Abstract
Cytotoxic T cells are essential mediators of protective immunity to viral infection and malignant tumours and are a key target of immunotherapy approaches. However, prolonged exposure to cognate antigens often attenuates the effector capacity of T cells and limits their therapeutic potential1,2,3,4. This process, known as T cell exhaustion or dysfunction1, is manifested by epigenetically enforced changes in gene regulation that reduce the expression of cytokines and effector molecules and upregulate the expression of inhibitory receptors such as programmed cell-death 1 (PD-1)5,6,7,8. The underlying molecular mechanisms that induce and stabilize the phenotypic and functional features of exhausted T cells remain poorly understood9,10,11,12. Here we report that the development and maintenance of populations of exhausted T cells in mice requires the thymocyte selection-associated high mobility group box (TOX) protein13,14,15. TOX is induced by high antigen stimulation of the T cell receptor and correlates with the presence of an exhausted phenotype during chronic infections with lymphocytic choriomeningitis virus in mice and hepatitis C virus in humans. Removal of its DNA-binding domain reduces the expression of PD-1 at the mRNA and protein level, augments the production of cytokines and results in a more polyfunctional T cell phenotype. T cells with this deletion initially mediate increased effector function and cause more severe immunopathology, but ultimately undergo a massive decline in their quantity, notably among the subset of TCF-1+ self-renewing T cells. Altogether, we show that TOX is a critical factor for the normal progression of T cell dysfunction and the maintenance of exhausted T cells during chronic infection, and provide a link between the suppression of effector function intrinsic to CD8 T cells and protection against immunopathology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Next-generation sequencing data, RNA microarray data, ATAC-sequencing data and whole-genome methylation datasets are available in the NCBI GEO repository with accession number GSE131643. All other datasets generated during the current study are available from the corresponding authors on reasonable request.
Code availability
Details and code for data-processing pipelines are available from https://gitlab.lrz.de/ImmunoPhysio/bulkSeqPipe and https://github.com/zehnlab/microarray_ngs_scripts.
References
Wherry, E. J. T cell exhaustion. Nat. Immunol. 12, 492–499 (2011).
Speiser, D. E. et al. T cell differentiation in chronic infection and cancer: functional adaptation or exhaustion? Nat. Rev. Immunol. 14, 768–774 (2014).
Schietinger, A. & Greenberg, P. D. Tolerance and exhaustion: defining mechanisms of T cell dysfunction. Trends Immunol. 35, 51–60 (2014).
Gallimore, A., Hengartner, H. & Zinkernagel, R. Hierarchies of antigen-specific cytotoxic T-cell responses. Immunol. Rev. 164, 29–32 (1998).
Barber, D. L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006).
Chihara, N. et al. Induction and transcriptional regulation of the co-inhibitory gene module in T cells. Nature 558, 454–459 (2018).
Youngblood, B. et al. Chronic virus infection enforces demethylation of the locus that encodes PD-1 in antigen-specific CD8+ T cells. Immunity 35, 400–412 (2011).
Ghoneim, H. E. et al. De novo epigenetic programs inhibit PD-1 blockade-mediated T cell rejuvenation. Cell 170, 142–157 (2017).
Pauken, K. E. et al. Epigenetic stability of exhausted T cells limits durability of reinvigoration by PD-1 blockade. Science 354, 1160–1165 (2016).
Utzschneider, D. T. et al. T cells maintain an exhausted phenotype after antigen withdrawal and population reexpansion. Nat. Immunol. 14, 603–610 (2013).
Philip, M. et al. Chromatin states define tumour-specific T cell dysfunction and reprogramming. Nature 545, 452–456 (2017).
Wieland, D. et al. TCF1+ hepatitis C virus-specific CD8+ T cells are maintained after cessation of chronic antigen stimulation. Nat. Commun. 8, 15050 (2017).
Aliahmad, P., Seksenyan, A. & Kaye, J. The many roles of TOX in the immune system. Curr. Opin. Immunol. 24, 173–177 (2012).
O’Flaherty, E. & Kaye, J. TOX defines a conserved subfamily of HMG-box proteins. BMC Genomics 4, 13 (2003).
Page, N. et al. Expression of the DNA-binding factor TOX promotes the encephalitogenic potential of microbe-induced autoreactive CD8+ T cells. Immunity 48, 937–950 (2018).
Wherry, E. J. et al. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 27, 670–684 (2007).
Utzschneider, D. T. et al. High antigen levels induce an exhausted phenotype in a chronic infection without impairing T cell expansion and survival. J. Exp. Med. 213, 1819–1834 (2016).
Martinez, G. J. et al. The transcription factor NFAT promotes exhaustion of activated CD8+ T cells. Immunity 42, 265–278 (2015).
Utzschneider, D. T. et al. T cell factor 1-expressing memory-like CD8+ T cells sustain the immune response to chronic viral infections. Immunity 45, 415–427 (2016).
Lin, W. W. et al. CD8+ T lymphocyte self-renewal during effector cell determination. Cell Rep. 17, 1773–1782 (2016).
Yu, S. et al. The TCF-1 and LEF-1 transcription factors have cooperative and opposing roles in T cell development and malignancy. Immunity 37, 813–826 (2012).
Delpoux, A., Lai, C. Y., Hedrick, S. M. & Doedens, A. L. FOXO1 opposition of CD8+ T cell effector programming confers early memory properties and phenotypic diversity. Proc. Natl Acad. Sci. USA 114, E8865–E8874 (2017).
Odorizzi, P. M., Pauken, K. E., Paley, M. A., Sharpe, A. & Wherry, E. J. Genetic absence of PD-1 promotes accumulation of terminally differentiated exhausted CD8+ T cells. J. Exp. Med. 212, 1125–1137 (2015).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Doedens, A. L. et al. Hypoxia-inducible factors enhance the effector responses of CD8+ T cells to persistent antigen. Nat. Immunol. 14, 1173–1182 (2013).
Frebel, H. et al. Programmed death 1 protects from fatal circulatory failure during systemic virus infection of mice. J. Exp. Med. 209, 2485–2499 (2012).
Pircher, H., Bürki, K., Lang, R., Hengartner, H. & Zinkernagel, R. M. Tolerance induction in double specific T-cell receptor transgenic mice varies with antigen. Nature 342, 559–561 (1989).
Dillon, S. R., Jameson, S. C. & Fink, P. J. Vβ5+ T cell receptors skew toward OVA + H-2Kb recognition. J. Immunol. 152, 1790–1801 (1994).
Puglielli, M. T. et al. In vivo selection of a lymphocytic choriomeningitis virus variant that affects recognition of the GP33-43 epitope by H-2Db but not H-2Kb. J. Virol. 75, 5099–5107 (2001).
Wlodarczyk, M. F., Kraft, A. R., Chen, H. D., Kenney, L. L. & Selin, L. K. Anti-IFN-γ and peptide-tolerization therapies inhibit acute lung injury induced by cross-reactive influenza A-specific memory T cells. J. Immunol. 190, 2736–2746 (2013).
Chen, H. D., Fraire, A. E., Joris, I., Welsh, R. M. & Selin, L. K. Specific history of heterologous virus infections determines anti-viral immunity and immunopathology in the lung. Am. J. Pathol. 163, 1341–1355 (2003).
Cornberg, M. et al. Clonal exhaustion as a mechanism to protect against severe immunopathology and death from an overwhelming CD8 T cell response. Front. Immunol. 4, 475 (2013).
Nitschke, K. et al. Tetramer enrichment reveals the presence of phenotypically diverse hepatitis C virus-specific CD8+ T cells in chronic infection. J. Virol. 89, 25–34 (2015).
Eisenberg, V. et al. Targeting multiple tumors using T-cells engineered to express a natural cytotoxicity receptor 2-based chimeric receptor. Front. Immunol. 8, 1212 (2017).
Tal, Y. et al. An NCR1-based chimeric receptor endows T-cells with multiple anti-tumor specificities. Oncotarget 5, 10949–10958 (2014).
Thurber, S. E., Khong, H. T., Kammula, U. S. & Rosenberg, S. A. Identification of endogenous HLA-A2-restricted reactivity against shared melanoma antigens in patients using the quantitative real-time polymerase chain reaction. J. Immunother. 25, 63–71 (2002).
Carey, T. E., Takahashi, T., Resnick, L. A., Oettgen, H. F. & Old, L. J. Cell surface antigens of human malignant melanoma: mixed hemadsorption assays for humoral immunity to cultured autologous melanoma cells. Proc. Natl Acad. Sci. USA 73, 3278–3282 (1976).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Silver, J. D., Ritchie, M. E. & Smyth, G. K. Microarray background correction: maximum likelihood estimation for the normal–exponential convolution. Biostatistics 10, 352–363 (2009).
Bolstad, B. M., Irizarry, R. A., Astrand, M. & Speed, T. P. A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19, 185–193 (2003).
Köster, J. & Rahmann, S. Snakemake—a scalable bioinformatics workflow engine. Bioinformatics 28, 2520–2522 (2012).
Andrews, S. FastQC: a quality control tool for high throughput sequence data. Version 0.11.6 http://www.bioinformatics.babraham.ac.uk/projects/fastqc (2017).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Anders, S., Pyl, P. T. & Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Ewels, P., Magnusson, M., Lundin, S. & Käller, M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–3048 (2016).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 289–300 (1995).
Kolde, R. pheatmap: Pretty Heatmaps. R package version 1.0.10 https://cran.r-project.org/web/packages/pheatmap/index.html (2012)
Zhang, Y. et al. Model-based analysis of ChIP–seq (MACS). Genome Biol. 9, R137 (2008).
Trinh, B. N., Long, T. I. & Laird, P. W. DNA methylation analysis by MethyLight technology. Methods 25, 456–462 (2001).
Youngblood, B. et al. Effector CD8 T cells dedifferentiate into long-lived memory cells. Nature 552, 404–409 (2017).
Xi, Y. & Li, W. BSMAP: whole genome bisulfite sequence MAPping program. BMC Bioinformatics 10, 232 (2009).
Feng, H., Conneely, K. N. & Wu, H. A Bayesian hierarchical model to detect differentially methylated loci from single nucleotide resolution sequencing data. Nucleic Acids Res. 42, e69 (2014).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Acknowledgements
We thank M. J. Bevan and M. Prlic for input, feedback and suggestions; T. Herbinger and B. Dötterböck; W. Schmid and C. Amette for technical assistance; and S. Schleicher and C. Lechner for animal husbandry. Work in the D.Z. laboratory was supported by a ‘European Research Council starting grant’ (ProtecTC) and subsequently a ‘European Research Council consolidator grant’ (ToCCaTa), grants from the Swiss National Science Foundation (CRSII3_160708, 310030E-164187, 51PHP0_157319 and PP00P3_144883), the Swiss Vaccine Research Institute (SVRI) and grants from the German Research Foundation (SFB1054). D.Z. and C.J.C. are supported by a German-Israeli Foundation for Scientific Research and Development (GIF) grant (I-1440-414.13/2017). K.S. is supported by German Research Foundation grants (SFB824 and SFB1335). R.T. and M.H. are funded by a German Research Foundation grant (TRR179-TP01). B.Y. is supported by the NIH (R01AI114442) and the American Lebanese Syrian Associated Charities (ALSAC). P.A.K. is supported by the Germany Center for Infection Research Munich site and the CRC TRR179.
Reviewer information
Nature thanks Axel Kallies and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
D.Z. and F.A. initiated the study; B.Y., D.Z. and F.A. planned and designed the core experiments; F.A. performed the core experiment in mice; M.v.H. performed the immunohistochemistry experiments and related graphical analyses; D.T.U. performed the experiments related to the microarray data; K.S. performed the histopathological analysis; and K.K. performed next-generation sequencing. C.J.C. and V.E. contributed the tumour-related studies in humans, M.H. and R.T. carried out the HCV-related work; B.Y., H.E.G. and Y.F. provided the epigenetics work and data analysis. M.W., P.R., F.A., M.D. and K.K. performed or supervised the bioinformatics analysis. D.W., D.M., P.A.K. and J.G.C. provided critical expertise and contributed specific analysis. The manuscript was written by B.Y., D.Z. and F.A., with contributions from R.T. and P.A.K.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 TOX expression is robustly linked to TCR signalling and the presence or absence of an exhausted phenotype.
a, C57BL/6 mice received P14 T cells and were infected with 2 × 106 PFU wild-type clone 13 or 0.66 × 106 PFU wild-type clone 13 mixed with 1.33 × 106 PFU gp33-deficient LCMV clone 13. P14 T cells were re-isolated 28 days later and sorted by flow cytometry. Transcriptional profiles were determined using RNA microarrays. Lines in the volcano plots indicate thresholds of log2 = ± 0.5 and P = 0.05 thresholds; genes with expression levels above or below these values are coloured and their numbers are depicted. Genes with |log2| ≥ 2 (shown as log2(FC)) and P ≤ 0.05 are listed on the right. b, C57BL/6 mice received P14 T cells and were infected with LCMV Armstrong or clone 13. P14 T cells were re-isolated 28 days later, transferred into naive host mice, and the secondary hosts were infected with LCMV Armstrong. Both types of donor P14 T cells were analysed 8 days later for differential gene expression as described in a. Microarray analyses were performed with n = 5 mice infected with wild-type clone 13 (a), n = 4 mice for all other treatments (a, b). c, Datasets from a and b were filtered for average expression >6, |log2| ≥ 0.5 and P ≤ 0.05. The Venn diagram represents the overlap between the filtered genes sets and the graph illustrates the fold change in expression that these genes have in both datasets. This reveals that almost all overlapping DEGs are synchronously expressed and either correlated or anti-correlated with a dysfunctional phenotype. d, e, Thymocytes from 5-week-old C57BL/6 mice (n = 4 mice) were stained for CD4, CD8, CD5, CD69 and TOX. Cells were preselected into CD8brightCD4bright or CD8dullCD4dull populations and further separated into CD5highCD69high and CD5lowCD69low populations. The representative histograms and graphs, which include data for all analysed mice, show TOX expression in CD8brightCD4brightCD5lowCD69low, CD8dullCD4dullCD5lowCD69low and CD8dullCD4dullCD5highCD69high cells. The latter group represents cells after positive selection, which show higher TOX levels. Data represent two independent experiments. Representative histograms are shown in d, and data for all mice (n = 4 mice) are shown in e. f, Analysis of TOX locus methylation obtained from previously published whole-genome bisulfite-sequencing data8,52 of naive, day 7 KLRG1highCD127low (TE), day 7 KLRG1lowCD127high (MP) and day 30 memory P14 T cells isolated from hosts infected with LCMV Armstrong; and day 8 (early) or 35 (late) P14 T cells isolated from hosts infected with clone 13. Vertical lines indicate CpG positions, red and black indicate the fraction of unmethylated versus methylated reads.
Extended Data Fig. 2 Wild-type and TOX-mutated T cells become similarly activated, yet show distinct phenotypes even with high titre LCMV infections, but this difference arises only following early, but not late, TOX disruption.
a, Adoptively transferred P14 T cells and total endogenous T cells were stained for TOX and KLRG1 expression 8 days after infection with LCMV Armstrong (Arm; n = 5 mice) or clone 13 (n = 5 mice). b, Extension of the data shown in Fig. 1d. Histograms represent TOX and PD-1 expression among HCV-specific tetramer+ T cells in patients with chronic, pharmacologically cured or spontaneously resolved HCV infection as well as in control influenza memory T cells. Representative histograms are shown (number of samples are indicated in Fig. 1d). c, d, Toxflox/floxCD4creRosa26-STOP-eYFP P14 (n = 3 mice) and CD4creRosa26-STOP-eYFP P14 (n = 4 mice) were directly infected with clone 13. Representative histograms and graphs with data from all analysed mice show CD69 (c) and PD-1 (d) expression on splenocytes 2.5 days after infection. e–j, C57BL/6 (B6; i, j) or Vβ5 hosts (e–h) received wild-type or TOX∆ P14 T cells and were infected with LCMV clone 13 or Armstrong and analysed as indicated on day 7, 8 or 20 of infection. Representative histograms or dot plots and corresponding graphs for all mice in one of two (or three (e, j)) experiments show wild-type (throughout in dark blue) and TOX∆ (in light blue) P14 T cells isolated from the spleen and analysed for PD-1 (e, f) and KLRG1 (g) expression (n = 4 mice in all groups except for day 8 TOX∆ P14 T cells for which n = 5 mice, grey shades are total host cells), for TNF and INFγ expression (h, i) after ex vivo gp33 re-stimulation (n = 5 mice for day 8 TOX∆ P14 in Vβ5 hosts and for wild-type and TOX∆ in C57BL/6 hosts, n = 4 mice for all other groups) or for GzmB levels 8 days after infection (j; n = 4 mice for Armstrong and n = 5 mice for clone 13). k–o, TOXlox/loxGzmbcreERT2 (n = 4 mice) and GzmbcreERT2 (n = 5 mice) mice received clone 13 and 2 mg tamoxifen on days 20–25 after infection, which specifically inactivates TOX in GzmB-expressing T cells. Representative histograms or dot plots and corresponding graphs for all mice in one of two experiments show day 35 splenic wild-type (in dark blue) and TOX∆ CD8 T cells (in light blue) analysed for the frequency of gp33-tetramer+ cells (l), the length of a DNA fragment amplified by PCR from gp33-tetramer+ cells, which includes the fifth exon of Tox, note that the fifth exon covers 231 bp (m), and PD-1 (n) and KLRG1 (o) expression on gp33-tetramer+ T cells. Symbols in all data plots and n values in the legend indicate number of mice, horizontal bars show the mean; P values originate from two-tailed unpaired t-tests.
Extended Data Fig. 3 TOX affects the expression of a large number of genes that are tightly linked to a dysfunctional phenotype, whereas TOX disruption has a minor effect during acute infections.
Extension of the dataset shown in Fig. 2e, wild-type (n = 5 mice) and TOX∆ (n = 5 mice) P14 T cells were transferred into C57BL/6 hosts and infected with LCMV clone 13 or Armstrong. Cells were isolated 8 or 20 days later and analysed by next-generation sequencing. a, Volcano plots comparing wild-type and TOX∆ P14 T cells isolated from hosts infected with clone 13 after 8 or 20 days, lines indicate filter criteria of log2 ± 1 and adjusted P = 0.05; genes upregulated in wild-type (orange) or ∆TOX (green) are highlighted and numbers above the thresholds are indicated. b, z-scores (DESeq2 package) depicting heat maps are a supervised selection of molecules. Wald statistics and Benjamini and Hochberg corrections were used for data analysis. c, Venn diagrams showing the overlap between three DEG lists defined by filtering the data shown in Extended Data Fig. 1a (dysfunctional versus normal T cells; red) and Extended Data Fig. 1b (re-expanded dysfunctional versus re-expanded memory T cells; blue) for average expression >6, P < 0.05 and |log2(WT/TOX∆)| ≥ 0.3 and the dataset shown in a, b for mean expression >50, P < 0.05 and |log2(WT/TOX∆)| ≥ 0.3. Numbers depict the DEG overlap between the datasets. d, The datasets from a and Extended Data Fig. 1a were filtered as indicated in c and loaded into the IPA software (Qiagen). Build-in algorithms (core analysis) were used to identify regulators that are linked to these datasets. Note that these analyses were performed independently for both datasets. Yet, there was a >60% overlap among the top-50 ranked regulators that were identified for both datasets (data not shown). The graph shows the regulators that are shared between both datasets and depicts the P values that are assigned to the regulator in the two datasets (x axis data are linked to Extended Data Fig. 1a, y axis data are linked to a and Fig. 2e). Highlighted in colour are regulators with P ≤ 10−8 in both datasets. e, The number of genes that are linked to the regulator identified for the dataset shown in Extended Data Fig. 1a (dysfunctional versus normal T cells) and the fraction of the linked genes that are differentially expressed in ∆TOX T cells. f, The Venn diagram is derived from c and compares only the datasets shown in a, Extended Data Fig. 1a and Fig. 2e. The 269 overlapping DEGs were imported into IPA and a core analysis was performed. Plots show the P values for the associated upstream transcription factors or transcription regulators (jointly referred to as TR) plus the number of genes linked to these molecules. g, Day 8 LCMV Armstrong gene expression profiles as explained for a and b.
Extended Data Fig. 4 Overexpression of TOX in human and mouse T cells augments PD-1 expression, restricts cytokine production and promotes acquisition of a dysfunctional phenotype, whereas absence of TOX exacerbates T-cell-mediated immunopathology.
a, Representative histograms (top) and the percentage of PD-1 expressing cells (bottom) on ex vivo activated TOX or control transduced T cells from healthy donors, the P value originates from two-tailed unpaired t-tests, n = 5 donor, all data are shown. b, Similar to a, TOX- and TCR-transduced T cells (TOX group) or TCR-only transduced T cells (Ctrl group) were co-cultured with SK-Mel-23 (grey) or 888-A2 (orange) cells for 16 h and TNF and IFNγ production in the supernatant was measured using specific ELISAs. Baseline cytokine production following co-culture with antigen-negative T cells was subtracted. Lines combine samples from the same donor with TOX or control overexpression. Two-tailed paired t-tests comparing cytokine levels for all donors were performed, all data are shown. c–f, P14 T cells were transduced with empty (dark green) or TOX-encoding retroviruses (light green), transferred into Vβ5 mice (n = 4 mice for both conditions). Hosts were infected with a mix of 0.5 × 106 PFU LCMV wild-type clone 13 and 4.5 × 106 gp-33-deficient clone 13. Representative histograms and graphs depicting data from all mice in one of two similar experiments show splenocytes collected 21 days after infection and analysed for GFP and TOX expression, GFP low and high gates illustrate the direct correlation between transduction efficacy and TOX expression (c), TNF (top) and IFNγ (bottom) production following ex vivo gp33 re-stimulation (d), PD-1 and KLRG1 (e), and TCF-1 (f) expression levels. Two-tailed unpaired t-tests were performed to calculate P values. g–k, Vβ5 and C57BL/6 mice received wild-type or ∆TOX P14 T cells and were infected with clone 13. Day 7 virus titres in liver and kidney of Vβ5 hosts (g, n = 6 mice) and representative day-11 tissue sections of the lungs (h) or liver (i) from C57BL/6 hosts are shown. Scale bars, 50 μm. Blue dashed lines mark the periportal regions with large cellular infiltrates in wild-type p14 recipients and the green dotted lines highlight necrotic zones found only in mice that received TOX∆ T cells. j, k, Semi-quantitative assessments of lung pathology scoring (j) and liver necrosis (Ishak score, k). A single experimental setup is presented in g–k, with n = 5 mice for wild-type and n = 6 mice for TOXΔ, all symbols are data for individual mice or human donors; solid horizontal lines represent the mean.
Extended Data Fig. 5 TOXΔ causes similar phenotypic and kinetics alterations in high- and low-affinity T cells in chronic infections, but it does not affect T cell differentiation in acute infections.
a–c, C57BL/6 mice were engrafted with wild-type (dark colour) and TOX∆ (light colour) P14 T cells and infected with wild-type LCMV clone 13 expressing the normal high-affinity peptide ligand (blue colour) or a low-affinity altered peptide ligand (C6, red colour) for P14 T cells17 (n = 5 mice in all groups except for C6 TOX∆, in which n = 4 mice). Blood was collected 7 or 13 days after infection and analysed for the ratios of TOX∆ and wild-type P14 T cells at both time points (a) and PD-1 (b) and KLRG1 (c) expression 13 days after infection. Representative histograms and plots depict data from all mice. d–f, C57BL/6 hosts received wild-type (dark blue) and TOX∆ (light blue, both n = 5 mice) P14 T cells and were infected with LCMV Armstrong. A fraction of the cells recovered from the spleen 30 days after infection were analysed and the remaining cells were transferred into naive C57BL/6 hosts and re-expanded by another LCMV Armstrong infection as depicted in the experimental schematic in d. Representative histograms and plots depicting data from all mice show KLRG1, CD62L and CD127 expression 30 days after infection (e) and absolute numbers of TCF-1+ P14 T cells in the primary hosts and magnitude of re-expansion of the transfer cells in the second host, the calculation assumes a 10% transfer efficacy (f). g, Extension of the dataset shown in Fig. 3b with wild-type (dark blue) and TOX∆ (light blue, both n = 5) P14 T cells in hosts infected with LCMV clone 13. The mean fluorescence intensity of TCF-1+ P14 T cells is shown. All data are representative of two (or three (g)) independent experiments. Two-tailed unpaired t-tests were performed. Symbols in graphs and n values in the legend are data for individual mice; solid horizontal lines represent the mean. ns, not significant.
Extended Data Fig. 6 Tox-deficient populations show similar splenic localization as wild-type T cells and on a per-cell basis have similar proliferation activity.
C57BL/6 mice were engrafted with wild-type or TOX∆ P14 T cells and infected with LCMV clone 13. Localization of the engrafted P14 T cells was determined 8 days (a; n = 5 mice for wild-type, n = 3 mice for TOX∆) and 14 days (b; n = 3 mice for wild-type, n = 4 mice for TOX∆) after infection. Sections were stained with anti-B220 (blue), anti-CD3 (green) and anti-CD45.1 (red) antibodies. Images of spleen sections were acquired using a fluorescence microscope. Top, representative microscopy images of spleen sections. Middle, a density threshold-based algorithm was used to vectorize the whole section and to identify the red pulp and separate the white pulp into the T and B cell zone. Dark grey represents the B cell zone, light grey the T cell zone, the clear background the red pulp. Red dots mark the location of individual P14 T cells. Bottom, relative distribution of wild-type and TOX∆ P14 T cells in the different anatomical locations. A single experiment is presented. c, C57BL/6 hosts received wild-type (dark blue) and TOX∆ P14 T cells (light blue, n = 5 mice in both groups) and were infected with LCMV clone 13. After 13 days, an EdU bolus was intraperitoneally injected and splenic P14 T cells were collected 6 h later and analysed for EdU incorporation. Representative flow cytometry plots and data from all mice in one of two representative experiments show the frequency of EdU+ P14 T cells. Symbols in graphs and n values in the legend are individual mice; solid horizontal lines represent the mean. Two-tailed unpaired t-tests were performed to calculate P values.
Extended Data Fig. 7 Absence of TOX alters genes expression preferentially in the TIM-3− population in the early infection phase.
a, b, Extension of the data shown in Fig. 4e; transferred wild-type and TOX∆ P14 T cells were isolated from C57BL/6 hosts 8 days after infection with clone 13, sorted into TIM-3+ (a) and TIM-3− (b) populations, and analysed for differential gene expression (n = 5 mice). Lines in the volcano indicate thresholds of log2(WT/TOX∆) ± 1 and adjusted P = 0.05; orange, genes upregulated in wild-type; green, upregulated in ∆TOX; numbers represent genes above the threshold. Heat maps include a supervised selection of molecules and show differential gene expression z-scores (DESeq2 package). Wald statistics and Benjamini and Hochberg corrections were used for data analysis. c, DEG list defined by MeanExpression >50, P < 0.05 and |log2(WT/TOX∆)| ≥ 0.5 was determined for the TIM-3− TOX∆ versus wild-type P14 T cells dataset and submitted to IPA core analysis. Depicted in decreasing order are the top pathways determined by the software. Details are shown for the ‘cellular growth and proliferation’ and ‘cell death and survival’ pathways. Orange, increased activity in TOX∆ compared to wild-type P14 T cells; blue, decreased activity in TOX∆ compared with wild-type P14 T cells. d, IPA was used to build a regulatory network connecting the top 30 transcriptional regulators identified in Extended Data Fig. 3f. Colours indicate differential expression of these regulators comparing TIM-3− wild-type versus TOX∆ (left network) and TIM-3+ wild-type versus TOX∆ (righ network). Red, upregulation; green, downregulation; white, unchanged or not detected. The data show that more regulators are differentially expressed in the TIM-3− than the TIM-3+ population. Data from a single measurement with n = 5 donor mice are shown.
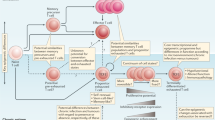
Extended Data Fig. 8 Proposed dynamics of wild-type and Tox-deficient antigen-specific T cell populations in chronic infection.
We consider that an initial wave of effector-committed T cells acquire certain signs of dysfunction independently of TOX. However, absence of TOX results in a failure to epigenetically enforce a dysfunctional phenotype in the critical TCF-1-expressing (TIM-3−) memory-like population. The over-time-increasing phenotypic changes at the level of the entire population are then the consequence of the continuous replacement of the effector-committed TCF-1− T cells from the improperly programmed TCF-1+ population. This process happen in parallel to the decline of the total population caused by decreased absolute numbers of TCF-1+ T cells in the absence of TOX.
Extended Data Fig. 9 Tox-deficient T cells show largely altered global methylation pattern and retain a methylation signature that is reminiscent of naive T cells.
a–f, C57BL/6 mice engrafted with wild-type (n = 3 mice) or TOX∆ (n = 3 mice) P14 T cells were infected with clone 13. On day 8 after infection, splenic wild-type and TOX∆ P14 T cells were sorted into TIM-3+ and TIM-3− populations and whole-genome DNA methylation analysis was performed after pooling cells from the three donor mice. a, Heat map showing cluster analysis of the top 3,000 differentially methylated regions (DMRs) between TOX∆ and wild-type P14 T cells. Colour intensity scales from red (methylated region) to blue (unmethylated region). b, Absolute number of methylated DMRs of TOX∆ and wild-type P14 T cells (left, red) or demethylated DMRs (green, right) population. c, Venn diagrams show the absolute number of overlapping DMRs that are more methylated in P14 TOX∆ and in naive P14 T cells relative to wild-type P14 on day 8 after infection for the TIM-3+ (left) or TIM-3− (right) population. d, Correlation analysis of DMRs and DEGs on day 8 after infection for TIM-3+ TOX∆ and wild-type P14 T cells (left) and for TIM-3− TOX∆ and wild-type P14 T cells (right). The DMR values from a, b, are plotted against matching data from the differential gene expression analysis shown in Fig. 4. e, f, Pathways from the NCI-Nature Pathway Interaction Database55 that are significantly enriched among genes undergoing DNA demethylation (>20% change in DNA methylation frequency) in the TIM-3+ (e) or TIM-3− (f) subsets of wild-type versus TOX∆ antigen-specific CD8 T cells isolated on day 8 after chronic LCMV infection. Data are derived from a single experiment.
Extended Data Fig. 10 Global and locus-specific chromatin accessibility difference in the absence of TOX.
C57BL/6 mice are engrafted with either TOX∆ (n = 3 mice) or wild-type (n = 3 mice) P14 T cells before infection with LCMV clone 13. Splenocytes are collected 8 and 13 days after infection and ATAC sequencing was performed as indicated in the Methods. The experiment was done once, samples were merged to obtain a better resolution. a, Summary of the numbers of differentially open chromatin regions (OCRs) among TOX∆ versus wild-type P14 T cells on day 8 (left) and 13 (right) after infection. b, Genome browser snapshots of ATAC-seq peak intensity in the Pdcd1 (top) and Tnf (bottom) loci for TOX∆ (pink) or wild-type (black) P14 T cells. Notable differences in chromatin accessibility are highlighted by red boxes. Note, open chromatin region is defined as a genomic region with differential chromatin accessibility of |log2(TOX∆/WT)| ≥ 2 and |log2(TOX∆/WT)| ≥ 1 adjusted P ≤ 0.05 as a cut-off. c, d, Biocarta pathways that are significantly enriched among genes with ATAC-seq peaks that gained (c; green) or lost (d; red) chromatin accessibility (|log2(WT/TOX∆)| ≥ 1) in TOX∆ versus wild-type antigen-specific CD8 T cells.
Supplementary information
Supplementary Figure
This file contains the uncropped blot shown in Extended Data Fig. 2.
Rights and permissions
About this article
Cite this article
Alfei, F., Kanev, K., Hofmann, M. et al. TOX reinforces the phenotype and longevity of exhausted T cells in chronic viral infection. Nature 571, 265–269 (2019). https://doi.org/10.1038/s41586-019-1326-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-019-1326-9
This article is cited by
-
Microfluidics-enabled fluorinated assembly of EGCG-ligands-siTOX nanoparticles for synergetic tumor cells and exhausted t cells regulation in cancer immunotherapy
Journal of Nanobiotechnology (2024)
-
FOXP1 and KLF2 reciprocally regulate checkpoints of stem-like to effector transition in CAR T cells
Nature Immunology (2024)
-
Transcription factors in chimeric antigen receptor T-cell development
Human Cell (2024)
-
Intra-tumoral T cells in pediatric brain tumors display clonal expansion and effector properties
Nature Cancer (2024)
-
An oncolytic virus delivering tumor-irrelevant bystander T cell epitopes induces anti-tumor immunity and potentiates cancer immunotherapy
Nature Cancer (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.