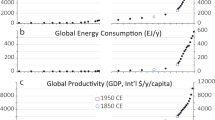
Abstract
Humanity has become a dominant force in shaping the face of Earth1,2,3,4,5,6,7,8,9. An emerging question is how the overall material output of human activities compares to the overall natural biomass. Here we quantify the human-made mass, referred to as ‘anthropogenic mass’, and compare it to the overall living biomass on Earth, which currently equals approximately 1.1 teratonnes10,11. We find that Earth is exactly at the crossover point; in the year 2020 (± 6), the anthropogenic mass, which has recently doubled roughly every 20 years, will surpass all global living biomass. On average, for each person on the globe, anthropogenic mass equal to more than his or her bodyweight is produced every week. This quantification of the human enterprise gives a mass-based quantitative and symbolic characterization of the human-induced epoch of the Anthropocene.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All data used in this study are available on GitHub, at https://github.com/milo-lab/anthropogenic_mass. Anthropogenic mass data are available from ref. 22 and at https://boku.ac.at/wiso/sec/data-download. TRENDY Dynamic Global Vegetation Models outputs are available at https://sites.exeter.ac.uk/trendy. Leaves dry matter content measurements were obtained via TryDB, at https://www.try-db.org/. Other datasets used in this study are available from the published literature, as detailed in the Methods and Supplementary Information.
Code availability
All code used in this study is available on GitHub, at https://github.com/milo-lab/anthropogenic_mass.
References
Ramankutty, N. & Foley, J. A. Estimating historical changes in global land cover: croplands from 1700 to 1992. Glob. Biogeochem. Cycles 13, 997–1027 (1999).
Krausmann, F. et al. Growth in global materials use, GDP and population during the 20th century. Ecol. Econ. 68, 2696–2705 (2009).
Matthews, E. The Weight of Nations: Material Outflows from Industrial Economies (World Resources Inst., 2000).
Smil, V. Harvesting the Biosphere: What We Have Taken from Nature (MIT Press, 2013).
Smil, V. Making the Modern World: Materials and Dematerialization (John Wiley & Sons, 2013).
Haff, P. K. Technology as a geological phenomenon: implications for human well-being. Geol. Soc. Lond. Spec. Publ. 395, 301–309 (2014).
Zalasiewicz, J. et al. Scale and diversity of the physical technosphere: a geological perspective. Anthropocene Rev. 4, 9–22 (2017).
Zalasiewicz, J., Waters, C. N., Williams, M. & Summerhayes, C. The Anthropocene as a Geological Time Unit: A Guide to the Scientific Evidence and Current Debate (Cambridge Univ. Press, 2018).
Stephens, L. et al. Archaeological assessment reveals Earth’s early transformation through land use. Science 365, 897–902 (2019).
Erb, K.-H. et al. Unexpectedly large impact of forest management and grazing on global vegetation biomass. Nature 553, 73–76 (2018).
Bar-On, Y. M., Phillips, R. & Milo, R. The biomass distribution on Earth. Proc. Natl Acad. Sci. USA 115, 6506–6511 (2018).
Pan, Y. et al. A large and persistent carbon sink in the world’s forests. Science 333, 988–993 (2011).
Reddington, C. L. et al. Air quality and human health improvements from reductions in deforestation-related fire in Brazil. Nat. Geosci. 8, 768–771 (2015).
Ceballos, G. & Ehrlich, P. R. Mammal population losses and the extinction crisis. Science 296, 904–907 (2002).
WWF. Living Planet Report–2018: Aiming Higher (WWF, 2018).
Bar-On, Y. M. & Milo, R. Towards a quantitative view of the global ubiquity of biofilms. Nat. Rev. Microbiol. 17, 199–200 (2019).
Pauliuk, S. & Hertwich, E. G. Socioeconomic metabolism as paradigm for studying the biophysical basis of human societies. Ecol. Econ. 119, 83–93 (2015).
Haberl, H. et al. Contributions of sociometabolic research to sustainability science. Nat. Sustainability 2, 173–184 (2019).
Fischer-Kowalski, M. et al. Methodology and indicators of economy-wide material flow accounting. J. Ind. Ecol. 15, 855–876 (2011).
Krausmann, F., Schandl, H., Eisenmenger, N., Giljum, S. & Jackson, T. Material flow accounting: measuring global material use for sustainable development. Annu. Rev. Environ. Resour. 42, 647–675 (2017).
Krausmann, F. et al. Global socioeconomic material stocks rise 23-fold over the 20th century and require half of annual resource use. Proc. Natl Acad. Sci. USA 114, 1880–1885 (2017).
Krausmann, F., Lauk, C., Haas, W. & Wiedenhofer, D. From resource extraction to outflows of wastes and emissions: the socioeconomic metabolism of the global economy, 1900–2015. Glob. Environ. Change 52, 131–140 (2018).
Steffen, W., Broadgate, W., Deutsch, L., Gaffney, O. & Ludwig, C. The trajectory of the Anthropocene: the great acceleration. Anthropocene Rev. 2, 81–98 (2015).
Vitousek, P. M., Ehrlich, P. R., Ehrlich, A. H. & Matson, P. A. Human appropriation of the products of photosynthesis. Bioscience 36, 368–373 (1986).
Haberl, H. et al. Quantifying and mapping the human appropriation of net primary production in Earth’s terrestrial ecosystems. Proc. Natl Acad. Sci. USA 104, 12942–12947 (2007).
Haberl, H., Erb, K.-H. & Krausmann, F. Human appropriation of net primary production: patterns, trends, and planetary boundaries. Annu. Rev. Environ. Resour. 39, 363–391 (2014).
Vitousek, P. M. Human domination of Earth’s ecosystems. Science 277, 494–499 (1997).
Dirzo, R. et al. Defaunation in the Anthropocene. Science 345, 401–406 (2014).
Crutzen, P. J. in Earth System Science in the Anthropocene (eds. Ehlers, E. & Kraft, T.) 13–18 (Springer, 2006).
Steffen, W., Crutzen, J. & McNeill, J. R. The Anthropocene: are humans now overwhelming the great forces of Nature? Ambio 36, 614–621 (2007).
Lewis, S. L. & Maslin, M. A. Defining the Anthropocene. Nature 519, 171–180 (2015).
Waters, C. N. et al. The Anthropocene is functionally and stratigraphically distinct from the Holocene. Science 351, aad2622 (2016).
Krausmann, F. et al. Economy-wide Material Flow Accounting. Introduction and Guide Version 1, Social Ecology Working Paper 151 (Alpen-Adria Univ., 2015).
Miatto, A., Schandl, H., Fishman, T. & Tanikawa, H. Global patterns and trends for non-metallic minerals used for construction. J. Ind. Ecol. 21, 924–937 (2017).
Cooper, A. H., Brown, T. J., Price, S. J., Ford, J. R. & Waters, C. N. Humans are the most significant global geomorphological driving force of the 21st century. Anthropocene Rev. 5, 222–229 (2018).
Food and Agriculture Organization of the United Nations. Global Forest Resources Assessment 2010: Main Report (FAO, 2010).
Liu, Y. Y. et al. Recent reversal in loss of global terrestrial biomass. Nat. Clim. Chang. 5, 470–474 (2015).
Sitch, S. et al. Recent trends and drivers of regional sources and sinks of carbon dioxide. Biogeosciences 12, 653–679 (2015).
Friedlingstein, P. et al. Global carbon budget 2019. Earth Syst. Sci. Data 11, 1783–1838 (2019).
Food and Agriculture Organization of the United Nations FAOSTAT http://faostat.fao.org.
Magnabosco, C. et al. The biomass and biodiversity of the continental subsurface. Nat. Geosci. 11, 707–717 (2018).
Haverd, V. et al. A new version of the CABLE land surface model (Subversion revision r4601) incorporating land use and land cover change, woody vegetation demography, and a novel optimisation-based approach to plant coordination of photosynthesis. Geosci. Model Dev. 11, 2995–3026 (2018).
Melton, J. R. & Arora, V. K. Competition between plant functional types in the Canadian Terrestrial Ecosystem Model (CTEM) v. 2.0. Geosci. Model Dev. 9, 323–361 (2016).
Lawrence, D. M. et al. The community land model version 5: description of new features, benchmarking, and impact of forcing uncertainty. J. Adv. Model. Earth Syst. 11, 4245–4287 (2019).
Tian, H. et al. North American terrestrial CO2 uptake largely offset by CH4 and N2O emissions: toward a full accounting of the greenhouse gas budget. Clim. Change 129, 413–426 (2015).
Meiyappan, P., Jain, A. K. & House, J. I. Increased influence of nitrogen limitation on CO2 emissions from future land use and land use change. Glob. Biogeochem. Cycles 29, 1524–1548 (2015).
Mauritsen, T. et al. Developments in the MPI-M Earth System Model version1.2 (MPI-ESM1.2) and its response to increasing CO2. J. Adv. Model. Earth Syst. 11, 998–1038 (2019).
Clark, D. B. et al. The Joint UK Land Environment Simulator (JULES), model description – Part 2: Carbon fluxes and vegetation dynamics. Geosci. Model Dev. 4, 701–722 (2011).
Poulter, B., Frank, D. C., Hodson, E. L. & Zimmermann, N. E. Impacts of land cover and climate data selection on understanding terrestrial carbon dynamics and the CO2 airborne fraction. Biogeosciences 8, 2027–2036 (2011).
Smith, B. et al. Implications of incorporating N cycling and N limitations on primary production in an individual-based dynamic vegetation model. Biogeosciences 11, 2027–2054 (2014).
Lienert, S. & Joos, F. A Bayesian ensemble data assimilation to constrain model parameters and land-use carbon emissions. Biogeosciences 15, 2909–2930 (2018).
Zaehle, S. & Friend, A. D. Carbon and nitrogen cycle dynamics in the O-CN land surface model: 1. Model description, site-scale evaluation, and sensitivity to parameter estimates. Glob. Biogeochem. Cycles 24, GB1005 (2010).
Krinner, G. et al. A dynamic global vegetation model for studies of the coupled atmosphere–biosphere system. Glob. Biogeochem. Cycles 19, GB1015 (2005).
Goll, D. S. et al. Carbon–nitrogen interactions in idealized simulations with JSBACH (version 3.10). Geosci. Model Dev. 10, 2009–2030 (2017).
Walker, A. P. et al. The impact of alternative trait-scaling hypotheses for the maximum photosynthetic carboxylation rate (Vcmax) on global gross primary production. New Phytol. 215, 1370–1386 (2017).
Kato, E., Kinoshita, T., Ito, A., Kawamiya, M. & Yamagata, Y. Evaluation of spatially explicit emission scenario of land-use change and biomass burning using a process-based biogeochemical model. J. Land Use Sci. 8, 104–122 (2013).
Tang, Z. et al. Patterns of plant carbon, nitrogen, and phosphorus concentration in relation to productivity in China’s terrestrial ecosystems. Proc. Natl Acad. Sci. USA 115, 4033–4038 (2018).
Poorter, H. et al. Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol. 193, 30–50 (2012).
Heldal, M., Norland, S. & Tumyr, O. X-ray microanalytic method for measurement of dry matter and elemental content of individual bacteria. Appl. Environ. Microbiol. 50, 1251–1257 (1985).
von Stockar, U. & Liu, J. Does microbial life always feed on negative entropy? Thermodynamic analysis of microbial growth. Biochim. Biophys. Acta 1412, 191–211 (1999).
Guo, L., Lin, H., Fan, B., Cui, X. & Chen, J. Impact of root water content on root biomass estimation using ground penetrating radar: evidence from forward simulations and field controlled experiments. Plant Soil 371, 503–520 (2013).
Glass, S. V. & Zelinka, S. L. in Wood Handbook: Wood as an Engineering Material Vol. 190, 4.1–4.19 (US Department of Agriculture, 2010).
Loveys, B. R. et al. Thermal acclimation of leaf and root respiration: an investigation comparing inherently fast- and slow-growing plant species. Glob. Change Biol. 9, 895–910 (2003).
Sheremetev, S. N. Herbs on the Soil Moisture Gradient (Water Relations and the Structural-Functional Organization) (KMK, 2005).
Michaletz, S. T. & Johnson, E. A. A heat transfer model of crown scorch in forest fires. Can. J. For. Res. 36, 2839–2851 (2006).
Messier, J., McGill, B. J. & Lechowicz, M. J. How do traits vary across ecological scales? A case for trait-based ecology. Ecol. Lett. 13, 838–848 (2010).
Boucher, F. C., Thuiller, W., Arnoldi, C., Albert, C. H. & Lavergne, S. Unravelling the architecture of functional variability in wild populations of Polygonum viviparum L. Funct. Ecol. 27, 382–391 (2013).
Dahlin, K. M., Asner, G. P. & Field, C. B. Environmental and community controls on plant canopy chemistry in a Mediterranean-type ecosystem. Proc. Natl Acad. Sci. USA 110, 6895–6900 (2013).
Kattge, J. et al. TRY–a global database of plant traits. Glob. Change Biol. 17, 2905–2935 (2011).
Lebigot, E. O. Uncertainties: a Python package for calculations with uncertainties. https://pythonhosted.org/uncertainties/ (2010).
Wiedenhofer, D., Fishman, T., Lauk, C., Haas, W. & Krausmann, F. Integrating material stock dynamics into economy-wide material flow accounting: concepts, modelling, and global application for 1900–2050. Ecol. Econ. 156, 121–133 (2019).
Acknowledgements
We thank U. Alon, S. Dan, G. Eshel, T. Fishman, E. Gelbrieth, T. Kaufmann, T. Klein, A. Knoll, E. Noor, N. Page, R. Phillips, J. Pongratz, M. Shamir, M. Shtein, B. Smith, C. Waters, T. Wiesel, M. Williams and members of our laboratory for help and discussions, and S. Sitch and the TRENDY DGVM community for access to their simulation outputs. This research was supported by the European Research Council (Project NOVCARBFIX 646827); Beck-Canadian Center for Alternative Energy Research; Dana and Yossie Hollander; Ullmann Family Foundation; Helmsley Charitable Foundation; Larson Charitable Foundation; Wolfson Family Charitable Trust; Charles Rothschild; and Selmo Nussenbaum. R.M. is the Charles and Louise Gartner professional chair. Y.M.B.-O is an Azrieli Fellow.
Author information
Authors and Affiliations
Contributions
E.E., L.B.-U. and R.M. wrote the manuscript. E.E. performed the bulk of the research and data analysis. L.B.-U. contributed to the anthropogenic mass analysis and biomass estimation. Y.M.B.-O. contributed to the biomass estimation and carbon content calculation. J.G. contributed to the water content calculation. E.E., J.G. and Y.M.B.-O. performed the uncertainty analysis. E.E., L.B.-U. and R.M. conceived the study. R.M. supervised the study. All authors discussed the results, and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Fridolin Krausmann, Dominik Wiedenhofer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Sensitivity analysis of the anthropogenic mass definition.
a–f, The effect of adding the following to the anthropogenic mass (dark purple): a, mass of the human population, b, mass of livestock, c, mass of crops and agroforestry, d, mass of earthworks, dredging and waste/overburden from mineral and metal production, and f, mass of anthropogenic atmospheric CO2 stocks, as well as e, the exclusion of the mass of industrial roundwood. The total biomass weight is depicted by the green line. Black dot indicates the year of intersection based on the alternative anthropogenic mass definition. Violet area and light green-dashed line indicate extrapolated anthropogenic mass and biomass estimates, respectively. Full description of the sensitivity analysis is provided in Supplementary Information section 1.
Extended Data Fig. 2 Anthropogenic mass composition since the year 1900, divided into material groups.
Dataset is based on ref. 22.
Extended Data Fig. 3 Anthropogenic mass relative annual change, with highlights of notable global events.
Relative annual change is calculated as the difference between two consecutive years divided by the earlier year anthropogenic mass value.
Extended Data Fig. 6 Calculation steps in plant biomass estimation for 1990–2017.
As further detailed in the Methods section ‘Biomass change over the years 1900–2017’.
Supplementary information
Supplementary Information
Supplementary Discussions: This file contains Supplementary Section 1 & 2. It includes the sensitivity analysis of the anthropogenic mass definition, as well as an additional discussion on the biomass trend in recent years.
Rights and permissions
About this article
Cite this article
Elhacham, E., Ben-Uri, L., Grozovski, J. et al. Global human-made mass exceeds all living biomass. Nature 588, 442–444 (2020). https://doi.org/10.1038/s41586-020-3010-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-3010-5
This article is cited by
-
A QGIS Plugin for GIS-based Multicriteria Decision Analysis: an Application of Developing Alternative Future Land-Use Scenarios in Ghana
Applied Spatial Analysis and Policy (2024)
-
Towards a bioinformational understanding of AI
AI & SOCIETY (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.