Abstract
High-pressure transitions are thought to modify hydrogen molecules to a molecular metallic solid and finally to an atomic metal1, which is predicted to have exotic physical properties and the topology of a two-component (electron and proton) superconducting superfluid condensate2,3. Therefore, understanding such transitions remains an important objective in condensed matter physics4,5. However, measurements of the crystal structure of solid hydrogen, which provides crucial information about the metallization of hydrogen under compression, are lacking for most high-pressure phases, owing to the considerable technical challenges involved in X-ray and neutron diffraction measurements under extreme conditions. Here we present a single-crystal X-ray diffraction study of solid hydrogen at pressures of up to 254 gigapascals that reveals the crystallographic nature of the transitions from phase I to phases III and IV. Under compression, hydrogen molecules remain in the hexagonal close-packed (hcp) crystal lattice structure, accompanied by a monotonic increase in anisotropy. In addition, the pressure-dependent decrease of the unit cell volume exhibits a slope change when entering phase IV, suggesting a second-order isostructural phase transition. Our results indicate that the precursor to the exotic two-component atomic hydrogen may consist of electronic transitions caused by a highly distorted hcp Brillouin zone and molecular-symmetry breaking.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Wigner, E. & Huntington, H. B. On the possibility of a metallic modification of hydrogen. J. Chem. Phys. 3, 764–770 (1935).
Babaev, E., Sudbø, A. & Ashcroft, N. W. A superconductor to superfluid phase transition in liquid metallic hydrogen. Nature 431, 666–668 (2004).
Bonev, S. A., Schwegler, E., Ogitsu, T. & Galli, G. A quantum fluid of metallic hydrogen suggested by first-principles calculations. Nature 431, 669–672 (2004).
McMahon, J. M., Morales, M. A., Pierleoni, C. & Ceperley, D. M. The properties of hydrogen and helium under extreme conditions. Rev. Mod. Phys. 84, 1607–1653 (2012).
Mao, H.-K., Chen, X.-J., Ding, Y., Li, B. & Wang, L. Solids, liquids, and gases under high pressure. Rev. Mod. Phys. 90, 015007 (2018).
Mao, H.-K. & Hemley, R. J. Ultrahigh-pressure transitions in solid hydrogen. Rev. Mod. Phys. 66, 671–692 (1994).
Lorenzana, H. E., Silvera, I. F. & Goettel, K. A. Orientational phase transitions in hydrogen at megabar pressures. Phys. Rev. Lett. 64, 1939 (1990).
Liu, X. D., Howie, R. T., Zhang, H. C., Chen, X. J. & Gregoryanz, E. High-pressure behavior of hydrogen and deuterium at low temperatures. Phys. Rev. Lett. 119, 065301 (2017).
Hemley, R. J. & Mao, H. K. Phase transition in solid molecular hydrogen at ultrahigh pressures. Phys. Rev. Lett. 61, 857–860 (1988).
Howie, R. T., Guillaume, C. L., Scheler, T., Goncharov, A. F. & Gregoryanz, E. Mixed molecular and atomic phase of dense hydrogen. Phys. Rev. Lett. 108, 125501 (2012).
Eremets, M. I. & Troyan, I. A. Conductive dense hydrogen. Nat. Mater. 10, 927–931 (2011).
Howie, R. T., Scheler, T., Guillaume, C. L. & Gregoryanz, E. Proton tunneling in phase IV of hydrogen and deuterium. Phys. Rev. B 86, 214104 (2012).
Dalladay-Simpson, P., Howie, R. T. & Gregoryanz, E. Evidence for a new phase of dense hydrogen above 325 gigapascals. Nature 529, 63–67 (2016).
Hazen, R. M., Mao, H. K., Finger, L. W. & Hemley, R. J. Single-crystal X-ray diffraction of n-H2 at high pressure. Phys. Rev. B 36, 3944–3947 (1987).
Loubeyre, P. et al. X-ray diffraction and equation of state of hydrogen at megabar pressures. Nature 383, 702–704 (1996).
Mao, H.-K. et al. Synchrotron X-ray diffraction measurements of single-crystal hydrogen to 26.5 gigapascals. Science 239, 1131–1134 (1988).
Mazin, I. I., Hemley, R. J., Goncharov, A. F., Hanfland, M. & Mao, H.-K. Quantum and classical orientational ordering in solid hydrogen. Phys. Rev. Lett. 78, 1066–1069 (1997).
Liu, H., Zhu, L., Cui, W. & Ma, Y. Room-temperature structures of solid hydrogen at high pressures. J. Chem. Phys. 137, 074501 (2012).
Pickard, C. J., Martinez-Canales, M. & Needs, R. J. Density functional theory study of phase IV of solid hydrogen. Phys. Rev. B 85, 214114 (2012).
Monserrat, B. et al. Structure and metallicity of phase V of hydrogen. Phys. Rev. Lett. 120, 255701 (2018).
Akahama, Y., Mizuki, Y., Nakano, S., Hirao, N. & Ohishi, Y. Raman scattering and X-ray diffraction studies on phase III of solid hydrogen. J. Phys. Conf. Ser. 950, 042060 (2017).
Prescher, C. et al. Beyond sixfold coordinated Si in SiO2 glass at ultrahigh pressures. Proc. Natl Acad. Sci. USA 114, 10041–10046 (2017).
Wang, L. et al. Nanoprobe measurements of materials at megabar pressures. Proc. Natl Acad. Sci. USA 107, 6140–6145 (2010).
Akahama, Y. et al. Evidence from X-ray diffraction of orientational ordering in phase III of solid hydrogen at pressures up to 183 GPa. Phys. Rev. B 82, 060101 (2010).
Magdău, I. B., Marqués, M., Borgulya, B. & Ackland, G. J. Simple thermodynamic model for the hydrogen phase diagram. Phys. Rev. B 95, 094107 (2017).
Goncharov, A. F., Eggert, J. H., Mazin, I. I., Hemley, R. J. & Mao, H.-K. Raman excitations and orientational ordering in deuterium at high pressure. Phys. Rev. B 54, R15590–R15593 (1996).
Glazyrin, K. et al. Importance of correlation effects in hcp iron revealed by a pressure-induced electronic topological transition. Phys. Rev. Lett. 110, 117206 (2013).
Kvashnin, Y. O., Sun, W., Marco, I. D. & Eriksson, O. Electronic topological transition and noncollinear magnetism in compressed hcp Co. Phys. Rev. B 92, 134422 (2015).
Feng, Q., Ekholm, M., Tasnádi, F., Jönsson, H. J. M. & Abrikosov, I. A. Topological transitions of the Fermi surface of osmium under pressure: an LDA+DMFT study. New J. Phys. 19, 033020 (2017).
Sun, W., Luo, W., Feng, Q. & Ahuja, R. Anisotropic distortion and Lifshitz transition in α-Hf under pressure. Phys. Rev. B 95, 115130 (2017).
Anderson, O. L., Isaak, D. G. & Yamamoto, S. Anharmonicity and the equation of state for gold. J. Appl. Phys. 65, 1534 (1989).
Turnbull, R. et al. Reactivity of hydrogen–helium and hydrogen–nitrogen mixtures at high pressures. Phys. Rev. Lett. 121, 195702 (2018).
Hrubiak, R., Sinogeikin, S., Rod, E. & Shen, G. The laser micro-machining system for diamond anvil cell experiments and general precision machining applications at the High Pressure Collaborative Access Team. Rev. Sci. Instrum. 86, 072202 (2015).
Akahama, Y. & Kawamura, H. Pressure calibration of diamond anvil Raman gauge to 410 GPa. J. Phys. Conf. Ser. 215, 012195 (2010).
Speziale, S., Zha, C.-S., Duffy, T. S., Hemley, R. J. & Mao, H.-K. Quasi-hydrostatic compression of magnesium oxide to 52 GPa: implications for the pressure-volume-temperature equation of state. J. Geophys. Res. 106, 515–528 (2001).
Prescher, C. & Prakapenka, V. B. DIOPTAS: a program for reduction of two-dimensional X-ray diffraction data and data exploration. High Press. Res. 35, 223–230 (2015).
Hrubiak, R., Smith, J. S. & Shen, G. Multimode scanning X-ray diffraction microscopy for diamond anvil cell experiments. Rev. Sci. Instrum. 90, 025109 (2019).
Wojdyr, M. Fityk: a general-purpose peak fitting program. J. Appl. Cryst. 43, 1126–1128 (2010).
Gonzalez-Platas, J., Alvaro, M., Nestola, F. & Angel, R. EosFit7-GUI: a new graphical user interface for equation of state calculations, analyses and teaching. J. Appl. Cryst. 49, 1377–1382 (2016).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys Rev B 47, 558–561 (1993).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Mostofi, A. A. et al. An updated version of wannier90: a tool for obtaining maximally-localised Wannier functions. Comput. Phys. Commun. 185, 2309–2310 (2014).
Goncharov, A. F., Gregoryanz, E., Hemley, R. J. & Mao, H.-K. Spectroscopic studies of the vibrational and electronic properties of solid hydrogen to 285 GPa. Proc. Natl Acad. Sci. USA 98, 14234–14237 (2001).
Pickard, C. J. & Needs, R. J. Structure of phase III of solid hydrogen. Nat. Phys. 3, 473–476 (2007).
Monserrat, B., Needs, R. J., Gregoryanz, E. & Pickard, C. J. Hexagonal structure of phase III of solid hydrogen. Phys. Rev. B 94, 134101 (2016).
Acknowledgements
We thank R. Ferry and M. Somayazulu for help with the gas loading systems; Y. Wang for the use of the MCC of Geo Soil Enviro Consortium for Advanced Radiation Sources (GSECARS); and E. Gregoryanz, A. Goncharov, D. Kim, D. Popov, Y. Ma, H. Liu and J. Tse for discussions. We acknowledge financial support from the National Natural Science Foundation of China (NSFC) under grant numbers U1530402 and 11811530001. This research was also supported by the US Department of Energy (DOE), Office of Basic Energy Science, Division of Materials Sciences and Engineering under award DE-FG02-99ER45775. Portions of this work were performed at the 34 IDE, 16 IDB (HPCAT), 13 IDD (GSECARS) and 1-BM beamlines of APS, ANL in USA, as well as the BL15U1 beamline of SSRF in China. HPCAT operations are supported by the Office of Experimental Sciences of the DOE National Nuclear Security Administration under award DE-NA0001974. V.B.P. is grateful to the National Science Foundation (NSF) for award MRI EAR/IF1531583. GSECARS is supported by NSF–Earth Sciences (grant number EAR-1128799) and DOE–Geosciences (grant number DE-FG02-94ER14466). This research used resources of the APS, a US DOE Office of Science User Facility operated for the DOE Office of Science by ANL under contract number DE-AC02-06CH11357. A.M., R.A. and W. Luo acknowledge support from Carl Tryggers Stiftelse for Vetenskaplig Forskning (CTS) and the Swedish Research Council (VR). SNIC and HPC2N are also acknowledged for providing computing time.
Author information
Authors and Affiliations
Contributions
H.-K.M. conceived and supervised the project; C.J., and B.L. conducted the synchrotron XRD measurements; W. Liu, R.X., J.S.S., S.S., Y.M., V.B.P., E.G. and G.S. developed the techniques for the H2 XRD measurements at the synchrotron beamlines; J.W., J.S., B.L., W.Y., X.H. and C.J. developed the DAC-related techniques to achieve the target ultrahigh-pressure experimental conditions; C.J. conducted the Raman experiments; C.J. and J.S. prepared the DAC samples; C.J., H.-K.M., G.S. and W.Y. performed the analysis; H.-K.M. and W.L.M. conceived and developed the ETT interpretation. A.M., W. Luo and R.A. conducted the ab-initio calculations. H.-K.M. and C.J. wrote the manuscript in consultation with W.L.M., G.S., W.Y., V.B.P., E.G., B.L., W. Liu, J.S.S., A.M., W. Luo, R.A. and Y.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
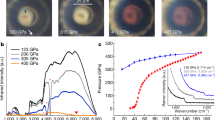
Extended Data Fig. 1 Raw XRD images obtained using a pure Re gasket and a composite gasket at the same beamline.
a, XRD image obtained at 45 GPa with the Re gasket. The inset shows a microscope image of the sample after gas loading, with the chamber diameter being 17 µm. At 45 GPa, the chamber shrinks to a diameter of approximately 10 µm. b, Raw XRD image of the sample obtained at 162 GPa using a composite gasket insert (cBN and epoxy); the chamber diameter is 7 µm. The setup of the beamline focusing device (Kirkpatrick–Baez mirrors) was similar in both measurements, with clean-up pinholes of 20 µm (a) and 60 µm (b) in diameter. It must be emphasized that even though the X-ray beam used in b has a larger tail (owing to the larger clean-up pinhole), b has a substantially lower background than a. The MgO and epoxy insert produces a similar level of background to the cBN and epoxy insert. Red masks in a and b cover the gaps between the sensor chips on the Pilatus 1M detector.
Extended Data Fig. 2 Definition of χ angle in the XRD image.
χ is the azimuthal angle of the XRD spot within the detector plane. The blue arrow points to a diffraction spot inside the red circle.
Extended Data Fig. 3 Features of Raman vibrons of hydrogen under high pressures.
a, Pressure-dependent frequency shift of vibrational modes of H2 at room temperature. b, Pressure-dependent shift of the widths (FWHM) of H2 vibrational modes at room temperature. v1 and v2 represent the two fundamental vibrational modes of H2, as shown in Extended Data Fig. 4. Circles and triangles represent the v1 and v2 modes, respectively; different colours represent different runs. Dashed lines are visual guides. In phase III, the frequency of v1 softens substantially with pressure compared to that in phase I, and the Raman peak broadens considerably simultaneously. In phase IV, the behaviour of v1 is similar to that in phase III, and a new fundamental vibrational mode, v2, appears at higher frequency. Phase IV also exhibits new low-frequency modes, as shown in Extended Data Fig. 4.
Extended Data Fig. 4 Complete Raman spectrum of the H2 sample at 232 GPa and room temperature.
This is the complete version of the spectrum shown in the inset of Fig. 2a. The spectrum clearly shows the characteristic Raman features of phase IV (marked by blue triangles), the low-frequency mode at 303 cm−1 and the second vibron at 4,149 cm−1. The inset shows the diamond Raman edge with a pressure of 234 GPa, which shows a difference of 2 GPa compared to our d100–P scale.
Extended Data Fig. 5 XRD measurements for the d100–P scale and immiscibility of H2 and He determined by Raman measurements.
a, Evolution of d spacing of the (100) reflection of H2, measured in the H2–He samples with Au as the pressure marker. The (111) reflection of the Au standard was used to calculate the pressure31. The inset shows the sample configuration. b, Raman spectra of the H2–He samples at selected pressures. Different colours mark different runs. c, Comparison of the pressure-dependent shift of vibron frequency between the H2–He samples (solid symbols) and the pure H2 samples (open symbols). Samples with names starting with HP and HX, as well as H27, are pure H2 samples. All other samples are H2–He samples.
Extended Data Fig. 6 2D diffraction contrast imaging of MgO and H2 (submicron crystallites).
a, 2D diffraction contrast imaging based on the intensity of the MgO (200) Bragg peak. Darker colour indicates higher intensity of the MgO (200) peak. The white area represents the hydrogen sample. b, Microscope image of the same sample area, illuminated by both transmitted and reflected light. c–h, 2D diffraction contrast images based on intensities of selected XRD spots of H2 samples at 97 GPa (c–e), 163 GPa (f, g) and 162 GPa (h). Darker colour indicates higher peak intensity. The step size of the 2D scan is 500 nm in both the horizontal and the vertical directions. The data analysis was performed using XDI software37.
Extended Data Fig. 7 Raw XRD images obtained using MCCs with two different vertical opening angles.
a, b, Images obtained using MCCs with 4θ angles of 10° (a) and 30° (b) with the Pilatus 1M area detector. The MCC with 10° vertical opening (located at GSECARS, APS, ANL) is mainly designed for the Large Volume Press and for DACs used to study amorphous or powder samples. With the 10° MCC in place, only about one third of the area detector can be exposed to X-rays (area enclosed in the red box in a). This limited vertical opening is not a problem when studying amorphous or powder samples, because the X-ray scattering or diffraction is uniformly distributed about the X-ray beam and can thus be captured by the area detector through the small window. However, for SXRD data collection, the XRD spots appear at specific azimuth angles on the area detector, and the limited vertical opening becomes an issue. As a result, data collection using this MCC was not trivial, and consisted of the following steps: (1) identify XRD spots without the MCC at low pressure; (2) rotate the sample about the χ axis to move the target XRD spot into the MCC opening; (3) increase the pressure and track the XRD spot with the MCC. It would be extremely time-consuming to directly search for a diffraction spot of hydrogen with the 10° MCC at pressures greater than 1 Mb. However, measurements of the c/a ratio using the 10° MCC are subject to relatively large systematic errors between different samples, as can be seen in the MCC data of Fig. 3d (most of these data above 100 GPa were measured with the 10° MCC). Because searching for the XRD spot using the 10° MCC is difficult, as explained above, and data collection with an MCC requires extended exposure time, usually one (100) peak and one (101) peak were measured to determine the c/a ratio. These peaks are from different crystals and have different crystal orientations under different stress conditions, resulting in the scattering of the c/a values of different samples. The MCC with vertical opening angle of 30° was specially designed by HPCAT to overcome the above problems. When using the 30° MCC, almost the entire area detector can be exposed (red box in b). This considerably facilitates the collection of SXRD data of H2 at pressures of several megabars, and is also suitable for performing SXRD studies of other low-atomic-number materials in the DAC at very high pressures.
Extended Data Fig. 8 Effects of using the MCC and the submicron probe for XRD measurements of solid hydrogen at megabar pressures.
a, Integrated hydrogen (100) peak measured with and without the MCC at 143 GPa. With the MCC, the background is reduced by a factor of 5. b, c, Corresponding raw images of the (100) peak. Integrations were performed using the same region of interest, which contains the target peak (shown in b and c), and the same setups in Dioptas36. The data collection times with and without the MCC were equivalent (the actual exposure in the measurement with the MCC was several times longer owing to the limited throughput of the MCC). The data were collected using a 2 × 1 µm2 focused X-ray beam at beamline 16 IDB of APS. d–f, XRD images corresponding to the real beam size of 300 nm (d) and equivalent beam sizes of 1 µm (e) and 2 µm (f). The data were collected using a 300-nm nano-beam. The data in d were collected at a single sample position with 60 s exposure. The image in e was acquired by merging nine images measured by a 3 × 3 2D scan with the same exposure time for each image (60 s). The image in f was obtained by merging 49 images collected by a 7 × 7 2D scan with the same exposure time for each image (60 s). The step size in the 2D scans was 500 nm. Red dots represent the X-ray beam. It is obvious that a larger beam size, which includes more background, results in a deterioration in data quality.
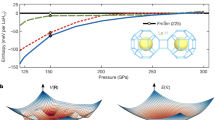
Extended Data Fig. 9 Calculated electronic band structures.
a–e, Band structures calculated at 50 GPa (a), 100 GPa (b), 156 GPa (c), 193 GPa (d) and 240 GPa (e). Eg denotes the electronic bandgap energy.
Extended Data Fig. 10
Enthalpy differences of various phases of hydrogen as a function of pressure according to DFT calculations.
Rights and permissions
About this article
Cite this article
Ji, C., Li, B., Liu, W. et al. Ultrahigh-pressure isostructural electronic transitions in hydrogen. Nature 573, 558–562 (2019). https://doi.org/10.1038/s41586-019-1565-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1565-9
This article is cited by
-
Synthesis and superconductivity in yttrium-cerium hydrides at high pressures
Nature Communications (2024)
-
Quantum phase diagram of high-pressure hydrogen
Nature Physics (2023)
-
Universal diamond edge Raman scale to 0.5 terapascal and implications for the metallization of hydrogen
Nature Communications (2023)
-
Nested order-disorder framework containing a crystalline matrix with self-filled amorphous-like innards
Nature Communications (2022)
-
Thermal conductivity of materials under pressure
Nature Reviews Physics (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.