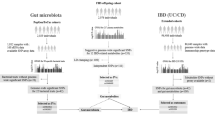
Abstract
Microbiome community typing analyses have recently identified the Bacteroides2 (Bact2) enterotype, an intestinal microbiota configuration that is associated with systemic inflammation and has a high prevalence in loose stools in humans1,2. Bact2 is characterized by a high proportion of Bacteroides, a low proportion of Faecalibacterium and low microbial cell densities1,2, and its prevalence varies from 13% in a general population cohort to as high as 78% in patients with inflammatory bowel disease2. Reported changes in stool consistency3 and inflammation status4 during the progression towards obesity and metabolic comorbidities led us to propose that these developments might similarly correlate with an increased prevalence of the potentially dysbiotic Bact2 enterotype. Here, by exploring obesity-associated microbiota alterations in the quantitative faecal metagenomes of the cross-sectional MetaCardis Body Mass Index Spectrum cohort (n = 888), we identify statin therapy as a key covariate of microbiome diversification. By focusing on a subcohort of participants that are not medicated with statins, we find that the prevalence of Bact2 correlates with body mass index, increasing from 3.90% in lean or overweight participants to 17.73% in obese participants. Systemic inflammation levels in Bact2-enterotyped individuals are higher than predicted on the basis of their obesity status, indicative of Bact2 as a dysbiotic microbiome constellation. We also observe that obesity-associated microbiota dysbiosis is negatively associated with statin treatment, resulting in a lower Bact2 prevalence of 5.88% in statin-medicated obese participants. This finding is validated in both the accompanying MetaCardis cardiovascular disease dataset (n = 282) and the independent Flemish Gut Flora Project population cohort (n = 2,345). The potential benefits of statins in this context will require further evaluation in a prospective clinical trial to ascertain whether the effect is reproducible in a randomized population and before considering their application as microbiota-modulating therapeutics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Raw amplicon sequencing data used in this study have been deposited in the EMBL-EBI European Nucleotide Archive (ENA) under accession number PRJEB37249. The metadata and processed microbiome data required for the reanalysis of results presented in the manuscript are respectively provided as Supplementary Table 2 and available for download at http://raeslab.org/software/BMIS/. For clinical cohort-related questions, contact K.C.
References
Vandeputte, D. et al. Quantitative microbiome profiling links gut community variation to microbial load. Nature 551, 507–511 (2017).
Vieira-Silva, S. et al. Quantitative microbiome profiling disentangles inflammation- and bile duct obstruction-associated microbiota alterations across PSC/IBD diagnoses. Nat. Microbiol. 4, 1826–1831 (2019).
Probert, C. S., Emmett, P. M. & Heaton, K. W. Some determinants of whole-gut transit time: a population-based study. QJM 88, 311–315 (1995).
Ford, E. S. Body mass index, diabetes, and C-reactive protein among U.S. adults. Diabetes Care 22, 1971–1977 (1999).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Sze, M. A. & Schloss, P. D. Looking for a signal in the noise: revisiting obesity and the microbiome. MBio 7, e01018-16 (2016).
Walters, W. A., Xu, Z. & Knight, R. Meta-analyses of human gut microbes associated with obesity and IBD. FEBS Lett. 588, 4223–4233 (2014).
Le Chatelier, E. et al. Richness of human gut microbiome correlates with metabolic markers. Nature 500, 541–546 (2013).
Karlsson, F. H. et al. Symptomatic atherosclerosis is associated with an altered gut metagenome. Nat. Commun. 3, 1245 (2012).
Falony, G. et al. Population-level analysis of gut microbiome variation. Science 352, 560–564 (2016).
Vieira-Silva, S. et al. Species-function relationships shape ecological properties of the human gut microbiome. Nat. Microbiol. 1, 16088 (2016).
Alberti, K. G. M. M., Zimmet, P. & Shaw, J. Metabolic syndrome—a new world-wide definition. A consensus statement from the International Diabetes Federation. Diabet. Med. 23, 469–480 (2006).
Depommier, C. et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat. Med. 25, 1096–1103 (2019).
Yun, Y. et al. Comparative analysis of gut microbiota associated with body mass index in a large Korean cohort. BMC Microbiol. 17, 151 (2017).
Quévrain, E. et al. Identification of an anti-inflammatory protein from Faecalibacterium prausnitzii, a commensal bacterium deficient in Crohn’s disease. Gut 65, 415–425 (2016).
Louis, P. et al. Restricted distribution of the butyrate kinase pathway among butyrate-producing bacteria from the human colon. J. Bacteriol. 186, 2099–2106 (2004).
Litvak, Y., Byndloss, M. X. & Bäumler, A. J. Colonocyte metabolism shapes the gut microbiota. Science 362, eaat9076 (2018).
Kriss, M., Hazleton, K. Z., Nusbacher, N. M., Martin, C. G. & Lozupone, C. A. Low diversity gut microbiota dysbiosis: drivers, functional implications and recovery. Curr. Opin. Microbiol. 44, 34–40 (2018).
Ding, T. & Schloss, P. D. Dynamics and associations of microbial community types across the human body. Nature 509, 357–360 (2014).
Gardiner, B. J. et al. Clinical and microbiological characteristics of Eggerthella lenta bacteremia. J. Clin. Microbiol. 53, 626–635 (2015).
Mazmanian, S. K., Liu, C. H., Tzianabos, A. O. & Kasper, D. L. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122, 107–118 (2005).
Wexler, H. M. Bacteroides: the good, the bad, and the nitty-gritty. Clin. Microbiol. Rev. 20, 593–621 (2007).
Ridker, P. M. et al. Reduction in C-reactive protein and LDL cholesterol and cardiovascular event rates after initiation of rosuvastatin: a prospective study of the JUPITER trial. Lancet 373, 1175–1182 (2009).
Muscogiuri, G. et al. The good and bad effects of statins on insulin sensitivity and secretion. Endocr. Res. 39, 137–143 (2014).
Khan, T. J. et al. Effect of atorvastatin on the gut microbiota of high fat diet-induced hypercholesterolemic rats. Sci. Rep. 8, 662 (2018).
Maier, L. et al. Extensive impact of non-antibiotic drugs on human gut bacteria. Nature 555, 623–628 (2018).
Forslund, K. et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 528, 262–266 (2015).
Zeiser, R. Immune modulatory effects of statins. Immunology 154, 69–75 (2018).
Grip, O., Janciauskiene, S. & Bredberg, A. Use of atorvastatin as an anti-inflammatory treatment in Crohn’s disease. Br. J. Pharmacol. 155, 1085–1092 (2008).
Ungaro, R. et al. Statins associated with decreased risk of new onset inflammatory bowel disease. Am. J. Gastroenterol. 111, 1416–1423 (2016).
Valles-Colomer, M. et al. The neuroactive potential of the human gut microbiota in quality of life and depression. Nat. Microbiol. 4, 623–632 (2019).
Touch, S. et al. Mucosal-associated invariant T (MAIT) cells are depleted and prone to apoptosis in cardiometabolic disorders. FASEB J. 32, 5078–5089 (2018).
Criscuolo, A. & Brisse, S. AlienTrimmer: a tool to quickly and accurately trim off multiple short contaminant sequences from high-throughput sequencing reads. Genomics 102, 500–506 (2013).
Li, J. et al. An integrated catalog of reference genes in the human gut microbiome. Nat. Biotechnol. 32, 834–841 (2014).
Cotillard, A. et al. Dietary intervention impact on gut microbial gene richness. Nature 500, 585–588 (2013).
Prifti, E. & Le Chatelier, E. MetaOMineR: a quantitative metagenomics data analyses pipeline. R package v.1.1 (2015).
Kultima, J. R. et al. MOCAT2: a metagenomic assembly, annotation and profiling framework. Bioinformatics 32, 2520–2523 (2016).
Nielsen, H. B. et al. Identification and assembly of genomes and genetic elements in complex metagenomic samples without using reference genomes. Nat. Biotechnol. 32, 822–828 (2014).
Prest, E. I., Hammes, F., Kötzsch, S., van Loosdrecht, M. C. M. & Vrouwenvelder, J. S. Monitoring microbiological changes in drinking water systems using a fast and reproducible flow cytometric method. Water Res. 47, 7131–7142 (2013).
Kultima, J. R. et al. MOCAT: a metagenomics assembly and gene prediction toolkit. PLoS ONE 7, e47656 (2012).
Falony, G., Vieira-Silva, S. & Raes, J. Microbiology meets big data: the case of gut microbiota-derived trimethylamine. Annu. Rev. Microbiol. 69, 305–321 (2015).
Darzi, Y., Falony, G., Vieira-Silva, S. & Raes, J. Towards biome-specific analysis of meta-omics data. ISME J. 10, 1025–1028 (2016).
Oksanen, J. et al. vegan: Community Ecology Package. R package v.2.2-1 (2015).
McMurdie, P. J. & Holmes, S. phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8, e61217 (2013).
Ogle, D. H. FSA: Fisheries Stock Analysis. R package v.0.8.13. (2017).
Hothorn, T., Hornik, K., van de Wiel, M. A. & Zeileis, A. A Lego system for conditional inference. Am. Stat. 60, 257–263 (2006).
Morgan, M. DirichletMultinomial: Dirichlet-multinomial mixture model machine learning for microbiome data. R package v.1.18.0 (2017).
Harrell, F. E. Hmisc: Harrell Miscellaneous. R package v.4.1-1 (2018).
Fox, J. & Weisberg, S. An R Companion to Applied Regression. (Sage, 2011).
Lüdecke, D. sjstats: Statistical Functions for Regression Models v.0.17.5 (2019).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics with S. (Springer, 2002).
Holmes, I., Harris, K. & Quince, C. Dirichlet multinomial mixtures: generative models for microbial metagenomics. PLoS ONE 7, e30126 (2012).
Duvallet, C., Gibbons, S. M., Gurry, T., Irizarry, R. A. & Alm, E. J. Meta-analysis of gut microbiome studies identifies disease-specific and shared responses. Nat. Commun. 8, 1784 (2017).
Grant, R. L. Converting an odds ratio to a range of plausible relative risks for better communication of research findings. Br. Med. J. 348, f7450 (2014).
Acknowledgements
We thank the study participants and nurses for their contributions to the project. MetaCardis was funded by European Union’s Seventh Framework Programme for research, technological development and demonstration under grant agreement HEALTH-F4-2012-305312 (MetaCardis project) and the French National Agency of Research (ANR; ‘Investissement d’Avenir’ FORCE, Metagenopolis grant ANR-11-DPBS-0001 and ICAN ANR-10-IAHU-05). The promotor of the clinical study was the Assistance Publique Hôpitaux de Paris (APHP). S.V.-S. was supported by a post-doctoral fellowship from the Research Foundation Flanders (FWO Vlaanderen). The Raes laboratory is supported by the VIB Grand Challenges programme, KU Leuven, the Rega Institute for Medical Research, and the FWO EOS program (30770923). The Novo Nordisk Foundation Center for Basic Metabolic Research is an independent research institution at the University of Copenhagen partially funded by an unrestricted donation from the Novo Nordisk Foundation. M.-E.D. was funded by the NIHR Imperial Biomedical Research Centre.
Author information
Authors and Affiliations
Consortia
Contributions
M.-E.D., S.D.E., P.G., J.P.G., T.H., J.J.H., L.K., I.L., J.N., J.-M.O., M.S., H.V., J.-D.Z., P.B., O.P., F.B., K.C. (the MetaCardis Consortium coordinator) and J.R. conceived the MetaCardis study protocol, including clinical standard operating procedures, study objectives and study design. T.N., J.A.-W. and R.C. coordinated recruitment and sample collection efforts over the different cohorts. T.N., J.A.-W., R.C. and K.A. curated and harmonized the clinical metadata. S.V.-S., G.F., E.B., T.N., J.A.-W., S.K.F., K.A., R.C., M.V.-C., S.P., E.P., V.T., N.P., E.L.C., F.A., J.-P.B., L.P.C., N.G., T.H.H., J.-S.H., C.L., H.K.P., B.Q., C.R., H.R., J.-E.S., N.B.S., S.T. and the MetaCardis Consortium assisted in sample collection, analyses, and/or data pre-processing and exploration. Faecal microbial DNA extraction and shotgun sequencing was performed by N.P., E.L.C. and S.F. Flow-cytometry-based faecal microbial load estimations were performed by T.T.D.N. Statistical analyses were designed and executed by S.V.-S., G.F., E.B., K.A., S.K.F. and M.V.-C. The manuscript was drafted by G.F., S.V.-S., K.C. and J.R. All authors revised the article and approved the final version for publication.
Corresponding authors
Ethics declarations
Competing interests
J.R., S.V.-S., G.F. and M.V.-C. are listed as inventors on patent application PCT/EP2018/084920, in the name of VIB VZW, Katholieke Universiteit Leuven, KU Leuven R&D and Vrije Universiteit Brussel, covering the features of the microbiome associated with inflammation described in ref. 2.
Additional information
Peer review information Nature thanks Peter Turnbaugh and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Microbiome variation in the BMIS cohort (n = 888 participants).
a, Percentage of subjects in the BMIS cohort taking medication of the stated drug classes. ACE inhibitors, angiotensin converting enzyme inhibitors; ARB, angiotensin II receptor blockers; ASA, acetylsalicylic acid; PPI, proton-pump inhibitors. b, Best model explaining inter-individual microbiome variation based on obesity-defining and metabolic-syndrome-defining variables as well as on most frequently disclosed therapeutics (taken by more than 10% of participants; Supplementary Table 4). Explanatory power of the included variables are reported for the variables taken individually (black bars; n = 888 biologically independent samples, univariate dbRDA)) or in a multivariate model (grey bars; n = 888 biologically independent samples, multivariate dbRDA). c, Principal coordinates analysis of inter-individual differences (genus level Bray–Curtis dissimilarity) in the microbiome profiles of the BMIS cohort (n = 888 biologically independent samples, data points coloured by enterotypes (Extended Data Fig. 4)) with the rest of the MetaCardis dataset in the background (n = 1,134, grey dots). Full and open circles corresponding to statin-medicated (Stat(+)) and non-statin-medicated participants (Stat(−)), respectively. Arrows represent the effect sizes of a post hoc fit of significant microbiome covariates identified in the multivariate model in b. d, Same principal coordinates analysis as in c, with the statin intake variable split into the separate statin classes (n = 888 biologically independent samples, simvastatin (n = 51), atorvastatin (n = 33) and other statins (n = 22); Supplementary Table 4). In c, d, the body of the box plot represents the first and third quartiles of the distribution, the line represents the median, and the whiskers extend from the quartiles to the last data point within 1.5× the interquartile range (IQR), with outliers beyond.
Extended Data Fig. 2 The association of BMI, fat mass percentage and serum fasting triglyceride levels with faecal microbial gene richness and faecal microbial load in the non-statin-medicated BMIS cohort (n = 782 participants).
All three covariates were found to be associated with both microbiome gene richness (n = 711 biologically independent samples, Spearman’s ρ = −0.45 to −0.26, Padj = 4.0 × 10−39 to 1.6 × 10−13), a proxy for microbial biodiversity previously suggested as a marker of metabolic health in obese individuals8, and faecal microbial load (n = 711 biologically independent samples, Spearman’s ρ = −0.17 to −0.13, Padj = 4.1 × 10−6 to 3.1 × 10−4; Supplementary Table 7). Adjustment for multiple testing (Padj) was performed using the Benjamini–Hochberg method. Least square linear regression lines (dashed line) with 95% confidence interval (grey shading) are provided for visual representation of the non-parametric testing provided in Supplementary Table 7. Data points are coloured by enterotype classification.
Extended Data Fig. 3 Association between the variation in quantitative butyrate production potential and the BMI, fat mass percentage and triglycerides levels of participants, or the enterotype classification of the samples, in the non-statin-medicated BMIS cohort (n = 782 participants).
Quantitative functional microbiome profiles were constructed by multiplication of relative proportions to an indexing factor proportional to the microbial load of the samples. The module ‘butyrate production II’ describes butyrate production from the butyryl-CoA–acetate CoA-transferase pathway—the most common among colon bacteria. a–d, The abundance of the butyrate production II module was negatively correlated with BMI (n = 771 biologically independent samples, Spearman’s ρ = −0.27, Padj = 3.1 × 10−13) (a), fat mass percentage (n = 771 biologically independent samples, Spearman’s ρ = −0.21, Padj = 6.0 × 10−8) (b) and tryglyceride levels (n = 771 biologically independent samples, Spearman’s ρ = −0.20, Padj = 6.4 × 10−8) (c), and significantly decreased in the Bact2 enterotype compared with the others (Bact2 < Prev < Bact1 = Rum; n = 771 biologically independent samples, Kruskal–Wallis Padj = 4.71 × 10−35; different letters denote enterotypes with a significant pairwise difference (post hoc Dunn tests provided in Supplementary Table 10) (d). The body of the box plot represents the first and third quartiles of the distribution, the line represents the median, and the whiskers extend from the quartiles to the last data point within 1.5× IQR, with outliers beyond. In a–d, adjustment for multiple testing (Padj) was performed using the Benjamini–Hochberg method.
Extended Data Fig. 4 Enterotyping of the MetaCardis dataset (n = 2,022 biologically independent samples).
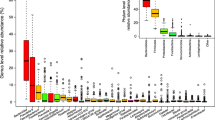
a, Principal coordinates visualization of the four enterotypes resulting from community typing was performed using DMM52 on genus-level faecal microbiome profiles. b, Information criteria (minimum Laplace) used to determine the optimal number of clusters (enterotypes) for the MetaCardis dataset (n = 2,022 biologically independent samples) DMM-based community typing. c, Average relative composition of the enterotypes for key genera, used to label the enterotypes Bacteroides1 (Bact1; high percentages of Bacteroides and Faecalibacterium), Bacteroides2 (Bact2; high percentages of Bacteroides and low percentages of Faecalibacterium), Prevotella (Prev; high percentages of Prevotella) and Ruminococcaceae (Rum; low percentages of Bacteroides and Prevotella), on the basis of their respective genus-level proportional abundance profiles.
Extended Data Fig. 5 Increased quantitative abundance of Eggerthella in the Bact2 enterotype of the non-statin-medicated BMIS cohort.
a, Difference in quantitative Eggerthella abundances between enterotypes (Prev = Rum < Bact1 < Bact2; n = 771 biologically independent samples, Kruskal–Wallis Padj = 4.10 × 10−47; different letters denote enterotypes with a significant pairwise difference (post hoc Dunn tests provided in Supplementary Table 10)). Adjustment for multiple testing (Padj) was performed using the Benjamini–Hochberg method. b, Difference in the proportion of Eggerthella (normalized by the sample total microbial load) between enterotypes, showing a comparable trend to that seen in a (n = 771 biologically independent samples). The body of the box plot represents the first and third quartiles of the distribution, the line represents the median, and the whiskers extend from the quartiles to the last data point within 1.5× IQR, with outliers beyond.
Extended Data Fig. 6 Species dominating the Bacteroides fraction in the different enterotypes of the non-statin-medicated BMIS cohort.
The top associations with the Bact2 enterotype—with the proportions they contribute to the total fraction shown in the ring chart—were the depletion in B. caccae (n = 768 biologically independent samples, Kruskal–Wallis, Padj = 1.3 × 10−15) and B. cellulosilyticus (n = 768 biologically independent samples, Kruskal–Wallis, Padj = 5.3 × 10−13) when compared with the Rum, Prev and Bact1 enterotypes, and the enrichment in B. fragilis (n = 768 biologically independent samples, Kruskal–Wallis, Padj = 3.5 × 10−11; Supplementary Table 11). Species were defined by species-level annotation of metagenomic species, and their proportional abundances were defined relative to the genus abundance. Samples for which the genus had a low total abundance (below the 20% quantile for all species belonging to the top 10 genera) were excluded from the analysis (n = 768 biologically independent samples were included). Adjustment for multiple testing (Padj) was performed using the Benjamini–Hochberg method.
Extended Data Fig. 7 Systemic inflammation and its relation to enterotypes and to BMI in the non-statin-medicated BMIS cohort.
a, Individuals with faecal samples enterotyped as Bact2 displayed more pronounced systemic inflammation levels as assessed through fasting serum hsCRP concentrations when compared with participants classified as Rum, Prev and Bact1 (n = 763 biologically independent samples, Kruskal–Wallis P = 1.37 × 10−10; Rum = Bact1 < Prev < Bact2; different letters denote enterotypes with a significant pairwise difference (post hoc Dunn tests provided in Supplementary Table 13)). The body of the box plot represents the first and third quartiles of the distribution, the line represents the median, and the whiskers extend from the quartiles to the last data point within 1.5× IQR, with outliers beyond. b, Linear model of the correlation between host systemic inflammation (hsCRP concentration, log10-transformed) and BMI, fitted by least squares regression (n = 763 biologically independent samples; estimated intercept = −0.8681, estimated slope = 0.0379, R2 = 0.47, P = 1.5 × 10−108).
Extended Data Fig. 8 Control for the effect of additional medication taken by obese statin-medicated or non-statin-medicated individuals of the BMIS cohort (n = 888 participants) on the association between reduced Bact2 prevalence and statin intake.
a, List of drugs taken by non-statin-medicated and statin-medicated obese BMIS participants separated into 5 groups: those reporting no (co-)medication (beyond statin intake) (+0), and those reporting one (+1), two (+2), three (+3) and more than three (more) (co-)medications. The size and colour of the dots represent the fraction of the non-statin-medicated or statin-medicated obese BMIS participants falling within that group. b, Difference in prevalence of the Bact2 enterotype in statin-medicated compared with non-statin-medicated obese BMIS participants, with decreasing co-medication threshold for inclusion of participants. For ‘all’, the total number of statin-medicated and non-statin-medicated obese BMIS participants were included (n = 474 biologically independent samples); then only subjects reporting three or fewer (≤3; n = 419), two or fewer (≤2; n = 369), one or fewer (≤1; n = 296) or no (0; n = 226) (co-)medications were included. The relative risk and respective significance level associated with the prevalence of the Bact2 enterotype given statin intake is provided above the bar plots (Fisher’s exact test, two-sided, *P < 0.05, relative risk = P(Bact2|Statin = Yes)/P(Bact2|Statin = No)).
Extended Data Fig. 9 Variation in prevalence of the Bact2 enterotype with BMI and statin intake in the BMIS discovery cohort, and in the FGFP and CVD validation cohorts.
a–c, Variation in the prevalence of the Bact2 enterotype with BMI for statin-medicated and non-statin-medicated individuals, showing the significant effect (represented by the range bar with an asterisk; Supplementary Table 16) of statin intake given individuals’ BMI, in the BMIS obese participants (n = 474 biologically independent samples, multivariate binomial logistic regression, Statin | BMI, relative risk = 0.34, *Padj = 0.025) (a); the FGFP cohort, a population-level recruitment with a much narrower BMI range than the BMIS cohort (n = 2,345 biologically independent samples, multivariate binomial logistic regression, Statin | BMI, relative risk = 0.72, *Padj = 0.045) (b) and the MetaCardis CVD cohort (n = 271 biologically independent samples, excluding 11 individuals for which BMI was not known, multivariate binomial logistic regression, Statin | BMI, relative risk = 0.29, *Padj = 0.021) (c). In a–c, the fit lines were obtained by multinomial logistic regression of enterotypes as predicted by BMI, for statin-medicated and non-statin-medicated individuals separately, with the shaded area corresponding to the 95% confidence intervals for the Bact2 regression. Adjustment for multiple testing (Padj) was performed using the Benjamini–Hochberg method.
Extended Data Fig. 10 Probability of carrying a Bact2 enterotype microbiota as a function of CRP levels and statin intake in the obese BMIS cohort.
Association between systemic inflammation (measured by hsCRP levels) and having a faecal microbiota of the Bact2 enterotype, according to statin medication status. Binomial logistic regression (lines with 95% confidence intervals as shaded area) was performed for statin-medicated and non-statin-medicated individuals separately (n = 462 biologically independent samples).
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-2. STROBE flow chart of MetaCardis participant recruitment and illustration of flow cytometry gating strategy.
Supplementary Tables
This file contains Supplementary Tables 1-19.
Rights and permissions
About this article
Cite this article
Vieira-Silva, S., Falony, G., Belda, E. et al. Statin therapy is associated with lower prevalence of gut microbiota dysbiosis. Nature 581, 310–315 (2020). https://doi.org/10.1038/s41586-020-2269-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2269-x
This article is cited by
-
Cluster-specific associations between the gut microbiota and behavioral outcomes in preschool-aged children
Microbiome (2024)
-
Oral-gut microbial transmission promotes diabetic coronary heart disease
Cardiovascular Diabetology (2024)
-
High-throughput transcriptomics of 409 bacteria–drug pairs reveals drivers of gut microbiota perturbation
Nature Microbiology (2024)
-
Targeting the Gut Microbiome to Treat Cardiometabolic Disease
Current Atherosclerosis Reports (2024)
-
Four functional profiles for fibre and mucin metabolism in the human gut microbiome
Microbiome (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.