Abstract
Global trends in the occurrence, toxicity and risk posed by harmful algal blooms to natural systems, human health and coastal economies are poorly constrained, but are widely thought to be increasing due to climate change and nutrient pollution. Here, we conduct a statistical analysis on a global dataset extracted from the Harmful Algae Event Database and Ocean Biodiversity Information System for the period 1985–2018 to investigate temporal trends in the frequency and distribution of marine harmful algal blooms. We find no uniform global trend in the number of harmful algal events and their distribution over time, once data were adjusted for regional variations in monitoring effort. Varying and contrasting regional trends were driven by differences in bloom species, type and emergent impacts. Our findings suggest that intensified monitoring efforts associated with increased aquaculture production are responsible for the perceived increase in harmful algae events and that there is no empirical support for broad statements regarding increasing global trends. Instead, trends need to be considered regionally and at the species level.
Similar content being viewed by others
Introduction
Among the 5000 species of known extant marine phytoplankton1, ~200 taxa can harm human society through the production of toxins that threaten seafood security and human health. These toxins are also responsible for wild or aquaculture fish-kills, may interfere with recreational use of coastal or inland waters, or cause economic losses. The toxin-producing species are listed in the IOC-UNESCO Taxonomic Reference List of Harmful MicroAlgae2, which currently includes 105 dinoflagellates, 37 marine cyanobacteria, 31 diatoms, 8 haptophytes, 6 raphidophytes, 3 dictyochophytes and 2 pelagophytes. In addition to toxic species, Harmful Algal Blooms (HABs) can be related to non-toxic microalgae producing high biomass and causing seawater discolorations, anoxia and mucilage that negatively affect the environment and human activities. The occurrences of harmful microalgal species are now routinely recorded in the Ocean Biodiversity Information System, OBIS (https://www.obis.org), a global database on the diversity, distribution and abundance of all marine organisms. Since 1985, reports of harmful algal events impacting on human society have been collated in the Harmful Algae Event Database, HAEDAT (http://haedat.iode.org).
HABs are by no means a new phenomenon but have been reported since historic times. Japanese archives reported coastal ‘red waters’ (‘akashiwo’) as early as the year 731 (putatively by the dinoflagellate Noctiluca scintillans3). Spanish explorers in the 1500 s described ‘red water’ (now presumed to be due to the dinoflagellate Karenia brevis) from the Gulf of Mexico4; Captain Vancouver lost a crew member in 1793 after consumption of blue mussels from Poison Cove, Southeast Alaska, USA, contaminated by PST from the dinoflagellate Alexandrium catenella5; in 1774 Captain Cook and his crew suffered from the symptoms of Ciguatera Poisoning (CP) after consumption of red bass from the New Hebrides (Vanuatu) (now known to be due to species of the dinoflagellate Gambierdiscus)6; Cattle deaths from cyanobacterial toxins (from the brackish water Nodularia spumigena) have been known from Lake Alexandrina, Australia, since 18787. However, several other HAB phenomena such as Diarrhetic Shellfish Poisoning (from the dinoflagellate genus Dinophysis and some Prorocentrum species; a syndrome newly described in 19788), Amnesic Shellfish Poisoning (from some species of the diatom Pseudo-nitzschia; newly described in 19879) and Azaspiracid Shellfish Poisoning (from the dinoflagellates Azadinium and Amphidoma; described in 199610) have only been recently recognised and have triggered monitoring programmes for HAB species and associated toxins across the globe.
One of the most frequently asked questions about HABs is whether they are increasing and expanding11,12,13,14,15,16, and, if so, what are the mechanisms behind the observed trends. Eutrophication, human- and climate-mediated introduction of alien harmful species, climatic variability, and aquaculture developments have all been put forward as causes of a possible expansion and intensification of HABs. It also is apparent that increased monitoring frequency, scientific knowledge and communication, coupled with new methods for identifying and quantifying cells and toxins, have led to more effective detection of blooms and impacts that are not new, but that previously had gone unnoticed or were underestimated. While some workers referred to an ‘apparent global increase’12, driven by increased awareness; or questioned the lack of evidence for HAB increase13, others have alluded to a ‘global epidemic’14 emphasising eutrophication as the key driver. Similarly, the 2019 Intergovernmental Panel for Climate Change assessment report, referring to both marine and freshwater HABs, concluded that ‘the occurrence of HABs, their toxicity and risk on natural and human systems are projected to continue to increase with warming and rising CO2 in the 21st century’17.
A quantitative global assessment of the present status of phenomena related to harmful algae is long overdue, but has not been achieved previously because data on the presence and abundance of harmful species and the trends of HABs were scattered in the increasing literature. Such knowledge-based standpoint on the trends and distribution patterns of harmful species and events is fundamental to enable effective management of HABs at a time that some 3.3 billion people rely on seafood for a fifth of their animal protein and with terrestrial systems becoming less hospitable for traditional agriculture (Food and Agricultural Organisation, FAO18). We here mine the OBIS database to scrutinise trends in phytoplankton and HAB monitoring efforts, and compare them with trends in HAEDAT events impacting human society. Throughout this study, we discriminate between trends in (i) occurrence of the causative microalgal organisms, (ii) detection of toxins in seafood, (iii) incidence of human poisonings, and (iv) fish kills and marine mortalities.
Methods
We explored the following three databases: IOC-UNESCO Taxonomic Reference List of Harmful Microalgae2; HABMAP-OBIS: Database on the geographic range of Harmful Species (https://www.obis.org); and notably HAEDAT, the Harmful Algal Event Database (http://haedat.iode.org). HAEDAT is the only existing database holding information about harmful algal events from across the globe, though the coverage of different regions varies substantially (Supplementary Table 1). HAEDAT data are classified into ‘events’ that lead to management actions or cause any negative economic impact or have ecological consequences of variable magnitude. A harmful algal event can include (i) water discoloration, scum or foam with socio-economic impact either due to the presence of toxic or non-toxic microalgae, (ii) biotoxin accumulation in various seafoods exceeding levels safe for human consumption, (iii) harmful algae abundance resulting in shellfish or other invertebrate harvesting ban or a beach closure as a precautionary measure to protect human health and (iv) any event where humans, animals or other organisms are negatively impacted by algae, either with or without human health consequences. HAEDAT does not hold records when there are no harmful events or no monitoring activities. Not all HAEDAT events have equal societal impacts. This study excluded freshwater harmful algae and macrophyte (seaweed) HABs.
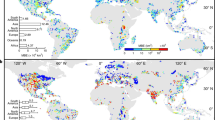
Global HAEDAT events were partitioned into 12 OBIS provinces (Fig. 1A): 1. East Coast USA/Canada, Greenland (abbreviated as ECA), 2. Florida, Caribbean, Central America, Venezuela (CCA), 3. South America (SAM), 4. West Coast USA/Canada, Alaska (WCA), 5. Australia/New Zealand (ANZ), 6. South East Asia (SEA), 7. North Asia (NAS), 8. Indian Ocean (IND), 9. Benguela (BENG, South Africa and Namibia), 10. Mediterranean (MED), 11. West/North Europe including Scandinavia (EUR) and 12. Pacific/Oceania (PAC). Each region was assigned a regional editorial team tasked with collating information on HAB species occurrences and impacts, with the data and interpretations published in a special issue of the journal Harmful Algae (vol. 102, February 2021)19,20,21,22,23,24,25,26,27,28,29,30. As more sampling could lead to more observations of HABs, we estimated sampling effort using OBIS, the most comprehensive dataset on marine species records, as a proxy for sampling effort. The following approach was utilised to estimate sampling effort from OBIS data. A total of 5,944,392 microalgal records were extracted from OBIS, including 289,668 distribution records of harmful species as defined by the IOC-UNESCO Taxonomic Reference list of Harmful Microalgae. OBIS records of microalgal species occurrence were matched with the HAEDAT regions using shapefiles (https://github.com/iobis/hab-regions) and a hexagonal discrete global grid constructed using https://github.com/r-barnes/dggridR. Within each region and each month, the number of grid cells with phytoplankton records was counted, and summed per year to obtain an estimate of sampling effort for that year. In many areas consistent phytoplankton and seafood toxin monitoring only commenced in the period 1985–1990s, and hence for the trend analyses (Figs. 2, 3) we only considered the 30+ year period from 1985 to 2018, covering 7000 HAEDAT records. In some regions, reliable data sets commenced in 1985–1987 (ECA, MED), in others in 1990 (EUR), but the NAS data only start in 2000. At the time of analysis (December 2019), data sets for West Africa, the Benguela Current, Indian Ocean were too sparse and therefore excluded from trend analyses.
A Twelve different geographic regions, with total number of recorded HAEDAT events indicated; B Different types of harmful algal phenomena; and C Different types of seafood toxin syndromes. Paralytic Shellfish Toxins were dominant in East Coast America (ECA), South America (SAM), West Coast America (WCA), South East Asia (SEA) and North East Asia (NAS), Diarrhetic Shellfish Toxins were prevalent in the Mediterranean (MED) and Europe (EUR), and Ciguatera was predominant in the Indian Ocean (IND) and tropical Pacific (PAC). Australia/New Zealand (ANZ) and Central America/Caribbean (CCA) displayed mixtures of events.
A Changes in nine geographic regions in the period 1985–2018 in annual HAEDAT events (algal blooms impacting on human society); 95% confidence intervals are shown in grey; B Meta-analysis of relationship between number of HAEDAT events and Year; and C Meta-analysis of relationship between number of geographic grids with 1 or more HAEDAT events and Year. Weighted mean correlations (filled circles) are shown with 99% confidence limits (bars). Only the Overall Global Totals (bottom) showed statistically significant increases (bars not overlapping zero). Nine geographic regions are considered separately because of great disparity in intensities of monitoring efforts. Data sets for West Africa, the Benguela Current, Indian Ocean, Pacific Ocean were too sparse and therefore excluded from analysis. For definition of HAEDAT regions see Fig. 1A.
A Changes in the period 1985–2018 in the number of annual OBIS microalgal observations as a proxy of scientific and monitoring sampling efforts; B Relative number of HAEDAT events adjusted for OBIS sampling effort; and C Meta-analysis of HAEDAT events adjusted for OBIS records vs. Year. Statistically significant increases in B were recorded for five regions (CCA, SAM, MED, NAS, EUR), while significant decreases occurred in two regions (WCA, ANZ), and two other regions (ECA, SEA) exhibited no significant change. The Overall Global Total (bottom) showed no statistically significant increase in HAEDAT numbers adjusted for OBIS sampling effort; 95% confidence intervals are shown in grey in (A, B). Weighted mean correlations (filled circles) are shown with 99% confidence limits (bars are shown in C).
To reach a consensus concerning the direction of the overall trend in HAB events, we used meta-analysis to combine correlation results across different regions. Meta-analysis is a powerful way to synthesise scientific results and conduct tests of a hypothesis across multiple studies or regions31,32. We conducted four separate meta-analyses. The first was on the ‘number of HAEDAT events’ over years to answer the question whether reported HAB events are increasing. Our analysis followed the methods of Worm and Myers32. We tested the assumption of homogeneity of effect sizes in HAEDAT events across different regions using the \(Q\) statistic (their equation 9) and found effect sizes were significantly different across regions, as is common in nature31. We thus conducted a random effects meta-analysis of correlations, so used the weighted-sample estimate of the unconditional variance for each region (their equation 11) to calculate the mean effect size (their equation 5) and then converted this back to a combined correlation coefficient using the inverse of the \(z\)-transform (their equation 8). We then tested the significance of this combined correlation coefficient by calculating the \(Z\) statistic for a two-tailed test at \(\alpha \,=\,0.05\) (their equation 7). Confidence limits of correlation coefficients were estimated based on equations in Zar33. All meta-analyses were conducted using the same procedures.
In the second meta-analysis, we tried to adjust for some of the potential bias associated with sampling effort by examining the number of geographic grids (global total was 623) with one or more HAB events per year. Geographic grids defined in HAEDAT are areas of coastline (in Europe typically 200 km in length), the central point of which is the focus of HAB monitoring. However, the size of grids varies globally with investment in monitoring and aquaculture developments. We thus conducted a meta-analysis on the time series of the number of geographic grids over years.
The third meta-analysis also tried to correct for some of the potential bias associated with sampling effort by using the OBIS data on phytoplankton sampling. This was to adjust for the potential increase in sampling effort that could lead to a perceived increase in HAEDAT events. We used the ratio of the number of HAEDAT events to OBIS microalgal sampling over years to assess whether standardised HAB events might be increasing.
Finally, the fourth meta-analysis tested the hypothesis that HAEDAT events were increasing as aquaculture has expanded. Our index of aquaculture intensity was the total tonnage of aquaculture produced seafood including fish, molluscs, crustaceans and aquatic plants in a region each year. Suitable aquaculture production data from FAO18, (https://ourworldindata.org/grapher/aquaculture-farmed-fish-production) were available for ANZ, SEA, MED and for the combined regions of ECA and WCA, and SAM and CCA (Supplementary Table 2). Data points were removed where there were zero OBIS observations for a year within a region.
In all meta-analyses, we adjusted for temporal autocorrelation in the time series in each region using the method of Pyper and Peterman34, which does not alter the correlation coefficient, but reduces the effective number of degrees of freedom and thus reduces statistical significance. This approach has previously been applied successfully to assessing changes in plankton time series in the North Atlantic35. Although we could have used generalised linear mixed models to estimate the overall mean relationship between variables, using meta-analysis provides a straightforward way to adjust for temporal autocorrelation in each time series, and thus minimise the risk of inflating the Type I error rate (the probability of incorrectly rejecting the null hypothesis when it is true).
Results and discussion
The global database
As of 10 December 2019 a total of 9503 HAEDAT events had been entered from across the globe, comprising 48% seafood biotoxin, 43% high phytoplankton counts and/or water discolorations causing a socio-economic impact, 7% mass animal or plant mortalities and 2% others (including foam and mucilage production). In a number of HAEDAT records, a single incident was categorised into multiple event types, such as both water discoloration and high phytoplankton count (11% were multiple event types). Among all events linked to seafood toxin syndromes, Paralytic Shellfish Toxins (PST) accounted for 35%, Diarrhetic Shellfish Toxins (DST) 30%, Ciguatera Poisoning (CP) and marine and brackish water cyanobacterial toxins each 9%, Amnesic Shellfish Toxins (AST) 7% and others 10% (including Neurotoxic Shellfish Toxins (NST), Azaspiracid Shellfish Toxins (AZA) and toxic aerosols).
Different OBIS regions (Fig. 1A) contained varying numbers of HAEDAT reports, with the largest number of records available for Europe, followed in order by North Asia, Mediterranean, the east and west coast of North America, the Caribbean, Pacific/Oceania, South East Asia and more limited data sets for South America, and Australia/New Zealand (Supplementary Table 1).
All geographic regions were impacted by multiple HAB types, but in varying proportions (Fig. 1B). High phytoplankton density problems comprised more than 50% of regional HAEDAT records in the Caribbean, Benguela, Mediterranean Sea, North and South East Asia, while seafood toxins and fish kill impacts dominated in all other regions. The productive Benguela upwelling system is prone to mass benthic mortalities linked to high algal biomass and low oxygen19. Among toxin-related impacts, those caused by PST prevailed in North America, the Caribbean, South America, South East Asia and North Asia, whereas DST were the most frequently recorded in Europe and the Mediterranean (Fig. 1C), and are an emerging threat in the USA. NST were confined to Florida (US), with a single outbreak also reported from New Zealand. Human poisonings from Ciguatera were prominent in the tropical Pacific, the Indian Ocean, Australia and the Caribbean. While HAEDAT records of PST, DST and AST mostly relate to the incidence of phycotoxins in seafood, causing closures of shellfish harvesting areas but rarely being associated with human poisonings, records for Ciguatera (CP) refer almost exclusively to human poisonings reported by medical practitioners. HAB events related to marine cyanotoxins were mainly reported from Australia/New Zealand (ANZ), the Indian Ocean (IND) and the Baltic Sea area in NW Europe (EUR).
Temporal trends in HAB events
The number of HAEDAT events reported for each region per year generally showed increases (Fig. 2A). Specifically, eight of the nine regions showed increases of which six were statistically significant (ECA, CCA, WCA, SEA, MED, EUR; Supplementary Table 1). The meta-analysis of the relationship between HAEDAT events and Year, plotted for each region with the significance level and the confidence intervals adjusted by the effective degrees of freedom (reduced because of autocorrelation) is shown in Fig. 2B. Most of the correlations for the individual regions overlapped zero (i.e. they were not significant), but the overall global total meta-analytic correlation was significant (r = 0.37, z = 2.97, p = 0.003) because it combined the information from each individual region and thus provided more statistical power. This suggests that the number of HAEDAT events is increasing over time. The meta-analysis of the relationship between number of geographic grids with one or more HAB events and Year had similar results but weaker statistical significance (r = 0.27, z = 2.16, p = 0.031, Fig. 2C).
While the number of geographic grids with HAB events is less prone to inconsistencies in what constitutes a HAB event and less affected by sampling effort, to try and adjust more specifically for sampling effort we used OBIS data on microalgae sampling. OBIS data generally showed an increase in sampling effort, although the SAM and NAS regions did not follow this trend (Fig. 3A). Once HAEDAT events were adjusted relative to OBIS observations, there were contrasting trends in standardised HAB events over time, with four regions (SAM, WCA, ANZ and NAS) changing direction compared to the unadjusted HAEDAT data (Fig. 3B vs. Fig. 3A). The meta-analysis of the standardised HAB events showed five of the nine regions with a substantially increasing trend (two flat, and two declining), but there was no statistically significant trend overall when all regions were combined (r = 0.35, z = 1.33, p = 0.18), (Fig. 3C). This implies there is insufficient evidence to conclude that HABs are increasing across all the regions analysed, but it is clear there are contrasting trends in individual regions.
In the period studied, aquaculture production increased 16-fold from a global total 11.35M tonnes in 1985 up to 178.5M tonnes in 2018, with the largest increases occurring SEA and SAM + CCA and with North America (ECA + WCA) and EUR stabilising (Fig. 4A). The number of HAEDAT events over time was significantly correlated with aquaculture production, with all regions with suitable data exhibiting more HAEDAT events as aquaculture expanded, with a strongly significant relationship overall (r = 0.43, z = 3.59, p = 0.0003; Fig. 4B bottom; Supplementary Table 2).
A Changes in 1985 to 2018 in five regions (ECA + WCA; SAM + CCA; ANZ; SEA; EUR) of tonnage of Aquaculture Production of fish, molluscs, crustaceans and aquatic plants; and B. Meta-analysis of HAEDAT events over time vs. Aquaculture. The overall number of HAEDAT events over time was significantly correlated with aquaculture production (bottom). Weighted mean correlations (filled circles) are shown with 99% confidence limits (bars) in (B).
Selected HAB case studies
Further exploring the influence of monitoring efforts, Fig. 5 depicts a 4× fold increase of positive global records between 1985 and 2018 of the main causative organisms of Diarrhetic Shellfish Poisoning (DSP; 84,392 OBIS records of the dinoflagellate genus Dinophysis; Fig. 5A), a 7× fold increase of global observations of the main causative organisms of Amnesic Shellfish Poisoning (ASP; 128,282 records of the diatom genus Pseudo-nitzschia; Fig. 5B) and 6× fold increase of global observations of one of the causative organisms of Paralytic Shellfish Poisoning (PSP; 9887 records of the dinoflagellate genus Alexandrium; Fig. 5C). It should be noted that records for Dinophysis, Pseudo-nitzschia and Alexandrium may also include non-toxic species or strains. In all three cases the clear increase in the number of observations through time of causative organisms is paralleled by the increase of HAEDAT records of the associated toxin syndromes (Fig. 5D–F) which in the case of PSP are contributed also by other species, namely the tropical Pyrodinium bahamense and widespread Gymnodinium catenatum. The occurrence of the causative toxigenic HAB species is not always an accurate predictor for the incidence of human shellfish poisonings (indicated by the shellfish icons in Fig. 5G–I). This reflects the efficiency of the food safety risk management strategies implemented in many of the affected countries. Globally, some 11,000 cases of nonfatal events related to DSP were reported, mostly from Europe, South America and Japan (Fig. 5G). These events mainly include closures of shellfish harvesting areas due to observed levels of DST above regulatory limits to protect human health. It is noted that, despite the widespread distribution of Pseudo-nitzschia species (Fig. 5H), there have been no human fatalities from Amnesic Shellfish Poisoning since the original 1987 incident in Prince Edward Island, Canada (150 illnesses with 3 fatalities), even though associated mortalities of marine mammals of high conservation value are of increasing concern in western North America, including in the climate hotspot of Arctic Alaska36. AST has also been associated with marine mammal calf mortalities in Argentina37. Of the global total of 3800 human Paralytic Shellfish Poisonings during the 1985–2018 period (Fig. 5I), the largest number occurred in the Philippines, a country strongly dependent on aquaculture for human food protein, with 2555 poisonings recorded between 1983 and 2013 of which 165 were fatalities23,38, predominantly caused by highly toxic Pyrodinium bahamense. Because of the increased use of molecular detection methods our knowledge on the global distribution of ciguatera- causing organisms, selected species of the benthic dinoflagellates Gambierdiscus and Fukuyoa, has increased considerably (Fig. 6A). The database on the presence of ciguatoxins in fish (Fig. 6B) is still limited because of the complexity of the chemical analysis used to confirm the presence of ciguatoxins. Exploring trends of human CP, in Hawaii poisonings have been decreasing, in French Polynesia and the Caribbean numbers remained stable, whereas CP is a new phenomenon in the Canary Islands (Fig. 6C). Globally CP affects 10,000–50,000 people annually but fatalities are rare20.
A–C Total number of global observations from OBIS of causative microalgal organisms of Dinophysis spp., Pseudo-nitzschia spp. and Alexandrium spp.; D–F The number of records of HAEDAT Toxic Events of DST, AST and PST.; G–I Global distribution maps (as red dots, from OBIS) as of 2018 of Dinophysis spp. (DSP), Pseudo-nitzschia spp. (ASP) and Alexandrium, Pyrodinium, Gymnodinium catenatum (PSP). The locations of toxic events resulting in human poisonings are indicated by the size of the shellfish icons. The first number shows number of poisonings, the second number indicates fatalities. For ASP, 150/3 signals 150 clinical cases with three fatalities. No human fatalities have ever occurred from DSP.
A Distribution of the dinoflagellate genera Gambierdiscus and Fukuyoa (blue and orange dots); B Ciguatoxins in fish (red) and shellfish (orange); and C Trends between 2000 and 2018 in human ciguatera poisonings in Hawaii, French Polynesia, Canary Islands, the Caribbean (light green) and Mexico (dark green). Adapted from Chinain et al.20.
Aquacultured finfish mortalities caused by the taxonomically unrelated microalgal genera Chattonella, Pseudochattonella, Heterosigma, Karenia, Karlodinium, Margalefidinium (Cochlodinium) and Prymnesium/ Chrysochromulina globally account for much greater economic damage than HABs contaminating seafood39. While most shellfish toxins have now been well characterised and are effectively monitored and regulated, finfish held captive in intensive aquaculture operations continue to be vulnerable to HABs (USD71M loss in Japan in 1972, USD70M in Korea in 1995, USD290M in China in 2012, USD100M in Norway in 201919,20,21,22,23,24,25,26,27,28,29,30,31,32), even though the causative ichthyotoxins usually are of no human health significance. The 2016 Chilean salmon mortality that caused a record USD800M loss led to major social unrest40. Again, the incidence of fish-killing HAB species is not an accurate predictor of economic losses. For example, Heterosigma blooms occur both on the west and east coast of North America, but fish mortalities are mostly confined to the west coast29. In large part, this reflects locations where blooms occur relative to the location and size of the aquaculture operations. The dinoflagellate Karlodinium australe never caused any problems in its Australian lagoon type locality41 but in 2014 killed 50,000 caged fish in Malaysia and is now also known from Japan and the Philippines42. In the wild, finfish can swim away from bloom areas, hence aquaculture finfish mortality is largely a human-generated problem.
Conclusions
Our analysis of HAB species and event distribution showed no conclusive evidence for a uniform trend of ‘increased global frequency and distribution of HABs’ in the period 1985–2018. When adjusted for sampling effort proxies, global HAB trends were not statistically significant. This suggests that the increase in HAEDAT event numbers, and hence the widespread perception of a global HAB increase, can be attributed largely to the intensification of observational efforts through scientific and monitoring activities. As for the period prior to 1985, data are sparse and unreliable and do not allow us to exclude that a HAB increase had occurred before that time.
Apart from increased observational efforts, the widely held impression of increased HABs derives from the expanding scale and nature of societal impacts. Increased use of coastal waters for aquaculture has been a key driver for occasionally disastrous, long-lasting economic impacts from HABs, in turn driving an awareness of new harmful algal species and new toxin types43. In the period studied, aquaculture production increased 16-fold, with the largest increases occurring in East Asia, Latin America and the Caribbean and with North America and Europe stabilising18. Alterations in toxin monitoring techniques or changes in regulatory levels also play a key role in the impact levels. Closures from 1998 to 2005 of the Scottish scallop fishery due to high concentrations of AST in the gonad tissues were effectively reduced by an amendment to the European Union shellfish hygiene directive facilitating end product testing and sale of adductor muscle only24. When new HAB phenomena affect previously poorly monitored areas or product (e.g. the 1972 Seto Inland Sea Chattonella associated aquaculture fish kill28, 1987 ASP outbreak in Canada9, 1994 NSP outbreak in New Zealand27, 1997 DSP outbreak in Australia27), economic impacts can initially be severe but subsequently decrease in most cases. Unlike for DST, AST and PST, we do not have adequate monitoring or regulatory tools in place for ciguatera toxins nor fish-killing HABs. Shellfish toxin outbreaks are now well-managed in developed countries and economies can recover swiftly, while finfish aquaculture industries can take many years to rebuild after mass mortalities due to HABs. The perceived positive HAB trend is thus mainly related to the growing need to exploit marine resources driven by human population increase (from 4.85B in 1985 to 7.71B in 201844), which acts as a multiplier13 of the impacts of HABs, independently of their actual trend.
Notwithstanding the lack of evidence for a global increase, variations in HAB occurrence were evident in our study at the regional scale, with statistically supported increasing, decreasing or stable trends and within-region variations of the HAB types driving those trends. As exemplified by selected HAB case studies, increases concerned AST and PST in marine mammals in the Arctic Pacific36, CP in the Canary Islands20, and green45 and red Noctiluca surface water discolorations27 in the Indian Ocean and South East Australia, respectively; benthic Ostreopsis mats and aerosol poisoning since the 2000s in the Mediterranean22, AST on the northeastern and west coast USA29,46,47, and Pseudochattonella blooms causing fish mortalities in northern Europe and Chile25,40. By contrast, in other previous HAB hotspots, harmful events have remained stable or dissipated e.g. Pyrodinium bahamense PST stabilising in the Philippines after 1990s23, and Alexandrium minutum PST disappearing from Egypt after 199448.
The scenario of high variability of impacts and trends observed in our study reflects the wide range of harmful event types caused by more than 200 different microalgae occurring in diversified coastal environments. Such highly variable species and regionally specific patterns render broad statements on HAB occurrence inappropriate. Global and regional assessments are only a first step towards an improved management of HAB impacts, which however is not possible due to lack of understanding of the ecological mechanisms driving global and local HAB trends and patterns. So far, no satisfactory ecological explanation has been found13 for the restricted distribution of phenomena related to specific toxins, such as AST (dominant on the West Coast of North America), AZA (Ireland), DST (dominant in Europe), NST (dominant in Florida), in spite of the wide distribution of the causative organisms. Differences in HAB occurrence among apparently similar sites, and various examples of trends and range expansions mentioned above also remain in most cases unexplained, which calls for ecological studies of individual phenomena at the local scale. Similarly, climate change and eutrophication impacts will vary from species to species16,49,50, and climate change drives different ocean regions to change at different rates51. While some of the apparently new HAB phenomena recognised here occurred in demonstrated temperature increase hotspots40 (Arctic Pacific, Chile, South East Australia), other climate factors such as ocean acidification, nutrient alterations and lowered oxygenation also play a role16. Climate change thus adds a new level of uncertainty to seafood security and safety for an ever-growing human population.
Being at the crossroad of several different societal and scientific issues, questions on HABs are best addressed on a species-by-species and site-by-site basis, and considering the respective impacts on local human activities, rather than handled as aggregates of microalgal HAB cell or species numbers, phycotoxin concentrations, or their global distributions. This is only the first of hopefully many future metadata analyses of the available HAB information, which has clearly shown the potential of the HAEDAT and OBIS data. However, the expansion and integration of these databases52 with other information are needed to capture the ecology of individual noxious species, explain their trends and quantify their economic and health impacts to society. Only with ever improving and better harmonised global data sets can we answer questions on the relationships between HABs, climate, eutrophication and aquaculture with absolute confidence and predict future trends.
Data availability
All individual OBIS and HAEDAT event data used in the present analysis are available via https://www.obis.org and http://haedat.iode.org, respectively. The FAO aquaculture production data are available from https://ourworldindata.org/grapher/aquaculture-farmed-fish-production. The full collated HAEDAT, OBIS and FAO dataset used for the present 1985–2018 meta-analyses are available from https://github.com/iobis/paper-hab-trends. The results of the statistical analyses on these data sets are provided as Supplementary Tables 1, 2 to this publication.
References
Sournia, A. et al. Marine phytoplankton: how many species in the world ocean? J. Plankton Res.13, 1093–1099 (1991).
Moestrup, Ø. et al. (eds). IOC-UNESCO Taxonomic Reference List of Harmful Micro Algae; www.marinespecies.org/hab (2009 onwards). UNESCO, Paris.
Okaichi, T. (ed). Red Tides. 439 (Terra Scientific Publishing Company/Kluwer Academic Publishers, 2004).
Steidinger, K. A. Historical perspective on Karenia brevis red tide research in the Gulf of Mexico. Harmful Algae 8, 549–561 (2009).
Vancouver, G. Voyage of discovery to the North Pacific Ocean, and round the world in the years 1791–95. (John Stockdale London, 1798).
Anderson, W. An account of some poisonous fish in the South Seas. Philos. Trans. R. Soc. London 66, 544 (1776).
Francis, G. Poisonous Australian lake. Nature 18, 11–12 (1878).
Yasumoto, T. et al. Identification of Dinophysis fortii as the causative organism of diarrhetic shellfish poisoning. Bull. Jpn. Soc. Sci. Fish. 46, 1405–1411 (1978).
Bates, S. S. et al. Pennate diatom Nitzschia pungens as the primary source of domoic acid, a toxin in shellfish from eastern Prince Edward Island, Canada. Can. J. Fish. Aquat. Sci. 46, 1203–1215 (1989).
McMahon, T. & Silke, J. West coast of Ireland; winter toxicity of unknown aetiology in mussels. Harmful Algae News 14, 2 (1996).
Anderson, D. M. Toxic algal blooms and red tides: a global perspective. In Red Tides: Biology, Environmental Science and Toxicology (eds. Anderson, D. M. et al.), 11–16 (Elsevier, New York, 1989).
Hallegraeff, G. M. A review of harmful algal blooms and their apparent global increase. Phycologia 32, 79–99 (1993).
Zingone, A., Wyatt, T. Harmful algal blooms: keys to the understanding of phytoplankton ecology. In (eds Robinson, A. R. et al.) The Global Coastal Ocean. Multiscale Interdisciplinary Processes. The Sea: Ideas And Observations On Progress In The Study Of The Seas, 13. 867–926 (Harvard University Press, 2005).
Smayda, T. J. Novel and nuisance phytoplankton blooms in the sea: evidence for a global epidemic. In (eds Graneli, E., Sundström, B., Edler, L. Anderson, D. M.) Toxic Marine Phytoplankton, 29–40 (Elsevier New York, 1990).
Anderson, D. M. HABs in a changing world: a perspective on harmful algal blooms, their impacts, and research and management in a dynamic era of climatic and environmental change. In Proc. 15th International Conference on Harmful Algae (ed. Kim, H. G. et al.), 3–17 (International Society for the Study of Harmful Algae, and IOC of UNESCO, Paris, 2014).
Wells, M. L. et al. Future HAB science: directions and challenges in a changing climate. Harmful Algae 91, https://doi.org/10.1016/j.hal.2019.101632 (2019).
Pörtner, H.-O. et al. IPCC Special Report on the Ocean and Cryosphere in a Changing Climate. https://www.ipcc.ch/srocc (2019).
Food and Agricultural Organisation. The State of World Fisheries and Aquaculture 2020. Sustain. Action https://doi.org/10.4060/ca9229en (2020).
Pitcher, G. C., Louw, D. C. Harmful algal blooms of the Benguela Eastern Boundary upwelling system. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101898 (2021).
Chinain, M. et al. Ciguatera poisonings: a global review of occurrences and trends. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101873 (2021).
McKenzie, C. H. et al. Three decades of Canadian marine harmful algal events: Phytoplankton and phycotoxins of concern to human and ecosystem health. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101852 (2021).
Zingone, A. et al. Toxic microalgae and noxious blooms in the Mediterranean Sea: a contribution to the global HAB status report. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101843 (2021).
Yñiguez, A. T. et al. Over 30 years of HABs in the Philippines and Malaysia: What have we learned? Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101776 (2021).
Bresnan, E. et al. Diversity and regional distribution of harmful algal events along the Atlantic margin of Europe. Harmful Algae 102 https://doi.org/10.1016/j.hal.2021.101976 (2021).
Karlson B. et al. Harmful algal blooms and their effects in coastal seas of northern Europe. Harmful Algae 102 https://doi.org/10.1016/j.hal.2021.101989 (2021).
Sunesen, I., et al. The Latin America and Caribbean HAB status report based on OBIS and HAEDAT maps and databases. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101920 (2021).
Hallegraeff, G. M. et al. Overview of Australian and New Zealand HAB species occurrences and HAEDAT events in the period 1985-2018, including a compilation of historic records. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101848 (2021).
Sakamoto, S. et al. Harmful algal blooms and associated fisheries damage in East Asia: Current status and trends in China, Japan, Korea and Russia. Harmful Algae 102 https://doi.org/10.1016/j.hal.2020.101787 (2021).
Anderson, D. M. et al. Marine harmful algal blooms (HABs) in the United States: history, current status and future trends. Harmful Algae 102 https://doi.org/10.1016/j.hal.2021.101975 (2021).
Belin, C., Soudant, D., Amzil, Z. Three decades of data on phytoplankton and phycotoxins on the French coast: Lessons from REPHY and REPHYTOX. Harmful Algae 102 https://doi.org/10.1016/j.hal.2019.101733
Gurevitch, J., Koricheva, J., Nakagawa, S., Stewart, G. Meta-analysis and the science of research synthesis. Nature 555, 175–182. https://doi.org/10.1038/nature25753 (2018).
Worm, B. & Myers, R. A. Meta-analysis of cod-shrimp interactions reveals top-down control in oceanic food webs. Ecology 84, 162–173 (2003).
Zar, J. H. Biostatistical Analysis (5th edn) (Prentice-Hall/Pearson, 2010).
Pyper, B. J. & Peterman, R. M. Comparison of methods to account for autocorrelation in correlation analyses of fish data. Can. J. Fish. Aquat. Sci. 55, 2127–2140 (1998).
Richardson, A. J. & Schoeman, D. S. Climate impact on plankton ecosystems in the Northeast Atlantic. Science 305, 1609–1612 (2004).
Lefebvre, K. A. et al. Prevalence of algal toxins in Alaskan marine mammals foraging in a changing arctic and subarctic environment. Harmful Algae 55, 13–24 (2016).
Wilson, C. et al. Southern right whale (Eubalaena australis) calf mortality at Peninsula Valdes, Argentina: Are harmful algal blooms to blame? Mar. Mamm. Sci. 32, 423–451 (2016).
Azanza, R. V. & Taylor, F. J. R. Are Pyrodinium blooms in the Southeast Asian region recurring and spreading? A view at the end of the millennium. AMBIO J. Hum. Environ. 30, 356–364 (2001).
Hallegraeff, G. M. et al. Review of progress in our understanding of fish-killing microalgae: implications for management and mitigation. In Proc. 17th International Conference on Harmful Algae, 148–153 (eds Proença, L. A. O., Hallegraeff, G. M.) (International Society for the Study of Harmful Algae, and IOC of UNESCO, Paris, 2017).
Trainer, V. L. et al. Pelagic harmful algal blooms and climate change: lessons from nature’s experiments with extremes. Harmful Algae 91, 101591 (2019).
de Salas, M. F. et al. Karlodinium australe sp.nov. (Gymnodiniales, Dinophyceae), a new potentially ichthyotoxic unarmoured dinoflagellate from lagoonal habitats of south- eastern Australia. Phycologia 44, 640–650 (2005).
Lim, H. C. et al. A bloom of Karlodinium australe (Gymnodiniales, Dinophyceae) associated with mass mortality of cage-cultured fishes in West Johor Strait, Malaysia. Harmful Algae 40, 51–62 (2014).
Hess P. What’s new in Toxins? In Proc. 12th International Conference on Harmful Algae, International Society for the Study of Harmful Algae and IOC of UNESCO, 360–370 (eds. Moestrup, Ø. et al.) (Paris 2008).
Roser, M., Ritchie, H., Ortiz-Ospina, E. World population growth. https://ourworldindata.org/ (2019).
Gomes, H. et al. Massive outbreaks of Noctiluca scintillans blooms in the Arabian Sea due to spread of hypoxia. Nat. Commun. 5, 4862 (2014).
Clark, S. et al. Pseudo-nitzschia bloom dynamics in the Gulf of Maine: 2012–2016. Harmful Algae 88, 101656 (2019).
McKibben, S. M. et al. Climatic regulation of the neurotoxin domoic acid. Proc. Natl Acad. Sci. 114, 239–244 (2017).
Ismael, A. & Halim, Y. Occurrence and succession of potentially harmful phytoplankton species in the Eastern Harbour of Alexandria. In Proc. 9th International Conference Harmful Algal Blooms, Hobart (Australia), 141–143 (eds. Hallegraeff, G. et al.) (Hobart, Australia, 2001).
Davidson, K. et al. Anthropogenic nutrients and harmful algae in coastal waters. J. Environ. Manag. 146, 206–216 (2014).
Hallegraeff, G. Ocean climate change, phytoplankton community responses, and harmful algal blooms: a formidable predictive challenge. J. Phycol. 46, 220–235 (2010).
Bopp, L. et al. Multiple stressors of ocean ecosystems in the 21st century: projections with CMIP5 models. Biogeosciences 10, 6225–6245 (2013).
Zingone, A. et al. Databases for the study of harmful algae, their global distribution and their trends. (eds. Wells, M. et al.) Best Practice Guidelines for the Study of HABs and Climate Change. Paris, UNESCO-IOC, (IOC Manuals and Guides, 88, 2021).
Acknowledgements
This work formed part of the Global Harmful Algae Status Reporting (GHSR) initiative, conducted under the auspices of IOC (Intergovernmental Oceanographic Commission of UNESCO), ICES (International Council for the Exploration of the Sea), PICES (North Pacific Marine Science Organisation), IAEA (International Atomic Energy Agency), and was sponsored by the Government of Flanders (Belgium).
Author information
Authors and Affiliations
Contributions
G.M.H., A.Z., D.M.A., P.A.T. and V.L.T. drafted the main body of the paper. A.R. conducted the statistical analyses, P.P. assisted with data extraction and graphics. H.E. secured funding and facilitated meetings and training workshops. All authors (G.M.H., D.M.A., C.B., M-Y.D.B., E.B., M.C., H.E., M.I., B.K., C.H.McK., I.S., G.C.P., P.P., A.R., L.S., P.A.T., V.L.T., A.T.Y. and A.Z.) contributed to H.A.B. database entry, the writing of regional summaries and checked the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Primary handling editors: Joseph Aslin, Clare Davis.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hallegraeff, G.M., Anderson, D.M., Belin, C. et al. Perceived global increase in algal blooms is attributable to intensified monitoring and emerging bloom impacts. Commun Earth Environ 2, 117 (2021). https://doi.org/10.1038/s43247-021-00178-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43247-021-00178-8
This article is cited by
-
Metagenomic mapping of cyanobacteria and potential cyanotoxin producing taxa in large rivers of the United States
Scientific Reports (2023)
-
Toxic and non-toxic dinoflagellates host distinct bacterial communities in their phycospheres
Communications Earth & Environment (2023)
-
Global river water quality under climate change and hydroclimatic extremes
Nature Reviews Earth & Environment (2023)
-
Coastal algal blooms have intensified over the past 20 years
Nature (2023)
-
Coastal phytoplankton blooms expand and intensify in the 21st century
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.