Abstract
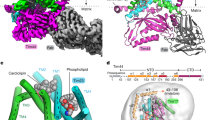
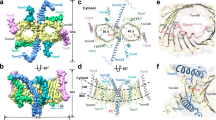
The translocase of the outer mitochondrial membrane (TOM) is the main entry gate for proteins1,2,3,4. Here we use cryo-electron microscopy to report the structure of the yeast TOM core complex5,6,7,8,9 at 3.8-Å resolution. The structure reveals the high-resolution architecture of the translocator consisting of two Tom40 β-barrel channels and α-helical transmembrane subunits, providing insight into critical features that are conserved in all eukaryotes1,2,3. Each Tom40 β-barrel is surrounded by small TOM subunits, and tethered by two Tom22 subunits and one phospholipid. The N-terminal extension of Tom40 forms a helix inside the channel; mutational analysis reveals its dual role in early and late steps in the biogenesis of intermembrane-space proteins in cooperation with Tom5. Each Tom40 channel possesses two precursor exit sites. Tom22, Tom40 and Tom7 guide presequence-containing preproteins to the exit in the middle of the dimer, whereas Tom5 and the Tom40 N extension guide preproteins lacking a presequence to the exit at the periphery of the dimer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The cryo-EM density maps have been deposited in the Electron Microscopy Data Bank under accession code EMD-9851. The atomic coordinates have been deposited in the Protein Data Bank under accession code 6JNF. All the data from this study are available in the Article, its Extended Data and Supplementary Information. The uncropped blots, gels and plates are provided in Supplementary Fig. 2.
References
Endo, T. & Yamano, K. Multiple pathways for mitochondrial protein traffic. Biol. Chem. 390, 723–730 (2009).
Neupert, W. A perspective on transport of proteins into mitochondria: a myriad of open questions. J. Mol. Biol. 427, 1135–1158 (2015).
Pfanner, N., Warscheid, B. & Wiedemann, N. Mitochondrial proteins: from biogenesis to functional networks. Nat. Rev. Mol. Cell Biol. 20, 267–284 (2019).
Shiota, T. et al. Molecular architecture of the active mitochondrial protein gate. Science 349, 1544–1548 (2015).
Sakaue, H. et al. Porin associates with Tom22 to regulate the mitochondrial protein gate assembly. Mol. Cell 73, 1044–1055 (2019).
Künkele, K.-P. et al. The preprotein translocation channel of the outer membrane of mitochondria. Cell 93, 1009–1019 (1998).
Ahting, U. et al. The TOM core complex: the general protein import pore of the outer membrane of mitochondria. J. Cell Biol. 147, 959–968 (1999).
Model, K., Meisinger, C. & Kühlbrandt, W. Cryo-electron microscopy structure of a yeast mitochondrial preprotein translocase. J. Mol. Biol. 383, 1049–1057 (2008).
Bausewein, T. et al. Cryo-EM structure of the TOM core complex from Neurospora crassa. Cell 170, 693–700 (2017).
Van den Berg, B. et al. X-ray structure of a protein-conducting channel. Nature 427, 36–44 (2004).
Voorhees, R. M., Fernández, I. S., Scheres, S. H. W. & Hegde, R. S. Structure of the mammalian ribosome–Sec61 complex to 3.4 Å resolution. Cell 157, 1632–1643 (2014).
Gogala, M. et al. Structures of the Sec61 complex engaged in nascent peptide translocation or membrane insertion. Nature 506, 107–110 (2014).
Itskanov, S. & Park, E. Structure of the posttranslational Sec protein–translocation channel complex from yeast. Science 363, 84–87 (2019).
Wu, X., Cabanos, C. & Rapoport, T. A. Structure of the post-translational protein translocation machinery of the ER membrane. Nature 566, 136–139 (2019).
Baud, C. et al. Translocation path of a substrate protein through its Omp85 transporter. Nat. Commun. 5, 5271 (2014).
Lundquist, K., Bakelar, J., Noinaj, N. & Gumbart, J. C. C-terminal kink formation is required for lateral gating in BamA. Proc. Natl Acad. Sci. USA 115, E7942–E7949 (2018).
Bamert, R. S. et al. Structural basis for substrate selection by the translocation and assembly module of the β-barrel assembly machinery. Mol. Microbiol. 106, 142–156 (2017).
Model, K. et al. Protein translocase of the outer mitochondrial membrane: role of import receptors in the structural organization of the TOM complex. J. Mol. Biol. 316, 657–666 (2002).
Dolezal, P., Likic, V., Tachezy, J. & Lithgow, T. Evolution of the molecular machines for protein import into mitochondria. Science 313, 314–318 (2006).
Fukasawa, Y., Oda, T., Tomii, K. & Imai, K. Origin and evolutionary alteration of the mitochondrial import system in eukaryotic lineages. Mol. Biol. Evol. 34, 1574–1586 (2017).
Maćasev, D. et al. Tom22′, an 8-kDa trans-site receptor in plants and protozoans, is a conserved feature of the TOM complex that appeared early in the evolution of eukaryotes. Mol. Biol. Evol. 21, 1557–1564 (2004).
Senes, A., Gerstein, M. & Engelman, D. M. Statistical analysis of amino acid patterns in transmembrane helices: the GxxxG motif occurs frequently and in association with β-branched residues at neighboring positions. J. Mol. Biol. 296, 921–936 (2000).
Harner, M., Neupert, W. & Deponte, M. Lateral release of proteins from the TOM complex into the outer membrane of mitochondria. EMBO J. 30, 3232–3241 (2011).
Höhr, A. I. C. et al. Membrane protein insertion through a mitochondrial β-barrel gate. Science 359, eaah6834 (2018).
Yamano, K., Tanaka-Yamano, S. & Endo, T. Tom7 regulates Mdm10-mediated assembly of the mitochondrial import channel protein Tom40. J. Biol. Chem. 285, 41222–41231 (2010).
Ellenrieder, L. et al. Separating mitochondrial protein assembly and endoplasmic reticulum tethering by selective coupling of Mdm10. Nat. Commun. 7, 13021 (2016).
Esaki, M. et al. Mitochondrial protein import. Requirement of presequence elements and TOM components for precursor binding to the TOM complex. J. Biol. Chem. 279, 45701–45707 (2004).
Court, D. A. et al. Role of the intermembrane-space domain of the preprotein receptor Tom22 in protein import into mitochondria. Mol. Cell. Biol. 16, 4035–4042 (1996).
Moczko, M. et al. The intermembrane space domain of mitochondrial Tom22 functions as a trans binding site for preproteins with N-terminal targeting sequences. Mol. Cell. Biol. 17, 6574–6584 (1997).
Kanamori, T. et al. Uncoupling of transfer of the presequence and unfolding of the mature domain in precursor translocation across the mitochondrial outer membrane. Proc. Natl Acad. Sci. USA 96, 3634–3639 (1999).
Rapaport, D., Neupert, W. & Lill, R. Mitochondrial protein import. Tom40 plays a major role in targeting and translocation of preproteins by forming a specific binding site for the presequence. J. Biol. Chem. 272, 18725–18731 (1997).
Yamamoto, H. et al. Tim50 is a subunit of the TIM23 complex that links protein translocation across the outer and inner mitochondrial membranes. Cell 111, 519–528 (2002).
Mokranjac, D. et al. Tim50, a novel component of the TIM23 preprotein translocase of mitochondria. EMBO J. 22, 816–825 (2003).
Shiota, T., Mabuchi, H., Tanaka-Yamano, S., Yamano, K. & Endo, T. In vivo protein-interaction mapping of a mitochondrial translocator protein Tom22 at work. Proc. Natl Acad. Sci. USA 108, 15179–15183 (2011).
Tamura, Y. et al. Tim23–Tim50 pair coordinates functions of translocators and motor proteins in mitochondrial protein import. J. Cell Biol. 184, 129–141 (2009).
Chacinska, A. et al. Mitochondrial presequence translocase: switching between TOM tethering and motor recruitment involves Tim21 and Tim17. Cell 120, 817–829 (2005).
Naoé, M. et al. Identification of Tim40 that mediates protein sorting to the mitochondrial intermembrane space. J. Biol. Chem. 279, 47815–47821 (2004).
Gornicka, A. et al. A discrete pathway for the transfer of intermembrane space proteins across the outer membrane of mitochondria. Mol. Biol. Cell 25, 3999–4009 (2014).
Chacinska, A. et al. Essential role of Mia40 in import and assembly of mitochondrial intermembrane space proteins. EMBO J. 23, 3735–3746 (2004).
Mesecke, N. et al. A disulfide relay system in the intermembrane space of mitochondria that mediates protein import. Cell 121, 1059–1069 (2005).
Kurz, M. Martin, H. Rassow, J., Pfanner, N. & Ryan, M. T. Biogenesis of Tim proteins of the mitochondrial carrier pathway: differential targeting mechanisms and crossing over with the main import pathway. Mol. Biol. Cell 10, 2461–2474 (1999).
Stojanovski, D. et al. Mitochondrial protein import: precursor oxidation in a ternary complex with disulfide carrier and sulfhydryl oxidase. J. Cell Biol. 183, 195–202 (2008).
Bien, M. et al. Mitochondrial disulfide bond formation is driven by intersubunit electron transfer in Erv1 and proofread by glutathione. Mol. Cell 37, 516–528 (2010).
Rapaport, D. et al. Structural requirements of Tom40 for assembly into preexisting TOM complexes of mitochondria. Mol. Biol. Cell 12, 1189–1198 (2001).
Habich, M. et al. Vectorial import via a metastable disulfide-linked complex allows for a quality control step and import by the mitochondrial disulfide relay. Cell Rep. 26, 759–774 (2019).
Becker, T. et al. Role of phosphatidylethanolamine in the biogenesis of mitochondrial outer membrane proteins. J. Biol. Chem. 288, 16451–16459 (2013).
Zheng, W. et al. Adaptive optics improves multiphoton super-resolution imaging. Nat. Methods 14, 869–872 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: Fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife 7, e42166 (2018).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Sikorski, R. S. & Hieter, P. A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 122, 19–27 (1989).
Model, K. et al. Multistep assembly of the protein import channel of the mitochondrial outer membrane. Nat. Struct. Biol. 8, 361–370 (2001).
Opaliński, Ł. et al. Recruitment of cytosolic J-proteins by TOM receptors promotes mitochondrial protein biogenesis. Cell Rep. 25, 2036–2043 (2018).
Qiu, J. et al. Coupling of mitochondrial import and export translocases by receptor-mediated supercomplex formation. Cell 154, 596–608 (2013).
Ando, T., Uchihashi, T. & Fukuma, T. High-speed atomic force microscopy for nano-visualization of dynamic biomolecular processes. Prog. Surf. Sci. 83, 337–437 (2008).
Uchihashi, T., Kodera, N. & Ando, T. Guide to video recording of structure dynamics and dynamic processes of proteins by high-speed atomic force microscopy. Nat. Protoc. 7, 1193–1206 (2012).
Milenkovic, D. et al. Identification of the signal directing Tim9 and Tim10 into the intermembrane space of mitochondria. Mol. Biol. Cell 20, 2530–2539 (2009).
Thornton, N. et al. Two modular forms of the mitochondrial sorting and assembly machinery are involved in biogenesis of α-helical outer membrane proteins. J. Mol. Biol. 396, 540–549 (2010).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Waterhouse, A. M., Procter, J. B., Martin, D. M. A., Clamp, M. & Barton, G. J. Jalview Version 2—a multiple sequence alignment editor and analysis workbench. Bioinformatics 25, 1189–1191 (2009).
Ashkenazy, H. et al. ConSurf 2016: an improved methodology to estimate and visualize evolutionary conservation in macromolecules. Nucleic Acids Res. 44, W344–W350 (2016).
Acknowledgements
We thank the members of the Endo and Pfanner labs for discussion and critical comments on the manuscript. This work was supported by JSPS KAKENHI to T.E. (15H05705 and 2222703), K.I. (16K21680 and 18K11543), T.S. (19K16077 and 18KK0197) and T.A. (26119003) and JST CREST grants to T.E. (JPMJCR12M1) and to M.K. (JPMJCR14M1); a European Research Council (ERC) Consolidator grant to N.W. (no. 648235); Deutsche Forschungsgemeinschaft grants to T.B. (BE 4679/2-2) and N.P. (PF 202/9-1); and Germany’s Excellence Strategy to T.B., N.W. and N.P. (CIBSS – EXC-2189—Project ID 390939984; GSC-4 Spemann Graduate School). The following grants are also acknowledged: a grant from Takeda Science Foundation (to T.E.); Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)) from AMED under grant numbers JP19am01011115 (to M.K.) and JP19am0101114 (to K.I.); grants from the Ichiro Kanehara Foundation for the Promotion of Medical Sciences and Medical Care, Waksman Foundation of Japan, Tokyo Biochemical Research Foundation, Sumitomo Foundation, Naito Foundation, and Uehara Memorial Foundation (to T.S.); and the Australian Research Council Discovery Project DP160100227 (to T.L.). We acknowledge the Cryo-EM Facility of the University of Tokyo. Y.A. was supported by a Research Fellowship for Young Scientists from the Japan Society of the Promotion of Science (15J07687). Work included in this study has also been performed in partial fulfillment of the requirements for the doctoral thesis of C.L. at the University of Freiburg. We thank T. Nishizawa for discussion and advice on cryo-EM data processing, M. H. Schuler for analysis of yeast mutants, R. Beckmann and A. Heuer for advice on cryo-EM measurements and H. Nishitoh and Y. Inaba for instruments and reagents for T.S.
Author information
Authors and Affiliations
Contributions
Y.A. performed most of the experiments and wrote the paper. A.T. performed and M.K. supervised cryo-EM measurements and data processing. J.Q., J. Song, C.L., L.-S.W., M.W. and C.S. performed functional studies on the role of TOM proteins in preprotein import into mitochondria, characterized yeast mutants, studied the import of precursor proteins into isolated mitochondria and analysed data. K.I. performed bioinformatics analyses and wrote a part of the paper. T.S. performed crosslinking and T.L. designed and supervised crosslinking and wrote a part of the paper. H.S. analysed yeast cell growth of double mutants of tom5Δ and Tom40 N-terminal truncation. K.Y., S.K. and J. Suzuki assisted with designing preparation of the materials including the yeast TOM complex. H.A. and T.A. performed HS-AFM measurements. N.W., T.B. and N.P. designed and supervised functional studies on the role of TOM proteins in preprotein import into mitochondria, analysed data and wrote a part of the paper. T.E. designed the research and wrote the paper. All authors discussed the results of the experiments and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Purification of the TOM complex.
a, Forms of the TOM complex. Newly imported Tom40 takes a β-barrel structure and forms an assembly ‘dimer’ lacking Tom22 and Tom20. The dimer is matured into the ‘trimer’ with the receptors Tom22 and Tom204. Even after the maturation, the trimer dynamically exchanges with a minor population of the assembly dimer4. After solubilization of mitochondria, the TOM complex can be found as a trimer, containing Tom20 at least partly. Our previous in vivo crosslinking analyses revealed the subunit arrangement of the trimer4,32, and the present study revealed the high-resolution cryo-EM structure of the dimeric TOM core complex that contains Tom22, but lacks Tom20. b, Elution profile of gel filtration of the purified TOM complex. c, Blue native electrophoresis gel of the TOM complex stained with Coomassie brilliant blue. The apparent molecular weight of the TOM complex is ~440 kDa on blue native electrophoresis, which corresponds to the trimer. d, SDS–PAGE gel of the TOM complex stained with Coomassie brilliant blue. The bands for Tom40, Tom22–His10, Tom20 and Tom5 were assigned by western blotting with antibodies against each subunits. The presence of Tom5, Tom6 and Tom7 were confirmed by mass spectrometry (MS) analyses. The bands for Tom5 and Tom7 were assigned by MS/MS analyses of the tryptic fragments of the proteins contained in each band. e, MALDI–TOF MS of purified TOM complex in the m/z range below 10,000. Calculated molecular weights of yeast small TOM subunits are 5986.0 (Tom5), 6407.6 (Tom6), and 6871.0 (Tom7). Tom7 appears to lack the N-terminal Met, and Tom6 may be acetylated. The data are representative of six (b, c), four (d) and two (e) independent experiments. f, Typically observed particles of HS-AFM images of the TOM complex trimer and dimer on mica.
Extended Data Fig. 2 Cryo-EM analysis of the TOM complex.
a, A micrograph of the TOM complex. b, Selected 2D classes showing various orientations. c, FSC curve after the application of soft mask in RELION, showing FSC of 0.143. d, The schematic workflow of the steps followed in data processing leading to the final structure of the TOM core complex. e, Angular particle distribution in the final reconstruction. f, Local map resolution of the final TOM complex structure with colours according to the scale on the right side. g, FSC curve between the EM map and the atomic model of the TOM complex. h, An example of the fit of the model into the density map; the region around β-strands 11–18 of Tom40 is shown.
Extended Data Fig. 3 β-barrel structure of Tom40.
a, Hydrogen bonds between the 19 β-strands on the basis of the EM structure. Circles, pentagons and squares indicate residues in loops, α-helices and β-strands, respectively. Disordered regions are coloured in grey. Hydrogen bond, CO–NH distance < 4.0 Å; weak hydrogen bond, 4.0 Å ≤ CO–NH distance ≤ 4.5 Å; no hydrogen bond, 4.5 Å < CO–NH distance. b, Relative positions of the other TOM subunits and phospholipid in contact with the Tom40 β-barrel. c, Summary of previous crosslinking results of Tom404. BPA-incorporated residues are labelled in bold. Crosslinked partners are indicated by different colours. Residues labelled with a red asterisk are not within a distance of 5 Å from their crosslinked partners in the EM structure.
Extended Data Fig. 4 Model for transition between the Tom22-containing dimer and trimer.
a, Comparison of the cryo-EM structure of Tom22-containing dimer (D) and crosslinked-based structure model of Tom22-containing trimer (T). b, The residues whose proximity relationships are inconsistent between the trimer (crosslinking) and the dimer (EM structure) are mapped on the EM structure. Top view from the cytosolic side (left) and side view (right) of the EM dimer structure (cartoon model) with the side chains (space-filling model) of the residues that were crosslinked to other subunits4 but were not within a distance of 4.5 Å from the crosslinked partners in the EM structure (Supplementary Table 1), and therefore judged to be important for formation of the mature trimer4. c, The residues suggesting the possible difference in the subunit interfaces between the trimer and dimer are mapped on the EM structure. Although Tom22A P112 and Tom40B Y309 face each other in the EM structure, the Tom22 transmembrane helix was crosslinked to two Tom40 molecules through Tom22 P112 and Tom40 Y3094, suggesting that these two residues should not face each other in the trimer. Tom22 E120 and Tom40 R310 are evolutionary conserved (Extended Data Fig. 7 and Supplementary Table 2) and electrostatic interaction between the two residues would contribute to stabilization of the dimer. d, e, Upon formation of the trimer from the dimer, the Tom22 transmembrane helix in the EM structure may rotate (with a possible conformational change), so that Tom22A P112 faces the Tom40A molecule and Tom22A S114 interacts with the Tom40B molecule. This rotation would lead to a switch of the electrostatic-interaction pairs from Tom22 E120 and Tom40 R310 to Tom22 E120 and Tom40 K90 with possible concomitant movement of the Tom7 C-terminal segment; the second short C-terminal helix of Tom7 appears to be stabilized by the hydrogen bond between the carbonyl O of L51 in Tom7 and the side-chain NH of K90 in Tom40 in the dimer, which is probably lost in the trimer. The rotation of the Tom22 transmembrane helix would also allow K94 and R89 of Tom22 to interact with D350 of the neighbouring Tom40 molecule. Basic and acidic residues are coloured in blue and red, respectively.
Extended Data Fig. 5 Conserved interactions between the Tom40 β-barrel and the α-helical subunits.
a–d, Interactions are shown between the Tom40 β-barrel and the α-helical subunits Tom22 (a), Tom7 (b), Tom6 (c) and Tom5 (d). Tom40 residues of the β-barrel outer wall and loops are shown in space-filling form and coloured by evolutionary conservation (darker blue represents more conserved residues). Tom40 residues at ‘conserved amino acid’ (conserved) positions within 4.5 Å distance of the α-helical subunits are labelled. The main chains of the α-helical subunits are shown in ribbon form, and residues at conserved positions within 4.5 Å distance of Tom40 are shown in stick form, labelled, and coloured by evolutionary conservation (deeper colour represents more highly conserved residues). Residues in parentheses are the most frequently observed residue at each conserved position. Pairs of residues at conserved positions of Tom40 β-barrel and α-helical subunits in proximity (≤4.5 Å) are underlined and bolded: Tom40B R310 (R) and Tom22 E120 (E), Tom40B S312 (S) and Tom22 S116 (A), Tom40B Y314 (F) and Tom22 L109 (L), Tom40B V324 (V) and Tom22 L109 (L), Tom40B V324 (V) and Tom22 L108 (L), Tom40B V324 (V) and Tom22 T105 (S), Tom40B L328 (L) and Tom22 P112 (P), Tom40B R330 (K) and Tom22 A119 (E), Tom40A L336 (F) and Tom22 L115 (L), Tom40B H346 (H) and Tom22 T105 (S), Tom40B H346 (H) and Tom22 T104 (T), and Tom40B H346 (H) and Tom22 W100 (W) (a); Tom40 F98 (F) and Tom7 G40 (G) and Tom40 G129 (G) and Tom7 I33 (I) (b); Tom40 L259 (Y) and Tom6 V46 (V), Tom40 L259 (Y) and Tom6 Q50 (Q), Tom40 G299 (G) and Tom6 N38 (S) and Tom40 S320 (S) and Tom6 N38(S) (c). No conserved proximate residue pair was found between Tom40 and Tom5 (d). Phospholipid (modelled as phosphatidylethanolamine) is shown in space-filling form; carbon, nitrogen, oxygen and phosphorus are coloured light green, blue, red and magenta, respectively (a).
Extended Data Fig. 6 Formation of the two exit sites of the TOM channel for different classes of precursor proteins.
a, Disordered regions of Tom22 and Tom40 facing the IMS. b, The TOM complex viewed from the IMS side shows that the trans presequence binding site (orange) is formed by the hydrophilic loop of Tom7 (residues 43–49), the hydrophilic β1–β2 loop (residues 92–96, M94 is disordered) and C-terminal domain (residues 363–387, residues after L374 are disordered, G363–E366 form a loop and E367–G373 form a helix) of Tom40 and the acidic IMS domain of Tom22 (residues 120–152, residues after 131 are disordered, the transmembrane helix is extended up to F130). Disordered parts are shown with beads. c, Crosslinked products of Tom22 containing BPA at the indicated positions were detected by the indicated antibodies. Crosslinked products between Tom22 and Tim50 (22–50) are indicated. Representative data of two independent experiments. d, Radiolabelled Oxa1 precursor was imported into wild-type and Tom40(Δ364–387) mitochondria for the indicated periods in the absence or presence of a membrane potential (Δψ) and analysed by blue native electrophoresis and autoradiography. Representative data of three independent experiments. Quantification of Oxa1 bound to the TOM complex; mean values ± s.e.m. (n = 3 biologically independent experiments); the amount of bound Oxa1 in wild-type mitochondria after 20 min (−Δψ) was set as 100% (control). e, Mitochondria isolated from wild-type and Tom40(Δ364–387) yeast grown at 30 °C were analysed by SDS–PAGE and western blotting using the indicated antisera. Representative data of four independent experiments. Amount of total mitochondrial protein is indicated. f, Mitochondria isolated from wild-type and Tom40(Δ364–387) yeast grown at 30 °C were analysed by blue native electrophoresis and western blotting with Tom40 antiserum. Representative data of four independent experiments. g, Radiolabelled Tim9 was imported and assembled into the TIM22 complex in wild-type and Tom40(Δ364–387) mitochondria; data are mean ± s.e.m. (n = 3 biologically independent experiments); the amount in wild-type mitochondria after 40 min import was set as 100% (control). h, The trans presequence binding sites for presequence-containing proteins (b) and the N extension of Tom40 recruiting small TIM chaperones (for presequence-lacking β-barrel and carrier proteins) and Mia40 (for MIA substrates) are spatially separated to form distinct precursor exit sites at the outlet of the TOM channel. The two exit sites are connected to the different translocation paths (yellow broken lines) that include either the aligned acidic patches (red) or the aligned hydrophobic patches (green) inside the Tom40 β-barrel pore (Extended Data Fig. 9). Disordered parts are shown with beads.
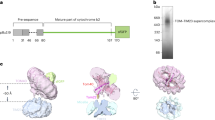
Extended Data Fig. 7 Roles of Tom5 and the N extension of Tom40 in MIA import and assembly.
a–e, Import of radiolabelled Tim9 precursor into isolated wild-type, tom5∆, tom6∆, tom7∆, tom22∆, tom20∆ and tom70∆ mitochondria, analysed by blue native electrophoresis (top) and SDS–PAGE (bottom). TIM22, TIM22 complex; Tim9–Mia40, mixed disulfide intermediate of Tim9 with Mia40; TIM9–10, Tim9–Tim10 complex. Representative data of three (a, b, top), four (d, top) or two (other panels) independent experiments. f, Mitochondria were isolated from wild-type, Tom40(∆58) and Tom40(∆59) yeast cells, which were grown at 30 °C and, where indicated, additionally shifted to 37 °C for 12 h, and analysed by SDS–PAGE and western blotting using the indicated antisera. Representative data of two independent experiments. g, Radiolabelled Tom5 precursor was imported into mitochondria isolated from wild-type and Tom40(∆59) yeast cells, which were grown at 30 °C and shifted to 37 °C for 12 h, followed by incubation with sodium carbonate. The carbonate-resistant pellets were analysed by SDS–PAGE and autoradiography. Representative data of three independent experiments. h, Mitochondria of wild-type, Tom40(∆58) and Tom40(∆59) cells grown at 30 °C were analysed by blue native electrophoresis and western blotting using Tom40- and Mia40-specific antisera. Representative data of two independent experiments. i, Imports of radiolabelled Atp2 and b2(167)∆DHFR into wild-type, Tom40(Δ58) and Tom40(Δ59) mitochondria were analysed by SDS–PAGE. Representative data of three independent experiments. ∆ψ, membrane potential; p, precursor form; m, mature form; i, intermediate form. j, Mitochondria of wild-type, Tom40(∆58) and Tom40(∆59) cells grown at 30 °C were subjected to hypo-osmotic swelling and treatment with proteinase K (Prot. K) as indicated. Proteins were analysed by SDS–PAGE and western blotting using the indicated antisera. Representative data of two independent experiments. The IMS protein Tim13 shows that the outer membranes of (non-swollen) Tom40(∆58) and Tom40(∆59) mitochondria were intact like those of wild-type mitochondria. k, Import of radiolabelled Su9–DHFR precursor (top) or AAC (bottom) into wild-type, Tom40(L57A) and Tom40(L57A/T62A) mitochondria was analysed by SDS–PAGE (top) or blue native gel (bottom). Representative data of three (Su9–DHFR) or two (AAC) independent experiments. l, Mitochondria of wild-type, Tom40(L57A) and Tom40(L57A/T62A) strains grown at 30 °C and, where indicated, additionally shifted to 37 °C for 29.5 h, were analysed by SDS–PAGE and western blotting using the indicated antisera. Representative data of two independent experiments.
Extended Data Fig. 8 MIA import and assembly are disturbed in point mutants of the Tom40 N extension.
a, Wild-type, Tom40(L57A) and Tom40(L57A/T62A) mitochondria from cells grown at 30 °C were analysed by blue native electrophoresis and western blotting using Tom40- and Mia40-specific antisera. Representative data of two independent experiments. b, Wild-type, tom5∆, Tom40(L202A) and Tom40(L57A/T62A) mitochondria were treated with DTT or 4-DPS and the redox state of Mia40 was analysed by non-reducing SDS–PAGE and western blotting. Representative data of three independent experiments. Red, reduced form; oxid., oxidized form. c, Growth analysis of yeast strains expressing the indicated Tom40 variants on YPG. Representative data of two independent experiments. d, Radiolabelled Tim9 was imported into isolated wild-type and Tom40(L202A) mitochondria for 5 min, followed by addition of iodoacetamide (IA), and incubated further as indicated. The import was analysed by blue native electrophoresis. Representative data of two independent experiments. TIM22, TIM22 complex; Tim9–Mia40, mixed disulfide intermediate of Tim9 with Mia40; TIM9–10, Tim9–Tim10 complex. e, Import of radiolabelled Tim9(C55S) into wild-type and Tom40(L202A) mitochondria followed by treatment with proteinase K as indicated. The import was analysed by blue native electrophoresis (top) and non-reducing SDS–PAGE (bottom). Representative data of two independent experiments. Tim9–Mia40, mixed disulfide intermediate of Tim9(C55S) with Mia40. f, Wild-type and Tom40(L202A) mitochondria from cells grown at 30 °C were analysed by blue native electrophoresis and western blotting using Tom40 and Mia40 specific antisera. Representative data of two independent experiments. g, Wild-type and Tom40(L202A) mitochondria from cells grown at 30 °C were treated with proteinase K where indicated. Representative data of two independent experiments. The stability of IMS-exposed proteins indicates that the outer membranes of Tom40(L202A) mitochondria were intact like those of wild-type mitochondria. h, Import of radiolabelled Su9–DHFR (top) and AAC (bottom) into wild-type and Tom40(L202A) mitochondria was analysed by SDS–PAGE (top) or blue native electrophoresis (bottom). Representative data of three independent experiments.
Extended Data Figure 9 The pore interior of Tom40 and precursor import paths.
a, b, The views from the cytosol (top) and cut-away views (bottom) for Tom40. Section planes for the cut-away views are indicated with broken lines in the top panels. The side chains of the Tom40 residues crosslinked to the translocation intermediates of a presequence-containing precursor pSu9–DHFR (a) and of a hydrophobic presequence-lacking precursor AAC–DHFR (b) are shown on the EM structure with colour reflecting the amount of crosslinked products detected4. The acidic patches (red) and hydrophobic patches (green) are shown in the cut-away views for the β-barrel side of the pore interior of Tom40. Possible precursor translocation paths are indicated with arrows by broken lines (bottom). c, Section curved plane for the cut-away views (centre, bottom) is indicated with a broken line in the view from the cytosolic side (top). The acidic patches (red), hydrophobic patches (green) and possible precursor translocation paths (broken lines) are shown (bottom).
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1 (alignment of the N-terminus to β-strand 14 of Tom40 from various organisms) and 2 (the uncropped version of all the blots, gels and plates in this study).
Supplementary Tables
This file contains Supplementary Tables 1-4.
Video 1
HS-AFM observation of the purified TOM complex on a mica plate (200 nm × 200 nm).
Video 2
HS-AFM observation of the purified TOM complex scattered on a mica plate showing conversion of the trimer to the dimer (50 nm × 50 nm).
Source data
Rights and permissions
About this article
Cite this article
Araiso, Y., Tsutsumi, A., Qiu, J. et al. Structure of the mitochondrial import gate reveals distinct preprotein paths. Nature 575, 395–401 (2019). https://doi.org/10.1038/s41586-019-1680-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1680-7
This article is cited by
-
Mitochondrial stress: a key role of neuroinflammation in stroke
Journal of Neuroinflammation (2024)
-
The ER-SURF pathway uses ER-mitochondria contact sites for protein targeting to mitochondria
EMBO Reports (2024)
-
The architecture of substrate-engaged TOM–TIM23 supercomplex reveals preprotein proximity sites for mitochondrial protein translocation
Cell Discovery (2024)
-
NLG1, encoding a mitochondrial membrane protein, controls leaf and grain development in rice
BMC Plant Biology (2023)
-
Molecular pathway of mitochondrial preprotein import through the TOM–TIM23 supercomplex
Nature Structural & Molecular Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.