Abstract
The biological basis of male–female brain differences has been difficult to elucidate in humans. The most notable morphological difference is size, with male individuals having on average a larger brain than female individuals1,2, but a mechanistic understanding of how this difference arises remains unknown. Here we use brain organoids3 to show that although sex chromosomal complement has no observable effect on neurogenesis, sex steroids—namely androgens—lead to increased proliferation of cortical progenitors and an increased neurogenic pool. Transcriptomic analysis and functional studies demonstrate downstream effects on histone deacetylase activity and the mTOR pathway. Finally, we show that androgens specifically increase the neurogenic output of excitatory neuronal progenitors, whereas inhibitory neuronal progenitors are not increased. These findings reveal a role for androgens in regulating the number of excitatory neurons and represent a step towards understanding the origin of sex-related brain differences in humans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All sequencing data (bulk and single cell) have been deposited at the NCBI Gene Expression Omnibus (GEO) under accession code GSE187877. The following open access databases were used in this study: SZDB: A Database for Schizophrenia Genetic Research (http://szdb.org/) and SFARI gene (https://gene.sfari.org/).
Code availability
Delboy code is available at https://github.com/alextkalinka/delboy.
References
Ritchie, S. J. et al. Sex differences in the adult human brain: evidence from 5216 UK Biobank participants. Cereb. Cortex 28, 2959–2975 (2018).
Ruigrok, A. N. V. et al. A meta-analysis of sex differences in human brain structure. Neurosci. Biobehav. Rev. 39, 34–50 (2014).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Green, T., Flash, S. & Reiss, A. L. Sex differences in psychiatric disorders: what we can learn from sex chromosome aneuploidies. Neuropsychopharmacol. 44, 9–21 (2018).
McCarthy, M. M. Multifaceted origins of sex differences in the brain. Phil. Trans. R. Soc. B 371, 20150106 (2016).
Loomes, R., Hull, L. & Mandy, W. P. L. What is the male-to-female ratio in autism spectrum disorder? A systematic review and meta-analysis. J. Am. Acad. Child Adolesc. Psychiatry 56, 466–474 (2017).
Abel, K. M., Drake, R. & Goldstein, J. M. Sex differences in schizophrenia. Int. Rev. Psychiatry 22, 417–428 (2010).
Hines, M. Neuroscience and sex/gender: looking back and looking forward. J. Neurosci. 40, 37–43 (2020).
McCarthy, M. M. & Arnold, A. P. Reframing sexual differentiation of the brain. Nat. Neurosci. 14, 677–683 (2011).
Woodson, J. C. & Gorski, R. A. in Sexual Differentiation of the Brain (ed. Matsumoto, A.) Ch. 13 (CRC Press, 1999).
Rabinowicz, T., Dean, D. E., Petetot, J. M.-C. & Courten-Myers, G. M. D. E. Gender differences in the human cerebral cortex: more neurons in males; more processes in females. J. Child Neurol. 14, 98–107 (1999).
Knickmeyer, R. C. et al. Impact of sex and gonadal steroids on neonatal brain structure. Cereb. Cortex 24, 2721–2731 (2014).
Arnold, A. P. A general theory of sexual differentiation. J. Neurosci. Res. 95, 291–300 (2017).
Arnold, A. P. The organizational–activational hypothesis as the foundation for a unified theory of sexual differentiation of all mammalian tissues. Horm. Behav. 55, 570–578 (2009).
McCarthy, M. M. Estradiol and the developing brain. Physiol. Rev. 88, 91–134 (2008).
Wallen, K. Hormonal influences on sexually differentiated behavior in nonhuman primates. Front. Neuroendocrin. 26, 7–26 (2005).
Miller, W. L. & Auchus, R. J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 32, 81–151 (2011).
Pollen, A. A. et al. Molecular identity of human outer radial glia during cortical development. Cell 163, 55–67 (2015).
Davey, R. A. & Grossmann, M. Androgen receptor structure, function and biology: from bench to bedside. Clin. Biochem. Rev. 37, 3–15 (2016).
Wang, F. et al. RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J. Mol. Diagn. 14, 22–29 (2012).
Quartier, A. et al. Genes and pathways regulated by androgens in human neural cells, potential candidates for the male excess in autism spectrum disorder. Biol. Psychiat. 84, 239–252 (2018).
Qu, Y. et al. Constitutively active AR-V7 plays an essential role in the development and progression of castration-resistant prostate cancer. Sci. Rep. 5, 7654 (2015).
Jeselsohn, R. et al. Emergence of constitutively active estrogen receptor-α mutations in pretreated advanced estrogen receptor-positive breast cancer. Clin. Cancer Res. 20, 1757–1767 (2014).
Kalinka, A. T. Improving the sensitivity of differential-expression analyses for under-powered RNA-seq experiments. Preprint at https://doi.org/10.1101/2020.10.15.340737 (2020).
Tang, T. et al. HDAC1 and HDAC2 regulate intermediate progenitor positioning to safeguard neocortical development. Neuron 101, 1117–1133 (2019).
Li, L., Jin, J. & Yang, X.-J. Histone deacetylase 3 governs perinatal cerebral development via neural stem and progenitor cells. Iscience 20, 148–167 (2019).
Dey, A. et al. YB-1 is elevated in medulloblastoma and drives proliferation in Sonic hedgehog-dependent cerebellar granule neuron progenitor cells and medulloblastoma cells. Oncogene 35, 4256–4268 (2016).
Kielar, M. et al. Mutations in Eml1 lead to ectopic progenitors and neuronal heterotopia in mouse and human. Nat. Neurosci. 17, 923–933 (2014).
Abrahams, B. S. et al. SFARI Gene 2.0: a community-driven knowledgebase for the autism spectrum disorders (ASDs). Mol. Autism 4, 36–36 (2013).
Hackinger, S. et al. Evidence for genetic contribution to the increased risk of type 2 diabetes in schizophrenia. Transl. Psychiatry 8, 252 (2018).
Udawela, M. et al. SELENBP1 expression in the prefrontal cortex of subjects with schizophrenia. Transl. Psychiatry 5, e615 (2015).
Fatemi, S. H., Folsom, T. D. & Thuras, P. D. Deficits in GABAB receptor system in schizophrenia and mood disorders: a postmortem study. Schizophr. Res. 128, 37–43 (2011).
Narayan, S., Head, S. R., Gilmartin, T. J., Dean, B. & Thomas, E. A. Evidence for disruption of sphingolipid metabolism in schizophrenia. J. Neurosci. Res. 87, 278–288 (2009).
Raudvere, U. et al. g:Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res. 47, W191–W198 (2019).
Göttlicher, M. et al. Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. EMBO J. 20, 6969–6978 (2001).
Boissinot, M. et al. Induction of differentiation and apoptosis in leukaemic cell lines by the novel benzamide family histone deacetylase 2 and 3 inhibitor MI-192. Leukemia Res. 36, 1304–1310 (2012).
Saito, A. et al. A synthetic inhibitor of histone deacetylase, MS-27-275, with marked in vivo antitumor activity against human tumors. Proc Natl Acad. Sci. USA 96, 4592–4597 (1999).
Alexeyenko, A. et al. Comparative interactomics with Funcoup 2.0. Nucleic Acids Res. 40, D821–D828 (2012).
Mayer, C. & Grummt, I. Ribosome biogenesis and cell growth: mTOR coordinates transcription by all three classes of nuclear RNA polymerases. Oncogene 25, 6384–6391 (2006).
Kim, W.-Y. Brain size is controlled by the mammalian target of rapamycin (mTOR) in mice. Commun. Integr. Biol. 8, e994377 (2015).
Saxton, R. A. & Sabatini, D. M. mTOR signaling in growth, metabolism, and disease. Cell 169, 361–371 (2017).
Voss, M. H. et al. Phase 1 study of mTORC1/2 inhibitor sapanisertib (TAK-228) in advanced solid tumours, with an expansion phase in renal, endometrial or bladder cancer. Br. J. Cancer 123, 1590–1598 (2020).
Choi, Y. J. et al. Inhibitory effect of mTOR activator MHY1485 on autophagy: suppression of lysosomal fusion. PLoS ONE 7, e43418 (2012).
Sohal, V. S. & Rubenstein, J. L. R. Excitation–inhibition balance as a framework for investigating mechanisms in neuropsychiatric disorders. Mol. Psychiatr. 24, 1248–1257 (2019).
Marín, O. & Müller, U. Lineage origins of GABAergic versus glutamatergic neurons in the neocortex. Curr. Opin. Neurobiol. 26, 132–141 (2014).
O’Shaughnessy, P. J. et al. Alternative (backdoor) androgen production and masculinization in the human fetus. PLoS Biol. 17, e3000002 (2019).
Martínez‐Cerdeño, V., Noctor, S. C. & Kriegstein, A. R. Estradiol stimulates progenitor cell division in the ventricular and subventricular zones of the embryonic neocortex. Eur. J. Neurosci. 24, 3475–3488 (2006).
Eliot, L., Ahmed, A., Khan, H. & Patel, J. Dump the “dimorphism”: comprehensive synthesis of human brain studies reveals few male–female differences beyond size. Neurosci. Biobehav. Rev. 125, 667–697 (2021).
Knickmeyer, R. C. & Baron-Cohen, S. Fetal testosterone and sex differences. Early Hum. Dev. 82, 755–760 (2006).
Bahari-Javan, S. et al. HDAC1 links early life stress to schizophrenia-like phenotypes. Proc. Natl Acad. Sci. USA 114, E4686–E4694 (2017).
Ryskalin, L., Limanaqi, F., Frati, A., Busceti, C. L. & Fornai, F. mTOR-related brain dysfunctions in neuropsychiatric disorders. Int. J. Mol. Sci. 19, 2226 (2018).
Lancaster, M. A. et al. Guided self-organization and cortical plate formation in human brain organoids. Nat. Biotechnol. 35, 659–666 (2017).
Fromer, M. et al. Gene expression elucidates functional impact of polygenic risk for schizophrenia. Nat. Neurosci. 19, 1442–1453 (2016).
Bagley, J. A., Reumann, D., Bian, S., Lévi-Strauss, J. & Knoblich, J. A. Fused cerebral organoids model interactions between brain regions. Nat. Methods 14, 743–751 (2017).
Hines, M., Constantinescu, M. & Spencer, D. Early androgen exposure and human gender development. Biol. Sex Differ. 6, 3 (2015).
Reyes, F. I., Boroditsky, R. S., Winter, J. D. S. & Faiman, C. Studies on human sexual development. II. Fetal and maternalserum gonadotropin and sex steroid concentrations. J. Clin. Endocrinol. Metab. 38, 612–617 (1974).
McManus, J. M. & Sharifi, N. Structure-dependent retention of steroid hormones by common laboratory materials. J. Steroid Biochem. Mol. Biol. 198, 105572 (2020).
Shoskes, J. J., Wilson, M. K. & Spinner, M. L. Pharmacology of testosterone replacement therapy preparations. Transl. Androl. Urol. 5, 834–843 (2016).
Wright, A. S., Thomas, L. N., Douglas, R. C., Lazier, C. B. & Rittmaster, R. S. Relative potency of testosterone and dihydrotestosterone in preventing atrophy and apoptosis in the prostate of the castrated rat. J. Clin. Invest. 98, 2558–2563 (1996).
Iacopino, F. et al. Valproic acid activity in androgen-sensitive and -insensitive human prostate cancer cells. Int. J. Oncol. 32, 1293–1303 (1992).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Giandomenico, S. L. et al. Cerebral organoids at the air–liquid interface generate diverse nerve tracts with functional output. Nat. Neurosci. 22, 669–679 (2019).
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A. & Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 14, 417–419 (2017).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Leek, J. T., Johnson, W. E., Parker, H. S., Jaffe, A. E. & Storey, J. D. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics 28, 882–883 (2012).
Friedman, J., Hastie, T. & Tibshirani, R. Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. 33, 1–22 (2010).
Gerard, D. Data-based RNA-seq simulations by binomial thinning. BMC Bioinformatics 21, 206 (2020).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Trapnell, C. et al. The dynamics and regulators of cell fate decisions are revealed by pseudotemporal ordering of single cells. Nat. Biotechnol. 32, 381–386 (2014).
Cao, J. et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature 566, 496–502 (2019).
Benito-Kwiecinski, S. et al. An early cell shape transition drives evolutionary expansion of the human forebrain. Cell 184, 2084–2102 (2021).
Noctor, S. C., Martínez‐Cerdeño, V. & Kriegstein, A. R. Distinct behaviors of neural stem and progenitor cells underlie cortical neurogenesis. J. Comp. Neurol. 508, 28–44 (2008).
Giandomenico, S. L., Sutcliffe, M. & Lancaster, M. A. Generation and long-term culture of advanced cerebral organoids for studying later stages of neural development. Nat. Protoc. 16, 579–602 (2021).
Acknowledgements
We thank D. Srivastava, S. Munro and M. Hegde for comments; the MRC LMB Light Microscopy facility for help with microscopy and Bioinformatics (P. Freire-Pritchett) for help with RNA-seq analysis, as well as the CRUK Genomics facility for library preparation and sequencing; M. Mancini, M. Marcelli and E. Wilson for depositing their plasmids in Addgene; and the other members of the Lancaster laboratory and the MRC LMB Cell Biology division for discussions. Work in the Lancaster laboratory is supported by the Medical Research Council (MC_UP_1201/9) and the European Research Council (ERC STG 757710).
Author information
Authors and Affiliations
Contributions
I.K. conceived the study, performed experiments, analysed data and wrote the paper. I.C. performed experiments and analysed the data. L.P. performed experiments. A.T.K. performed bioinformatics analysis. M.A.L. supervised the study, analysed data and co-wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
M.A.L. is an inventor on several patents related to cerebral organoids, is co-founder and member of the scientific advisory board of a:head bio, and is a member of the scientific advisory board of the Roche Institute for Translational Bioengineering.
Peer review information
Nature thanks Giorgia Quadrato and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Treatment and phenotypic characterization of cerebral organoids.
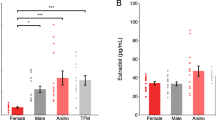
a) Illustration of the determination of the progenitor zone for quantifications. The progenitor zone (VZ+SVZ) was determined based on immunostaining and histological landmarks (see Methods). Immunostaining of XX 35d organoids stained for TBR2 (yellow, white), co-stained with DAPI (blue). Dashed line indicates the progenitor zone. Schematic of an organoid ventricle with different progenitor populations of interest. b) Quantification of thicknesses of different morphological zones between male (XY) and female (XX) brain organoids at 35d (above) and 52d (below). Significance values (Mann-Whitney, two-tailed) of the measurements from XX organoids, as compared to XY organoids, are indicated by different shades of grey. VZ-ventricular zone, SVZ-subventricular zone. c) Actual measured concentration of testosterone (left) and oestradiol (right) in the medium after the addition of 100nM (calculated) on day 0, as measured by ELISA assay. d) Actual measured concentration of testosterone over 4 days of organoid culture after addition of 100nM (calculated) on day 0, as measured by ELISA. e) Sections of whole XX organoids at 35d (left), and XY organoids at 52d (right), stained for TBR2 (white). Co-stained with DAPI (blue). f) XY 35d organoids stained for TBR2 (yellow, white), co-stained with DAPI (blue), treated with DHT, T or E. g) Quantification of non-apical mitotic, PH3+ cells in XY and XX, 35d and 52d organoids, treated with DHT, T and E. Scale bars: e) 500µm, a), f) 100 µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 2 Basal progenitors are increased after androgen treatment.
a) Quantification of TBR2+ cells per mm2 progenitor zone (see Extended Data Fig. 1a) at 35d. b) Image of the ventricular zone/subventricular zone (VZ/SVZ) border in XX 35d organoids stained for Ki67 (green, white) and TBR2 (white). Cells positive for both Ki67 and TBR2 are indicated with magenta arrowheads. Double positive mitotic cells are indicated with yellow arrowheads. A white dashed line delineates VZ/SVZ border with the ventricular zone below. c) Immunostaining for proliferation marker Ki67 (white) on XX 35d control (left) and DHT treated (right) organoids. Dashed yellow lines represent the apical surface (bottom line) and the VZ/SVZ boundary. Note the difference in Ki67+ cells in the SVZ of DHT-treated organoids. d) Quantification of Ki67/TBR2 double positive cells, out of all TBR2+ cells, in XX 35d and 52d organoids. e) Immunostaining for HOPX (fire LUT) of XX 52d organoids. Yellow dashed line indicates the ventricular surface. Note the HOPX signal in radial glia. Images are single, 1.2 µm optical planes. f) Quantification of HOPX+ basal radial glia per mm2 SVZ of XX 52d organoids. g) Immunostaining for phosphorylated histone H3 (PH3) (green) on XX 35d and XY 52d old organoids. Co-stained with DAPI (white). h) Quantification of apical mitotic cells (PH3+), normalized per mm length of ventricle in male (XY) and female (XX) organoids, at 35d and 52d, treated with DHT, T and E. i) Quantification of ventricular length in male (XY) and female (XX) organoids, at 35d and 52d, treated with DHT, T and E. Scale bars: g) 100 µm, e) 50µm, b), c) 20µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 3 Clonal labelling reveals increased radial glial proliferation after androgen treatment.
a) Timeline and schematic of the Sendai/Lenti virus lineage tracing and analysis, with virus encoding emGFP injected at day 45 and virus encoding RFP injected at day 53. b) Representative images of GFP-labelled clones (white) 8 days post Sendai emGFP labelling in control (above) and DHT-treated (below) XX organoids. DAPI is in blue. c) Quantification of GFP+ clone size (left side) at 51d and 53d and RFP+ clone size at 61d. d) XX control and DHT organoids injected with Lenti-RFP at 53d, and fixed at 61d. RFP (magenta). Co-stained with DAPI (blue). e) Quantification of TBR2+ cells per mm2 of progenitor layer in control and DHT-treated XX organoids, at 8 days post-labelling with Sendai emGFP. Note that TBR2+ intermediate progenitors were not yet increased at this time point, as the radial glia which produce them were still in the VZ. Scale bars: d) 50µm, b) 25µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 4 AR activity in radial glia promotes their proliferation rather than differentiation.
a) RNA Scope (fluorescent in situ) for AR of XY organoids at 45d. Single AR mRNA puncta (white). Co-stained with DAPI (blue). Yellow dashed lines demarcate ventricular zone (VZ) and the subventricular zone (SVZ). a’) Portion of the VZ from a. a”) Portion of the SVZ from a. b) RNA Scope for AR of XX organoids at 45d. Single AR mRNA puncta (white). Co-stained with DAPI (blue). b’) Portion of the VZ from b. b”) Portion of the SVZ from b. Note the difference in the amount of AR mRNA in the VZ and SVZ in both cell lines. c) RNA Scope for AR of XX 21d (top) and 35d (bottom) organoids. Single AR mRNA puncta (white). Immunostaining for TBR2 (magenta). Co-stained with DAPI (blue). Yellow dashed line demarcates the apical surface c’) Portion of the ventricular zone (VZ) from XX 21d organoid. c”) Portion of the VZ from XX 35d organoid. d) RNA Scope negative (above) and positive (below) control (fire look up table) in female (XX) 35d organoids. DAPI is in white. e) Quantification of AR mRNA distribution in GFP+ pairs of cells, depending on the stage of cell division. f) RNAscope of AR mRNA (white puncta), together with GFP signal (green) from EmGFP Sendai Fluorescence Reporter showing examples of three daughter cell pairs. Yellow dashed lines indicate GFP+ cell body. g) Western blot for AR on control, DHT and E treated organoids at 17-25 days. AR specific band is predicted to be around 110 kDa. Note the increased AR signal in DHT-treated organoids. Asterisk indicates a lower amount of protein loaded for the 75d organoid lane (see Methods). For gel source data, see Supplementary Fig. 1. h) Quantification of levels of AR protein from g), normalized by GAPDH expression. i) Droplet digital PCR (ddPCR) results showing the decrease in AR transcription between days 17 and 25. ref = housekeeping gene EIF2B2. j) Representative images of the cortical wall at 21, 35 and 52d, with DAPI-labelled nuclei visible, showing the relative reduction in the radial glial progenitor layer (ventricular zone - VZ) and an increase in the thickness of the neuronal layer over time. k) Quantification of the percentage (%) of cells (AR+ DAPI), containing AR mRNA puncta (as detected by RNA Scope), out of all cells (DAPI) at 21, 35 and 52d. Cells counted: 21d – 1222, 35d – 1067, 52d – 1201. Co-stained with DAPI (white). l) XX organoids electroporated (EP) at 45d and fixed at 50d. EGFP-C1: control; EGFP-C1-AR-V7: constitutively active AR. Immunostaining for GFP and TBR2. Yellow dashed lines demarcate the apical and basal boundaries of the VZ. Note increased GFP+ nuclei in the VZ in EGFP-C1-AR-V7. m) Immunostaining for GFP (green) and Ki67 (magenta) on XX 50d organoids, electroporated at 45d. Co-stained with DAPI (blue). EGFP-C1: control plasmid; EGFP-C1-AR-V7: plasmid expressing constitutively active AR; EGFP-C1-ERaY537S: plasmid expressing constitutively active ERa. n) Quantification of the proportion of GFP+ cells co-staining for the proliferation marker Ki67 at 5 days post electroporation in XX organoids electroporated with the indicated plasmid at 45d. o) Quantification of the proportion of GFP+ cells co-staining for the intermediate progenitor marker TBR2 at 2- and 5 days post electroporation in XX organoids electroporated at 45d. At 5 days post electroporation, most cells electroporated with EGFP-C1-ERaY537S died indicating a later effect on cell survival. p) Quantification of the proportion of GFP+ cells co-staining for the neuronal marker HuC/D 2- and 5 days post electroporation in XX organoids electroporated at 45d. At 5 days post electroporation, most cells electroporated with EGFP-C1-ERaY537S died. q) XX 47d organoids, electroporated at 45d. Immunostaining for GFP (green), NGN2 (cyan) and BRN2 (magenta). Co-stained with DAPI (white). Yellow arrowheads: GFP+/NGN2+ cells. White arrowheads: GFP+/BRN2+ cells. Yellow dashed line demarcates the apical surface. Note the increased expression of differentiation markers upon expression of ERaY537S perhaps indicating a premature cell cycle exit and relating to the cell death observed. r) Quantification of the proportion of GFP+ cells co-staining for the neurogenic marker NGN2 2 days post electroporation in XX organoids electroporated at 45d. s) Quantification of the proportion of GFP+ cells co-staining for the upper layer neurogenesis marker BRN2 2 days post electroporation in XX organoids electroporated at 45d. Scale bars: a), b), l), m) 50µm, c), d), q) 25µm, a’), a”), b’), b”), j) 20µm, c’), c”), f) 5µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 5 Live imaging of radial glial division modes after activation and androgen signalling.
a) Still images from live imaging of organoids electroporated at 31d and imaged beginning at 2 days post-electroporation (see Methods for details of image acquisition). pCAG-mCherry+ cells are shown in magenta, and EGFP-C1-AR-V7+ are shown in green. Green arrowheads: GFP+ cells. Yellow arrowheads: mCherry+/GFP– cells. Filled in arrowheads: one of the tracked daughter cells. a) Comparison of the behaviour of mCherry+ and GFP+ only cells at 33d. Basal surface is up. mCherry+ only cell, upon division (06:11) produces two daughter cells, one of which migrates basally, indicating a more differentiated identity, whereas the other stays in the ventricular zone (VZ), representing an example of asymmetric division. GFP+ cell divides (07:28), but both of the daughter cells continue to reside in the VZ. b) Still images from live imaging of organoids electroporated at 54d and imaged at 56d. Basal surface is down. After division (15:50), both of the daughter cells remain in the VZ. Yellow dashed lines demarcate the apical surfaces. Time scale: hours:minutes. c) Schematic of the proposed mechanism leading to increased basal progenitors. Radial glia can divide symmetrically, increasing their numbers, or asymmetrically, generating one radial glia and one basal progenitor. Application of androgen supports symmetric, proliferative divisions, thus increasing the size of radial glial clones. After androgen is withdrawn, these radial glia can start producing basal progenitors in increased numbers, as shown in Fig. 1b, c. Basal progenitors represented as a single population (yellow cells), for clarity. Scale bars: a), b) 20µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 6 Transcriptomics reveals key signalling pathways and cell types.
a) Principle component analysis (PCA) plots of XX 35d bulk RNA-seq, uncorrected for batch effects. b) Principle component analysis (PCA) plots of XX 35d bulk RNA-seq, corrected for batch effects using the R package ComBat. Filtering of lowly expressed genes was performed prior to batch correction, resulting in different PC value scales between a and b. See Methods. c) Quantification of SRD5A1, an upregulated DEG under androgen treatment. SRD5A1 was quantified as the number of SRD5A1+ puncta per 10µm2 of the ventricular zone (VZ) in XX 35d control and DHT-treated organoids. d) Venn diagram representing the overlap of up- and downregulated DEGs in androgen treated XX 35d organoids with genes differentially expressed in patients with schizophrenia73. P values represent significance of a hypergeometric test of the intersection with up- and downregulated genes (P=3.6E-4 and P=0.0012, respectively). DE: differentially expressed. e) Top significant GO34 term enrichments in XX 35d androgen upregulated genes. f) Left, UMAP plot showing different clusters identified by scRNA-seq in 35d old treated and control XX organoids. Right, UMAP plot showing the distribution of cells belonging to control, DHT and E treatments. Note the high degree of overlap indicating reproducibility. g) Heat map showing scaled expression levels of cluster specific genes identified through differential gene expression analysis across clusters. The top 10 genes ordered by average log fold change are shown for each cluster. h) Dot plot showing the relative expression of cell type-specific markers. i) Feature scatter plot for percent ribosome (percent.ribo) by percent mitochondrial (percent.mt) reads. Left, plotted data points grouped by RG subcluster. Right, plotted data points grouped by treatment status. −0.27 refers to the correlation coefficient. j) Pseudotime analysis using Monocle3 mapped onto the reclustered seurat neural clusters (all clusters except ChP/Hem) UMAP. k) Feature plot of the neurogenic marker BTG2 (Tis21) and more mature marker synaptotagmin (SYT1) which was also identified as upregulated in RG2 cluster. l) Dot plot of the subclustering of the IP/N cell population showing the relative expression of cell type-specific markers and clear separation of different cell subpopulations. ImmN = immature neurons, MatN = maturing neurons. m) Stacked bar plot of radial glial (RG) cluster proportions detected by scRNA-seq at 35d. Upper right: chi-square with Monte Carlo simulation for overall distribution (Supplementary Table 4). Note increased RG1 and bRG, but decrease in more committed RG2, with no significant difference compared with expected counts in total RG (combined four RG clusters) and non-RG clusters (IP/N and ChP/Hem) in DHT treatment as determined by chi-square test (not shown) indicating specificity for RG subclusters. *P<0.05 and **P<0.00005 n) Point range plot of Monte Carlo permutation test of statistical significance between cell cluster proportions in DHT compared with control (left) and E compared with control (right) displaying bootstrapped confidence intervals for the difference in cluster proportion (reported as observed log2 fold-change) (see Methods). o) Dot plot showing the relative expression of bRG markers in different treatments, detected by scRNA-seq, in 35d old organoids. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 7 HDAC activity interacts with androgen signalling to influence progenitor behaviour.
a) Dot plot showing the relative expression of HDACs in different treatments, detected by scRNA-seq, in 35d old organoids. b) UMAP plots showing the relative expression of HDAC2 in cells from control, DHT and E treated organoids. Right - UMAP plot showing the radial glia clusters identified by scRNA-seq. Yellow circle demarcates the bRG subpopulation. Note the increased expression of HDAC2 in DHT bRG cluster. c) Immunostaining for TBR2 (green) and HuC/D (magenta) in XX 35d organoids, treated with VPA from 17d. Co-stained with DAPI (white, blue). c’) c”) c”’) insets from c), as indicated. Yellow asterisks indicate neural rosettes forming above the VZ in some VPA-treated organoids. d) Quantification of ventricular length for XX 35d old organoids treated with VPA and VPA+DHT. e) Immunostaining for CTIP2 (magenta), and SATB2 (green, white) on XX 35d control and VPA-treated organoids. Co-stained with DAPI (blue). e’), e”) insets from e) showing just SATB2 in fire look-up table. e”) Note the SATB2+ cells (yellow arrowheads) and SATB2+ staining present in the VZ in VPA-treated organoids. Yellow dashed lines demarcate the apical and basal boundaries of the VZ. f) Quantification of SATB2+ cells in the cortical wall of control, VPA- and VPA+DHT-treated organoids in XX organoids at 35d. g) Quantification of the thickness of VZ for XX 35d organoids treated with VPA and VPA+DHT. h) Immunostaining for TBR2 (yellow) in 35d old XX organoids with the following treatments: DHT, MI-192 (HDAC2/3 inhibitor), MI-192+DHT, MS-275 (HDAC1/3 inhibitor), MS-275+DHT. White dashed line demarcates the apical surface. i) Quantification of TBR2+ cells per mm2 of the control and the following treatments: DHT, MI-192 (HDAC2/3 inhibitor), MI-192+DHT, MS-275 (HDAC1/3 inhibitor), MS-275+DHT in 35d old XX organoids. Scale bar: c) 100 µm, c’), c’’), c’’’), e), h) 50µm, e’), e”) 20µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 8 Downstream mTOR activity influences progenitor expansion.
a) Heat map showing scaled expression of differentially expressed genes in DHT-treated compared to control organoids, detected by scRNA-seq. Cell identities were assigned to their treatment group and differential expression analysis performed in Seurat as performed for comparison across clusters (see Methods). b) Dot plot showing the relative expression of bRG markers, overlapping markers of RG1 cells and DE transcripts in DHT, and genes of the mTOR pathway in cells from control, DHT and E treated organoids, detected by scRNA-seq. c) Protein interaction network of upregulated DEG directly interacting genes, obtained by bulk RNA seq. Translation/ribosome biogenesis genes are in red. d) Immunostaining for phosphorylated S6 (PS6) (fire look-up table), an indicator of mTOR activity, in 35d old XX organoids, treated with DHT and E. White arrowheads indicate PS6+ cell bodies in the ventricular zone (VZ). e) Quantification of PS6+ cells in the VZ of control, DHT- and E-treated 35d old XX organoids. f) Immunostaining for TBR2 (white) in control and MHY-1485-treated 35d XX organoids. Co-stained with DAPI (blue). g) Quantification of TBR2+ cells per mm2 progenitor layer in control and MHY-1485-treated 35d XX organoids. Scale bars: d), f) 50µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 9 Ventral progenitors and mouse organoids exhibit differential responses to those of human excitatory neurogenic progenitors.
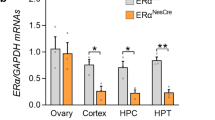
a) Schematic of neurogenesis in the human developing brain at ~12 gestation weeks. Excitatory neurons (green arrows) are born within the dorsal cortex and migrate basally to form the cortical plate. Inhibitory neurons (orange arrow) are born in the ventral telencephalon and migrate towards the dorsal side to incorporate themselves into the cortical plate. Yellow: progenitor zone, dark pink: cortical plate. b) RNA Scope (fluorescent in situ hybridization) for AR mRNA (white) in XX and XY 45d old ventral organoids. Co-stained with DAPI (blue). b’) and b”) - portions of the VZ, as indicated. Note the decreased levels compared with Extended Data Fig. 4a, b. c) Above: Heat map of steroidogenic enzyme expression from XX 35d bulk RNA-seq. Values are in tpm. Below: Schematic of the steroidogenic pathway. Purple – SRD5A1. ESR1, ESR2-estrogen receptors a and b, respectively. d) Comparison of SRD5A1 (fire LUT) immunostaining on XX and XY dorsal 35d and ventral 45d organoids. Both anti-SRD5A1 and anti-TBR2 primary antibodies were used in XY samples, and the secondary antibody recognized both primary antibodies but are discernible by their different subcellular localisations. Co-stained with DAPI (white). Yellow dashed lines demarcate the apical and basal boundaries of the VZ. d’) Portion of the VZ in XX dorsal 35d organoids stained for SRD5A1. Note the SRD5A1+ puncta. d”) Portion of the VZ in XX ventral 45d organoids stained for SRD5A1. d’’’) Portion of the VZ in XY dorsal 35d organoids stained for SRD5A1. Note the SRD5A1+ puncta. d”’’) Portion of the VZ in XY ventral 45d organoids stained for SRD5A1. White arrowhead - SRD5A1+ punctum. e) Timeline of the mouse organoid generation protocol. See Methods for details. f) Immunostaining for TBR2 (yellow) on 11d old mouse organoids in control, DHT- and E- treated organoids. Co-stained with DAPI (blue). g) Quantification of TBR2+ cells per mm2 of progenitor layer in mouse organoids at 9d and 11d. Scale bars: b), d), f) 50µm, b’), b”) 20µm, d’), d”), d’’’), d””) 10µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Extended Data Fig. 10 Excitatory neurons are increased after release of androgen signalling.
a) Immunostaining of XX 52d continuously treated control and DHT organoids stained for NEUROD2 (white). b) Quantification of NEUROD2+ cells per mm2 of cortical wall at 35 days, in XY and XX organoids. c) Quantification of NEUROD2+ cells per mm2 of cortical wall at 52 days, in XY and XX organoids. d) Timeline of the pulse-chase experiment. Hormones were administered between 17-35d, then removed. Organoids were fixed at 52d and 75d. e) NEUROD2+ cells per mm2 cortical wall after pulse-chase treatment. f) Immunostaining for CTIP2 (white) of XX 52d organoids after pulse-chase treatment (control and DHT). Co-stained with DAPI (blue). g) Quantification of CTIP2+ cells per mm2 cortical wall after pulse-chase treatment. h) Quantification of SATB2+ cells per 0.5mm2 cortical wall at 75d after pulse-chase treatment. Scale bars: a) 100µm, f) 50µm. See Methods for details of statistics and Supplementary Table 5 for details of n numbers.
Supplementary information
Supplementary Information
This file contains a Supplementary Guide, Supplementary Tables 1–5 and Supplementary Fig. 1.
Supplementary Video 1
Live imaging of XX organoids, electroporated at 31 d, and imaged at 33 d for >48 h. Cells labelled with pCAG-mCherry are in magenta. Cells electroporated with EGFP-C1-AR-V7 are in green. Still images from this video are shown in Extended Data Fig. 5a. Time scale: hours:minutes.
Supplementary Video 2
Live imaging of XX organoids, electroporated at 54 d, and imaged at 56 d for >48 h. Cells labelled with pCAG-mCherry are in magenta. Cells electroporated with EGFP-C1-AR-V7 are in green. Still images from this video are shown in Extended Data Fig. 5b. Time scale: hours:minutes.
Supplementary Video 3
Green channel (EGFP-C1-AR-V7) from Supplementary Video 2 for easier tracking of electroporated, GFP+ cells. Time scale: hours:minutes.
Rights and permissions
About this article
Cite this article
Kelava, I., Chiaradia, I., Pellegrini, L. et al. Androgens increase excitatory neurogenic potential in human brain organoids. Nature 602, 112–116 (2022). https://doi.org/10.1038/s41586-021-04330-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-04330-4
This article is cited by
-
Sex differences in placenta-derived markers and later autistic traits in children
Translational Psychiatry (2023)
-
Androgen signal for brain size
Nature Reviews Neuroscience (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.