Abstract
The fungal microbiota (mycobiota) is an integral part of the complex multikingdom microbial community colonizing the mammalian gastrointestinal tract and has an important role in immune regulation1,2,3,4,5,6. Although aberrant changes in the mycobiota have been linked to several diseases, including inflammatory bowel disease3,4,5,6,7,8,9, it is currently unknown whether fungal species captured by deep sequencing represent living organisms and whether specific fungi have functional consequences for disease development in affected individuals. Here we developed a translational platform for the functional analysis of the mycobiome at the fungal-strain- and patient-specific level. Combining high-resolution mycobiota sequencing, fungal culturomics and genomics, a CRISPR–Cas9-based fungal strain editing system, in vitro functional immunoreactivity assays and in vivo models, this platform enables the examination of host–fungal crosstalk in the human gut. We discovered a rich genetic diversity of opportunistic Candida albicans strains that dominate the colonic mucosa of patients with inflammatory bowel disease. Among these human-gut-derived isolates, strains with high immune-cell-damaging capacity (HD strains) reflect the disease features of individual patients with ulcerative colitis and aggravated intestinal inflammation in vivo through IL-1β-dependent mechanisms. Niche-specific inflammatory immunity and interleukin-17A-producing T helper cell (TH17 cell) antifungal responses by HD strains in the gut were dependent on the C. albicans-secreted peptide toxin candidalysin during the transition from a benign commensal to a pathobiont state. These findings reveal the strain-specific nature of host–fungal interactions in the human gut and highlight new diagnostic and therapeutic targets for diseases of inflammatory origin.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
ITS sequencing data are publicly available in NCBI Sequence Read Archive (SRA) under the BioProject ID PRJNA610042. The data from whole-genome sequencing of human gut-derived C. albicans isolates are publicly available in the NCBI SRA under BioProject ID PRJNA702809. Raw sequencing data for representative C. albicans strains were downloaded from the NCBI SRA under BioProject ID PRJNA432884 (ref. 43). Other data necessary to evaluate the paper are provided within the Article and its Supplementary Information. Source data are provided with this paper.
Change history
20 July 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41586-022-05102-4
References
The Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Lloyd-Price, J. et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 569, 655–662 (2019).
Chehoud, C. et al. Fungal signature in the gut microbiota of pediatric patients with inflammatory bowel disease. Inflamm. Bowel Dis. 21, 1948–1956 (2015).
Hoarau, G. et al. Bacteriome and mycobiome interactions underscore microbial dysbiosis in familial Crohn’s disease. mBio 7, e01250–16 (2016).
Liguori, G. et al. Fungal dysbiosis in mucosa-associated microbiota of Crohn’s disease patients. J. Crohn’s Colitis 10, 296–305 (2016).
Sokol, H. et al. Fungal microbiota dysbiosis in IBD. Gut 66, 1039–1048 (2017).
Ott, S. J. et al. Fungi and inflammatory bowel diseases: alterations of composition and diversity. Scand. J. Gastroenterol. 43, 831–841 (2008).
Limon, J. J. et al. Malassezia is associated with Crohn’s disease and exacerbates colitis in mouse models. Cell Host Microbe 25, 377–388 (2019).
Jain, U. et al. Debaryomyces is enriched in Crohn’s disease intestinal tissue and impairs healing in mice. Science 371, 1154–1159 (2021).
Kaplan, G. G. The global burden of IBD: from 2015 to 2025. Nat. Rev. Gastroenterol. Hepatol. 12, 720–727 (2015).
Israeli, E. et al. Anti-Saccharomyces cerevisiae and antineutrophil cytoplasmic antibodies as predictors of inflammatory bowel disease. Gut 54, 1232–1236 (2005).
Lewis, J. D. et al. Inflammation, antibiotics, and diet as environmental stressors of the gut microbiome in pediatric Crohn’s disease. Cell Host Microbe 18, 489–500 (2015).
Schaffer, T. et al. Anti-Saccharomyces cerevisiae mannan antibodies (ASCA) of Crohn’s patients crossreact with mannan from other yeast strains, and murine ASCA IgM can be experimentally induced with Candida albicans. Inflamm. Bowel dis. 13, 1339–1346 (2007).
Standaert-Vitse, A. et al. Candida albicans colonization and ASCA in familial Crohn’s disease. Am. J. Gastroenterol. 104, 1745–1753 (2009).
Doron, I. et al. Human gut mycobiota tune immunity via CARD9-dependent induction of anti-fungal IgG antibodies. Cell 184, 1017–1031 (2021).
Leonardi, I. et al. CX3CR1+ mononuclear phagocytes control immunity to intestinal fungi. Science 359, 232–236 (2018).
Cohen, R., Roth, F. J., Delgado, E., Ahearn, D. G. & Kalser, M. H. Fungal flora of the normal human small and large intestine. N. Engl. J. Med. 280, 638–641 (1969).
Sovran, B. et al. Enterobacteriaceae are essential for the modulation of colitis severity by fungi. Microbiome 6, 152 (2018).
Danese, S. & Fiocchi, C. Ulcerative colitis. N. Engl. J. Med. 365, 1713–1725 (2011).
Fan, D. et al. Activation of HIF-1α and LL-37 by commensal bacteria inhibits Candida albicans colonization. Nat. Med. 21, 808–814 (2015).
Jawhara, S. et al. Colonization of mice by Candida albicans is promoted by chemically induced colitis and augments inflammatory responses through galectin-3. J. Infect. Dis. 197, 972–980 (2008).
Marakalala, M. J. et al. Differential adaptation of Candida albicans in vivo modulates immune recognition by dectin-1. PLoS Pathog. 9, e1003315 (2013).
Liang, S. H. et al. Hemizygosity enables a mutational transition governing fungal virulence and commensalism. Cell Host Microbe 25, 418–431.e6 (2019).
Schonherr, F. A. et al. The intraspecies diversity of C. albicans triggers qualitatively and temporally distinct host responses that determine the balance between commensalism and pathogenicity. Mucosal Immunol. 10, 1335–1350 (2017).
Forche, A. et al. Selection of Candida albicans trisomy during oropharyngeal infection results in a commensal-like phenotype. PLoS Genet. 15, e1008137 (2019).
Tso, G. H. W. et al. Experimental evolution of a fungal pathogen into a gut symbiont. Science 362, 589–595 (2018).
Kasper, L. et al. The fungal peptide toxin candidalysin activates the NLRP3 inflammasome and causes cytolysis in mononuclear phagocytes. Nat. Commun. 9, 4260 (2018).
Wellington, M., Koselny, K., Sutterwala, F. S. & Krysan, D. J. Candida albicans triggers NLRP3-mediated pyroptosis in macrophages. Eukaryot. Cell 13, 329–340 (2014).
Uwamahoro, N. et al. The pathogen Candida albicans hijacks pyroptosis for escape from macrophages. mBio 5, e00003–14 (2014).
Moyes, D. L. et al. Candidalysin is a fungal peptide toxin critical for mucosal infection. Nature 532, 64–68 (2016).
Verma, A. H. et al. Oral epithelial cells orchestrate innate type 17 responses to Candida albicans through the virulence factor candidalysin. Sci. Immunol. 2, eaam8834 (2017).
Naglik, J. R., Gaffen, S. L. & Hube, B. Candidalysin: discovery and function in Candida albicans infections. Curr. Opin. Microbiol. 52, 100–109 (2019).
Pierce, J. V. & Kumamoto, C. A. Variation in Candida albicans EFG1 expression enables host-dependent changes in colonizing fungal populations. mBio 3, e00117–12 (2012).
Witchley, J. N. et al. Candida albicans morphogenesis programs control the balance between gut commensalism and invasive infection. Cell Host Microbe 25, 432–443 (2019).
Li, X. et al. Response to fungal dysbiosis by gut-resident CX3CR1+ mononuclear phagocytes aggravates allergic airway disease. Cell Host Microbe 24, 847–856 (2018).
Rohde, C. M. et al. Metabonomic evaluation of Schaedler altered microflora rats. Chem. Res. Toxicol. 20, 1388–1392 (2007).
Schaedler, R. W., Dubos, R. & Costello, R. The development of the bacterial flora in the gastrointestinal tract of mice. J. Exp. Med. 122, 59–66 (1965).
Witchley, J. N., Basso, P., Brimacombe, C. A., Abon, N. V. & Noble, S. M. Recording of DNA-binding events reveals the importance of a repurposed Candida albicans regulatory network for gut commensalism. Cell Host Microbe 29, 1002–1013 (2021).
Vyas, V. K. et al. New CRISPR mutagenesis strategies reveal variation in repair mechanisms among fungi. mSphere 3, e00154–18 (2018).
Vyas, V. K., Barrasa, M. I. & Fink, G. R. A Candida albicans CRISPR system permits genetic engineering of essential genes and gene families. Sci. Adv. 1, e1500248 (2015).
Selmecki, A., Forche, A. & Berman, J. Genomic plasticity of the human fungal pathogen Candida albicans. Eukaryot. Cell 9, 991–1008 (2010).
MacCallum, D. M. et al. Property differences among the four major Candida albicans strain clades. Eukaryot. Cell 8, 373–387 (2009).
Ropars, J. et al. Gene flow contributes to diversification of the major fungal pathogen Candida albicans. Nat. Commun. 9, 2253 (2018).
Butler, G. et al. Evolution of pathogenicity and sexual reproduction in eight Candida genomes. Nature 459, 657–662 (2009).
Drummond, R. A. et al. CARD9+ microglia promote antifungal immunity via IL-1β- and CXCL1-mediated neutrophil recruitment. Nat. Immunol. 20, 559–570 (2019).
Shouval, D. S. et al. Interleukin 1β mediates intestinal inflammation in mice and patients with interleukin 10 receptor deficiency. Gastroenterology 151, 1100–1104 (2016).
Friedrich, M. et al. IL-1-driven stromal–neutrophil interactions define a subset of patients with inflammatory bowel disease that does not respond to therapies. Nat. Med. 27, 1970–1981 (2021).
Mogavero, S. et al. Candidalysin delivery to the invasion pocket is critical for host epithelial damage induced by Candida albicans. Cell. Microbiol. 23, e13378 (2021).
Longman, R. S. et al. CX3CR1+ mononuclear phagocytes support colitis-associated innate lymphoid cell production of IL-22. J. Exp. Med. 211, 1571–1583 (2014).
Hepworth, M. R. et al. Group 3 innate lymphoid cells mediate intestinal selection of commensal bacteria-specific CD4+ T cells. Science 348, 1031–1035 (2015).
Tang, J., Iliev, I. D., Brown, J., Underhill, D. M. & Funari, V. A. Mycobiome: approaches to analysis of intestinal fungi. J. Immunol. Methods 421, 112–121 (2015).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Zheng, X. et al. A high-performance computing toolset for relatedness and principal component analysis of SNP data. Bioinformatics 28, 3326–3328 (2012).
Gu, Z., Gu, L., Eils, R., Schlesner, M. & Brors, B. circlize implements and enhances circular visualization in R. Bioinformatics 30, 2811–2812 (2014).
Danecek, P. et al. Twelve years of SAMtools and BCFtools. Gigascience 10, giab008 (2021).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Bankevich, A. et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 19, 455–477 (2012).
Wick, R. R., Schultz, M. B., Zobel, J. & Holt, K. E. Bandage: interactive visualization of de novo genome assemblies. Bioinformatics 31, 3350–3352 (2015).
Acknowledgements
We thank the members of the Iliev laboratory for suggestions relating to the manuscript; W. Jin and C. Guo, who shared cloning reagents and equipment; G. Fink, V. K. Vyas and D. A. Bernstein for providing plasmids and protocols and all contributing members of the JRI IBD Live Cell Bank Consortium; and the staff at the Microbiome Core Laboratory of Weill Cornell Medicine. Support for sample acquisition through the JRI IBD Live Cell Bank is provided by the JRI, Jill Roberts Center for IBD, Cure for IBD, the Rosanne H. Silbermann Foundation and Weill Cornell Medicine Division of Pediatric Gastroenterology and Nutrition. X.V.L. is supported by fellowship from the Charles H. Revson Foundation. I.L. is supported by fellowships from the Crohn’s and Colitis Foundation (568319). W.D.F. is supported by a fellowship from the NIH (F32DK120228). J.R.N is supported by the Wellcome Trust (214229_Z_18_Z), NIH (R37-DE022550) and IS-BRC-1215-20006. B.H. is supported by the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG) within the Cluster of Excellence ‘Balance of the Microverse’, under Germany’s Excellence Strategy, EXC 2051, Project-ID 390713860. Research in the Iliev laboratory is supported by US National Institutes of Health (R01DK113136, R01DK121977, R21AI146957 and R01AI163007), the Irma T. Hirschl Career Scientist Award, the Kenneth Rainin Foundation, the Crohn’s and Colitis Foundation, the Leona M. and Harry B. Helmsley Charitable Trust and the Burroughs Wellcome Fund Investigators in the Pathogenesis of Infectious Disease (PATH) Award.
Author information
Authors and Affiliations
Contributions
X.V.L. and I.D.I. conceived and designed experiments. X.V.L. performed experiments. I.L., A.S., W.D.F., T.K., W.-Y.L., I.H.G., I.D., A.G.-G., M.B.D., G.M.C. and M.M. helped with experiments and assays. A.S. and X.V.L. established strain collection and identification. A.S. and I.L. performed the ITS sequencing and analysis. G.G.P. performed the whole-genome analysis. C.Y. performed histology scoring. X.V.L., I.L. and G.G.P. generated figures from analysed data. X.V.L. designed, performed and validated CRISPR–Cas9-mediated mutagenesis of C. albicans isolates. J.R.N. and B.H. provided the ece1Δ/Δ mutant, a parental strain and contributed to related data interpretation. E.J.S. provided samples and advice. X.V.L. and I.D.I. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
I.D.I. and X.L. are inventors on a US provisional patent application US 63/287,233 filed by Cornell University, covering inventions described in this manuscript. The other authors declare no competing interests.
Peer review
Peer review information
Nature thanks Gordon Brown and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
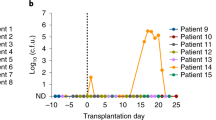
Extended Data Fig. 1 Relative abundance of intestinal fungal genera in non-IBD and patients with UC.
a, Alpha diversity analysis was analysed using the Shannon diversity index among fungi communities at the fungal OTU level in colonic mucosa (MUC) enriched samples from non-IBD or ulcerative colitis-affected (UC) individuals. Based on quality control one non-IBD sample was excluded from further mycobiome sequencing and analysis. b, Non-metric multidimensional scaling (NMDS) plot of distance ordination based on Bray–Curtis dissimilarities for fungal ITS1 OTUs in colonic mucosa (MUC) enriched samples from non-IBD or ulcerative colitis-affected (UC) individuals. Analysis of similarities (ANOSIM) statistics. c, Relative genus abundance of intestinal fungal genera. Boxplots in a and c, the lower and upper hinges correspond to the first and third quartiles (the 25th and 75th percentiles). The horizontal line shows the median. a–c, Each dot represents an individual human subject, non-IBD (n = 37) or ulcerative colitis-affected (UC, n = 40) individuals. Analysis performed with two-sided, non-paired, Mann-Whitney test, Benjamini-Hochberg (BH) corrected for data in a and c.
Extended Data Fig. 2 Intestinal colonization by C. albicans does not cause spontaneous colitis during homeostasis nor does it aggravate DSS-induced colitis.
a, Faecal C. albicans burdens were assessed after 3 days of C. albicans colonization in mice that received either control feeding water (n = 4) or DSS water (n = 5) for 4 days. Dots represent individual mice. b–g, Mice, after being gavaged with PBS (n = 5) or C. albicans (n = 5) for 14 days, were induced by 3% DSS water for 7 days. b, Schematic figure of DSS-induced colitis model of mice with intestinal colonization by C. albicans. c, H&E staining and histology score of colon sections. n= 9 (DSS + control PBS) and n = 7 (DSS + C. albicans). The scale bar 200 μm. Data in c are pooled from two independent experiments with similar results. d, e, Representative flow cytometry plot and quantification of Foxp3+ (d) and RORγt+ CD4+ T (e) cells. f, g, Mice were gavaged twice per week with PBS (n = 5) or C. albicans WT SC5314 (n = 5). Mice were sacrificed four months later for colon length (f) and histology evaluation, the scale bar 100 μm (g). Each dot represents an individual mouse. Data in a–e are representative of three independent experiments with similar results. Data in f, g representative of two independent experiments with similar results. Data in a, c, and d–g are shown as mean ± s.e.m., unpaired, two-tailed, Mann-Whitney test.
Extended Data Fig. 3 C. albicans expands and promotes intestinal inflammation under immunosuppression therapy for UC.
a, Medication data summary for UC patients (n = 5609) who visited New York-Presbyterian Hospital from 2015–2018. Corticosteroids (CS, n = 2522); Others include Mercaptopurine, Azathioprine, Methotrexate, Tacrolimus, Cyclosporine and biologics (n = 3057). Some individuals were being treated with two or more treatments. b, Faecal C. albicans burdens were measured after 3 days of C. albicans colonization in mice that received either PBS (n = 5) or prednisolone daily (10 mg/kg/day, n = 5). c–e, WT SPF mice were fed PBS (n = 5), P. kudriavzevii (Pk, n = 5), or Candida albicans (n = 5) while receiving prednisolone treatment (prednisolone + PBS, prednisolone + P. kudriavzevii, or prednisolone + C. albicans). 3% DSS drinking water was used to induce colitis for 7 days. Mice were sacrificed three days after the DSS water was removed. c, Faecal fungal burdens upon sacrifice. d, Colon length was assessed. e, Representative flow cytometry plots and quantification of the frequency IL-17A+CD4+ T cells in the colons. Results in b–f are shown as mean ± s.e.m. Each dot represents an individual mouse. Data in b–e are representative of three independent experiments with similar results. Unpaired, two-tailed, Mann-Whitney test (b, c) or one-way ANOVA followed by the Tukey’s post hoc test (d, e).
Extended Data Fig. 4 Gut-derived C. albicans isolates exhibit different phenotypic responses in a filamentation assay.
a, Gut C. albicans isolates were cultured on Spider agar at 37 °C for 5 days followed by assessment of the edge of wrinkled and smooth colonies. Percentage of the filamentation phenotypes of gut C. albicans isolates in Fig. 2a on spider agar. b–i, Filamentation phenotype of representative gut C. albicans isolates used in Fig. 2a. b, C. albicans IDB311, IDB312, and IDB313 isolates from UC patient IDB31. c, C. albicans IDB831, IDB832, and IDB833 isolates from UC patient IDB83. d, C. albicans IDB101, IDB102, and IDB104 isolates from UC patient IDB10. e, C. albicans IDC481, IDC482, and IDC483 isolates from UC patient IDC48. f, C. albicans IDB071, IDB072, and IDB073 isolates from UC patient IDB07. g, C. albicans IDA651, IDA652, and IDA653 isolates from UC patient IDA65. h, C. albicans IDA921, IDA922, and IDA923 isolates from UC patient IDA92. i, C. albicans IDC561, IDC562, and IDC563 isolates from non-IBD individual IDC56. Data are representative of three independent experiments with same results.
Extended Data Fig. 5 High-damaging strains induce greater proinflammatory immune responses.
a–c, WT germ-free (GF) mice were colonized with C. albicans strains for three weeks. PBS (n = 5), LD-Ca (IDC561, n = 5), HD-Ca (IDB311, n = 6). a, Gating strategy to analyse CD4+ T cells in colonic lamina propria cells. Representative flow cytometry plots and quantification of CD4+IL-17A+IL-17F+ (b, from left to right) and CD4+RORγt+ cells (c, from left to right) in the colon. d–f, ASF mice were colonized with Ca strains for three weeks. PBS (n = 6), LD-Ca (IDC561, n = 6), and HD-Ca (IDB311, n = 6). d, C. albicans burden in the faeces at day 21. e, Frequency of colonic CD4+IL-17A+ TH17 cells. f, Frequency of colonic CD4+RORγt+ cells. g, h, ASF mice were colonized with LD-Ca (IDB891, n = 5) or HD-Ca (IDB101, n = 6) for three weeks. g, C. albicans burden in the faeces at day 21. h, Frequency of CD4+IL-17A+ TH17 cells in the colon were assessed. i, j, ASF mice were colonized with LD-Ca (IDC662, n = 6) or HD-Ca (IDC711, n = 6) for three weeks. i, C. albicans burden in the faeces at day 21. j, Frequencies of CD4+IL-17A+ and CD4+IL-17A+ IL-17F+ TH17 cells in the colon were assessed. k, SPF WT mice were with Ca strains colonized and treated with prednisolone followed by DSS-induced murine colitis. PBS (n = 10), HD-Ca IDB311 (n = 9) and LD-Ca IDC561 (n = 7). Faecal C. albicans burdens were assessed upon sacrifice. Results in b–k are shown as mean ± s.e.m. Each dot represents an individual mouse. Data in b, c are representative of three independent experiments with similar results. Data in d–k are representative of three independent experiments with similar results. Unpaired, two-tailed, Mann-Whitney test (d–k) or one-way ANOVA followed by the Tukey’s post hoc test (b, c).
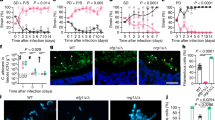
Extended Data Fig. 6 Efg1-dependent candidalysin is required for cell damage in high-damaging C. albicans strains from the human gut.
a, Caco-2 cells were infected with live C. albicans for 12 h, and both the EFG1 expression of C. albicans. HD/C.a IDB311 and LD/C.a IDC561 strains, and respective efg1Δ/Δ (efg1) strains were used in this experiment. Data shown as mean ± s.d. Data points indicate technical well replicates (n = 3). Data are representative of three independent experiments with similar results. b, Dendrogram showing SNP-based distances between EFG1 sequences of isolates. Each isolate was obtained from an individual subject from the non-IBD (n = 8) or UC patient (n = 10) group. c, Caco-2 cells were infected with C. albicans and LDH release of Caco-2 cells was assessed. HD-Ca IDB101, HD-Ca IDB101 efg1Δ/Δ mutant (efg1), LD-Ca IDB891, and LD-Ca IDB891 efg1Δ/Δ mutant (efg1) were used. d, C. albicans HD-Ca IDB311, HD-Ca IDB101 and the respective EFG1 mutants were cultured on Spider agar at 37 °C for 5 days followed by assessment of the filamentation phenotype. Data are representative of two independent experiments with same results. e, f, GF mice were colonized with PBS (GF, n = 5), HD-Ca IDB311 (HD, n = 6) or HD-Ca IDB311 efg1 (HD efg1, n = 6) strains for three weeks. e, Frequencies of IL-17A+CD4+ T cells; f, Frequencies, and total cell numbers of IL-17A+IL-17F+CD4+ T cells, in the colon were assessed by flow cytometry. g, ECE1 gene expression in C. albicans cells were assessed upon EFG1 gene deletion. Data shown as mean ± s.d. Data points indicate technical well replicates (n = 3). Data are representative of three independent experiments with similar results. h, Log2 transformed ratio of gene expression between EFG1 mutant and WT C. albicans strain upon colonization of the large intestine represented as a volcano plot. Data were obtained and analysed from a recent resource dataset from Witchley et al. 2021. i, LDH release were assessed from Caco-2 cells. HD-Ca IDB101, LD-Ca IDB891 and respective ECE1 mutant strains were used. Data shown as mean ± s.d. Data points indicate technical well replicates (n = 3). Data are representative of three independent experiments with similar results. j–m, GF mice were colonized with C a strains for three weeks. PBS GF (n = 8), HD-Ca IDB311 (HD, n = 10), HD ece1 (n = 10), LD-Ca IDC561 (LD, n = 7), and LD ece1 (n = 7). j, Quantification of the frequencies of IL-17A+CD4+ T cells; k, Quantification of the frequencies, and total cell numbers of IL-17A+IL-17F+CD4+ T cells in the colon. l, Faecal C. albicans burden were measured at day 21. Each dot represents an individual mouse. m, HD and LD C. albicans strains both colonize the intestine and form a mixture of yeast and hyphal morphotypes. Fluorescence in situ hybridization (FISH) was utilized to visualize the morphology of C. albicans HD-Ca IDB311, LD-Ca IDC561, and respective ECE1 mutant strains (HD-Ca IDB311 ece1Δ/Δ and LD-Ca IDC561 ece1Δ/Δ) in the colon tissue after 21 days of mono-colonization of C. albicans. The nuclei of colonic epithelial cells were stained with DAPI (blue), colonic mucin was stained with a FITC- conjugated Ulex europaeus agglutinin (UEA-I, lectin) (green), and C. albicans was stained with a Cy3-coupled pan-fungal-specific probe (red). The colon tissue of germ-free mice was used as a control. The scale bar 25 μm. Results in e, f and j–l are shown as mean ± s.e.m. Data in e, f are representative of three independent experiments with similar results. Data in j–m are representative of two independent experiments with similar results. Unpaired, two-tailed, Mann-Whitney test (a, g), One-way ANOVA followed by the Tukey’s post hoc test (e, f, j, k, l).
Extended Data Fig. 7 Genomic comparative analysis of genetic polymorphisms in CPH1, UME6, FLO8 and ECE1 among high- and low-damaging strains.
a–c, Dendrogram showing SNP-based distances between CPH1 (a), UME6 (b), FLO8 (c) and ECE1 (d) sequences of isolates. Each isolate was obtained from an individual subject from the non-IBD (n = 8) or UC patient (n = 10) group.
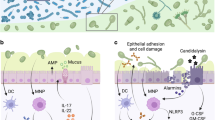
Extended Data Fig. 8 Gut C. albicans promotes intestinal pro-inflammatory immunity through candidalysin.
a, b, Cell damage by C. albicans. mBMDMs (a) and Caco-2 cells (b) were incubated with live C. albicans (m.o.i. = 5) wild-type parental (Ca Pare) or C. albicans ece1Δ/Δ (Ca ece1) strains for 16 h and LDH release in the supernatants was measured. Results are shown as mean ± s.d. Data points indicate technical well replicates (n = 3). Data are representative of three independent experiments. Unpaired, two-tailed, student test. c–e, ASF mice were colonized with C. albicans Pare or C. albicans ece1 strains for three weeks. n = 6 mice in each group. c, d, Frequency and total cell numbers of CD11b+Ly6G+ neutrophils (c, from left to right) and IL-17A+CD4+ T cells (d, from left to right) in the colon. e, Faecal C. albicans burdens were measured at day 21. f–j, WT SPF mice were colonized with or without C. albicans Pare or C. albicans ece1 strain, DSS colitis was induced followed by treatment with prednisolone. f, Faecal C. albicans burden were measured upon sacrifice. g, representative H&E colon section. The scale bar 200 μm. h, Histology scores. PBS (n = 14), C. albicans Pare (n = 15), and C. albicans ece1 (n = 13). Data are pooled from two independent experiment with similar results. i, Frequency of colonic lamina propria neutrophils. j, IL-17A+CD4+ T cells were assessed upon sacrifice. Results in c–f and h–j are shown as mean ± s.e.m. Each dot represents an individual mouse. Data in c–f and i, j are representative of two independent experiments with similar results. Unpaired, two-tailed, Mann-Whitney test (e, f) or one-way ANOVA followed by the Tukey’s post hoc test (c, d, h–j).
Extended Data Fig. 9 C. albicans clonal expansion, and microevolution occur in the human gut.
a, Dendrogram showing genome-wide SNP-based distances among two or three human gut C. albicans isolates obtained from the same individual subject. Strains isolated from non-IBD subjects (nIBD), UC patients (UC), and refence strain C. albicans SC5314 (Ref. Strain). b, Heatmap showing genome-wide density of heterozygous SNPs from two or three human gut C. albicans isolates obtained from the same individual subject. Strains isolated from non-IBD subjects (nIBD), UC patients (UC), and refence strain C. albicans SC5314 (Ref. Strain) are labelled. Color density indicates the number of heterozygous SNPs detected in each 10 kbp window of an isolate’s genome. Arrows point to the genomic locations of ECE1 and EFG1 genes. c, Multiple sequence alignment of candidalysin (SK1 peptide) amino acid sequences across multiple isolates of C. albicans. Each isolate was obtained from an individual subject from the non-IBD or UC patient group. Four variants of candidalysin across strains show no association/clustering of specific variant with HD or LD strain. Two haplotypes are shown for isolates that are heterozygous in this region.
Extended Data Fig. 10 Immune mediators released by macrophage upon infection with gut -derived C. albicans strains.
a, LDH release measured in culture supernatants of hMDM after infection with C. albicans parental strain (Pare), C. albicans ece1Δ/Δ (Ca ece1) and an untreated group (Ctrl) for 16 h. b, Macrophage-released mediators measured by cytometric bead assays from cultures in Extended Data Fig. 10a. c, IL-1β release measured in culture supernatants of LPS-primed hMDMs infected with C. albicans parental strain, C. albicans ece1, and in uninfected group. Parallel experiments were performed with a. Results a–c are shown as mean ± s.d. Data are representative of three independent experiments with similar results. d–f, Cytokine release in culture supernatants of unprimed human monocyte-derived macrophages (hMDMs) after incubation with live gut derived-C. albicans isolates (m.o.i. = 5). d, IL-6 cytokine production was measured by ELISA. hMDM damage measured (LDH assay) in the same experiment was correlated with specific cytokine release. e, f, Correlation between TNF-α and IL-6 cytokine from hMDM induced by patient-specific gut C. albicans and Mayo score in corresponding UC patients (n = 10). Dot is shown as an average value of three technical repeats, Data are representative of three independent experiments with similar results. The simple linear regression was performed, where P-value calculated by a F test. g, h, Mice colonized with or without HD-Ca IBD311 were treated with prednisolone (Pred) followed by DSS-mediated induction of colitis. Each mouse further treated with 1 mg anti-IL-1R1 IgG (αIL1R) or isotype IgG (IgG) at the time point indicated in the schematic figure of experimental layout (g). IgG+PBS (n = 8), IgG+C. albicans (n = 9) and αIL-1R1+C. albicans (n = 7). h, Representative flow cytometry plots of IL-17A+IL-17F+CD4+ T cells. Data in d–h are representative of three independent experiments with similar results. One-way ANOVA followed by the Tukey’s post hoc test (a–c).
Supplementary information
Supplementary Tables
Supplementary Tables 1–4.
Source data
Rights and permissions
About this article
Cite this article
Li, X.V., Leonardi, I., Putzel, G.G. et al. Immune regulation by fungal strain diversity in inflammatory bowel disease. Nature 603, 672–678 (2022). https://doi.org/10.1038/s41586-022-04502-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-022-04502-w
This article is cited by
-
Altered gut mycobiome in patients with end-stage renal disease and its correlations with serum and fecal metabolomes
Journal of Translational Medicine (2024)
-
The hyphal-specific toxin candidalysin promotes fungal gut commensalism
Nature (2024)
-
Fungi in cancer
Nature Reviews Cancer (2024)
-
WAY-262611 ameliorates the inflammatory bowel disease by activating Wnt/β-catenin pathway
In Vitro Cellular & Developmental Biology - Animal (2024)
-
The Microbiome Matters: Its Impact on Cancer Development and Therapeutic Responses
Journal of Microbiology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.