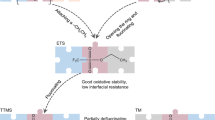
Abstract
The ideal electrolyte for the widely used LiNi0.8Mn0.1Co0.1O2 (NMC811)||graphite lithium-ion batteries is expected to have the capability of supporting higher voltages (≥4.5 volts), fast charging (≤15 minutes), charging/discharging over a wide temperature range (±60 degrees Celsius) without lithium plating, and non-flammability1,2,3,4. No existing electrolyte simultaneously meets all these requirements and electrolyte design is hindered by the absence of an effective guiding principle that addresses the relationships between battery performance, solvation structure and solid-electrolyte-interphase chemistry5. Here we report and validate an electrolyte design strategy based on a group of soft solvents that strikes a balance between weak Li+–solvent interactions, sufficient salt dissociation and desired electrochemistry to fulfil all the aforementioned requirements. Remarkably, the 4.5-volt NMC811||graphite coin cells with areal capacities of more than 2.5 milliampere hours per square centimetre retain 75 per cent (54 per cent) of their room-temperature capacity when these cells are charged and discharged at −50 degrees Celsius (−60 degrees Celsius) at a C rate of 0.1C, and the NMC811||graphite pouch cells with lean electrolyte (2.5 grams per ampere hour) achieve stable cycling with an average Coulombic efficiency of more than 99.9 per cent at −30 degrees Celsius. The comprehensive analysis further reveals an impedance matching between the NMC811 cathode and the graphite anode owing to the formation of similar lithium-fluoride-rich interphases, thus effectively avoiding lithium plating at low temperatures. This electrolyte design principle can be extended to other alkali-metal-ion batteries operating under extreme conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available within this article and its Supplementary information. Additional data are available from the corresponding authors upon reasonable request.
References
Goodenough, J. B. & Kim, Y. Challenges for rechargeable Li batteries. Chem. Mater. 22, 587–603 (2010).
Winter, M., Barnett, B. & Xu, K. Before Li ion batteries. Chem. Rev. 118, 11433–11456 (2018).
Liu, Y., Zhu, Y. & Cui, Y. Challenges and opportunities towards fast-charging battery materials. Nat. Energy 4, 540–550 (2019).
Rodrigues, M.-T. F. et al. A materials perspective on Li-ion batteries at extreme temperatures. Nat. Energy 2, 17108 (2017).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4418 (2004).
Smart, M. et al. Improved low-temperature performance of lithium-ion cells with quaternary carbonate-based electrolytes. J. Power Sources 119, 349–358 (2003).
Herreyre, S. et al. New Li-ion electrolytes for low temperature applications. J. Power Sources 97, 576–580 (2001).
Dong, X., Guo, Z., Guo, Z., Wang, Y. & Xia, Y. Organic batteries operated at −70 °C. Joule 2, 902–913 (2018).
Rustomji, C. S. et al. Liquefied gas electrolytes for electrochemical energy storage devices. Science 356, eaal4263 (2017).
Holoubek, J. et al. Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Nat. Energy 6, 303–313 (2021).
Zhang, S., Xu, K. & Jow, T. A new approach toward improved low temperature performance of Li-ion battery. Electrochem. Commun. 4, 928–932 (2002).
Huang, C. K., Sakamoto, J., Wolfenstine, J. & Surampudi, S. The limits of low-temperature performance of Li-ion cells. J. Electrochem. Soc. 147, 2893 (2000).
Zhang, S., Xu, K. & Jow, T. Low temperature performance of graphite electrode in Li-ion cells. Electrochim. Acta 48, 241–246 (2002).
Petzl, M., Kasper, M. & Danzer, M. A. Lithium plating in a commercial lithium-ion battery—a low-temperature aging study. J. Power Sources 275, 799–807 (2015).
Cai, W. et al. The boundary of lithium plating in graphite electrode for safe lithium-ion batteries. Angew. Chem. Int. Ed. 133, 13117–13122 (2021).
Johnson, L. et al. The role of LiO2 solubility in O2 reduction in aprotic solvents and its consequences for Li–O2 batteries. Nat. Chem. 6, 1091–1099 (2014).
Burke, C. M., Pande, V., Khetan, A., Viswanathan, V. & McCloskey, B. D. Enhancing electrochemical intermediate solvation through electrolyte anion selection to increase nonaqueous Li–O2 battery capacity. Proc. Natl Acad. Sci. USA 112, 9293–9298 (2015).
Holoubek, J. et al. An all-fluorinated ester electrolyte for stable high-voltage Li metal batteries capable of ultra-low-temperature operation. ACS Energy Lett. 5, 1438–1447 (2020).
Ihara, M. et al. Properties of carbon anodes and thermal stability in LiPF6/methyl difluoroacetate electrolyte. J. Electrochem. Soc. 150, A1476 (2003).
Cho, Y.-G. et al. Enabling the low-temperature cycling of NMC||graphite pouch cells with an ester-based electrolyte. ACS Energy Lett. 6, 2016–2023 (2021).
Henderson, W. A. Glyme−lithium salt phase behavior. J. Phys. Chem. B 110, 13177–13183 (2006).
Chu, H. et al. Achieving three-dimensional lithium sulfide growth in lithium–sulfur batteries using high-donor-number anions. Nat. Commun. 10, 188 (2019).
Linert, W., Camard, A., Armand, M. & Michot, C. Anions of low Lewis basicity for ionic solid state electrolytes. Coord. Chem. Rev. 226, 137–141 (2002).
Li, Y., Lu, Y., Adelhelm, P., Titirici, M.-M. & Hu, Y.-S. Intercalation chemistry of graphite: alkali metal ions and beyond. Chem. Soc. Rev. 48, 4655–4687 (2019).
Xue, W. et al. FSI-inspired solvent and “full fluorosulfonyl” electrolyte for 4 V class lithium-metal batteries. Energy Environ. Sci. 13, 212–220 (2020).
Seo, D. M., Borodin, O., Han, S.-D., Boyle, P. D. & Henderson, W. A. Electrolyte solvation and ionic association II. Acetonitrile-lithium salt mixtures: highly dissociated salts. J. Electrochem. Soc. 159, A1489 (2012).
Chen, J. et al. Electrolyte design for LiF-rich solid–electrolyte interfaces to enable high-performance microsized alloy anodes for batteries. Nat. Energy 5, 386–397 (2020).
Bogle, X., Vazquez, R., Greenbaum, S., Cresce, A. V. W. & Xu, K. Understanding Li+–solvent interaction in nonaqueous carbonate electrolytes with 17O NMR. J. Phys. Chem. Lett. 4, 1664–1668 (2013).
Chen, S. et al. High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30, 1706102 (2018).
Ren, X. et al. Role of inner solvation sheath within salt–solvent complexes in tailoring electrode/electrolyte interphases for lithium metal batteries. Proc. Natl Acad. Sci. USA 117, 28603–28613 (2020).
Zhang, X. et al. Advanced electrolytes for fast-charging high-voltage lithium-ion batteries in wide-temperature range. Adv. Energy Mater. 10, 2000368 (2020).
Yamaki, J.-I. et al. Thermal studies of fluorinated ester as a novel candidate for electrolyte solvent of lithium metal anode rechargeable cells. J. Power Sources 102, 288–293 (2001).
Beltrop, K. et al. Enabling bis(fluorosulfonyl)imide-based ionic liquid electrolytes for application in dual-ion batteries. J. Power Sources 373, 193–202 (2018).
Waldmann, T. et al. Interplay of operational parameters on lithium deposition in lithium-ion cells: systematic measurements with reconstructed 3-electrode pouch full cells. J. Electrochem. Soc. 163, A1232 (2016).
Hammersley, A., Svensson, S., Hanfland, M., Fitch, A. & Hausermann, D. Two-dimensional detector software: from real detector to idealised image or two-theta scan. High Pressure Res. 14, 235–248 (1996).
Qiu, X., Thompson, J. W. & Billinge, S. J. PDFgetX2: a GUI-driven program to obtain the pair distribution function from X-ray powder diffraction data. J. Appl. Crystallogr. 37, 678 (2004).
Borodin, O. et al. Insights into the structure and transport of the lithium, sodium, magnesium, and zinc bis(trifluoromethansulfonyl)imide salts in ionic liquids. J. Phys. Chem. C 122, 20108–20121 (2018).
Glaser, R., Borodin, O., Johnson, B. R., Jhulki, S. & Yushin, G. Minimizing long-chain polysulfide formation in Li–S batteries by using localized low concentration highly fluorinated electrolytes. J. Electrochem. Soc. 168, 090543 (2021).
Borodin, O. & Smith, G. D. Quantum chemistry and molecular dynamics simulation study of dimethyl carbonate: ethylene carbonate electrolytes doped with LiPF6. J. Phys. Chem. B 113, 1763–1776 (2009).
Gaussian 16 Rev. C.01 (Gaussian Inc., Wallingford, CT, 2016).
Borodin, O. in Electrolytes for Lithium and Lithium-Ion Batteries: Modern Aspects of Electrochemistry Vol. 58 (eds Richard, T. et al.) 371–401 (Springer, 2014).
Jürgen, H. Ab-initio simulations of materials using VASP: density‐functional theory and beyond. J. Comput. Chem. 29, 2044–2078 (2008).
Dixit, M. et al. Thermodynamic and kinetic studies of LiNi0.5Co0.2Mn0.3O2 as a positive electrode material for Li-ion batteries using first principles. Phys. Chem. Chem. Phys. 18, 6799–6812 (2016).
Sun, J., Ruzsinszky, A. & Perdew, J. P. Strongly constrained and appropriately normed semilocal density functional. Phys. Rev. Lett. 115, 036402 (2015).
Larsen, A. H. et al. The atomic simulation environment—a Python library for working with atoms. J. Phys. Condens. Matter 29, 273002 (2017).
Ong, S. P. et al. Python Materials Genomics (pymatgen): a robust, open-source Python library for materials analysis. Comput. Mater. Sci. 68, 314–319 (2013).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
This work was supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technology Office of the US DOE through the Applied Battery Research for Transportation (ABRT) programme under contract number DE-SC0012704. Research was sponsored by the Army Research Laboratory and was accomplished under cooperative agreement number W911NF-20-2-0284. This research used beamline 28-ID-2 of the National Synchrotron Light Source II, a US DOE Office of Science user facility operated for the DOE Office of Science by Brookhaven National Laboratory under contract number DE-SC0012704.
Author information
Authors and Affiliations
Contributions
J.X. and C.W. conceived the idea for the project. J.X., J.Z., H.W. and S.H. prepared the materials and performed electrochemical experiments. T.P.P., K.X. and O.B. conducted the quantum chemistry calculations and MD simulations. F.C. carried out the NMR analysis. J.L. and H.H. performed the AFM measurements. S.T., E.H. and X.-Q.Y. conducted the PDF experiments. All the authors discussed the results, analysed the data and drafted the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Zheng Chen, Weijiang Xue and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Illustration of the capacity matching and kinetic matching.
a, Thick interface between the commercial carbonate-based electrolyte and lithiated graphite anodes. b, Self-limiting interface between the designed electrolyte and lithiated graphite anodes. c-e, Schematic illustration of (c) capacity mismatching and (d) capacity matching as a function of charge rate or charge temperature, as well as (e) resistance matching between anode and cathode.
Extended Data Fig. 2 X-ray measurements and MD simulation results.
a, b, A structure factor S(Q) from X-ray measurements (solid black line) and MD simulations (blue dash) for 1M LiTFSI in MDFA/MDFSA–TTE electrolyte and two dominating solvents MDFA and TTE. S(Q) for MDFA and TTE were shifted by −1.5 and 3.0.
Extended Data Fig. 3 MD simulation results.
a, b, A snapshot of MD simulation box of LiTFSI in MDFA (a) and 1M LiTFSI in MDFA/MDFSA–TTE (b) from MD simulations at 25 °C. Solvent is shown as wireframe. c, Li+(MDFA)4 free Li+ (solvent separated from TFSI−) from MD simulation of 1M LiTFSI in MDFA/MDFSA–TTE (b, c) at 25 °C. Jmol colour scheme are used: Li – purple, N – blue, F – green, S – yellow, C – grey, O –red, H – white. d, Ion and solvent self-diffusion coefficients from MD simulations of 1M LiTFSI in MDFA/MDFSA–TTE electrolyte. e, Conductivity of 1M LiTFSI in MDFA-MDFSA–TTE electrolyte from MD simulations and experiments.
Extended Data Fig. 4 Rate performance of polycrystalline NMC811||graphite pouch cells at 25 °C.
a-f, Pouch cells with different amount of 1M LiTFSI MDFA/MDFSA–TTE electrolyte under 3C (a, c, e), and 4C (b, d, f).
Extended Data Fig. 5 Cycling performance at ultralow temperature.
Cycling performance of NMC811||graphite full cells at 0.1C under −50/60 °C.
Extended Data Fig. 6 Kinetic analysis of the low-temperature process.
a, Cell illustration of three-electrode set up for kinetic analysis. b, Cell voltage and electrode potential of NMC811||graphite pouch cells in 0.2C under −30 °C. c, d, Nyquist impedance plots and fitted lines using the equivalent circuit of the three-electrode cells containing 1M LiTFSI MDFA/MDFSA–TTE electrolyte at (c) 25 °C and (d) −30 °C. Impedance spectra were obtained at 50% SOC.
Extended Data Fig. 7 DFT calculations of reaction energies.
a-c, Reaction energies from PBE+U (red) and SCAN (blue) (units: eV) for dehydrogenation of MDFA on Li0.5NiO2 (a), TTE on Li0.5NiO2 (b), and Li+-MDFSA on Li1.0NiO2 – a single F− ion is present on the surface as well to neutralize the cell (c). Refer to text for explanation of the large energy difference in (c).
Supplementary information
Supplementary Information
This file contains Supplementary Discussion and References.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, J., Zhang, J., Pollard, T.P. et al. Electrolyte design for Li-ion batteries under extreme operating conditions. Nature 614, 694–700 (2023). https://doi.org/10.1038/s41586-022-05627-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-05627-8
This article is cited by
-
Safe electrolyte for long-cycling alkali-ion batteries
Nature Sustainability (2024)
-
Ligand-channel-enabled ultrafast Li-ion conduction
Nature (2024)
-
Promoting high-voltage stability through local lattice distortion of halide solid electrolytes
Nature Communications (2024)
-
Tiny sheaths of solvent boost battery performance
Nature (2024)
-
Mechanism of interfacial effects in sodium-ion storage devices
Nano Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.