Abstract
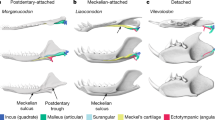
The evolution of the mammalian middle ear is thought to provide an example of ‘recapitulation’—the theory that the present embryological development of a species reflects its evolutionary history. Accumulating data from both developmental biology and palaeontology have suggested that the transformation of post-dentary jaw elements into cranial ear bones occurred several times in mammals1,2. In addition, well-preserved fossils have revealed transitional stages in the evolution of the mammalian middle ear1,3,4. But questions remain concerning middle-ear evolution, such as how and why the post-dentary unit became completely detached from the dentary bone in different clades of mammaliaforms. Here we report a definitive mammalian middle ear preserved in an eobaatarid multituberculate mammal, with complete post-dentary elements that are well-preserved and detached from the dentary bones. The specimen reveals the transformation of the surangular jaw bone from an independent element into part of the malleus of the middle ear, and the presence of a restricted contact between the columelliform stapes and the flat incus. We propose that the malleus–incus joint is dichotomic in mammaliaforms, with the two bones connecting in either an abutting or an interlocking arrangement, reflecting the evolutionary divergence of the dentary–squamosal joint4. In our phylogenetic analysis, acquisition of the definitive mammalian middle ear in allotherians such as this specimen was independent of that in monotremes and therians. Our findings suggest that the co-evolution of the primary and secondary jaw joints in allotherians was an evolutionary adaptation allowing feeding with unique palinal (longitudinal and backwards) chewing. Thus, the evolution of the allotherian auditory apparatus was probably triggered by the functional requirements of the feeding apparatus.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The specimen (IVPP V20778) reported here is housed in the Institute of Vertebrate Paleontology and Paleoanthropology, Beijing, China. Character matrices are given in the Supplementary Information.
References
Luo, Z. X. Developmental patterns in Mesozoic evolution of mammal ears. Annu. Rev. Ecol. Evol. Syst. 42, 355–380 (2011).
Maier, W. & Ruf, I. Evolution of the mammalian middle ear: a historical review. J. Anat. 228, 270–283 (2016).
Meng, J., Wang, Y. & Li, C. Transitional mammalian middle ear from a new Cretaceous Jehol eutriconodont. Nature 472, 181–185 (2011).
Han, G., Mao, F., Bi, S., Wang, Y. & Meng, J. A Jurassic gliding euharamiyidan mammal with an ear of five auditory bones. Nature 551, 451–456 (2017).
He, H. Y. et al. Timing of the Jiufotang Formation (Jehol Group) in Liaoning, northeastern China, and its implications. Geophys. Res. Lett. 31, L12605 (2004).
Millien, V. & Bovy, H. When teeth and bones disagree: body mass estimation of a giant extinct rodent. J. Mamm. 91, 11–18 (2010).
Wilson, G. P. et al. Adaptive radiation of multituberculate mammals before the extinction of dinosaurs. Nature 483, 457–460 (2012).
Allin, E. F. & Hopson, J. in The Evolutionary Biology of Hearing Ch. 28 (eds Webster, D. B. et al.) 587–614 (Springer, 1992).
Meng, J. The stapes of Lambdopsalis bulla (Multituberculata) and transformational analyses on some stapedial features in Mammaliaformes. J. Vertebr. Paleontol. 12, 459–471 (1992).
Meng, J. & Hou, S. L. Earliest known mammalian stapes from an Early Cretaceous eutriconodontan mammal and implications for the evolution of mammalian middle ear. Palaeontol. Pol. 67, 181–196 (2016).
Rougier, G. W., Wible, J. R. & Novacek, M. J. Middle-ear ossicles of the multituberculate Kryptobaatar from the Mongolian Late Cretaceous: implications for mammaliamorph relationships and the evolution of the auditory apparatus. Am. Mus. Novit. 3187, 1–43 (1996).
Fleischer, G. Evolutionary principles of the mammalian middle ear. Adv. Anat. Embryol. Cell Biol. 55, 3–70 (1978).
Bi, S. et al. An Early Cretaceous eutherian and the placental–marsupial dichotomy. Nature 558, 390–395 (2018).
Schultz, J. A., Ruf, I. & Martin, T. Oldest known multituberculate stapes suggests an asymmetric bicrural pattern as ancestral for Multituberculata. Proc. R. Soc. B 285, 20172779 (2018).
Tucker, A. S., Watson, R. P., Lettice, L. A., Yamada, G. & Hill, R. E. Bapx1 regulates patterning in the middle ear: altered regulatory role in the transition from the proximal jaw during vertebrate evolution. Development 131, 1235–1245 (2004).
Zeller, U. in Mammal Phylogeny: Mesozoic Differentiation, Multituberculates, Monotremes, Early Therians, and Marsupials Ch. 8 (eds Szalay, F. S. et al.) 95–107 (Springer, 1993).
Kermack, K. A., Mussett, F. & Rigney, H. W. The skull of Morganucodon. Zool. J. Linn. Soc. 71, 1–158 (1981).
Luo, Z. X. et al. New evidence for mammaliaform ear evolution and feeding adaptation in a Jurassic ecosystem. Nature 548, 326–329 (2017).
Anthwal, N., Urban, D. J., Luo, Z.-X., Sears, K. E. & Tucker, A. S. Meckel’s cartilage breakdown offers clues to mammalian middle ear evolution. Nature Ecol. Evol. 1, 0093 (2017).
Urban, D. J. et al. A new developmental mechanism for the separation of the mammalian middle ear ossicles from the jaw. Proc. R. Soc. B. 284, 20162416 (2017).
Lautenschlager, S., Gill, P. G., Luo, Z.-X., Fagan, M. J. & Rayfield, E. J. The role of miniaturization in the evolution of the mammalian jaw and middle ear. Nature 561, 533–537 (2018).
Rowe, T. Coevolution of the mammalian middle ear and neocortex. Science 273, 651–654 (1996).
Zeller, U. in Morphogenesis of the Mammalian Skull (eds Kuhn, H.-J. & Zeller, U.) 17–50 (Paul Parey, 1987).
Meng, J., Hu, Y. M., Wang, Y. Q. & Li, C. K. The ossified Meckel’s cartilage and internal groove in Mesozoic mammaliaforms: implications to origin of the definitive mammalian middle ear. Zool. J. Linn. Soc. 138, 431–448 (2003).
Wang, Y., Hu, Y., Meng, J. & Li, C. An ossified Meckel’s cartilage in two Cretaceous mammals and origin of the mammalian middle ear. Science 294, 357–361 (2001).
Ramírez-Chaves, H. E. et al. Mammalian development does not recapitulate suspected key transformations in the evolutionary detachment of the mammalian middle ear. Proc. R. Soc. Lond. B 283, 20152606 (2016).
Crompton, A. W. & Parker, P. Evolution of the mammalian masticatory apparatus. Am. Sci. 66, 192–201 (1978).
Allin, E. F. Evolution of the mammalian middle ear. J. Morphol. 147, 403–437 (1975).
Crompton, A. W. & Hylander, W. L. in The Ecology and Biology of Mammal-like Reptiles (eds Hotton, N. et al.) 78–98 (Smithsonian Institute Press, 1986).
Abzhanov, A. von Baer’s law for the ages: lost and found principles of developmental evolution. Trends Genet. 29, 712–722 (2013).
Luo, Z. X., Crompton, A. W. & Sun, A. L. A new mammaliaform from the early Jurassic and evolution of mammalian characteristics. Science 292, 1535–1540 (2001).
Luo, Z. X., Ji, Q. & Yuan, C. X. Convergent dental adaptations in pseudo-tribosphenic and tribosphenic mammals. Nature 450, 93–97 (2007).
Rich, T. H. et al. The mandible and dentition of the Early Cretaceous monotreme Teinolophos trusleri. Alcheringa 40, 475–501 (2016).
Luo, Z. X., Gatesy, S. M., Jenkins, F. A. Jr, Amaral, W. W. & Shubin, N. H. Mandibular and dental characteristics of Late Triassic mammaliaform Haramiyavia and their ramifications for basal mammal evolution. Proc. Natl Acad. Sci. USA 112, E7101–E7109 (2015).
Hu, Y., Wang, Y., Luo, Z. & Li, C. A new symmetrodont mammal from China and its implications for mammalian evolution. Nature 390, 137–142 (1997).
Lillegraven, J. A. & Krusat, G. Cranio-mandibular anatomy of Haldanodon exspectatus (Docodonta; Mammalia) from the Late Jurassic of Portugal and its implications to the evolution of mammalian characters. Rocky Mountain Geol. 28, 39–138 (1991).
Rowe, T. Definition, diagnosis and origin of Mammalia. J. Vertebr. Paleontol. 8, 241–264 (1988).
Bi, S., Wang, Y., Guan, J., Sheng, X. & Meng, J. Three new Jurassic euharamiyidan species reinforce early divergence of mammals. Nature 514, 579–584 (2014).
Krause, D. W. et al. First cranial remains of a gondwanatherian mammal reveal remarkable mosaicism. Nature 515, 512–517 (2014).
Csiki-Sava, Z., Vremir, M., Meng, J., Brusatte, S. L. & Norell, M. A. Dome-headed, small-brained island mammal from the Late Cretaceous of Romania. Proc. Natl. Acad. Sci. USA 115, 4857–4862 (2018).
Ronquist, F. et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Goloboff, P. A., Farris, J. S. & Nixon, K. C. TNT, a free program for phylogenetic analysis. Cladistics 24, 774–786 (2008).
Yuan, C. X., Ji, Q., Meng, Q. J., Tabrum, A. R. & Luo, Z. X. Earliest evolution of multituberculate mammals revealed by a new Jurassic fossil. Science 341, 779–783 (2013).
Xu, L. et al. Largest known Mesozoic multituberculate from Eurasia and implications for multituberculate evolution and biology. Sci. Rep. 5, 14950 (2015).
Acknowledgements
We thank S.-H. Xie for specimen preparation; Y.-M. Hou and P.-F. Yin for help with computed laminography scans and virtual reconstructions; X. Jin and X.-C. Guo for help with photographing and drawing; and T. Martin and J. A. Schultz for access to Guimarota specimens in the University of Bonn. We benefited from discussions with D. W. Krause, Z. X. Luo, T. Martin, J. A. Schultz, N. Kusuhashi and J. K. O’Connor. Financial support was from the Strategic Priority Research Program of the Chinese Academy of Sciences (grants XDB18000000 and XDB26000000), the National Natural Science Foundation of China (grants 41802005 and 41688103), and the State Key Laboratory of Palaeobiology and Stratigraphy (Nanjing Institute of Geology and Palaeontology, CAS; grant 183121).
Author information
Authors and Affiliations
Contributions
Y.W. and H.W. designed the study. H.W. organized computed tomography scans and virtual reconstructions, performed phylogenetic analyses, and prepared the main text, figures and Supplementary Information. All authors contributed to revising the manuscript and figures. Y.W. supervised all research activities.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks Simone Hoffmann and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Fig. 1 Cranio-mandibular morphology of J. kielanae (holotype IVPP V20778).
a, Skull in dorsal view and right mandible in lateral view. b, Skull in ventral view, left mandible in lateral view, and right mandible in medial view. c, Close-up view of cranio-mandibular features. d, Close-up medial view of the right dentary. The flat glenoid fossa accommodates the mandibular condyle, which is positioned below the occlusal level of the lower molars and faces posteriorly in IVPP V20778. Together with the distinct masseteric fossa—which presumably provides attachment for a well developed masseteric muscle, inserting anteriorly below P4—the glenoid fossa produces a palinal (posterior) power stroke with distinct posterior chewing.
Extended Data Fig. 2 Dentition of J. kielanae (IVPP V20778).
a–c, Left upper cheek teeth (P1 to M2) in lingual (a), occlusal (b) and buccal (c) views. d, e, Right I1 in medial view (d) and lateral view (e). f, g, Right I2 in medial (f) and lateral (g) views. h–j, Right I3 in lingual (h), buccal (i) and posterior (j) views. k, Right upper premolars (P1 to P5) in occlusal view. l, Right upper incisors (I1 to I3) in medial view. m, Right lower molars (M1 and M2) in occlusal view. n, p, Right lower teeth (I1, P2 to M2) in lingual (n) and buccal (p) views. A, anterior; B, buccal; L, lingual; O, occlusal; P, posterior. Scale bars, 0.2 mm.
Extended Data Fig. 3 Computed laminography images and reconstructions of left middle-ear bones.
a–d, Computed laminography images on different levels. The path of the chorda tympani is marked with a yellow arrow in b. The stapedial foramen, identified by computed laminography, is shown in c, d. e, Three-dimensional reconstruction of left middle-ear bones in dorsal view. f, X-ray rendering of left middle ear, showing the differing thicknesses of different parts of the bones.
Extended Data Fig. 4 Articular configurations of the malleus–incus complex.
a, Left auditory bones of Ornithorhynchus in dorsal view (modified from ref. 16). b, Interpretive reconstruction of left auditory bones of Arboroharamiya in dorsal view (modified from ref. 4). c, Interpretive reconstruction of left auditory bones of Jeholbaatar in dorsal view. The yellow arrows in a–c show that the incus lies dorsal to the malleus in Ornithorhynchus, Arboroharamiya and Jeholbaatar, demonstrating the ‘abutting system’ (AS) arrangement of the malleus–incus complex. d, Left auditory bones of Didelphis in medial view (modified from ref. 28), showing that the malleus–incus complex maintains the interlocking system (IS) arrangement (yellow arrow), with a rostrocaudal contact between these two elements. e, Left auditory bones of Liaoconodon in medial view (modified from ref. 3). f, Left auditory bones of Morganucodon in medial view (modified from ref. 28). Here the incus (quadrate) has a medial trochlear facet to contact the concave surface of the malleus body (articular fossa) posteriorly.
Extended Data Fig. 5 Strict consensus of parsimony analysis based on data matrix A.
Tree length, 2,622; consistency index, 0.327; retention index, 0.795. On the basis of analysis using TNT 3.0, 14 most parsimonious trees are returned; tree length, 2,539, consistency index, 0.338; retention index, 0.804. The blue shading shows the monophyly of allotherians within crown mammals. Node supports are given as Bremer support values.
Extended Data Fig. 6 Results of Bayesian analysis of multituberculates.
This 50% majority-rule consensus was obtained from 10 million Markov Chain Monte Carlo generations with a 25% burn-in fraction. Node supports are listed as posterior probabilities (percentages). The blue rectangle shows the monophyly of eobaatarids, with Jeholbaatar closely related to Sinobaatar.
Extended Data Fig. 7 Manual and pedal structure, and ternary diagrams showing the proportions of phalanges from manual and pedal digit III.
a, b, Shoulder (a) and pelvic (b) girdles in dorsal view. c, d, Right manus (c) and pes (d) in lateral view. e, f, Ternary plots showing ratios of metapodial (metacarpal or metatarsal), proximal and intermediate phalanges for Jeholbaatar digit III from the manus (e) and pes (f), and comparison with some extant terrestrial and arboreal mammals. The lengths of these three phalanges are shown as ratios of the combined length of these elements. Mc, metacarpal; Mt, metatarsal. The lengths of Jeholbaatar manus and pes elements (in mm, with asterisks indicating damaged elements) are: Mc I, 2.76; Mc II, *2.84; Mc III, *3.70; Mc IV, *2.81; Mc V, 2.79; digit I proximal phalanx, 1.98; digit II proximal phalanx, 2.84; digit II intermediate phalanx, *1.60; digit III proximal phalanx, 2.40; digit III intermediate phalanx, 2.26; digit IV proximal phalanx, *2.22; digit IV intermediate phalanx, 1.83; digit V proximal phalanx, 1.92; digit V intermediate phalanx, 1.54; phalange index, that is, (proximal plus intermediate)/metacarpal, digit III, 126%; Mt I, 3.92; Mt II, 4.99; Mt III, 5.42; Mt IV, *1.69; Mt V, *3.33; digit I proximal phalanx, 3.51; digit II proximal phalanx, 3.58; digit II intermediate phalanx, 2.82; digit III proximal phalanx, 3.59; digit III intermediate phalanx, 3.46; digit IV proximal phalanx, *1.73; digit IV intermediate phalanx, 3.25; digit V intermediate phalanx, 2.63; phalanx index, that is, (proximal+intermediate phalanges)/metatarsal, digit III, 130%. The manual proportion of J. kielanae places it closer (than the other multituberculates in the sample) to the arboreal category; the pedal proportion clusters mostly with arboreal taxa. The data for extant taxa are derived from ref. 38.
Supplementary information
Supplementary Information
This file contains Supplementary Information, including (A) Methods; (B) Systematic Discussion; (C) Body Mass Estimation; (D) Estimation of Hearing Capability; (E) Phylogenetic Analysis; and (F) References.
Rights and permissions
About this article
Cite this article
Wang, H., Meng, J. & Wang, Y. Cretaceous fossil reveals a new pattern in mammalian middle ear evolution. Nature 576, 102–105 (2019). https://doi.org/10.1038/s41586-019-1792-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1792-0
This article is cited by
-
Middle ear innovation in Early Cretaceous eutherian mammals
Nature Communications (2023)
-
A new multituberculate from the latest Cretaceous of central China and its implications for multituberculate tooth homologies and occlusion
Journal of Mammalian Evolution (2023)
-
A New Mammal Skull from the Late Cretaceous of Romania and Phylogenetic Affinities of Kogaionid Multituberculates
Journal of Mammalian Evolution (2022)
-
A monotreme-like auditory apparatus in a Middle Jurassic haramiyidan
Nature (2021)
-
Early mammalian social behaviour revealed by multituberculates from a dinosaur nesting site
Nature Ecology & Evolution (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.