fundamental questions: (1) where and how frequently global coastal oceans have been affected by phytoplankton blooms; (2) whether the blooms have expanded or intensified over the past two decades, both globally and regionally; and (3) the identity of the potential drivers.
Mapping global coastal phytoplankton blooms
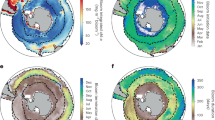
We generated a satellite-based dataset of phytoplankton bloom occurrence to characterize the spatial and temporal patterns of algal blooms in coastal oceans globally. The dataset was derived using global, 1-km resolution daily observations from the Moderate Resolution Imaging Spectroradiometer (MODIS) onboard NASA’s Aqua satellite, and all 0.76 million images acquired by this satellite mission between 2003 and 2020 were used. We developed an automated method to detect phytoplankton blooms using MODIS images (Extended Data Fig. 1) (Methods). In this study, we define a phytoplankton bloom as the accumulation of microscopic algae at the ocean surface that exhibits satellite-detectable fluorescence signals21. However, whether a bloom produces toxins or is harmful to humans or the marine environment is not distinguishable from satellite data. We delineated bloom-affected areas (that is, the areas where algal blooms were detected), and enumerated the bloom count at the 1-km pixel level (that is, the number of detected blooms per pixel) (Fig. 1). We further estimated the bloom frequency (dimensionless) by integrating the bloom count and affected areas within 1° × 1° grid cells (see Methods), and this metric was used to examine temporal dynamics in bloom intensity. Validation with independent satellite samples selected via several visual inspection techniques showed an overall accuracy level of more than 95% for our method, and comparisons using discrete events in HAEDAT6 indicated that we successfully identified bloom counts for 79.3% of the historical HAB events in that database (Extended Data Figs. 2–6). We examined phytoplankton blooms in the exclusive economic zones (EEZs) of 153 coastal countries and in 54 large marine ecosystems (LMEs) (Extended Data Fig. 7). Our study area encompasses global continental shelves and outer margins of coastal currents, which offer the majority of marine resources available for human use (see Methods). Out of the 153 coastal countries examined, 126 were observed to have phytoplankton blooms (Fig. 1). The total bloom-affected area was 31.47 million km2, equivalent to approximately 24.2% of the global land area and 8.6% of the global ocean area, with a median bloom count of 4.3 per year during the past 2 decades (Fig. 1b). Europe (9.52 million km2—30.3% of the total affected area) and North America (6.78 million km2—21.5% of the total affected area) contributed the largest bloom areas. By contrast, the most frequent blooms were found around Africa and South America (median bloom counts of more than 6.3 per year). Australia experienced the lowest frequency (2.4 per year) and affected area (2.84 million km2—9.0% of the total affected area) of blooms.
a, The spatial distribution of annual mean bloom count based on daily satellite detections. b, Continental and global statistics for annual mean bloom count (South America (SA), n = 3,846,125; Africa (AF), n = 2,516,225; Europe (EU), n = 17,703,949; North America (NA), n = 10,034,286; Asia (AS), n = 5,371,158; Australia (AU), n = 2,781,998 pixel observations). The centre line represents the median value, bottom and top bounds of boxes are first and third quartiles, and the whiskers show a maximum of 1.5 times the interquartile range. c, Continental statistics for the long-term annual mean of bloom-affected areas (n = 18 years). The percentages show the corresponding contributions to the global total. The bars represent s.d. Open circles are the affected areas during different years. Map created using Python 3.8.
Phytoplankton blooms occurred frequently in the eastern boundary current systems (that is, California, Benguela, Humboldt and Canary), northeastern USA, Latin America, the Baltic Sea, Northern Black Sea and the Arabian Sea (Fig. 1a). Five LMEs were found with the most frequent blooms (annual median bloom count over 15), including Patagonian Shelf, Northeast US continental Shelf, the Baltic Sea, Gulf of California and Benguela Current (Extended Data Fig. 7). These hotspots are often reported as having a high incidence of algal blooms, some of which are HABs, driven by either coastal upwelling or pronounced anthropogenic nutrient enrichment9,22,23,24,25,26. European LMEs showed mostly large proportions of bloom-affected areas, and some also showed frequent bloom occurrences. By contrast, Asian LMEs exhibited mainly infrequent blooms, given their large affected areas. We identified more bloom events in estuarine regions than along coasts in regions without major river discharge (P < 0.05; Extended Data Fig. 8), highlighting the critical role of terrestrial nutrient sources in coastal algal blooms3.
Long-term trends
The total global bloom-affected area has expanded by 3.97 million km2 (13.2%) between 2003 and 2020, equivalent to 0.14 million km2 yr−1 (P < 0.05; Fig. 2). Furthermore, the number of countries with significant bloom expansion was about 1.6 times those with a decreasing trend. The global median bloom frequency showed an increasing rate of 59.2% (+2.19% yr−1, P < 0.05) over the observed period. Spatially, areas showing significant increasing trends (P < 0.05) in bloom frequency were 77.6% larger than those with the opposite trends (Fig. 2). Globally, our analysis revealed overall consistent fluctuations between the bloom-affected area and bloom frequency between 2003 and 2020 (Fig. 2b). However, there was no significant relationship between bloom extent and frequency in 23 countries and 10 LMEs over the past two decades, underscoring the spatial and temporal variability of algal blooms and the importance of continuous satellite monitoring.
a, Spatial patterns of the trends in bloom frequency at a 1° × 1° grid scale. The latitudinal profiles show the fractions of grids with significant and insignificant trends (positive or negative) along the east–west direction. b, Interannual variability and trends in annual median bloom frequency and total global bloom-affected area. The linear slopes and P-value (two-sided t-test) are indicated. The shading associated with the bloom frequency data represents an uncertainty level of 5% in bloom detection. Map created using Python 3.8.
The entire Southern Hemisphere was primarily characterized by increased bloom frequency, although weakened blooms were also sometimes found. In the Northern Hemisphere, the low latitude (<30° N) coasts were mainly featured with strong bloom weakening (Fig. 2a), primarily in the California Current System and the Arabian Sea. Bloom strengthening was found in the northern Gulf of Mexico and the East and South China Seas, albeit at smaller magnitudes. At higher latitudes, weakening blooms were detected mainly in the northeastern North Atlantic and the Okhotsk Sea in the northwestern North Pacific. Globally, the largest increases in bloom frequency were observed in six major coastal current systems, including Oyashio (+6.31% yr−1), Alaska (+5.22% yr−1), Canary (+4.28% yr−1), Malvinas (+3.02% yr−1), Gulf Stream (+2.42% yr−1) and Benguela (+2.30% yr−1) (Figs. 2a and 3).
a,b, Global patterns of trends in SST gradient (a) and SST (b) from 2003 to 2020. c, Long-term changes in bloom frequency in the regions labelled in a and b, and their relationship to the SST and SST gradient. Linear slope (S) of bloom frequency and the correlation coefficient (r) between bloom frequency and the SST and the SST gradient (∇SST) are shown. Asterisks indicate statistically significant (P < 0.05) correlations. Maps created using ArcMap 10.4 and Python 3.8.
Natural and anthropogenic effects
Increases in sea surface temperature (SST) can stimulate bloom occurrence. We found significant positive correlations (P < 0.05) between the annual mean bloom frequency and the coincident SST (SST data were averaged over the growth window of algal blooms within a year (Methods and Extended Data Fig. 9)) in several high-latitude regions (>40° N), such as the Alaska Current (r = 0.44), the Oyashio Current (r = 0.48) and the Baltic Sea (r = 0.41) (Fig. 3). These findings agree with previous studies, in which the bloom-favourable seasons in these temperate seas have been extended under warmer temperatures27,28,29. However, this temperature-based mechanism did not apply to regions with inconsistent trends between SST and bloom frequency, particularly for the substantial bloom weakening in the tropical and subtropical areas (Figs. 2a and 3b).
Changes in climate can also affect ocean circulation, altering ocean mixing and the transport of nutrients that drive the growth of marine phytoplankton and bloom formation30,31,32. We used the spatial SST gradient (in °C m−1) as a proxy for the magnitude of oceanic mesoscale currents (the time-varying velocity of kinetic energy (also known as the eddy kinetic energy (EKE))) by following the methods of a previous study33, and examined its effects on algal blooms (Methods). The trend in the SST gradient appeared more spatially aligned to bloom frequency than SST. We found significant positive correlations (P < 0.05) between the SST gradient and bloom frequency for various coastal current systems, including the Canary (r = 0.84), Malvinas (r = 0.83), California (r = 0.81), Benguela (r = 0.73), Gulf Stream (r = 0.61) and Oyashio (r = 0.58) currents.
Trends in bloom frequency in subtropical eastern boundary upwelling regions (the California, Benguela and Canary currents) followed the changes in mesoscale currents (Fig. 3a,c). In the California Current System, the decrease in bloom frequency was probably due to the weakened upwelling (represented by a reduced SST gradient and increased SST) and thus lower nutrient supply25. Conversely, the Canary and Benguela currents were characterized by strengthened upwelling and increased bloom frequency. The two western boundary current systems at high latitudes (Malvinas and Oyashio)—although characterized by less pronounced upwelling34—exhibited a similar mechanism to the subtropical eastern boundary regions. For the subtropical western boundary Gulf Stream current, the strengthened current jets (manifested as a larger SST gradient) brought more nutrients from the continental shelf35, triggering more algal blooms. Nevertheless, whether these changes in oceanic mesoscale activities were responses to wind, stratification, the shear of ocean currents or other factors33 requires region-based investigations.
Global climate events, represented as the multivariate El Niño–Southern Oscillation index36 (MEI), also showed connections with coastal bloom frequency. The minimum MEI in 2010 (a strong La Niña year) was followed by a low bloom frequency in the following year, and the largest MEI in 2015 (a strong El Niño year) was followed by the strongest bloom frequency in 2016 (Fig. 2b and Extended Data Fig. 10a).
Changes in anthropogenic nutrient enrichment may have also contributed to the trends in blooms37. For example, the decline in bloom frequency in the Arabian Sea, without clear links to SST or SST gradient changes, could result from decreased fertilizer use in the surrounding countries (such as Iran) (Extended Data Fig. 10). By contrast, the bloom strengthening in some Asian countries could be attributed to surges in fertilizer use38. We examined trends in fertilizer usage (either nitrogen or phosphorus) and bloom frequency and found high positive correlations in China, Iran, Vietnam and the Philippines. Paradoxically, decreased fertilizer uses and increased bloom frequency were identified in some countries, suggesting that nutrient control efforts might have been counterbalanced by the stimulatory effects of climate warming or other factors. Furthermore, the intensified aquaculture industry in Finland, China, Algeria, Guinea, Vietnam, Argentina, Russia and Uruguay may also be linked to their increased bloom incidence, as suggested by the significant positive correlations (r > 0.5, P < 0.05) between their aquaculture production and bloom frequency. A similar relationship between aquaculture expansion and positive trends in HAB incidence was reported from an analysis of HAEDAT data6. However, analogous positive feedbacks for fertilizer or aquaculture were not found in many other countries. Thus, an ecosystem model incorporating terrestrial and oceanic nutrient transport and nutrient–plankton relationships of different species39 is required to quantify the contributions of natural and anthropogenic factors to algal blooms14.
Future implications
We acknowledge that our criteria for a detectable bloom event is operationally defined by sensor sensitivities and other factors, and that the bloom count metric used here may underestimate algal bloom incidence, particularly compared to harmful events entered in HAEDAT. For example, in a recent global analysis of the HAEDAT events, Hallegraeff et al.6 report a dozen or more events per year for each of nine regions over a 33-year study period, compared to the global median bloom count of 4.3 in this study. There are several possible explanations for this discrepancy, such as the many low-cell-concentration HABs that are not detectable from space but that can still cause harm, as well as sensor sensitivities and algorithm thresholds. Furthermore, our bloom count was averaged over all 1-km pixels within the EEZs, whereas HAEDAT entries are based on discrete sampling regions. This underestimation does not, however, alter the trends and other conclusions of this study, as the metrics used here were constant across time and space. Underestimates would have been uniform across regions globally. In this regard, it is of note that the study of Hallegraeff et al.6 found a significant link between the number of HAEDAT events over time and the global expansion of aquaculture production, similar to findings in our study.
The major contribution of our study is to provide a spatially and temporally consistent characterization of global coastal algal blooms between 2003 and 2020. Globally, increasing trends in algal bloom area and frequency are apparent. Regionally, however, trends were non-uniform owing to the compounded effects of changes in climate (such as changes in SST and SST gradient and climate extremes), anthropogenic eutrophication and aquaculture development. Our daily mapping of bloom events offers valuable baseline information to understand the mechanisms underlying the formation, maintenance, and dissipation of algal blooms5,40. This could aid in developing forecasting models (on either global or regional scales) that can help minimize the consequences of harmful blooms, and can also help in policy decisions relating to the control of nutrient discharges and other HAB-stimulatory factors. Noting again that many blooms are beneficial, particularly in terms of their positive effects on ecosystems as well as on wild and farmed fisheries, the results here can also contribute toward policies and management actions that sustain those beneficial blooms.
Methods
Data sources
MODIS on the Aqua satellite provides a global coverage within 1–2 days. All images acquired by this satellite mission from January 2003 to December 2020 were used in our study to detect global coastal phytoplankton blooms, with a total of 0.76 million images. MODIS Level-1A images were downloaded from the Ocean Biology Distributed Active Archive Center (OB.DAAC) at NASA Goddard Space Flight Center (GSFC), and were subsequently processed with SeaDAS software (version 7.5) to obtain Rayleigh-corrected reflectance (Rrc (dimensionless), which was converted using the rhos (in sr−1) product (rhos × π) from SeaDAS)41, remote sensing reflectance (Rrs (sr−1)) and quality control flags (l2_flags). If a pixel was flagged by any of the following, it was then removed from phytoplankton bloom detection: straylight, cloud, land, high sunglint, high solar zenith angle and high sensor zenith angle (https://oceancolor.gsfc.nasa.gov/atbd/ocl2flags/). MODIS level-3 product for aerosol optical thicknesses (AOT) at 869 nm was also obtained from OB.DAAC NASA GSFC (version R2018.0), which was used to examine the impacts of aerosols on bloom trends.
We examined the algal blooms in the EEZs of 153 ocean-bordering countries (excluding the EEZs in the Caspian Sea or around the Antarctic), 126 of which were found with at least one bloom in the past two decades. The EEZ dataset is available at https://www.marineregions.org/download_file.php?name=World_EEZ_v11_20191118.zip. The EEZs are up to 200 nautical miles (or 370 km) away from coastlines, which include all continental shelf areas and offer the majority of marine resources available for human use. Regional statistics of algal blooms were also performed for LMEs. LMEs encompass global coastal oceans and outer edges of coastal currents areas, which are defined by various distinct features of the oceans, including hydrology, productivity, bathymetry and trophically dependent populations42. Of the 66 LMEs identified globally, we excluded the Arctic and Antarctic regions and examined 54 LMEs. The boundaries of LMEs were obtained from https://www.sciencebase.gov/catalog/item/55c77722e4b08400b1fd8244.
We used HAEDAT to validate our satellite-detected phytoplankton blooms in terms of presence or absence. The HAEDAT dataset (http://haedat.iode.org) is a collection of records of HAB events, maintained under the UNESCO Intergovernmental Oceanographic Commission and with data archives since 1985. For each HAB event, the HAEDAT records its bloom period (ranging from days to months) and geolocation. We merged duplicate entries when both the recorded locations and times of the HAEDAT events were very similar to one another, and a total number of 2,609 HAEDAT events were ultimately selected between 2003 and 2020.
We used the ¼° resolution National Oceanic and Atmospheric Administration Optimum Interpolated SST (v. 2.1) data to examine the potential simulating effects of warming on the global phytoplankton trends. We also estimated the SST gradients following the method of Martínez-Moreno33. As detailed in ref. 33, the SST gradient can be used as a proxy for the magnitude of oceanic mesoscale currents (EKE). We used the SST gradient to explore the effects of ocean circulation dynamics on algal blooms.
Fertilizer uses and aquaculture production for different countries was used to examine the potential effects of nutrient enrichment from humans on global phytoplankton bloom trends. Annual data between 2003 and 2019 on synthetic fertilizer use, including nitrogen and phosphorus, are available from https://ourworldindata.org/fertilizers. Annual aquaculture production includes cultivated fish and crustaceans in marine and inland waters, and sea tanks, and the data between 2003 and 2018 are available from https://ourworldindata.org/grapher/aquaculture-farmed-fish-production.
The MEI, which combines various oceanic and atmospheric variables36, was used to examine the connections between El Niño–Southern Oscillation activities and marine phytoplankton blooms. The dataset is available from https://psl.noaa.gov/enso/mei/.
Development of an automated bloom detection method
A recent study by the UNESCO Intergovernmental Oceanographic Commission revealed that globally reported HAB events have increased6. However, such an overall increasing trend was found to be highly correlated with recently intensified sampling efforts6. Once this potential bias was accounted for by examining the ratio between HAB events to the number of samplings5, there was no significant global trend in HAB incidence, though there were increases in certain regions. With synoptic, frequent, and large-scale observations, satellite remote sensing has been extensively used to monitor algal blooms in oceanic environments17,18,19. For example, chlorophyll a (Chla) concentrations, a proxy for phytoplankton biomass, has been provided as a standard product by NASA since the proof-of-concept Coastal Zone Color Scanner (1978–1986) era43,44. The current default algorithm used to retrieve Chla products is based on the high absorption of Chla at the blue band45,46, which often shows high accuracy in the clear open oceans but high uncertainties in coastal waters. This is because, in productive and dynamic coastal oceans, the absorption of Chla in the blue band can be obscured by the presence of suspended sediments and/or coloured dissolved organic matter (CDOM)47. To address this problem, various regionalized Chla algorithms have been developed48. Unfortunately, the concentrations of the water constituents (CDOM, sediment and Chla) can vary substantially across different coastal oceans. As a result, a universal Chla algorithm that can accurately estimate Chla concentrations in global coastal oceans is not currently available.
Alternatively, many spectral indices have been developed to identify phytoplankton blooms instead of quantifying their bloom biomass, including the normalized fluorescence line height21 (nFLH), red tide index49 (RI), algal bloom index47 (ABI), red–blue difference (RBD)50, Karenia brevis bloom index50 (KBBI) and red tide detection index51 (RDI). In practice, the most important task for these index-based algorithms is to determine their optimal thresholds for bloom classification. However, such optimal thresholds can be regional-or image-specific20, due to the complexity of optical features in coastal waters and/or the contamination of unfavourable observational conditions (such as thick aerosols, thin clouds, and so on), making it difficult to apply spectral-index-based algorithms at a global scale.
To circumvent the difficulty in determining unified thresholds for various spectral indices across global coastal oceans, an approach from a recent study to classify algal blooms in freshwater lakes52 was adopted and modified here. In that study, the remotely sensed reflectance data in three visible bands (red, green and blue) were converted into two-dimensional colour space created by the Commission Internationale del’éclairage (CIE), in which the position on the CIE chromaticity diagram represented the colour perceived by human eyes (Extended Data Fig. 1a). As the algal blooms in freshwater lakes were manifested as greenish colours, the reflectance of bloom-containing pixels was expected to be distributed in the green gamut of the CIE chromaticity diagram; the stronger the bloom, the closer the distance to the upper border of the diagram (the greener the water).
Here, the colour of phytoplankton blooms in the coastal oceans can be greenish, yellowish, brownish, or even reddish53, owing to the compositions of bloom species (diatoms or dinoflagellates) and the concentrations of different water constituents. Furthermore, the Chla concentrations of the coastal blooms are typically lower than those in inland waters, thus demanding more accurate classification algorithms. Thus, the algorithm proposed by Hou et al.52 was modified when using the CIE chromaticity space for bloom detection in marine environments. Specifically, we used the following coordinate conversion formulas to obtain the xy coordinate values in the CIE colour space:
where R, G and B represent the Rrc at 748 nm, 678 nm (fluorescence band) and 667 nm in the MODIS Aqua data, respectively. By contrast, the R, G and B channels used in Hou et al.52 were the red, green and blue bands. We used the fluorescence band for the G channel because, for a given region, the 678 nm signal increases monotonically with the Chla concentration for blooms of moderate intensity21, which is similar to the response of greenness to freshwater algal blooms. Thus, the converted y value in the CIE coordinate system represents the strength of the fluorescence. In practice, for pixels with phytoplankton blooms, the converted colours in the chromaticity diagram will be located within the green, yellow or orange–red gamut (see Extended Data Fig. 1a); the stronger the fluorescence signal is, the closer the distance to the upper border of the CIE diagram (larger y value). By contrast, for bloom-free pixels without a fluorescence signal, their converted xy coordinates will be located in the blue or purple gamut. Therefore, we can determine a lower boundary in the CIE two-dimensional coordinate system to separate bloom and non-bloom pixels, similar to the method proposed by Hou et al.52.
We selected 53,820 bloom-containing pixels from the MODIS Rrc data as training samples to determine the boundary of the CIE colour space. These sample points were selected from nearshore waters worldwide where frequent phytoplankton blooms have been reported (Extended Data Fig. 2); the algal species included various species of dinoflagellates and diatoms20. A total of 80 images was used, which were acquired from different seasons and across various bloom magnitudes, to ensure that the samples used could almost exhaustively represent the different bloom conditions in the coastal oceans.
We combined the MODIS FLHRrc (fluorescence line height based on Rrc) and enhanced red–green–blue composite (ERGB) to delineate bloom pixels manually. The FLHRrc image was calculated as:
where Rrc667, Rrc678 and Rrc748 are the Rrc at 667, 678 and 748 nm, respectively, and F667, F678 and F748 are the corresponding extraterrestrial solar irradiance. ERGB composite images were generated using Rrc of three bands at 555 (R), 488 (G) and 443 nm (B). Although phytoplankton-rich and sediment-rich waters have high FLHRrc values, they appear as darkish and bright features in the ERGB images (Extended Data Fig. 3), respectively21. In fact, visual examination with fluorescence signals and ERGB has been widely accepted as a practical way to delineate coastal algal blooms on a limited number of images21,54,55. Note that the FLHRrc here was slightly different from the NASA standard nFLH product56, as the latter is generated using Rrs (corrected for both Rayleigh and aerosol scattering) instead of Rrc (with residual effects of aerosols). However, when using the NASA standard algorithm to further perform aerosol scattering correction over Rrc, 20.7% of our selected bloom-containing pixels failed to obtain valid Rrs (without retrievals or flagged as low quality), especially for those with strong blooms (see examples in Extended Data Fig. 4). Likewise, we also found various nearshore regions with invalid Rrs retrievals. By contrast, Rrc had valid data for all selected samples and showed more coverage in nearshore coastal waters. The differences between Rrs and Rrc were because the assumptions for the standard atmospheric correction algorithm do not hold for bloom pixels or nearshore waters with complex optical properties57. In fact, Rrc has been used as an alternative to Rrs in various applications in complex waters58,59.
We converted the Rrc data of 53,820 selected sample pixels into the xy coordinates in the CIE colour space (Extended Data Fig. 1a). As expected, these samples of bloom-containing pixels were located in the upper half of the chromaticity diagram (the green, yellow and orange–red gamut) (Extended Data Fig. 1a). We determined the lower boundary of these sample points in the chromaticity diagram, which represents the lightest colour and thus the weakest phytoplankton blooms; any point that falls above this boundary represents stronger blooms. The method to determine the boundary was similar to Hou et al.52: we first binned the sample points according to the x value in the chromaticity diagram and estimated the 1st percentile (Q1%) of the corresponding Y for each bin; then, we fit the Q1% using two-order polynomial regression. Sensitivity analysis with Q0.3% (the three-sigma value) resulted in minor changes (<1%) in the resulting bloom areas for single images. Notably, sample points were rarely located near white points (x = 1/3 and y = 1/3, represent equal reflection from three RGB bands) in the diagram, and we used two polynomial regressions to determine the boundaries for x values greater and less than 1/3, which can be expressed as:
Based on the above, if a pixel’s xy coordinate (converted from Rrc spectrum) satisfies the conditions of (x < 1/3 AND y > y1) or (x > 1/3 AND y > y2), it is classified as a ‘bloom’ pixel.
Depending on the local region and application purpose, the meaning of ‘phytoplankton bloom’ may differ. Here, for a global application, the pixelwise bloom classification is based on the relationship (represented using the CIE colour space) between Rrc in the 667-, 678- and 754-nm bands derived from visual interpretation of the 80 pairs of FLHRrc and ERGB imagery. Instead of a simple threshold, we used a lower boundary of the sample points in the chromaticity diagram to define a bloom. In simple words, a pixel is classified as a bloom if its fluorescence signal is detectable (the associated xy coordinate in the CIE colour space located above the lower boundary). Histogram of the nFLH values from the 53,820 training pixels demonstrated the minimum value of ~0.02 mW cm−2 μm−1 (Extended Data Fig. 1a), which is in line with the lower-bound signal of K. brevis blooms on the West Florida shelf21,47. Note that, such a minimum nFLH is determined from the global training pixels, and it does not necessarily represent a unified lower bound for phytoplankton blooms across the entire globe, especially considering that fluorescence efficiency may be a large variable across different regions. Different regions may have different lower bounds of nFLH to define a bloom, and such variability is represented by the predefined boundary in the CIE chromaticity diagram in our study. Correspondingly, although the accuracy of Chla retrievals may have large uncertainties in coastal waters, the histogram of the 53,820 training pixels shows a lower bound of ~1 mg m−3 (Extended Data Fig. 1a). Similarly to nFLH, such a lower bound may not be applicable to all coastal regions, as different regions may have different lower bounds of Chla for bloom definition.
Although the MODIS cloud (generated by SeaDAS with Rrc869 < 0.027) and associated straylight flags can be used to exclude most clouds, we found that residual errors from thin clouds or cloud shadows could affect the spectral shape and cause misclassification for bloom detections. Thus, we designed two spectral indices to remove such effects:
where Index1 and Index2 were used to remove cloud shadows and clouds, respectively. The nRrc443, nRrc488 and nRrc555 in index1 are the normalized Rrc, obtained by normalizing Rrc488. Similar calculations were performed for index2. The purpose of normalizations is to eliminate the effect of the absolute magnitude of the reflectance, so that the thresholds of these two indices are influenced by only the relative magnitude (spectral shape). We determined thresholds for Index1 (>0.12) and Index2 (<0.012) through trial-and-error and ensured that the misclassifications caused by residual errors from clouds and cloud shadows could be effectively removed. After applying the cloud/cloud shadow and various other masks that are associated with l2_flags, we obtained an annual mean valid pixel observation (Nvobs) of ~2.0 × 105 for global 1° × 1° grid cells, and the fluctuation patterns and trends of Nvobs, either annually or seasonally, are different from that of the global bloom frequency and affected areas (see Supplementary Fig. 1).
Assessments of the algorithm performance
In addition to phytoplankton blooms, macroalgal blooms (Sargassum and Ulva) frequently occur in many coastal oceans60,61,62,63. To verify whether our CIE-fluorescence algorithm could eliminate such impacts, we compared the spectra between micro-and macroalgal blooms (see Extended Data Fig. 1b). We found that the spectral shapes of Sargassum and Ulva are substantially different from those of microalgae, particularly for the three bands used for CIE coordinate conversion. The converted xy coordinates for macroalgae were located in the purple–red gamut of the CIE diagram, which was far below the predefined boundary (Extended Data Fig. 1). Moreover, our algorithm is not affected by highly turbid waters for the following two reasons: first, extremely high turbidity tends to saturate the MODIS ocean bands64, which can be easily excluded; second, without a fluorescence peak, the reflectance of unsaturated turbid waters, after conversion to CIE coordinates, will be located below the predefined boundary (see example in Extended Data Fig. 1b). We also confirmed that the spectral shapes of coccolithophore blooms are different from dinoflagellates and diatoms (see example in Extended Data Fig. 1b), and thus they are excluded from our algorithm.
Three different types of validation methods were adopted to demonstrate the reliability of the proposed CIE-fluorescence algorithm for phytoplankton bloom detection in global coastal oceans, including visual inspections of the RGB, ERGB and FLHRrc images, verifications using independent manually delineated algal blooms, and comparisons with the reported HAB events from the HAEDAT dataset.
First, we selected MODIS Aqua images from different locations where coastal phytoplankton blooms have been recorded in the published literature. We visually compared the RGB, ERGB, and FLHRrc images, and our algorithm detected bloom patterns (see examples in Extended Data Fig. 3). Comparisons from various images worldwide showed that our algorithm could successfully identify regions with high FLHRrc values and brownish-to-darkish features on the ERGB images, which can be considered phytoplankton blooms.
Second, we delineated additional 15,466 bloom-containing pixels from 35 images covering different coastal areas, using the same visual inspection and manual delineation method as for the training sample pixels. Moreover, we also selected 14,149 bloom-free pixels (bright or turquoise green colours on ERGB images or low FLHRrc values) within the same images as bloom-containing images. We applied our algorithm to all these pixels, and compared the algorithm-identified and manually delineated results. Our CIE-fluorescence algorithm showed high values in both producer and user accuracies (92.04% and 98.63%) (Supplementary Table 1), and appeared effective at identifying bloom pixels and excluding false negatives (blooms classified as non-blooms) and false positives (non-blooms classified as blooms).
Third, we validated the satellite-detected phytoplankton blooms using in situ reported HAB events from the HAEDAT dataset. For each HAB event in the HAEDAT dataset, we obtained all MODIS images over the reported bloom period (from days to months). Within each year, we estimated the ratio between the number of satellite images with ‘bloom detected’ against the number of valid images (see definition above) during the bloom periods across the entire globe (Supplementary Table 1). Moreover, we calculated the number of events with at least one successful satellite bloom detection (Ns), and then estimated the ratio between Ns and the total HAB events for each year. Results showed that substantial amounts (averaged at 51.2%) of satellite observations acquired during the HAB event periods were found with phytoplankton blooms by our algorithm. Overall, 79.3% of the global HAB events were successfully identified by satellite. The discrepancies between satellite and in situ observations could be explained by the following reasons: first, our study focused only on the phytoplankton blooms that are resolvable by satellite fluorescence signals; other types of HAB events in the HAEDAT dataset may not have been detectable by satellite observations, such as events with lower phytoplankton biomass but high toxicity, occurrences at the subsurface layers, or fluorescence signals overwhelmed by suspended sediments65,66,67. Second, although the HAEDAT recorded HAB events could be sustained for long periods, high biomass of surface algae may not have occurred every day within this period due to the changes in stratification, precipitation, wind, vertical migration of cells, and many other factors68. Third, the spatial scale of certain HAB events may have been too small to be identified using the 1-km resolution MODIS observations. Fourth, a reduced MODIS satellite observation frequency by the contaminations of clouds and land adjacency effects69. Therefore, we believe the underestimations of satellite-detected blooms compared to the in situ reported HAB events were mainly due to inconsistencies between the two observations rather than uncertainties in our algorithm.
Because Rrc depends not only on water colour but also on aerosols (type and concentration) and solar and viewing geometry, a sensitivity analysis was used to determine whether such variables could impact bloom detection. Aerosol reflectance (ρa) with different AOTs at 869 nm was simulated using the NASA-recommended maritime aerosol model (r75f02, with a relative humidity of 75% and a fine-mode fraction of 2%). Then, ρa of each MODIS band was added to Rrc images, and the resulting bloom areas with and without added ρa were compared. Results showed that even with a change of 0.02 in AOT at 869 nm, the bloom areas showed minor changes (<2%) in the tested images; minor changes were also found when we used different aerosol models to conduct ρa simulations70. Note that 0.02 represents the high end of the AOT intra-annual variability in coastal oceans (see Extended Data Fig. 5), and the associated interannual changes are much smaller. Thus, the use of Rrc instead of the fully atmospherically corrected reflectance Rrs could have limited impacts on our detected global bloom trend.
We also tried various index-based algorithms developed in previous studies. However, results showed that all these methods require image-specific thresholds to accurately determine algal bloom boundaries for different coastal regions (see Extended Data Fig. 6). By contrast, although our CIE-fluorescence algorithm may lead to different bloom thresholds for different regions, it can identify bloom pixels without adjusting the coefficients and, therefore, is more suitable for global-scale bloom assessment efforts.
We acknowledge that our satellite-detected algal blooms represent only high amounts of phytoplankton biomass on the ocean surfaces without distinguishing whether such blooms produce toxins or are harmful to marine environments. Furthermore, with only limited spectral information from MODIS, it is difficult to discriminate the phytoplankton species of algal blooms; such information could help to improve our understanding of the impacts of these phytoplankton blooms. However, we expect these challenges to be addressed soon with the scheduled launch of the Plankton, Aerosol, Cloud, ocean Ecosystem (PACE) mission by NASA in 2024, where the hyperspectral measurements over a broad spectrum (350–885 nm) will make species-level classifications possible71.
Exploring the patterns and trends of global coastal phytoplankton blooms
We applied the CIE-fluorescence algorithm to all MODIS Aqua level-2 Rrc images, and a total number of 0.76 million images between 2003 and 2020 were processed. We mapped all detected blooms into 1-km daily scale level-3 composites. The number of bloom counts within a year for each location can be easily enumerated, and the long-term annual mean values were then estimated (Fig. 1a). We further calculated the total global bloom-affected area (the areas where algal blooms were detected at least once) for each year and examined their changes over time (Fig. 2b).
We defined bloom frequency (dimensionless) to represent the density of phytoplankton blooms for a year by integrating the bloom count and bloom-affected areas within 1°×1° grid cells within that year, which is expressed as:
where Mi is the enumerated bloom count for each 1-km resolution pixel in a year within one 1° × 1° grid cell, and n represents the associated number of bloom-affected pixels in the same cell (the number of pixels with Mi > 0), and N is the total number of 1-km MODIS pixels in this grid cell. We estimated the bloom frequency for each year between 2003 and 2020, and determined the long-term trend over global EEZs through a linear least-squares regression (see Fig. 2a).
Continental and country-level statistics were performed for bloom count, bloom-affected areas, and bloom frequency (Fig. 1b,c and Supplementary Table 2), using boundaries for the EEZs of different ocean-bordering countries (see above). Similar statistics were also conducted for 54 LMEs (Extended Data Fig. 7 and Supplementary Table 3).
Correlations with SST and SST gradient
To assess the impacts of climate change on long-term trends in coastal phytoplankton blooms, we correlated the annual mean bloom frequency and the associated SST and SST gradient in various coastal current systems for grid cells with significant changes in bloom frequency (Fig. 3c). The SST and SST gradient were averaged over the growth window within a year, assuming that the changes within the growth window, either in water temperatures or ocean circulations, play more important roles in the bloom trends compared to other seasons32.
We determined the growth window of phytoplankton blooms for each 1° × 1° grid cell (Extended Data Fig. 9a) using the following method: first, we estimated the proportion of cumulative bloom-affected pixels within the grid cells for a year. Second, a generalized additive model72 was used to determine the shape of the phenological curves (Extended Data Fig. 9b), where a log link function and a cubic cyclic regression spline smoother were applied73,74. Third, the timing of maximum bloom-affected areas (TMBAA) was then determined by identifying the inflection point on the bloom growth curve (Extended Data Fig. 9c). To facilitate comparisons across Northern and Southern Hemispheres, the year in the Southern Hemisphere was shifted forward by 183 days (Extended Data Fig. 9c). We characterized the similarity of the bloom growth curve between different grid cells and grouped them into three distinct clusters using a fuzzy c-means cluster analysis method75,76. We found uniform distributions of the clusters over large geographic areas. Cluster I is mainly distributed in mid-low latitudes (<45° N and <30° S), where the maximum bloom-affected areas were expected in the early period of the year. Cluster II was mostly found in higher latitudes, with bloom developments (quasi-) synchronized with increases in SST. Cluster III was detected along the coastlines, where the bloom-affected areas increase throughout the entire year. In practice, the growth window for clusters I and III was set as the entire year, and that for cluster II was set from day 150 to day 270 within the year. We further found that the TMBAA for cluster II showed small changes over the entire period (Extended Data Fig. 9d), indicating relatively stable phenological cycles for those phytoplankton blooms32,77.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The satellite-based dataset of global coastal algal bloom at 1-km resolution and the associated code are available at https://doi.org/10.5281/zenodo.7359262. Source data are provided with this paper.
Change history
06 March 2023
In the version of this article initially published, there was a typo in the last term of equation (4), now corrected to read “x > 1/3,” where the greater than symbol was missing.
References
Breitburg, D. et al. Declining oxygen in the global ocean and coastal waters. Science 359, eaam7240 (2018).
Anderson, D. M. Turning back the harmful red tide. Nature 388, 513–514 (1997).
Beman, J. M., Arrigo, K. R. & Matson, P. A. Agricultural runoff fuels large phytoplankton blooms in vulnerable areas of the ocean. Nature 434, 211–214 (2005).
Heisler, J. et al. Eutrophication and harmful algal blooms: a scientific consensus. Harmful Algae 8, 3–13 (2008).
Anderson, D. M., Cembella, A. D. & Hallegraeff, G. M. Progress in understanding harmful algal blooms: paradigm shifts and new technologies for research, monitoring, and management. Annu. Rev. Mar. Sci. 4, 143–176 (2012).
Hallegraeff, G. M. et al. Perceived global increase in algal blooms is attributable to intensified monitoring and emerging bloom impacts. Commun. Earth Environ. 2, 117 (2021).
Smith, V. H. Eutrophication of freshwater and coastal marine ecosystems a global problem. Environ. Sci. Pollut. Res. 10, 126–139 (2003).
Fleming, L. E. et al. Review of Florida red tide and human health effects. Harmful Algae 10, 224–233 (2011).
Richlen, M. L., Morton, S. L., Jamali, E. A., Rajan, A. & Anderson, D. M. The catastrophic 2008–2009 red tide in the Arabian Gulf region, with observations on the identification and phylogeny of the fish-killing dinoflagellate Cochlodinium polykrikoides. Harmful Algae 9, 163–172 (2010).
Hallegraeff, G. & Bolch, C. Unprecedented toxic algal blooms impact on Tasmanian seafood industry. Microbiol. Aust. 37, 143–144 (2016).
Diaz, R. J. & Rosenberg, R. Spreading dead zones and consequences for marine ecosystems. Science 321, 926–929 (2008).
Barton, A. D., Irwin, A. J., Finkel, Z. V. & Stock, C. A. Anthropogenic climate change drives shift and shuffle in North Atlantic phytoplankton communities. Proc. Natl Acad. Sci. USA 113, 2964–2969 (2016).
Gobler, C. J. Climate change and harmful algal blooms: insights and perspective. Harmful Algae 91, 101731 (2020).
Zohdi, E. & Abbaspour, M. Harmful algal blooms (red tide): a review of causes, impacts and approaches to monitoring and prediction. Int. J. Environ. Sci. Technol. 16, 1789–1806 (2019).
Wells, M. L. et al. Future HAB science: directions and challenges in a changing climate. Harmful Algae 91, 101632 (2020).
Rabalais, N. N., Turner, R. E., Díaz, R. J. & Justić, D. Global change and eutrophication of coastal waters. ICES J. Mar. Sci. 66, 1528–1537 (2009).
Blondeau-Patissier, D., Gower, J. F., Dekker, A. G., Phinn, S. R. & Brando, V. E. A review of ocean color remote sensing methods and statistical techniques for the detection, mapping and analysis of phytoplankton blooms in coastal and open oceans. Prog. Oceanogr. 123, 123–144 (2014).
Wolny, J. L. et al. Current and future remote sensing of harmful algal blooms in the Chesapeake Bay to support the shellfish industry. Front. Mar. Sci. 7, 337 (2020).
Stumpf, R. P. et al. Monitoring Karenia brevis blooms in the Gulf of Mexico using satellite ocean color imagery and other data. Harmful Algae 2, 147–160 (2003).
Bernard, S., Kudela, R. M., Robertson Lain, L. & Pitcher, G. Observation of Harmful Algal Blooms with Ocean Colour Radiometry http://dx.doi.org/10.25607/OBP-1042 (IOCCG, 2021).
Hu, C. et al. Red tide detection and tracing using MODIS fluorescence data: a regional example in SW Florida coastal waters. Remote Sens. Environ. 97, 311–321 (2005).
Andersen, J. H. et al. Long-term temporal and spatial trends in eutrophication status of the Baltic Sea. Biol. Rev. 92, 135–149 (2017).
Gómez, F. & Boicenco, L. An annotated checklist of dinoflagellates in the Black Sea. Hydrobiologia 517, 43–59 (2004).
Townsend, D. W., Pettigrew, N. R. & Thomas, A. C. Offshore blooms of the red tide dinoflagellate, Alexandrium sp., in the Gulf of Maine. Cont. Shelf Res. 21, 347–369 (2001).
Pitcher, G. C., Figueiras, F. G., Hickey, B. M. & Moita, M. T. The physical oceanography of upwelling systems and the development of harmful algal blooms. Prog. Oceanogr. 85, 5–32 (2010).
López-Cortés, D. J. et al. The state of knowledge of harmful algal blooms of Margalefidinium polykrikoides (a.k.a. Cochlodinium polykrikoides) in Latin America. Front. Mar. Sci. 6, 463 (2019).
Anderson, D. M. et al. Evidence for massive and recurrent toxic blooms of Alexandrium catenella in the Alaskan Arctic. Proc. Natl Acad. Sci. USA 118, e2107387118 (2021).
Griffith, A. W., Doherty, O. M. & Gobler, C. J. Ocean warming along temperate western boundaries of the Northern Hemisphere promotes an expansion of Cochlodinium polykrikoides blooms. Proc. R. Soc. B 286, 20190340 (2019).
Conley, D. J. Save the Baltic Sea. Nature 486, 463–464 (2012).
Mahadevan, A., D’Asaro, E., Lee, C. & Perry, M. J. Eddy-driven stratification initiates North Atlantic spring phytoplankton blooms. Science 337, 54–58 (2012).
Chelton, D. B., Gaube, P., Schlax, M. G., Early, J. J. & Samelson, R. M. The influence of nonlinear mesoscale eddies on near-surface oceanic chlorophyll. Science 334, 328–332 (2011).
Boyce, D. G., Petrie, B., Frank, K. T., Worm, B. & Leggett, W. C. Environmental structuring of marine plankton phenology. Nat. Ecol. Evol. 1, 1484–1494 (2017).
Martínez-Moreno, J. et al. Global changes in oceanic mesoscale currents over the satellite altimetry record. Nat. Clim. Change 11, 397–403 (2021).
Kämpf, J. & Chapman, P. in Upwelling Systems of the World 31–65 (Springer, 2016).
Lee, T. N., Yoder, J. A. & Atkinson, L. P. Gulf Stream frontal eddy influence on productivity of the southeast US continental shelf. J. Geophys. Res. 96, 22191–22205 (1991).
Wolter, K. & Timlin, M. S. El Niño/Southern Oscillation behaviour since 1871 as diagnosed in an extended multivariate ENSO index (MEI. ext). Int. J. Climatol. 31, 1074–1087 (2011).
Glibert, P. M. & Burford, M. A. Globally changing nutrient loads and harmful algal blooms: recent advances, new paradigms, and continuing challenges. Oceanography 30, 58–69 (2017).
Lu, C. & Tian, H. Global nitrogen and phosphorus fertilizer use for agriculture production in the past half century: shifted hot spots and nutrient imbalance. Earth Syst. Sci. Data 9, 181–192 (2017).
Falkowski, P. G., Barber, R. T. & Smetacek, V. Biogeochemical controls and feedbacks on ocean primary production. Science 281, 200–206 (1998).
Wells, M. L. et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae 49, 68–93 (2015).
Hu, C. A novel ocean color index to detect floating algae in the global oceans. Remote Sens. Environ. 113, 2118–2129 (2009).
Sherman, K. Adaptive management institutions at the regional level: the case of large marine ecosystems. Ocean Coast. Manag. 90, 38–49 (2014).
Gordon, H. R., Clark, D. K., Mueller, J. L. & Hovis, W. A. Phytoplankton pigments from the Nimbus-7 coastal zone color scanner: comparisons with surface measurements. Science 210, 63–66 (1980).
Moore, J. K. & Abbott, M. R. Phytoplankton chlorophyll distributions and primary production in the Southern Ocean. J. Geophys. Res. 105, 28709–28722 (2000).
Hu, C. et al. Improving satellite global chlorophyll a data products through algorithm refinement and data recovery. J. Geophys. Res. 124, 1524–1543 (2019).
Hu, C., Lee, Z. & Franz, B. Chlorophyll a algorithms for oligotrophic oceans: a novel approach based on three-band reflectance difference. J. Geophys. Res. 117, C01011 (2012).
Hu, C. & Feng, L. Modified MODIS fluorescence line height data product to improve image interpretation for red tide monitoring in the eastern Gulf of Mexico. J. Appl. Remote Sens. 11, 012003 (2016).
Siswanto, E. et al. Empirical ocean-color algorithms to retrieve chlorophyll-a, total suspended matter, and colored dissolved organic matter absorption coefficient in the Yellow and East China Seas. J. Oceanogr. 67, 627–650 (2011).
Ahn, Y.-H. & Shanmugam, P. Detecting the red tide algal blooms from satellite ocean color observations in optically complex Northeast-Asia coastal waters. Remote Sens. Environ. 103, 419–437 (2006).
Amin, R. et al. Novel optical techniques for detecting and classifying toxic dinoflagellate Karenia brevis blooms using satellite imagery. Opt. Express 17, 9126–9144 (2009).
Shen, F., Tang, R., Sun, X. & Liu, D. Simple methods for satellite identification of algal blooms and species using 10-year time series data from the East China Sea. Remote Sens. Environ. 235, 111484 (2019).
Hou, X. et al. Global mapping reveals increase in lacustrine algal blooms over the past decade. Nat. Geosci. 15, 130–134 (2022).
Dierssen, H. M., Kudela, R. M., Ryan, J. P. & Zimmerman, R. C. Red and black tides: quantitative analysis of water-leaving radiance and perceived color for phytoplankton, colored dissolved organic matter, and suspended sediments. Limnol. Oceanogr. 51, 2646–2659 (2006).
Zhao, J., Temimi, M. & Ghedira, H. Characterization of harmful algal blooms (HABs) in the Arabian Gulf and the Sea of Oman using MERIS fluorescence data. ISPRS J. Photogramm. Remote Sens. 101, 125–136 (2015).
Qi, L. et al. Noctiluca blooms in the East China Sea bounded by ocean fronts. Harmful Algae 112, 102172 (2022).
Behrenfeld, M. J. et al. Satellite-detected fluorescence reveals global physiology of ocean phytoplankton. Biogeosciences 6, 779–794 (2009).
Gordon, H. R. Atmospheric correction of ocean color imagery in the Earth Observing System era. J. Geophys. Res. 102, 17081–17106 (1997).
Feng, L., Hou, X., Li, J. & Zheng, Y. Exploring the potential of Rayleigh-corrected reflectance in coastal and inland water applications: a simple aerosol correction method and its merits. ISPRS J. Photogramm. Remote Sens. 146, 52–64 (2018).
Feng, L., Hu, C., Han, X., Chen, X. & Qi, L. Long-term distribution patterns of chlorophyll-a concentration in China’s largest freshwater lake: MERIS full-resolution observations with a practical approach. Remote Sens. 7, 275–299 (2015).
Xiao, J. et al. An anomalous bi-macroalgal bloom caused by Ulva and Sargassum seaweeds during spring to summer of 2017 in the western Yellow Sea, China. Harmful Algae 93, 101760 (2020).
Teichberg, M. et al. Eutrophication and macroalgal blooms in temperate and tropical coastal waters: nutrient enrichment experiments with Ulva spp. Glob. Change Biol. 16, 2624–2637 (2010).
Viaroli, P. et al. Nutrient and iron limitation to Ulva blooms in a eutrophic coastal lagoon (Sacca di Goro, Italy). Hydrobiologia 550, 57–71 (2005).
Dierssen, H. M., Chlus, A. & Russell, B. Hyperspectral discrimination of floating mats of seagrass wrack and the macroalgae Sargassum in coastal waters of Greater Florida Bay using airborne remote sensing. Remote Sens. Environ. 167, 247–258 (2015).
Hu, C. et al. Dynamic range and sensitivity requirements of satellite ocean color sensors: learning from the past. Appl. Opt. 51, 6045–6062 (2012).
Trainer, V. L. et al. Pelagic harmful algal blooms and climate change: lessons from nature’s experiments with extremes. Harmful Algae 91, 101591 (2020).
Mardones, J. I. et al. Disentangling the environmental processes responsible for the world’s largest farmed fish-killing harmful algal bloom: Chile, 2016. Sci. Total Environ. 766, 144383 (2021).
Gilerson, A. et al. Fluorescence component in the reflectance spectra from coastal waters. II. Performance of retrieval algorithms. Opt. Express 16, 2446–2460 (2008).
Lee, J. H., Harrison, P. J., Kuang, C. & Yin, K. in The Environment in Asia Pacific Harbours (ed. Wolanski, E.) 187–206 (Springer, 2006).
Feng, L. & Hu, C. Cloud adjacency effects on top-of-atmosphere radiance and ocean color data products: a statistical assessment. Remote Sens. Environ. 174, 301–313 (2016).
Ahmad, Z. et al. New aerosol models for the retrieval of aerosol optical thickness and normalized water-leaving radiances from the SeaWiFS and MODIS sensors over coastal regions and open oceans. Appl. Opt. 49, 5545–5560 (2010).
Werdell, P. J. et al. The Plankton, Aerosol, Cloud, Ocean Ecosystem mission: status, science, advances. Bull. Am. Meteorol. Soc. 100, 1775–1794 (2019).
Hastie, T. J. Generalized Additive Models (Routledge, 2017).
Wood, S. N. Generalized Additive Models: An Introduction with R (CRC press, 2017).
Macgregor, C. J. et al. Climate-induced phenology shifts linked to range expansions in species with multiple reproductive cycles per year. Nat. Commun. 10, 4455 (2019).
Bezdek, J. C. Pattern Recognition with Fuzzy Objective Function Algorithms (Springer, 2013).
Bi, S. et al. Optical classification of inland waters based on an improved fuzzy C-means method. Opt. Express 27, 34838–34856 (2019).
Kheireddine, M., Mayot, N., Ouhssain, M. & Jones, B. H. Regionalization of the Red Sea based on phytoplankton phenology: a satellite analysis. J. Geophys. Res. 126, e2021JC017486 (2021).
Acknowledgements
We thank NASA for providing global MODIS satellite images, and the Intergovernmental Oceanographic Commission (IOC) of UNESCO for providing the HAEDAT dataset. L.F. was supported by the National Natural Science Foundation of China (no. 41890852, 42271322 and 41971304). D.M.A. was supported by the Woods Hole Center for Oceans and Human Health (National Science Foundation grant OCE-1840381 and National Institutes of Health grants NIEHS-1P01-ES028938-01).
Author information
Authors and Affiliations
Contributions
Y.D. and S.Y.: methodology, data processing and analyses, and writing. L.F.: conceptualization, methodology, funding acquisition, supervision and writing. D.Z.: data processing and analysis. C.H., W.X., D.M.A., Y.L., X.-P.S., D.G.B., L.G. and C.Z. participated in interpreting the results and refining the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Bryan Franz, Bingkun Luo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Development of the CIE-fluorescence algorithm to detect phytoplankton blooms using MODIS satellite imagery.
(a). A1: The density plot of manually delineated bloom-containing pixels in the CIE coordinate system (n = 53,820), and their distribution in the CIE color space (box in A2). A3: Histograms of nFLH and Chla for the delineated pixels, obtained using NASA standard algorithms47,57. (b) MODIS true color composites and selected spectra for phytoplankton blooms, macroalgal blooms (Ulva and Sargassum), coccolithophore blooms, and sediment-rich turbid waters. The x-y numbers indicate their corresponding positions in the CIE coordinate system. The black rectangular boxes in the three lower panels highlight different spectral shapes between phytoplankton blooms and other features near the fluorescence band. Maps created using ArcMap 10.4.
Extended Data Fig. 2 MODIS-detected bloom count within certain years for several coastal regions with frequently reported blooms.
The MODIS observational year is annotated within each panel, and overlaid points indicate in situ recorded harmful algal bloom events from the Harmful Algae Event Database (HAEDAT) within the same year. The lower right panel shows the locations of all the HAEDAT records that were used for algorithm validations in this study (Supplementary Table 1), which also demonstrates the increase in sampling effort in the most recent years. Created using ArcMap 10.4.
Extended Data Fig. 3 Performance of the CIE-fluorescence algorithm for phytoplankton bloom detection in 12 selected coastal oceans.
From left to right are the RGB-true color composite, ERGB composite, FLHRrc, and the bloom area (green pixels) detected by the CIE-fluorescence algorithm. Created using ArcMap 10.4.
Extended Data Fig. 4 Examples showing disadvantages of using NASA standard Rrs (i.e., with the removal of both Rayleigh and aerosol scattering) in algal bloom detection.
From left to right are the RGB composites, ERGB, nFLH, and the bloom areas (green pixels) detected by the CIE-fluorescence algorithm (based on Rrc, without the removal of aerosol scattering). Substantial amounts of invalid Rrs retrievals can be observed in the red-encircled areas in which severe blooms can be found. Additionally, nFLH shows high values at cloud edges (yellow-encircled areas), making it challenging to use a simple threshold to classify blooms. However, such problems can be circumvented in our CIE-fluorescence algorithm. Created using ArcMap 10.4.
Extended Data Fig. 5 Sensitivity analysis of the impacts of aerosols on bloom detection.
(a) Responses of bloom area (BA) to changes in aerosol optical thickness (AOT). Aerosol reflectance (ρa) with AOTs of 0.01 and 0.02 at 869-nm is simulated and added to the MODIS images, and the resulting bloom areas (green pixels) with and without added ρa are compared. The left columns show the RGB composites, and the right three columns show the bloom areas under different AOTs. The percentages of BA changes are annotated in the panels. (b) The standard deviation between the 12 monthly mean values of AOT in global coastal waters (i.e., 66.7% of the intra-annual variability), and the histogram is shown in (c). Maps created using ArcMap 10.4.
Extended Data Fig. 6 Comparison of different index-based algorithms in algal bloom detection in various coastal regions.
Image-specific thresholds (annotated within the panels) are required (labeled within the panels) for RI50, ABI (estimated with FLHRrc)48, RBD51, KBBI51, and RDI52 to delineate accurate bloom areas (i.e., high nFLH values, which appear as bright and darkish features on the ERGB images). The left panels are the bloom areas (green pixels) extracted using our CIE-fluorescence algorithm. The RGB-true color and ERGB composites are shown in Extended Data Fig. 3. Created using ArcMap 10.4.
Extended Data Fig. 7 Annual median bloom count and the proportion of bloom-affected areas for large marine ecosystems (LMEs).
(a) Annual median bloom count, (b) proportion of bloom-affected areas. The data are ordered from the largest to the smallest. The LMEs are grouped by continent, and their names, numbers, and locations are shown in (a) and (b). Map created using Python 3.8.
Extended Data Fig. 8 Comparison of bloom counts in the estuarine and non-estuarine regions.
Boxplots for long-term mean bloom count in the estuarine (n = 13,622 pixel observations) and non-estuarine (n = 361,604 pixel observations) regions. Comparison analysis was performed by two sided Welch’s t-test (P < 0.001).Upper and lower bounds are first and third quartiles, the bar in the middle represents the median value, and the whiskers show the minimum and maximum values. Sixty-two estuarine zones from large rivers were selected, and the boundary of each zone was manually delineated according to high-resolution satellite images.
Extended Data Fig. 9 Clusters of different bloom growth paths.
(a) The spatial distribution of different clusters. The fractions of different clusters across different latitudes are summarized. (b) The development of the maximum bloom-affected areas within a year within 1° × 1° grid cells, where all global grid cells are grouped into three distinct clusters according to the similarity of the bloom growth curve. The colored bond curves represent the mean values of all the grid cells, and their mean SST and associated standard deviations are shown with dashed lines and gray shading. The proportions of different clusters in the global bloom-affected areas are annotated. (c) and (f) The mean timing of the maximum bloom-affected areas (TMBAA) and the associated standard deviations between 2003 and 2019. The whole year in the Southern Hemisphere is shifted forward by 183 days in (c). Maps created using Python 3.8.
Extended Data Fig. 10 Changes in climate extremes, global fertilizer uses, and fishery production over the past two decades.
(a) Changes in the bi-monthly Multivariate El Niño–Southern Oscillation (ENSO) index (MEI) between 2002 and 2020. Positive and negative MEI values represent EI Niño and La Niña events, respectively. The dots show annual mean values. (b–c) Trends of nitrogen and phosphorus from 2003 to 2019 for different countries. (d) Trends of fishery production from 2003 to 2018. Gray indicates no data. Maps created using ArcMap 10.4.
Supplementary information
Supplementary Information
This file contains Supplementary Fig 1, and Supplementary Tables 1-3.
Supplementary Data
This file contains source data for Supplementary Fig. 1.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dai, Y., Yang, S., Zhao, D. et al. Coastal phytoplankton blooms expand and intensify in the 21st century. Nature 615, 280–284 (2023). https://doi.org/10.1038/s41586-023-05760-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-05760-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.