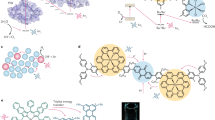
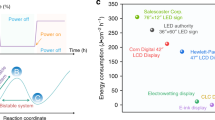
Abstract
Lighting based on light-emitting diodes (LEDs) not only is more energy efficient than traditional lighting, but also enables improved performance and control. The colour, intensity and distribution of light can now be controlled with unprecedented precision, enabling light to be used both as a signal for specific physiological responses in humans and plants, and as an efficient fuel for fresh food production. Here we show how a broad and improved understanding of the physiological responses to light will facilitate greater energy savings and provide health and productivity benefits that have not previously been associated with lighting.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Parker, A. In the Blink of an Eye: How Vision Sparked the Big Bang of Evolution (Basic Books, New York, 2003).
Gerkema, M. P., Davies, W. I., Foster, R. G., Menaker, M. & Hut, R. A. The nocturnal bottleneck and the evolution of activity patterns in mammals. Proc. R. Soc. Lond. B 280, 20130508 (2013).
Gregory, R. L. Eye and Brain: The Psychology of Seeing (Princeton Univ. Press, Princeton, 2015).
Lucas, R. J. et al. Measuring and using light in the melanopsin age. Trends Neurosci. 37, 1–9 (2014). A summary of the neurophysiology of the melanopsin ipRGC sensory pathway and of the implications for the measurement, production and application of light (includes a free measurement tool to calculate the photoreceptive inputs for circadian, neuroendocrine and neurobehavioral responses).
Figueiro, M. G., Brainard, G. C., Lockley, S. W., Revell, V. L. & White, R. Light and Human Health: An Overview of the Impact of Optical Radiation on Visual, Circadian, Neuroendocrine and Neurobehavioral Responses. Technical Memorandum IES TM-18-08 (Illuminating Engineering Society, 2008).
Bowers, B. & Anastas, P. Lengthening the Day: A History of Lighting Technology (Oxford Univ. Press, Oxford, 1998).
Boyce, P. R. Human factors in lighting (CRC Press, Boca Raton, 2014).
Schivelbusch, W. Disenchanted Night: The Industrialization of Light in the Nineteenth Century (Univ. California Press, Berkeley, 1995).
Steinmetz, C. P. Radiation, Light and Illumination: A Series of Engineering Lectures Delivered at Union College (McGraw-Hill, New York, 1918).
Tsao, J. Y., Han, J., Haitz, R. H. & Pattison, P. M. The blue LED Nobel prize: historical context, current scientific understanding, human benefit. Ann. Phys. 527, A53–A61 (2015). A succinct discussion of the Nobel-prize-winning breakthroughs that led to blue-LED and LED lighting and of the context of these breakthroughs in the history of semiconductor science and technology.
US DOE SSL Program. Solid-State Lighting R&D Plan. https://www.energy.gov/sites/prod/files/2018/09/f56/ssl_rd-plan_jun2016.pdf (2016).
Krames, M. R. et al. Status and future of high-power light-emitting diodes for solid-state lighting. J. Disp. Technol. 3, 160–175 (2007).
Tsao, J. Y. & Waide, P. The world’s appetite for light: Empirical data and trends spanning three centuries and six continents. Leukos 6, 259–281 (2010).
US DOE SSL Program. Energy Savings Forecast of Solid-State Lighting in General Illumination Applications. https://energy.gov/sites/prod/files/2016/09/f33/energysavingsforecast16_2.pdf (2016).
Schubert, E. F. & Kim, J. K. Solid-state light sources getting smart. Science 308, 1274–1278 (2005). The first paper to discuss the potential of solid-state lighting to be ‘smart’, in the sense of being able not only to provide energy savings but also to adjust to the specific environments and requirements of a wide range of applications.
Tsao, J. Y. et al. Toward smart and ultra-efficient solid-state lighting. Adv. Opt. Mater. 2, 809–836 (2014). A comprehensive review of the state of solid-state lighting in terms of its ultimate potential to be both ‘smart’ and ultra-efficient.
Watson, B. From light to bright: San Diego is building the world’s largest municipal Internet of Things. GE Reports https://www.ge.com/reports/light-bright-san-diego-leads-way-future-smart-cities/ (2017).
Tsao, J. Y., Schubert, E. F., Fouquet, R. & Lave, M. The electrification of energy: long-term trends and opportunities. MRS Energy Sustain. 5, E7 (2018).
US DOE SSL Program. Solid-State Lighting 2017 Suggested Research Topics Supplement: Technology and Market Context. https://energy.gov/sites/prod/files/2017/09/f37/ssl_supplement_suggested-topics_sep2017_0.pdf (2017).
Dijk, D. J. & von Schantz, M. Timing and consolidation of human sleep, wakefulness, and performance by a symphony of oscillators. J. Biol. Rhythms 20, 279–290 (2005).
Thapan, K., Arendt, J. & Skene, D. J. An action spectrum for melatonin suppression: evidence for a novel non-rod, non-cone photoreceptor system in humans. J. Physiol. 535, 261–267 (2001).
Brainard, G. C. et al. Action spectrum for melatonin regulation in humans: evidence for a novel circadian photoreceptor. J. Neurosci. 21, 6405–6412 (2001).
Brainard, G. C. & Hanifin, J. P. Photons, clocks, and consciousness. J. Biol. Rhythms 20, 314–325 (2005).
Provencio, I. et al. A novel human opsin in the inner retina. J. Neurosci. 20, 600–605 (2000).
Gooley, J. J., Lu, J., Chou, T. C., Scammell, T. E. & Saper, C. B. Melanopsin in cells of origin of the retinohypothalamic tract. Nat. Neurosci. 4, 1165 (2001).
Berson, D. M., Dunn, F. A. & Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 295, 1070–1073 (2002).
Hattar, S., Liao, H. W., Takao, M., Berson, D. M. & Yau, K. W. Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 295, 1065–1070 (2002).
Provencio, I., Jiang, G., De Grip, W. J., Hayes, W. P. & Rollag, M. D. Melanopsin: an opsin in melanophores, brain, and eye. Proc. Natl Acad. Sci. USA 95, 340–345 (1998). A landmark paper that details the discovery of melanopsin, ultimately leading to melanopsin being identified as a functional photopigment in the retinas of mammals, including humans, with roles in the regulation by light of circadian, neuroendocrine, neurobehavioral and visual responses.
Hannibal, J. et al. Central projections of intrinsically photosensitive retinal ganglion cells in the macaque monkey. J. Comp. Neurol. 522, 2231–2248 (2014).
Cajochen, C., Khalsa, S. B. S., Wyatt, J. K., Czeisler, C. A. & Dijk, D. J. EEG and ocular correlates of circadian melatonin phase and human performance decrements during sleep loss. Am. J. Physiol. 277, R640–R649 (1999).
Cajochen, C., Zeitzer, J. M., Czeisler, C. A. & Dijk, D. J. Dose-response relationship for light intensity and ocular and electroencephalographic correlates of human alertness. Behav. Brain Res. 115, 75–83 (2000).
Cajochen, C. et al. High sensitivity of human melatonin, alertness, thermoregulation, and heart rate to short wavelength light. J. Clin. Endocrinol. Metab. 90, 1311–1316 (2005).
Lockley, S. W. et al. Short-wavelength sensitivity for the direct effects of light on alertness, vigilance, and the waking electroencephalogram in humans. Sleep 29, 161–168 (2006).
Wright, K. P. Jr, Badia, P., Myers, B. L. & Plenzler, S. C. Combination of bright light and caffeine as a countermeasure for impaired alertness and performance during extended sleep deprivation. J. Sleep Res. 6, 26–35 (1997).
Chang, A. M., Scheer, F. A., Czeisler, C. A. & Aeschbach, D. Direct effects of light on alertness, vigilance, and the waking electroencephalogram in humans depend on prior light history. Sleep 36, 1239–1246 (2013).
Rüger, M., Gordijn, M. C., Beersma, D. G., de Vries, B. & Daan, S. Weak relationships between suppression of melatonin and suppression of sleepiness/fatigue in response to light exposure. J. Sleep Res. 14, 221–227 (2005).
Phipps-Nelson, J., Redman, J. R., Dijk, D. J. & Rajaratnam, S. M. Daytime exposure to bright light, as compared to dim light, decreases sleepiness and improves psychomotor vigilance performance. Sleep 26, 695–700 (2003).
Cajochen, C. et al. Evening exposure to a light-emitting diodes (LED)-backlit computer screen affects circadian physiology and cognitive performance. J. Appl. Physiol. 110, 1432–1438 (2011).
Rahman, S. A., St Hilaire, M. A. & Lockley, S. W. The effects of spectral tuning of evening ambient light on melatonin suppression, alertness and sleep. Physiol. Behav. 177, 221–229 (2017).
Segal, A. Y., Sletten, T. L., Flynn-Evans, E. E., Lockley, S. W. & Rajaratnam, S. M. Daytime exposure to short- and medium-wavelength light did not improve alertness and neurobehavioral performance. J. Biol. Rhythms 31, 470–482 (2016).
Sletten, T. L. et al. Randomised controlled trial of the efficacy of a blue-enriched light intervention to improve alertness and performance in night shift workers. Occup. Environ. Med. 74, 792–801 (2017).
Lam, R. W. & Tam, E. M. A Clinician’s Guide to Using Light Therapy (Cambridge Univ. Press, New York, 2009).
Wirz-Justice, A., Benedetti, F., Terman, M. & Basel, S. Chronotherapeutics for affective disorders: a clinician’s manual for light and wake therapy. Ann. Clin. Psychiatry 22, 67 (2010).
Brainard, G. C., Barger, L. K., Soler, R. R. & Hanifin, J. P. The development of lighting countermeasures for sleep disruption and circadian misalignment during spaceflight. Curr. Opin. Pulm. Med. 22, 535–544 (2016).
Gooley, J. J., et al. Spectral responses of the human circadian system depend on the irradiance and duration of exposure to light. Sci. Transl. Med. 2, 31ra33 (2010).
Lall, G. S. et al. Distinct contributions of rod, cone, and melanopsin photoreceptors to encoding irradiance. Neuron 66, 417–428 (2010).
Altimus, C. M. et al. Rod photoreceptors drive circadian photoentrainment across a wide range of light intensities. Nat. Neurosci. 13, 1107–1112 (2010).
Dacey, D. M. et al. Melanopsin-expressing ganglion cells in primate retina signal colour and irradiance and project to the LGN. Nature 433, 749–754 (2005).
Ecker, J. L. et al. Melanopsin-expressing retinal ganglion-cell photoreceptors: cellular diversity and role in pattern vision. Neuron 67, 49–60 (2010).
Brown, T. M., Wynne, J., Piggins, H. D. & Lucas, R. J. Multiple hypothalamic cell populations encoding distinct visual information. J. Physiol. 589, 1173–1194 (2011).
Brown, T. M. et al. Melanopsin-based brightness discrimination in mice and humans. Curr. Biol. 22, 1134–1141 (2012).
Estevez, M. E. et al. Form and function of the M4 cell, an intrinsically photosensitive retinal ganglion cell type contributing to geniculocortical vision. J. Neurosci. 32, 13608–13620 (2012).
Zaidi, F. H. et al. Short-wavelength light sensitivity of circadian, pupillary, and visual awareness in humans lacking an outer retina. Curr. Biol. 17, 2122–2128 (2007).
Zele, A. J., Feigl, B., Adhikari, P., Maynard, M. L. & Cao, D. Melanopsin photoreception contributes to human visual detection, temporal and colour processing. Sci. Rep. 8, 3842 (2018).
Horiguchi, H., Winawer, J., Dougherty, R. F. & Wandell, B. A. Human trichromacy revisited. Proc. Natl Acad. Sci. USA 110, E260–E269 (2013).
Spitschan, M., Datta, R., Stern, A. M., Brainard, D. H. & Aguirre, G. K. Human visual cortex responses to rapid cone and melanopsin-directed flicker. J. Neurosci. 36, 1471–1482 (2016).
Zhao, X., Stafford, B. K., Godin, A. L., King, W. M. & Wong, K. Y. Photoresponse diversity among the five types of intrinsically photosensitive retinal ganglion cells. J. Physiol. 592, 1619–1636 (2014).
Prigge, C. L. et al. M1 ipRGCs influence visual function through retrograde signaling in the retina. J. Neurosci. 36, 7184–7197 (2016).
Hannibal, J., Christiansen, A. T., Heegaard, S., Fahrenkrug, J. & Kiilgaard, J. F. Melanopsin expressing human retinal ganglion cells: Subtypes, distribution, and intraretinal connectivity. J. Comp. Neurol. 525, 1934–1961 (2017).
Brainard, G. C. & Hanifin, J. P. Handbook of Advanced Lighting Technology (Springer, Berlin, 2017).
Stevens, R. G., Brainard, G. C., Blask, D. E., Lockley, S. W. & Motta, M. E. Breast cancer and circadian disruption from electric lighting in the modern world. CA Cancer J. Clin. 64, 207–218 (2014). A summary of the empirical and epidemiological evidence relating to the potential health consequences of inappropriate night-time light exposure disrupting human circadian physiology, melatonin production and sleep.
Rao, D., Yu, H., Bai, Y., Zheng, X. & Xie, L. Does night-shift work increase the risk of prostate cancer? A systematic review and meta-analysis. OncoTargets Ther. 8, 2817–2826 (2015).
James, P. et al. Outdoor light at night and breast cancer incidence in the nurses’ health study II. Environ. Health Perspect. 125, 087010 (2017).
Blask, D. E. et al. Melatonin-depleted blood from premenopausal women exposed to light at night stimulates growth of human breast cancer xenografts in nude rats. Cancer Res. 65, 11174–11184 (2005).
World Health Organization, International Agency for Research on Cancer. Shiftwork. IARC Monogr. Eval. Carcinog. Risks Hum. 98, 561 (2010).
Scheer, F. A., Hilton, M. F., Mantzoros, C. S. & Shea, S. A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl Acad. Sci. USA 106, 4453–4458 (2009).
Buxton, O.M., et al. Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Sci. Transl. Med. 4, 129ra43 (2012).
Morris, C. J. et al. Endogenous circadian system and circadian misalignment impact glucose tolerance via separate mechanisms in humans. Proc. Natl Acad. Sci. USA 112, E2225–E2234 (2015).
Morris, C. J., Purvis, T. E., Mistretta, J. & Scheer, F. A. Effects of the internal circadian system and circadian misalignment on glucose tolerance in chronic shift workers. J. Clin. Endocrinol. Metab. 101, 1066–1074 (2016).
Leproult, R. & Van Cauter, E. in Pediatric Neuroendocrinology Vol. 17 (eds Loche, S. et al.) 11–21 (Karger, Basel, 2010).
Leproult, R., Holmbäck, U. & Van Cauter, E. Circadian misalignment augments markers of insulin resistance and inflammation, independently of sleep loss. Diabetes 63, 1860–1869 (2014).
Blask, D. et al. Light Pollution: Adverse Health Effects of Nighttime Lighting. CSAPH Report 4-A-12 (American Medical Association, 2012).
Czeisler, C. A., Chiasera, A. J. & Duffy, J. F. Research on sleep, circadian rhythms and aging: applications to manned spaceflight. Exp. Gerontol. 26, 217–232 (1991).
Stewart, K. T., Hayes, B. C. & Eastman, C. I. Light treatment for NASA shiftworkers. Chronobiol. Int. 12, 141–151 (1995).
Barger, L. K. et al. Learning to live on a Mars day: fatigue countermeasures during the Phoenix Mars Lander mission. Sleep 35, 1423–1435 (2012).
Brainard, G. C. et al. Solid-state lighting for the International Space Station: tests of visual performance and melatonin regulation. Acta Astronaut. 92, 21–28 (2013).
Bugbee, B. Toward an optimal spectral quality for plant growth and development: the importance of radiation capture. Acta Hortic. 1134, 1–12 (2016). A comprehensive review of the current research on spectral effects on photosynthesis and plant morphology.
Snowden, M. C., Cope, K. R. & Bugbee, B. Sensitivity of seven diverse species to blue and green light: interactions with photon flux. PLoS ONE 11, e0163121 (2016). A review of interactions between photon flux, photosynthesis and plant morphology.
Hoover, W. H. The dependence of carbon dioxide assimilation in a higher plant on wavelength of radiation. Smithson. Misc. Collect. 95, 1–13 (1937).
McCree, K. J. The action spectrum, absorptance and quantum yield of photosynthesis in crop plants. Agric. Meteorol. 9, 191–216 (1971).
Inada, K. Action spectra for photosynthesis in higher plants. Plant Cell Physiol. 17, 355–365 (1976).
Downs, R. J. Controlled Environments of Plant Research (Columbia Univ. Press, New York, 1975).
Kubota, C., Eguchi, T. & Kroggel, M. UV-B radiation dose requirement for suppressing intumescence injury on tomato plants. Sci. Hortic. 226, 366–371 (2017).
Raviv, M. & Antignus, Y. UV radiation effects on pathogens and insect pests of greenhouse-grown crops. Photochem. Photobiol. 79, 219–226 (2004).
Went, F. W. The Experimental Control of Plant Growth (Chronica Botanica, New York, 1957). A classic book on the early days of photobiology.
Höll, J. et al. Impact of pulsed UV-B stress exposure on plant performance: How recovery periods stimulate secondary metabolism while reducing adaptive growth attenuation. Plant Cell Environ. (2018).
Bugbee, B. in Light Emitting Diodes for Agriculture (ed. Gupta, D.) 81–99 (Springer, Singapore, 2017).
Kozai, T., Fujiwara, K. & Runkle, E. S. LED Lighting for Urban Agriculture (Springer, Singapore, 2016).
Carvalho, S. D. & Folta, K. M. Environmentally modified organisms – expanding genetic potential with light. Crit. Rev. Plant Sci. 33, 486–508 (2014). A comprehensive review of spectral effects on plant growth for 20 major crops.
Murchie, E. H. & Niyogi, K. K. Manipulation of photoprotection to improve plant photosynthesis. Plant Physiol. 155, 86–92 (2011).
Zielinska-Dabkowska, K. M. Make lighting healthier. Nature 553, 274–276 (2018).
LUXEON Rebel Color Line Product Datasheet DS68. https://www.lumileds.com/uploads/265/DS68-pdf (Lumileds Holding B.V., 2017).
Roederer, M. Conjugation of monoclonal antibodies. http://www.drmr.com/abcon/ (2004).
Pattison, P. M., Tsao, J. Y. & Krames, M. R. Light-emitting diode technology status and directions: opportunities for horticultural lighting. Acta Hortic. 1134, 413–426 (2016).
Acknowledgements
P.M.P. and J.Y.T. acknowledge support from the Department of Energy through its Office of Energy Efficiency and Renewable Energy’s Solid-State Lighting Program under contract DE-FE0025912. J.Y.T. acknowledges support from Sandia National Laboratories, a multi-program laboratory managed and operated by National Technology and Engineering Solutions of Sandia, LLC, a wholly owned subsidiary of Honeywell International, Inc., for the US Department of Energy’s National Nuclear Security Administration under contract DE-NA0003525. The work of SSLS, Inc. is carried out on behalf of the US Department of Energy SSL Program, Washington, DC. G.C.B. acknowledges J. Hanifin for discussions, editorial review and referencing; F. Scheer, K. Roecklein and R. Lucas for insights on portions of the text; and B. Warfield for help with the design of Fig. 3. G.C.B. was supported, in part, by NASA grants NNX15AC14, NNX08AD66A and NNX09AM68G; NSF grant EEC-0812056; DOE grant DE-EE0008207; The Institute for Integrative Health; and the Philadelphia Section of the Illuminating Engineering Society. B.B. is indebted to 35 years of discussions with colleagues from around the world including T. Volk, M. van Iersel, M. Blonquist, J. Frantz, R. Heins, R. Wheeler and C. Mitchell. B.B. also acknowledges support from NASA grant NNX17AJ31G, the USDA Specialty Crop Research Initiative and the Utah Agricultural Experiment Station. Any opinions, findings and conclusions or recommendations expressed in this manuscript are those of the authors and do not necessarily reflect the views of the authors’ funding agencies, including the US Department of Energy, Sandia National Laboratories and NASA.
Reviewer information
Nature thanks J.-H. Ryou, J. Wargent and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
P.M.P. outlined the overall article; J.Y.T., G.C.B., B.B. and P.M.P. wrote first drafts of the introductory, ‘Lighting for human health and productivity’, ‘Lighting for plants’ and ‘Conclusions’ sections, respectively; P.M.P., J.Y.T., G.C.B. and B.B. subsequently reviewed and edited all sections.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pattison, P.M., Tsao, J.Y., Brainard, G.C. et al. LEDs for photons, physiology and food. Nature 563, 493–500 (2018). https://doi.org/10.1038/s41586-018-0706-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0706-x
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.