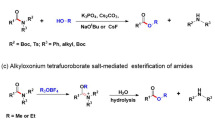
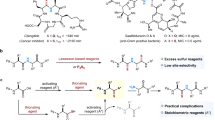
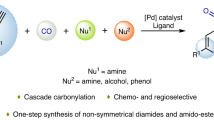
Abstract
Amides and related carbonyl derivatives are of central importance across the physical and life sciences1,2. As a key biological building block, the stability and conformation of amides affect the structures of peptides and proteins as well as their biological function. In addition, amide-bond formation is one of the most frequently used chemical transformations3,4. Given their ubiquity, a technology that is capable of modifying the fundamental properties of amides without compromising on stability may have considerable potential in pharmaceutical, agrochemical and materials science. In order to influence the physical properties of organic molecules—such as solubility, lipophilicity, conformation, pKa and (metabolic) stability—fluorination approaches have been widely adopted5,6,7. Similarly, site-specific modification with isosteres and peptidomimetics8, or in particular by N-methylation9, has been used to improve the stability, physical properties, bioactivities and cellular permeabilities of compounds. However, the N-trifluoromethyl carbonyl motif—which combines both N-methylation and fluorination approaches—has not yet been explored, owing to a lack of efficient methodology to synthesize it. Here we report a straightforward method to access N-trifluoromethyl analogues of amides and related carbonyl compounds. The strategy relies on the operationally simple preparation of bench-stable carbamoyl fluoride building blocks, which can be readily diversified to the corresponding N–CF3 amides, carbamates, thiocarbamates and ureas. This method tolerates rich functionality and stereochemistry, and we present numerous examples of highly functionalized compounds—including analogues of widely used drugs, antibiotics, hormones and polymer units.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information files.
References
Greenberg, A. et al. (eds) The Amide Linkage: Structural Significance in Chemistry, Biochemistry, and Materials Science (Wiley, 2000).
Pattabiraman, V. R. & Bode, J. W. Rethinking amide bond synthesis. Nature 480, 471–479 (2011).
Schneider, N., Lowe, D. M., Sayle, R. A., Tarselli, M. A. & Landrum, G. A. Big data from pharmaceutical patents: a computational analysis of medicinal chemists’ bread and butter. J. Med. Chem. 59, 4385–4402 (2016).
Brown, D. G. & Boström, J. Analysis of past and present synthetic methodologies on medicinal chemistry: where have all the new reactions gone? J. Med. Chem. 59, 4443–4458 (2016).
Müller, K., Faeh, C. & Diederich, F. Fluorine in pharmaceuticals: looking beyond intuition. Science 317, 1881–1886 (2007).
Purser, S., Moore, P. R., Swallow, S. & Gouverneur, V. Fluorine in medicinal chemistry. Chem. Soc. Rev. 37, 320–330 (2008).
Isanbor, C. & O’Hagan, D. Fluorine in medicinal chemistry: A review of anti-cancer agents. J. Fluor. Chem. 127, 303–319 (2006).
Vagner, J., Qu, H. & Hruby, V. J. Peptidomimetics, a synthetic tool of drug discovery. Curr. Opin. Chem. Biol. 12, 292–296 (2008).
Chatterjee, J., Gilon, C., Hoffman, A. & Kessler, H. N-methylation of peptides: a new perspective in medicinal chemistry. Acc. Chem. Res. 41, 1331–1342 (2008).
Pharmaceutical Products and Market. Statista https://www.statista.com/markets/412/topic/456/pharmaceutical-products-market/.
Metcalf, L. R. in Ullmann’s Encyclopedia of Industrial Chemistry (Wiley-VCH, 2000).
Holland, J. R. et al. A controlled trial of urethane treatment in multiple myeloma. Blood 27, 328–342 (1966).
Sijbesma, R. P. et al. Reversible polymers formed from self-complementary monomers using quadruple hydrogen bonding. Science 278, 1601–1604 (1997).
Valeur, E. & Bradley, M. Amide bond formation: beyond the myth of coupling reagents. Chem. Soc. Rev. 38, 606–631 (2009).
van der Werf, A., Hribersek, M. & Selander, N. N-trifluoromethylation of nitrosoarenes with sodium triflinate. Org. Lett. 19, 2374–2377 (2017).
Klöter, G. & Seppelt, K. Trifluoromethanol (CF3OH) and trifluoromethylamine (CF3NH2). J. Am. Chem. Soc. 101, 347–349 (1979).
Scattolin, T., Deckers, K. & Schoenebeck, F. Efficient synthesis of trifluoromethyl amines through a formal umpolung strategy from the bench-stable precursor (Me4N)SCF3. Angew. Chem. Int. Ed. 56, 221–224 (2017).
Hagooly, Y., Gatenyo, J., Hagooly, A. & Rozen, S. Toward the synthesis of the rare N-(trifluoromethyl)amides and the N-(difluoromethylene)-N-(trifluoromethyl)amines [RN(CF3)CF2R′] using BrF3. J. Org. Chem. 74, 8578–8582 (2009).
Handa, M. & Inoue, M. O-acyl-N-aryl-N-(trifluoromethyl)hydroxylamine derivative and method for producing the same. Japanese patent JP2012062284A (2010).
Ruppert, I. Organylisocyaniddifluoride R–N=CF2 durch direktfluorierung von isocyaniden. Tetrahedron Lett. 21, 4893–4896 (1980).
Sheppard, W. A. N-Fluoroalkylamines. I. Difluoroazomethines. J. Am. Chem. Soc. 87, 4338–4341 (1965).
Pajtás, D. et al. Optimization of the synthesis of flavone–amino acid and flavone–dipeptide hybrids via Buchwald–Hartwig reaction. J. Org. Chem. 82, 4578–4587 (2017).
Schindler, C. S., Forster, P. M. & Carreira, E. M. Facile formation of N-acyl-oxazolidinone derivatives using acid fluorides. Org. Lett. 12, 4102–4105 (2010).
Clark, R. D. et al. Synthesis and evaluation of ureido- and vinylureidopenicillins as inhibitors of intraruminal lactic acid production. J. Med. Chem. 24, 1250–1253 (1981).
Acknowledgements
We acknowledge RWTH Aachen University for financial support, and K. Deckers and T. Sperger for assistance and discussions.
Author information
Authors and Affiliations
Contributions
T.S. and S.B.-G. performed the experiments. S.B.-G. undertook the calculations. All authors analysed the data and contributed to the preparation of the manuscript. F.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
A patent application has been submitted by RWTH Aachen University for this methodology, with T.S. and F.S. as inventors (2018112315090400DE).
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Nature thanks Scott Bagley, Jonathan Clayden and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Supplementary information
Supplementary Information
This PDF file includes Materials and Methods, Experimental Procedures, Characterization Data, Stability Data, Racemization analyses, NMR study, Mechanistic Studies, Computational Details, NMR Spectra and Supplementary Figures S1 to S59.
Rights and permissions
About this article
Cite this article
Scattolin, T., Bouayad-Gervais, S. & Schoenebeck, F. Straightforward access to N-trifluoromethyl amides, carbamates, thiocarbamates and ureas. Nature 573, 102–107 (2019). https://doi.org/10.1038/s41586-019-1518-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1518-3
This article is cited by
-
Engineering site-selective incorporation of fluorine into polyketides
Nature Chemical Biology (2022)
-
Closing the gap between 19F and 18F chemistry
EJNMMI Radiopharmacy and Chemistry (2021)
-
Fluorinated compounds present opportunities for drug discovery
Nature (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.