Abstract
Transmission of SARS-CoV-2 is uncontrolled in many parts of the world; control is compounded in some areas by the higher transmission potential of the B.1.1.7 variant1, which has now been reported in 94 countries. It is unclear whether the response of the virus to vaccines against SARS-CoV-2 on the basis of the prototypic strain will be affected by the mutations found in B.1.1.7. Here we assess the immune responses of individuals after vaccination with the mRNA-based vaccine BNT162b22. We measured neutralizing antibody responses after the first and second immunizations using pseudoviruses that expressed the wild-type spike protein or a mutated spike protein that contained the eight amino acid changes found in the B.1.1.7 variant. The sera from individuals who received the vaccine exhibited a broad range of neutralizing titres against the wild-type pseudoviruses that were modestly reduced against the B.1.1.7 variant. This reduction was also evident in sera from some patients who had recovered from COVID-19. Decreased neutralization of the B.1.1.7 variant was also observed for monoclonal antibodies that target the N-terminal domain (9 out of 10) and the receptor-binding motif (5 out of 31), but not for monoclonal antibodies that recognize the receptor-binding domain that bind outside the receptor-binding motif. Introduction of the mutation that encodes the E484K substitution in the B.1.1.7 background to reflect a newly emerged variant of concern (VOC 202102/02) led to a more-substantial loss of neutralizing activity by vaccine-elicited antibodies and monoclonal antibodies (19 out of 31) compared with the loss of neutralizing activity conferred by the mutations in B.1.1.7 alone. The emergence of the E484K substitution in a B.1.1.7 background represents a threat to the efficacy of the BNT162b2 vaccine.
Similar content being viewed by others
Main
The BNT162b2 mRNA vaccine encodes the full-length trimerized spike protein of SARS CoV-22 and was designed against the Wuhan-1 isolate. Concerns have been raised as to whether vaccines will be effective against newly emergent SARS-CoV-2 variants, such as B.1.1.7 (N501Y.V1)3. In clinical studies of BNT162b2, the geometric mean titre (GMT) of neutralizing antibodies associated with 50% neutralization increased after the first dose and the vaccine provided high levels of protection against infection and severe disease after the second dose4.
Activity of vaccine and convalescent sera against B.1.1.7
Participants (n = 37) received the first dose of the BNT162b2 mRNA vaccine 3 weeks before blood was drawn for the collection of serum and peripheral blood mononuclear cells. The median age was 62 years (interquartile range, 47–84 years) and 35% of participants were female. Of these participants, 21 individuals also had a blood draw 3 weeks after receiving the second dose of the BNT162b2 mRNA vaccine. Serum IgG titres against nucleocapsid protein, the spike protein and the receptor-binding domain (RBD) of the spike protein were assayed (Extended Data Fig. 1a).
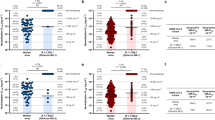
Using lentiviral pseudotyping, we studied the wild-type (wild-type spike bearing D614G) and mutant B.1.1.7 spike proteins (Fig. 1a) to measure the neutralization activity of vaccine-elicited sera. The vaccine sera exhibited a range of inhibitory dilutions that provided 50% neutralization (ID50) (Fig. 1b, c). The GMT against the wild-type spike protein after the second dose of vaccine was substantially higher than after the first dose (318 compared with 77) (Fig. 1b, e). There was correlation between total spike IgG titres and serum neutralization titres (Extended Data Fig. 1b). A broad range of T cell responses was measured by IFNγ FluoroSpot against SARS-CoV-2 peptides in samples from individuals who received the vaccine after the first dose. These cellular responses did not correlate with serum neutralization titres or IgG spike antibody titres (Extended Data Fig. 1c, d).
a, Spike in the open conformation with a single erect RBD (Protein Data Bank (PDB): 6ZGG) is shown in the vertical view of the trimer axis. The locations of mutated residues are shown as red spheres, with deletions indicated in a dashed outline, and are labelled on the monomer with an erect RBD. b–g, The 50% serum neutralization titres of the first dose of the vaccine (b, c, n = 37), the second dose of the vaccine (d, e, n = 21) and convalescent sera (f, g, n = 27) against the wild-type (WT) spike protein and the spike protein of the variant B.1.1.7 (containing the N501Y, A570D, ΔH69/ΔV70, ΔY144, P681H, T716I, S982A and D1118H mutations). HS, human serum control. b, d, f, Mean fold changes in ID50 are indicated above the graphs. Data points of the same individual are connected by lines. Data are GMT ± s.d. and individual values of two independent experiments each with two technical repeats. Two-tailed Wilcoxon matched-pairs signed-rank test with no adjustment for multiple comparisons; **P < 0.01, ****P < 0.0001. The cut-off for 50% neutralization was set to 4.
We then generated mutated pseudoviruses carrying the spike protein with the N501Y and A570D substitutions and the H69/V70 deletion (ΔH69/ΔV70). We observed a small increase in the ability of sera from individuals who were vaccinated or had recovered from COVID-19 to inhibit this triple-mutant virus (Extended Data Fig. 2a–c). We next included the full set of eight mutations in the spike protein that is present in the B.1.1.7 variant (Fig. 1a). Of the 29 sera with neutralization activity after the first dose, 20 showed evidence of a reduction in neutralization titres against the B.1.1.7 variant (Fig. 1b, c and Extended Data Fig. 3), with a fold change of 3.2 ± 5.7 (mean ± s.d.). After the second dose, the GMT was markedly increased compared with the first-dose titres, with a fold change of 1.9 ± 0.9 (mean ± s.d.) (Fig. 1d, e). Among sera from 27 individuals who had recovered from COVID-19, the GMT at 50% neutralization was 1,334 for the wild-type spike protein, which is significantly higher than the GMT after the second dose of the vaccine (Fig. 1f, g). The fold change in ID50 for neutralization of the B.1.1.7 compared with wild-type (D614G) spike protein was 4.5 ± 8.7 (Fig. 1f, g and Extended Data Fig. 4).
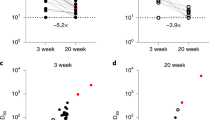
The E484K substitution (Fig. 2a) has been reported as an escape mutation for several monoclonal antibodies5, and is present in the B.1.351 (501Y.V2) and P.1 (501Y.V3) lineages. As of 11 February 2021, 30 B.1.1.7 sequences also had the E484K substitution (Fig. 2c). Phylogenetic analysis suggests that there have been multiple independent acquisitions, with one lineage appearing to expand over time, indicating active transmission (Fig. 2b). This has resulted in Public Health England naming this a variant of concern (VOC 202102/02)6. We therefore generated pseudoviruses that carried the B.1.1.7 spike mutations with or without the additional E484K substitution and tested these against sera obtained after the first and second dose of the BNT162b2 mRNA vaccine as well as against convalescent sera. After the second vaccine dose, we observed a considerable loss of neutralizing activity for the pseudovirus with the B.1.1.7 spike mutations and E484K (Fig. 3d, e). The mean fold change for the E484K-containing B.1.1.7 spike variant was 6.7 compared with 1.9 for the B.1.1.7 variant, relative to the wild-type spike protein (Fig. 3a–c and Extended Data Fig. 5). Similarly, when we tested a panel of convalescent sera with a range of neutralization titres (Fig. 1f, g and Extended Data Fig. 5), we observed additional loss of activity against the mutant B.1.1.7 spike with E484K, with fold change of 11.4 relative to the wild-type spike protein (Fig. 3f, g and Extended Data Fig. 5).
a, Representation of the spike RBM:ACE2 interface (PDB: 6M0J) with residues E484, N501 and K417 highlighted as spheres coloured by element. b, Maximum likelihood phylogeny of a subset of sequences from the UK with the E484K mutation (blue) and the B.1.1.7 lineage (green), with background sequences from the UK without RBD mutations shown in black. As of 11 February 2021, 30 sequences from the B.1.1.7 lineage (one cluster of 25 at the top of the phylogenetic tree) have acquired the E484K substitution (red). c, Sequence accumulation over time in GISAID for UK sequences of the B.1.1.7 and other variants with or without E484K.
All virus variants were in a spike(D614G) background. a, Example neutralization curves of vaccinated individuals (ID 5, 7, 18, 28). The inverse dilution is shown on a log scale. Data are mean ± s.e.m. representative of two independent experiments each with two technical replicates. b–g, The 50% neutralization titres of each virus against sera derived after the first vaccine dose (b, c, n = 37), the second vaccine dose (d, e, n = 21) and for convalescent sera (f, g, n = 20) expressed as fold change relative to the wild-type virus. b, d, f, Mean fold changes in ID50 are indicated above the graphs. Data are mean ± s.d. and individual values; error bars for negative values are not shown. c, e, g, Data are the mean fold change of two technical replicates and are representative of two independent experiments. Data points of the same individual are connected by lines. b, d, f, Two-tailed paired Student’s t-test; *P < 0.05, **P < 0.01, ****P < 0.0001; NS, not significant. The cut-off for 50% neutralization was set to 4.
Monoclonal antibody activity against B.1.1.7
We tested 60 monoclonal antibodies isolated from 15 individuals who had recovered from SARS-CoV-2 infection in early 2020 with an in vitro pseudotyped neutralization assay against the B.1.1.7 spike protein (Supplementary Table 1). Out of 60 monoclonal antibodies, 20 (33.3%) showed a greater than twofold loss of neutralizing activity against the B.1.1.7 variant compared to wild-type SARS-CoV-2 (Fig. 4a, b and Extended Data Fig. 6). The B.1.1.7 mutant virus fully escaped neutralization by 8 out of 10 monoclonal antibodies (80%) that target the N-terminal domain (NTD) (Fig. 4c). Of the 31 monoclonal antibodies that target the receptor-binding motif (RBM), 5 (16.1%) showed more than 100-fold decrease in B.1.1.7 neutralization, and additional 6 monoclonal antibodies (19.4%) had a partial 2–10-fold reduction (Fig. 4d). Finally, all RBD-specific non-RBM-targeting monoclonal antibodies that were tested fully retained neutralizing activity against B.1.1.7 (Fig. 4e).
a, Neutralization of pseudotyped SARS-CoV-2–murine leukemia virus (MLV) carrying wild-type spike (spike(D614G)) (grey), spike from B.1.1.7 (blue) or a triple-mutant spike protein (TM, carrying RBD mutations K417N, E484K and N501Y) (red) by three selected monoclonal antibodies (S2E12, S2X333 and S2H14) from one representative experiment. Data are mean ± s.d. of two technical replicates. b, Neutralization of SARS-CoV-2–MLVs carrying wild-type spike (spike(D614G)), spike from B.1.1.7 or a triple-mutant spike protein (spike(N501Y, E484K, K417N)) by 60 monoclonal antibodies targeting the NTD (n = 10), RBM (n = 31) or non-RBM sites in the RBD (n = 19). Data are the mean 50% inhibitory concentration (IC50) values (ng ml−1) of n = 2 independent experiments. c–e, Neutralization by NTD-specific (c), RBM-specific (d) and non-RBM-specific (e) monoclonal antibodies is shown as the mean IC50 values (top) and mean fold change in B.1.1.7 (blue) or the triple mutant (spike(N501Y, E484K, K417N)) (red) relative to the wild-type virus (bottom). The orange line shows the threshold for non-neutralizing titres. Top, data are mean ± s.d. IC50 values from two independent experiments. Bottom, data are mean ± s.d. fold change from two independent experiments. f–h, The kinetics of the binding of monoclonal antibodies to wild-type (black), N501Y (blue) and E484K (red) RBD as measured by biolayer interferometry. f, The four RBM-targeting monoclonal antibodies with no reduced binding to the RBD with N501Y or E484K are shown. g, h, Area under the curve (AUC) (g) and the fold change in the area under the curve (h) of 50 monoclonal antibodies tested against the wild-type, N501Y and E484K RBD. Monoclonal antibodies with a more than 1.3-fold (cut-off indicated by the orange line) change in area under the curve are shown in blue and red; orange dots show non-RBM-specific monoclonal antibodies.
To address the role of the N501Y substitution in B.1.1.7 in the neutralization escape from RBM-specific antibodies, we tested the binding of 50 RBD-specific monoclonal antibodies to the wild-type and N501Y-mutant RBD by biolayer interferometry (Fig. 4f and Extended Data Fig. 7). The 5 RBM-specific monoclonal antibodies that did not neutralize the B.1.1.7 variant (Fig. 4d) showed a complete loss of binding to the N501Y-mutant RBD (Fig. 4g, h), demonstrating a role for this mutation as an escape mechanism for certain RBM-targeting monoclonal antibodies.
To assess the effect of E484K on this panel of monoclonal antibodies, we generated a triple-mutant SARS-CoV-2 pseudotype virus carrying the K417N, E484K and N501Y mutations (spike(N501Y, E484K, K417N)). The inclusion of the K417N substitution was prompted by the observation that substitutions at this position have been found in five sequences from recent viral isolates within the B.1.1.7 lineage (K417 to Asn, Glu or Arg). This is in keeping with the convergent evolution of the virus to an RBD containing N501Y, E484K and K417N or K417T as evidenced by the B.1.351 and P.1 lineages. Notably, mutations at K417 are reported to escape neutralization by monoclonal antibodies, including the recently approved monoclonal antibody LY-CoV0165,7. Out of the 60 monoclonal antibodies tested, 20 (33.3%) showed a loss of neutralizing activity against the spike(N501Y, E484K, K417N) mutant of more than 10-fold compared to wild-type SARS-CoV-2 (Fig. 4a, b and Extended Data Fig. 6), and of these 19 are RBM-specific monoclonal antibodies. As above, we addressed the role of the E484K substitution in the escape from RBM-specific antibodies by testing the binding of 50 RBD-specific monoclonal antibodies to the RBD of the wild-type and E484K-mutant spike protein by biolayer interferometry (Fig. 4f and Extended Data Fig. 8). Out of the 19 RBM-specific monoclonal antibodies that showed reduced or loss of neutralization of the spike(N501Y, E484K, K417N) mutant (Fig. 4d), 16 showed a complete or partial loss of binding to the RBD of the E484K mutant (Fig. 4g, h), which is consistent with findings that E484K is an important mutation for viral escape8,9,10. In addition, 3 of these 16 monoclonal antibodies also lost the ability to bind to an RBD containing the N501Y substitution, indicating that a fraction of RBM-specific antibodies are sensitive to both the N501Y and E484K substitutions. Similarly, 3 of the 19 monoclonal antibodies that lost neutralization against the spike(N501Y, E484K, K417N) mutant (S2D8, S2H7 and S2X128) were previously shown to lose binding and neutralization to the K417V mutant, and are here shown to be sensitive to either the N501Y or the E484K substitution.
Binding of the RBD of the B.1.1.7 variant to ACE2
Using biolayer interferometry, we found that human ACE2 bound to the RBD of the B.1.1.7 variant with an affinity of 22 nM compared to an affinity of 133 nM for the wild-type RBD (Extended Data Fig. 9), in agreement with our previous deep-mutational scanning measurements using dimeric ACE211. Although ACE2 bound with comparable on rates to both RBDs, the observed dissociation rate constant was slower for B.1.1.7 than for the wild-type RBD (Extended Data Table 1). These findings could explain the efficient ongoing transmission of this newly emergent SARS-CoV-2 lineage and the possibly reduced opportunity for antibody binding. To understand the effect of the mutations in the triple mutant (K417N, E484K and N501Y), we evaluated the binding of ACE2 to the immobilized RBD of spike(N501Y, E484K, K417N). We determined an ACE2-binding affinity of 64 nM for the RBD of spike(N501Y, E484K, K417N), driven by a faster off rate than observed for the RBD of the B.1.1.7 variant but slower than for the wild-type RBD. We propose that the K417N mutation is slightly detrimental to ACE2 binding, which explains the intermediate affinity determined for the RBD of spike(N501Y, E484K, K417N) compared to the B.1.1.7 and wild-type RBDs, probably as a result of disrupting the salt bridge formed with ACE2 residue D30.
Discussion
Serum neutralizing activity is a correlate of protection for other respiratory viruses, including influenza12 and respiratory syncytial virus, for which prohylaxis with monoclonal antibodies has been used in at-risk groups13,14. Neutralizing antibody titres seemed to be highly correlated with vaccine protection against SARS-CoV-2 rechallenge in nonhuman primates15,16.
This study reports on neutralization by sera collected after both the first and second doses of the BNT162b2 vaccine. The participants of this study were older adults, in line with the targeting of this age group in the initial rollout of the vaccination campaign in the UK. We demonstrate that neutralization of a pseudovirus containing the spike protein with the full set of mutations that is present in the B.1.1.7 variant showed a small reduction using sera from individuals who received the BNT162b2 vaccine that was more marked after the first dose than the second dose. This could be related to the increased breadth, potency and/or concentration of antibodies after the boost dose. Other studies have reported a small reduction in neutralization against the B.1.1.7 variant in individuals vaccinated with two doses of BNT162b217 and mRNA-127318. The reduced neutralizing activity observed with polyclonal antibodies elicited by mRNA vaccines observed in this study is further supported by the loss of neutralizing activity observed with human monoclonal antibodies directed against both the RBD and, to a major extent, the NTD.
Multiple variants, including the 501Y.V2 and B.1.1.7 lineages, have multiple mutations as well as deletions in the NTD, most of which are located in a site of vulnerability that is targeted by all known NTD-specific neutralizing antibodies19,20. The role of NTD-specific neutralizing antibodies might be underestimated, in part by the use of neutralization assays based on target cells that overexpress ACE2 receptors. NTD-specific monoclonal antibodies were suggested to interfere with viral entry based on other accessory receptors, such as DC-SIGN and L-SIGN21, and their neutralization potency was found to be dependent on different in vitro culture conditions19. The observation that 9 out of 10 NTD-specific neutralizing antibodies did not show a complete or near-complete loss of neutralizing activity against B.1.1.7 indicates that this new variant may have also evolved to escape from this class of antibodies, which may have a yet unrecognized role in protective immunity. Taken together, the presence of multiple escape mutations in the NTD is supportive of the hypothesis that this region of the spike, in addition to the RBM, is also under immune pressure.
Worryingly, we have shown that there are multiple B.1.1.7 sequences in the UK that contain the E484K substitution with early evidence of transmission as well as independent acquisitions. We measured a further reduction in neutralization titres by vaccine sera when E484K was present alongside the B.1.1.7 spike mutations. A recent study18 has also shown that variants carrying the E484K substitution resulted in a 3–6-fold reduction in neutralization by sera from individuals who received the mRNA-1273 vaccine. Consistently, in this study we found that approximately 50% of the RBM-specific monoclonal antibodies tested lost neutralizing activity against SARS-CoV-2 carrying E484K. E484K has been shown to affect neutralization by monoclonal antibodies or convalescent sera, especially in combination with N501Y and K417N8,22,23,24.
Vaccines are a key part of a long-term strategy to bring SARS-CoV-2 transmission under control. Our data suggest that vaccine escape by the virus of current spike-directed vaccines designed against the Wuhan-1 strain will be inevitable, particularly given that E484K is emerging independently and recurrently on a B.1.1.7 (501Y.V1) background, and given the rapid global spread of B.1.1.7. Other major variants with E484K such as 501Y.V2 and V3 are also spreading regionally. This should be mitigated by designing next-generation vaccines with mutated spike sequences and using alternative viral antigens.
Methods
Data reporting
No statistical methods were used to predetermine sample size. The experiments were not randomized and the investigators were not blinded to allocation during experiments and outcome assessment.
Participant recruitment and ethics
Participants who had received the first dose of the BNT162b2 vaccine and individuals with COVID-19 were consented into the COVID-19 cohort of the NIHR Bioresource. The study was approved by the East of England–Cambridge Central Research Ethics Committee (17/EE/0025).
SARS-CoV-2 serology by multiplex particle-based flow cytometry
Recombinant SARS-CoV-2 nucleocapsid, spike and RBD proteins were covalently coupled to distinct carboxylated bead sets (Luminex) to form a triplex and were analysed as previously described25. Specific binding was reported as the mean fluorescence intensity.
Generation of spike mutants
Amino acid substitutions were introduced into the D614G pCDNA_SARS-CoV-2_S plasmid as previously described26 using the QuikChange Lightening Site-Directed Mutagenesis kit, following the manufacturer’s instructions (Agilent Technologies). Sequences were checked by Sanger sequencing.
The plasmid encoding the B.1.1.7 or triple-mutant (spike(N501Y, E484K, K417N)) SARS-CoV-2 spike glycoprotein was used to produce SARS-CoV-2–MLVs based on overlap extension PCR as follows. In brief, a modification of the overlap extension PCR protocol27 was used to introduce the eight mutations of the B.1.1.7 lineage or the three mutations of the triple mutant (spike(N501Y, E484K, K417N)) in the SARS-CoV-2 spike gene. In a first step, nine DNA fragments with overlapping sequences were amplified by PCR from a plasmid (phCMV1, Genlantis) encoding the full-length SARS-CoV-2 spike gene (BetaCoV/Wuhan-Hu-1/2019; accession number, mn908947). The mutations (ΔH69/ΔV70, Δ144, N501Y, A570D, D614G, P681H, S982A, T716I and D1118H or K417N, E484K and N501Y) were introduced by amplification with primers with a similar melting temperature. Deletion of the C-terminal 21 amino acids was introduced to increase surface expression of the recombinant spike protein28. Next, three contiguous overlapping fragments were fused by a first overlap PCR using the most external primers of each set, resulting in three larger fragments with overlapping sequences. A final overlap PCR was performed on the three large fragments using the most external primers to amplify the full-length spike gene and the flanking sequences including the restriction sites KpnI and NotI. This fragment was digested and cloned into the expression plasmid phCMV1. For all PCR reactions the Q5 Hot Start High Fidelity DNA polymerase (New England Biolabs) was used according to the manufacturer’s instructions and adapting the elongation time to the size of the amplicon. After each PCR step, the amplified regions were separated on an agarose gel and purified using an Illustra GFX PCR DNA and Gel Band Purification Kit (Merck).
Pseudotype virus preparation
Viral vectors were prepared by transfection of HEK293T cells using the Fugene HD transfection reagent (Promega). HEK293T cells were transfected with a mixture of 11 μl of Fugene HD, 1 μg of pCDNAD19spike-HA, 1 μg of p8.91 HIV-1 Gag-Pol expression vector29,30 and 1.5 μg of pCSFLW (expressing the firefly luciferase reporter gene with the HIV-1 packaging signal)31. Viral supernatants were collected at 48 h and 72 h after transfection, filtered through a 0.45-μm filter and stored at −80 °C. The 50% tissue culture infectious dose of the SARS-CoV-2 pseudovirus was determined using the Steady-Glo Luciferase assay system (Promega).
Serum and plasma pseudotype neutralization assay
Spike pseudotype assays have been shown to have similar characteristics to neutralization tests using fully infectious wild-type SARS-CoV-232. Virus neutralization assays were performed on HEK293T cells that were transiently transfected with ACE2 and TMPRSS2 using a SARS-CoV-2 spike pseudotyped virus that expressed luciferase33. Pseudotyped virus was incubated with a serial dilution of heat-inactivated human serum samples or sera from individuals who were vaccinated in duplicate for 1 h at 37 °C. Virus-only and cell-only controls were also included. Then, freshly trypsinized HEK293T ACE2- and TMPRSS2-expressing cells were added to each well. After incubation for 48 h in a 5% CO2 environment at 37 °C, luminescence was measured using the Steady-Glo or Bright-Glo Luciferase assay system (Promega). Neutralization was calculated relative to virus-only controls. Dilution curves are shown as the mean ± s.e.m. neutralization. ID50 values were calculated in GraphPad Prism. The ID50 values within groups were summarized as the GMT and statistical comparisons between groups were made with Wilxocon ranked-sign tests. In addition, the effects of the mutations on the neutralizing effect of the sera were expressed as fold change in ID50 of the wild-type compared to mutant pseudotyped virus. Statistical difference in the mean fold change between groups was determined using a two-tailed Student’s t-test.
IFNγ FluoroSpot assays
Frozen peripheral blood mononuclear cells (PBMCs) were rapidly thawed, and the freezing medium was diluted into 10 ml of TexMACS medium (Miltenyi Biotech), centrifuged and resuspended in 10 ml of fresh medium with 10 U ml−1 DNase (Benzonase, Merck-Millipore via Sigma-Aldrich), PBMCs were incubated at 37 °C for 1 h, followed by centrifugation and resuspension in fresh medium supplemented with 5% human serum (Sigma-Aldrich) before being counted. PBMCs were stained with 2 μl of each antibody: anti-CD3–fluorescein isothiocyanate (FITC), clone UCHT1; anti-CD4–phycoerythrin (PE), clone RPA-T4; anti-CD8a–peridinin-chlorophyll protein-cyanine 5.5 (PerCP-Cy5.5), clone RPA-8a (all BioLegend, London, UK), LIVE/DEAD Fixable Far Red Dead Cell Stain Kit (Thermo Fisher Scientific). PBMC phenotyping was performed on the BD Accuri C6 flow cytometer. Data were analysed with FlowJo v.10 (Becton Dickinson). In brief, 1.5–2.5 × 105 PBMCs were incubated in precoated Fluorospot plates (Human IFNγ FLUOROSPOT (Mabtech)) in triplicate with peptide mixes specific to spike, nucleocapsid and membrane proteins of SARS-CoV-2 (final peptide concentration 1 μg ml−1 per peptide, Miltenyi Biotech) and an unstimulated and positive control mix (containing anti-CD3 (Mabtech), Staphylococcus Enterotoxin B, phytohaemagglutinin (all Sigma-Aldrich)) at 37 °C in a humidified CO2 atmosphere for 48 h. The cells and medium were decanted from the plate and the assay was developed following the manufacturer’s instructions. Developed plates were read using an AID iSpot reader (Oxford Biosystems) and counted using AID EliSpot v.7 software (Autoimmun Diagnostika). All data were then corrected for background cytokine production and expressed as spot-forming units per million PBMCs or CD3+ T cells.
Antibody discovery and recombinant expression
Human monoclonal antibodies were isolated from plasma cells or memory B cells of donors who are immune to SARS-CoV or SARS-CoV-2 as previously described34,35,36,37. Recombinant antibodies were expressed in ExpiCHO cells at 37 °C and 8% CO2. Cells were transfected using ExpiFectamine. Transfected cells were supplemented 1 day after transfection with ExpiCHO Feed and ExpiFectamine CHO Enhancer. The cell culture supernatant was collected 8 days after transfection and filtered through a 0.2-μm filter. Recombinant antibodies were affinity purified on an ÄKTA xpress FPLC device using 5-ml HiTrap MabSelect PrismA columns followed by buffer exchange to histidine buffer (20 mM histidine, 8% sucrose, pH 6) using HiPrep 26/10 desalting columns.
Pseudovirus neutralization assay using monoclonal antibodies
MLV-based SARS-CoV-2 S-glycoprotein-pseudotyped viruses were prepared as previously described35. HEK293T/17 cells were cotransfected with a plasmid encoding the wild-type, B.1.1.7 or triple-mutant (spike(N501Y, E484K, K417N)) SARS-CoV-2 spike glycoprotein, an MLV Gag-Pol packaging construct and the MLV transfer vector encoding a luciferase reporter using X-tremeGENE HP transfection reagent (Roche) according to the manufacturer’s instructions. Cells were cultured for 72 h at 37 °C with 5% CO2 before collection of the supernatant. VeroE6 cells stably expressing human TMPRSS2 were cultured in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum, 1% penicillin–streptomycin (100 IU ml−1 penicillin, 100 μg ml−1), 8 μg ml−1 puromycin and plated into 96-well plates for 16–24 h. Pseudovirus with a serial dilution of monoclonal antibodies was incubated for 1 h at 37 °C and then added to the wells after washing twice with DMEM. After 2–3 h, DMEM containing 20% fetal bovine serum and 2% penicillin–streptomycin was added to the cells. After 48–72 h of infection, Bio-Glo (Promega) was added to the cells and incubated in the dark for 15 min before the the luminescence was read using a Synergy H1 microplate reader (BioTek). Measurements were done in duplicate and relative luciferase units were converted to the percentage of neutralization and plotted with a nonlinear regression model to determine the IC50 values using GraphPad Prism software (v.9.0.0).
Antibody binding measurements using biolayer interferometry
Monoclonal antibodies (Supplementary Table 1) were diluted to 3 μg ml−1 in kinetic buffer (PBS supplemented with 0.01% BSA) and immobilized on Protein A Biosensors (FortéBio). Antibody-coated biosensors were incubated for 3 min with a solution containing 5 μg ml−1 of wild-type, N501Y or E484K SARS-CoV-2 RBD in kinetic buffer, followed by a 3-min dissociation step. Changes in the molecules bound to the biosensors caused a shift in the interference pattern that was recorded in real time using an Octet RED96 system (FortéBio). The binding response over time was used to calculate the area under the curve using GraphPad Prism software (v.9.0.0).
Production of SARS-CoV-2 and B.1.1.7 RBDs and human ACE2
The SARS-CoV-2 RBD (BEI NR-52422) construct was synthesized by GenScript into CMVR with an N-terminal mu-phosphatase signal peptide, a C-terminal octa-histidine tag (GHHHHHHHH) and an avi tag. The boundaries of the construct are 328RFPN331 (N terminus) and 528KKST531 (C terminus)38. The B.1.1.7 RBD gene was synthesized by GenScript into pCMVR with the same boundaries and construct details with a mutation at N501Y. These plasmids were transiently transfected into Expi293F cells using Expi293F expression medium (Life Technologies) at 37 °C 8% CO2 while rotating at 150 rpm. The cultures were transfected using PEI cultivated for 5 days. Supernatants were clarified by centrifugation (10 min at 4,000g) before loading onto a nickel-NTA column (GE Healthcare). Purified protein was biotinylated overnight using BirA (Biotin ligase) before size-exclusion chromatography into PBS. Human ACE2–Fc (residues 1–615 with a C-terminal thrombin cleavage site and human Fc tag) was synthesized by Twist. Clarified supernatants were affinity-purified using a Protein A column (GE Life Sciences) that was directly neutralized and buffer exchanged. The Fc tag was removed by thrombin cleavage in a reaction mixture containing 3 mg of recombinant ACE2–Fc ectodomain and 10 μg of thrombin in 20 mM Tris-HCl pH 8.0, 150 mM NaCl and 2.5 mM CaCl2. The reaction mixture was incubated at 25 °C overnight and reloaded on a Protein A column to remove uncleaved protein and the Fc tag. The cleaved protein was further purified by gel filtration using a Superdex 200 column 10/300 GL (GE Life Sciences) equilibrated in PBS.
Protein affinity measurements using biolayer interferometry
Biotinylated RBDs (from wild-type spike, spike(N501Y) or spike(N501Y, E484K, K417N)) were immobilized at 5 ng μl in undiluted 10× Kinetics Buffer (Pall) to SA sensors until a load level of 1.1 nm. A dilution series of either monomeric ACE2 or Fab in undiluted kinetics buffer starting at 1,000 to 50 nM was used for 300–600 s to determine protein–protein affinity. The data were baseline subtracted and the plots fitted using the Pall FortéBio/Sartorius analysis software (v.12.0). Data were plotted in Graphpad Prism (v.9.0.2).
Phylogenetic analysis
All complete and low-coverage-excluded SARS-CoV-2 sequences were downloaded from the GISAID database (http://gisaid.org/)39 on 11 February 2021. All sequences were realigned to the SARS-CoV-2 reference strain MN908947.3, using MAFFT v.7.475 with automatic flavour selection and the --–keeplength --addfragments options40. Sequences were then deduplicated. Major SARS-CoV-2 clade memberships were assigned to all sequences using the Nextclade server v.0.12 (https://clades.nextstrain.org/).
Maximum likelihood phylogenetic trees were produced using the above curated dataset using IQ-TREE v.2.1.241. Evolutionary model selection for trees was inferred using ModelFinder10 and trees were estimated using the GTR + F + I model with 1,000 ultrafast bootstrap replicates42. All trees were visualized with Figtree v.1.4.4 (http://tree.bio.ed.ac.uk/software/figtree/) and manipulated and coloured with ggtree v.2.2.4. Phylogenies were rooted on the SARS-CoV-2 reference sequence (MN908947.3) and nodes arranged in descending order.
Statistical analysis
Linear regression was used to explore the association between the antibody response, T cell response and serum neutralization in Stata 13. The Pearson correlation coefficient was reported.
Neutralization data analysis
Neutralization was calculated relative to virus-only controls. Dilution curves were presented as a mean ± s.e.m. neutralization. IC50 values were calculated in GraphPad Prism. The ID50 values within groups were summarized as a GMT and statistical comparisons between groups were made using Wilxocon ranked-sign tests. In addition, the effects of the mutations on the neutralizing effect of the sera were expressed as the fold change in ID50 of the wild-type compared to the mutant pseudotyped virus. Statistical difference in the mean fold change between groups was determined using a two-tailed Student’s t-test.
IFNγ FluoroSpot assay data analysis
The association between the spike-associated T cell response, spike-specific antibody response and serum neutralization was determined using linear regression. The Pearson correlation coefficients between these variables were determined using Stata 13.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this paper.
Data availability
The neutralization and biolayer interferometry data shown in Fig. 4 and Extended Data Figs. 6–8 can be found in the Source Data for Fig. 4. All sequences are publicly available and were downloaded from http://gisaid.org. Deduplicated and subsampled data are freely available at https://github.com/StevenKemp/sequence_files/blob/main/vaccinepaper/with_background_subsampled_deduped_aligned_UKonly_484_vui.fasta.gz. Other data are available from the corresponding authors on request. Source data are provided with this paper.
Change history
21 July 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41586-022-05103-3
References
Volz, E. et al. Transmission of SARS-CoV-2 lineage B.1.1.7 in England: insights from linking epidemiological and genetic data. Preprint at https://doi.org/10.1101/2020.12.30.20249034 (2021).
Mulligan, M. J. et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 586, 589–593 (2020).
Kemp, S. A. et al. Recurrent emergence and transmission of a SARS-CoV-2 spike deletion H69/V70. Preprint at https://doi.org/10.1101/2020.12.14.422555 (2021).
Polack, F. P. et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 383, 2603–2615 (2020).
Wang, P. et al. Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7. Nature https://doi.org/10.1038/s41586-021-03398-2 (2021).
Public Health England. PHE statement on Variant of Concern and new Variant Under Investigation. https://www.gov.uk/government/news/phe-statement-on-variant-of-concern-and-new-variant-under-investigation (2021).
Thomson, E. C. et al. Circulating SARS-CoV-2 spike N439K variants maintain fitness while evading antibody-mediated immunity. Cell 184, 1171–1187 (2021).
Greaney, A. J. et al. Comprehensive mapping of mutations in the SARS-CoV-2 receptor-binding domain that affect recognition by polyclonal human plasma antibodies. Cell Host Microbe https://doi.org/10.1016/j.chom.2021.02.003 (2021).
Andreano, E. et al. SARS-CoV-2 escape in vitro from a highly neutralizing COVID-19 convalescent plasma. Preprint at https://doi.org/10.1101/2020.12.28.424451 (2020).
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., von Haeseler, A. & Jermiin, L. S. ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods 14, 587–589 (2017).
Starr, T. N. et al. Deep mutational scanning of SARS-CoV-2 receptor binding domain reveals constraints on folding and ACE2 binding. Cell 182, 1295–1310 (2020).
Verschoor, C. P. et al. Microneutralization assay titres correlate with protection against seasonal influenza H1N1 and H3N2 in children. PLoS ONE 10, e0131531 (2015).
Kulkarni, P. S., Hurwitz, J. L., Simões, E. A. F. & Piedra, P. A. Establishing correlates of protection for vaccine development: considerations for the respiratory syncytial virus vaccine field. Viral Immunol. 31, 195–203 (2018).
Goddard, N. L., Cooke, M. C., Gupta, R. K. & Nguyen-Van-Tam, J. S. Timing of monoclonal antibody for seasonal RSV prophylaxis in the United Kingdom. Epidemiol. Infect. 135, 159–162 (2007).
Mercado, N. B. et al. Single-shot Ad26 vaccine protects against SARS-CoV-2 in rhesus macaques. Nature 586, 583–588 (2020).
McMahan, K. et al. Correlates of protection against SARS-CoV-2 in rhesus macaques. Nature 590, 630–634 (2021).
Muik, A. et al. Neutralization of SARS-CoV-2 lineage B.1.1.7 pseudovirus by BNT162b2 vaccine-elicited human sera. Science https://doi.org/10.1126/science.abg6105 (2021).
Wu, K. et al. mRNA-1273 vaccine induces neutralizing antibodies against spike mutants from global SARS-CoV-2 variants. Preprint at https://doi.org/10.1101/2021.01.25.427948 (2021).
McCallum, M. et al. N-terminal domain antigenic mapping reveals a site of vulnerability for SARS-CoV-2. Preprint at https://doi.org/10.1101/2021.01.14.426475 (2021).
Suryadevara, N. et al. Neutralizing and protective human monoclonal antibodies recognizing the N-terminal domain of the SARS-CoV-2 spike protein. Preprint at https://doi.org/10.1101/2021.01.19.427324 (2021).
Soh, W. T. et al. The N-terminal domain of spike glycoprotein mediates SARS-CoV-2 infection by associating with L-SIGN and DC-SIGN. Preprint at https://doi.org/10.1101/2020.11.05.369264 (2020).
Tegally, H. et al. Detection of a SARS-CoV-2 variant of concern in South Africa. Nature https://doi.org/10.1038/s41586-021-03402-9 (2021).
Greaney, A. J. et al. Complete mapping of mutations to the SARS-CoV-2 spike receptor-binding domain that escape antibody recognition. Cell Host Microbe 29, 44–57 (2020).
Weisblum, Y. et al. Escape from neutralizing antibodies by SARS-CoV-2 spike protein variants. eLife 9, e61312 (2020).
Xiong, X. et al. A thermostable, closed SARS-CoV-2 spike protein trimer. Nat. Struct. Mol. Biol. 27, 934–941 (2020).
Gregson, J. et al. Human immunodeficiency virus-1 viral load is elevated in individuals with reverse-transcriptase mutation M184V/I during virological failure of first-line antiretroviral therapy and is associated with compensatory mutation L74I. J. Infect. Dis. 222, 1108–1116 (2019).
Forloni, M., Liu, A. Y. & Wajapeyee, N. Creating insertions or deletions using overlap extension polymerase chain reaction (PCR) mutagenesis. Cold Spring Harb. Protoc. https://doi.org/10.1101/pdb.prot097758 (2018).
Case, J. B. et al. Neutralizing antibody and soluble ACE2 inhibition of a replication-competent VSV-SARS-CoV-2 and a clinical isolate of SARS-CoV-2. Cell Host Microbe 28, 475–485 (2020).
Naldini, L., Blömer, U., Gage, F. H., Trono, D. & Verma, I. M. Efficient transfer, integration, and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector. Proc. Natl Acad. Sci. USA 93, 11382–11388 (1996).
Gupta, R. K. et al. Full-length HIV-1 Gag determines protease inhibitor susceptibility within in-vitro assays. AIDS 24, 1651–1655 (2010).
Kemp, S. A. et al. SARS-CoV-2 evolution during treatment of chronic infection. Nature https://doi.org/10.1038/s41586-021-03291-y (2021).
Schmidt, F. et al. Measuring SARS-CoV-2 neutralizing antibody activity using pseudotyped and chimeric viruses. J. Exp. Med. 217, e20201181 (2020).
Mlcochova, P. et al. Combined point of care nucleic acid and antibody testing for SARS-CoV-2 following emergence of D614G spike variant. Cell Rep. Med. 1, 100099 (2020).
Corti, D. et al. A neutralizing antibody selected from plasma cells that binds to group 1 and group 2 influenza A hemagglutinins. Science 333, 850–856 (2011).
Pinto, D. et al. Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody. Nature 583, 290–295 (2020).
Tortorici, M. A. et al. Ultrapotent human antibodies protect against SARS-CoV-2 challenge via multiple mechanisms. Science 370, 950–957 (2020).
Piccoli, L. et al. Mapping neutralizing and immunodominant sites on the SARS-CoV-2 spike receptor-binding domain by structure-guided high-resolution serology. Cell 183, 1024–1042 (2020).
Walls, A. C. et al. Elicitation of potent neutralizing antibody responses by designed protein nanoparticle vaccines for SARS-CoV-2. Cell 183, 1367–1382 (2020).
Shu, Y. & McCauley, J. GISAID: global initiative on sharing all influenza data — from vision to reality. Euro Surveill. 22, 30494 (2017).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Minh, B. Q. et al. IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 37, 1530–1534 (2020).
Minh, B. Q., Nguyen, M. A. & von Haeseler, A. Ultrafast approximation for phylogenetic bootstrap. Mol. Biol. Evol. 30, 1188–1195 (2013).
Acknowledgements
We thank the Cambridge University Hospitals NHS Trust Occupational Health Department; the NIHR Cambridge Clinical Research Facility and staff at CUH; E. Lim and G. Okecha; J. Voss for the gift of HeLa cells that stably express ACE2. R.K.G. is supported by a Wellcome Trust Senior Fellowship in Clinical Science (WT108082AIA). L.E.M. is supported by a Medical Research Council Career Development Award (MR/R008698/1). S.A.K. is supported by the Bill and Melinda Gates Foundation via PANGEA grant OPP1175094. D.A.C. is supported by a Wellcome Trust Clinical PhD Research Fellowship. K.G.C.S. is the recipient of a Wellcome Investigator Award (200871/Z/16/Z). This research was supported by the National Institute for Health Research (NIHR) Cambridge Biomedical Research Centre, the Cambridge Clinical Trials Unit (CCTU) and the NIHR BioResource. This study was supported by the National Institute of General Medical Sciences (R01GM120553 to D.V.), the National Institute of Allergy and Infectious Diseases (DP1AI158186 and HHSN272201700059C to D.V.), a Pew Biomedical Scholars Award (D.V.), an Investigators in the Pathogenesis of Infectious Disease Awards from the Burroughs Wellcome Fund (D.V.) and Fast Grants (D.V.). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. I.A.T.M.F. is funded by a SANTHE award (DEL-15-006).
Author information
Authors and Affiliations
Consortia
Contributions
D.C., R.K.G. and D.A.C. conceived the study. R.K.G., D.A.C., L.E.M., J. Bassi, M.W., L.C.-G., G.B.-M., R.D., B.G., N.K., A.E., M.S.P., D.V., L.P., A.D.M., J.R.B. and D.C. designed the study and experiments. B.M., D.A.C., N.T., R.P.D., I.A.T.M.F., A.C.W., L.C.-G., S.A.K. and G.B.-M. performed experiments. R.K.G., D.A.C., B.M., R.D., I.A.T.M.F., A.C.W., L.E.M., J. Bassi, K.G.C.S. and D.V. interpreted data. A.D.M., and C.S.F. carried out pseudovirus neutralization assays. D.P. produced pseudoviruses. M.S.P., L.P., W.H., D.V. and D.C. designed the experiments. M.A.T., J. Bassi and S.J. expressed and purified the proteins. K.C., S.J. and E.C. sequenced and expressed antibodies. E.C. and K.C. performed mutagenesis to create mutant expression plasmids. A.C.W. and S.B. performed binding assays. A.R., A.F.P. and C.G. contributed to the recruitment of donors and collection of samples related to the isolation of monoclonal antibodies. H.W.V., G.S., A.L., D.V., L.P., D.V. and D.C. analysed the data and prepared the manuscript with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
A.D.M., J. Bassi, D.P., C.S.F., S.B., K.C., N.S., E.C., G.S., S.J., A.L., H.W.V., M.S.P., L.P. and D.C. are employees of Vir Biotechnology and may hold shares in Vir Biotechnology. H.W.V. is a founder of PierianDx and Casma Therapeutics. Neither company provided funding for this work or is performing related work. D.V. is a consultant for Vir Biotechnology. The Veesler laboratory has received a sponsored research agreement from Vir Biotechnology. R.K.G. has received consulting fees from UMOVIS Lab, Gilead and ViiV. The other authors declare no competing interests.
Additional information
Peer review information Nature thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Immune responses three weeks after first dose of the BNT162b2 vaccine against SARS-CoV-2.
a, Serum IgG responses against the N protein, spike protein and the RBD of the spike protein of participants who received one vaccine dose (light green) or two vaccine doses (dark green), patients who had recovered from COVID-19 (red) and healthy control individuals (grey) were measured by a flow-cytometry-based Luminex assay. n = 25. MFI, mean fluorescence intensity. Data are GMT ± s.d. (lines and error bars) of two technical repeats and individual values (cirlces). b, Relationship between serum IgG responses, measured by flow cytometry, and serum neutralization ID50. n = 25. c, Relationship between serum neutralization ID50 and T cell responses against SARS-CoV-2 by IFNγ FluoroSpot. n = 24. SFU, spot-forming units. d, Relationship between serum IgG responses and T cell responses. n = 23. b–d, Simple linear regressions are shown with Pearson correlation (r), P value (p) and regression coefficient/slope (β).
Extended Data Fig. 2 Neutralization by the first dose of the BNT162b2 vaccine and convalescent sera against the wild-type and mutant (N501Y, A570D, ΔH69/ΔV70) SARS-CoV-2-pseudotyped viruses.
a, b, Dilution of the vaccine sera for 50% neutralization against the wild-type and spike mutant (N501Y, A570D, ΔH69/ΔV70) viruses. b, Data are GMT ± s.d. (lines and error bars) of two independent experiments with two technical repeats and individual values (circles). Two-tailed Wilcoxon matched-pairs signed-rank test with no adjustment for multiple comparisons; ***P < 0.001. c, d, Dilution of convalescent sera for 50% neutralization against the wild-type and spike mutant (N501Y, A570D, ΔH69/ΔV70) viruses. Data are GMT ± s.d. (dotted lines and error bars) of a representative experiment with two technical repeats and individual values (circles). Two-tailed Wilcoxon matched-pairs signed-rank test with no adjustment for multiple comparisons; ns, not significant. e, Representative curves of log10-transformed inverse dilutions of convalescent sera against the percentage of neutralization for the wild-type and spike mutant (N501Y, A570D, ΔH69/ΔV70) viruses. In cases in which a curve is shifted to the right the virus is less sensitive to the neutralizing antibodies in the serum. Data are mean ± s.e.m. of two technical replicates. Data are representative of two independent experiments. The cut-off for 50% neutralization was set to 4 (dotted lines in a, b). a, c, Data points of the same individual are connected by lines.
Extended Data Fig. 3 Representative neutralization curves of sera from individuals vaccinated with the BNT162b2 vaccine against pseudotyped virus bearing eight mutations in the spike protein that are present in the B.1.1.7 variant compared with the wild-type virus.
All virus variants were in a spike(D614G) background. The log10-transformed inverse dilutions of the sera are shown against the percentage of neutralization. In cases in which a curve is shifted to the right the virus is less sensitive to the neutralizing antibodies in the serum. Data are for the first dose of vaccine (D1). Data are mean ± s.e.m. representative of two independent experiments each with two technical replicates.
Extended Data Fig. 4 Representative neutralization curves of convalescent sera against wild-type and B.1.1.7 spike-mutant SARS-CoV-2 pseudotyped viruses.
The log10-transformed inverse dilutions of the sera are shown against the percentage of neutralization. In cases in which a curve is shifted to the right the virus is less sensitive to the neutralizing antibodies in the serum. Data are mean ± s.e.m. representative of two independent experiments each with two technical replicates.
Extended Data Fig. 5 Neutralization potency of mRNA vaccine sera and convalescent sera (before SARS-CoV-2 B.1.1.7) against pseudotyped virus bearing spike mutations in the B.1.1.7 lineage with and without the E484K substitution in the RBD.
All virus variants were in a spike(D614G) background. Neutralization potency of the sera from the first (left; n = 37) and the second (middle, n = 21) vaccine dose and of convalescent plasma (CP) (right; n = 27) against wild-type SARS-CoV-2, the B.1.1.7 variant with spike(N501Y, A570D, ΔH69/ΔV70, Δ144, P681H, T716I, S982A, D1118H) and the B.1.1.7 variant with spike(N501Y, A570D, ΔH69/ΔV70, Δ144, P681H, T716I, S982A, D1118H) and the additional E484K substitution. Data are GMT ± s.d. representative of two independent experiments each with two technical repeats. Wilcoxon matched-pairs signed-rank test; **P < 0.01, ***P < 0.001, **** P < 0.0001.
Extended Data Fig. 6 Neutralization of the wild-type spike(D614G), B.1.1.7 spike and spike(N501Y, E484K, K417N) proteins of the SARS-CoV-2-pseudotyped virus by a panel of 57 monoclonal antibodies.
a–c, Neutralization of the wild-type spike (black), B.1.1.7 spike (blue) and spike(N501Y, E484K, K417N) (TM) (red) SARS-CoV-2–MLV by 9 NTD-targeting (a), 29 RBM-targeting (b) and 19 non-RBM-targeting (c) monoclonal antibodies. Data are mean ± s.d. of two technical replicates from one representative experiment.
Extended Data Fig. 7 Kinetics of binding to the RBD of wild-type and spike(N501Y) SARS-CoV-2 for 43 RBD-specific monoclonal antibodies.
a-b, a, b, Binding to the RBD of wild-type (black) and spike(N501Y) (blue) SARS-CoV-2 by 22 RBM-targeting (a) and 21 non-RBM-targeting (b) monoclonal antibodies. An antibody of irrelevant specificity was included as a negative control.
Extended Data Fig. 8 Kinetics of binding to the RBD of wild-type and spike(E484K) SARS-CoV-2 for 46 RBD-specific monoclonal antibodies.
a, b, Binding to the RBD of wild-type (black) and spike(E484K) (red) SARS-CoV-2 by 27 RBM-targeting (a) and 19 non-RBM-targeting (b) monoclonal antibodies. An antibody of irrelevant specificity was included as a negative control.
Extended Data Fig. 9 Binding of human ACE2 to the RBDs of the wild-type SARS-CoV-2 spike, spike(N501Y) and spike(N501Y, E484K, K417N) proteins.
a–c, Biolayer interferometry binding analysis of the human ACE2 (huACE2) ectodomain (residues 1–615) to immobilized RBD of wild-type SARS-CoV-2 (a) and the RBD of B.1.1.7 spike(N501Y) (b) and spike (N501Y, E484K, K417N) proteins (c). Black lines correspond to a global fit of the data using a 1:1 binding model.
Supplementary information
Supplementary Table 1
Neutralization, V gene usage and other properties of tested mAbs.
Supplementary Table 2
GISAID acknowledgements.
Rights and permissions
About this article
Cite this article
Collier, D.A., De Marco, A., Ferreira, I.A.T.M. et al. Sensitivity of SARS-CoV-2 B.1.1.7 to mRNA vaccine-elicited antibodies. Nature 593, 136–141 (2021). https://doi.org/10.1038/s41586-021-03412-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-021-03412-7
This article is cited by
-
Characterization of intrinsic and effective fitness changes caused by temporarily fixed mutations in the SARS-CoV-2 spike E484 epitope and identification of an epistatic precondition for the evolution of E484A in variant Omicron
Virology Journal (2023)
-
Bafilomycin A1 inhibits SARS-CoV-2 infection in a human lung xenograft mouse model
Virology Journal (2023)
-
Some common deleterious mutations are shared in SARS-CoV-2 genomes from deceased COVID-19 patients across continents
Scientific Reports (2023)
-
In silico prediction of immune-escaping hot spots for future COVID-19 vaccine design
Scientific Reports (2023)
-
B-cell and antibody responses to SARS-CoV-2: infection, vaccination, and hybrid immunity
Cellular & Molecular Immunology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.