Abstract
Piezoelectricity—the direct interconversion between mechanical and electrical energies—is usually remarkably enhanced at the morphotropic phase boundary of ferroelectric materials1,2,3,4, which marks a transition region in the phase diagram of piezoelectric materials and bridges two competing phases with distinct symmetries1,5. Such enhancement has enabled the recent development of various lead and lead-free piezoelectric perovskites with outstanding piezoelectric properties for use in actuators, transducers, sensors and energy-harvesting applications5,6,7,8. However, the morphotropic phase boundary has never been observed in organic materials, and the absence of effective approaches to improving the intrinsic piezoelectric responses of polymers9,10 considerably hampers their application to flexible, wearable and biocompatible devices. Here we report stereochemically induced behaviour in ferroelectric poly(vinylidene fluoride-co-trifluoroethylene) (P(VDF-TrFE)) copolymers, which is similar to that observed at morphotropic phase boundaries in perovskites. We reveal that compositionally tailored tacticity (the stereochemical arrangement of chiral centres related to the TrFE monomers11,12) can lead to intramolecular order-to-disorder evolution in the crystalline phase and thus to an intermediate transition region that is reminiscent of the morphotropic phase boundary, where competing ferroelectric and relaxor properties appear simultaneously. Our first-principles calculations confirm the crucial role of chain tacticity in driving the formation of this transition region via structural competition between the trans-planar and 3/1-helical phases. We show that the P(VDF-TrFE) copolymer with the morphotropic composition exhibits a longitudinal piezoelectric coefficient of −63.5 picocoulombs per newton, outperforming state-of-the-art piezoelectric polymers10. Given the flexibility in the molecular design and synthesis of organic ferroelectric materials, this work opens up the way for the development of scalable, high-performance piezoelectric polymers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information.
References
Fu, H. & Cohen, R. E. Polarization rotation mechanism for ultrahigh electromechanical response in single-crystal piezoelectrics. Nature 403, 281–283 (2000).
Guo, R. et al. Origin of the high piezoelectric response in PbZr1-xTixO3. Phys. Rev. Lett. 84, 5423–5426 (2000).
Bellaiche, L., García, A. & Vanderbilt, D. Finite-temperature properties of Pb(Zr1-xTix)O3 alloys from first principles. Phys. Rev. Lett. 84, 5427–5430 (2000).
Kutnjak, Z., Petzelt, J. & Blinc, R. The giant electromechanical response in ferroelectric relaxors as a critical phenomenon. Nature 441, 956–959 (2006).
Ahart, M. et al. Origin of morphotropic phase boundaries in ferroelectrics. Nature 451, 545–548 (2008).
Park, S. E. & Shrout, T. R. Ultrahigh strain and piezoelectric behavior in relaxor based ferroelectric single crystals. J. Appl. Phys. 82, 1804–1811 (1997).
Saito, Y. et al. Lead-free piezoceramics. Nature 432, 84–87 (2004).
Zeches, R. J. et al. A strain-driven morphotropic phase boundary in BiFeO3. Science 326, 977–980 (2009).
Lovinger, A. J. Ferroelectric polymers. Science 220, 1115–1121 (1983).
Ramadan, K. S., Sameoto, D. & Evoy, S. A review of piezoelectric polymers as functional materials for electromechanical transducers. Smart Mater. Struct. 23, 033001 (2014).
Kolda, R. R. & Lando, J. B. The effect of hydrogen-fluorine defects on the conformational energy of polytrifluoroethylene chains. J. Macromol. Sci. B 11, 21–39 (1975).
Cais, R. E. & Kometani, J. M. Synthesis of pure head-to-tail poly(trifluoroethylenes) and their characterization by 470-MHz fluorine-19 NMR. Macromolecules 17, 1932–1939 (1984).
Jaffe, B., Roth, R. S. & Marzullo, S. Piezoelectric properties of lead zirconate-lead titanate solid-solution ceramics. J. Appl. Phys. 25, 809–810 (1954).
La-Orauttapong, D. et al. Phase diagram of the relaxor ferroelectric (1−x)Pb(Zn1/3Nb2/3)−xPbTiO3. Phys. Rev. B 65, 144101 (2002).
Noheda, B., Cox, D. E., Shirane, G., Guo, J. & Ye, Z.-G. Phase diagram of the ferroelectric relaxor (1−x)Pb(Mg1/3Nb2/3) −xPbTiO3. Phys. Rev. B 66, 054104 (2002).
Farmer, B. L., Hopfinger, A. J. & Lando, J. B. Polymorphism of poly(vinylidene fluoride): potential energy calculations of the effects of head-to-head units on the chain conformation and packing of poly(vinylidene fluoride). J. Appl. Phys. 43, 4293–4303 (1972).
Lovinger, A. J., Davis, D. D., Cais, R. E. & Kometani, J. M. The role of molecular defects on the structure and phase transitions of poly(vinylidene fluoride). Polymer 28, 617–626 (1987).
Lovinger, A. J., Davis, G. T., Furukawa, T. & Broadhurst, M. G. Crystalline forms in a copolymer of vinylidene fluoride and trifluoroethylene (52/48 mol%). Macromolecules 15, 323–328 (1982).
Davis, G. T., Furukawa, T., Lovinger, A. J. & Broadhurst, M. G. Structural and dielectric investigation on the nature of the transition in a copolymer of vinylidene fluoride and trifluoroethylene (52/48 mol %). Macromolecules 15, 329–333 (1982).
Zhang, Q. M., Bharti, V. & Zhao, X. Giant electrostriction and relaxor ferroelectric behavior in electron-irradiated poly(vinylidene fluoride-trifluoroethylene) copolymer. Science 280, 2101–2104 (1998).
Furukawa, T. & Takahashi, Y. Ferroelectric and antiferroelectric transitions in random copolymers of vinylidene fluoride and trifluoroethylene. Ferroelectrics 264, 1739–1748 (2001).
Furukawa, T. & Seo, N. Electrostriction as the origin of piezoelectricity in ferroelectric polymers. Jpn. J. Appl. Phys. 29, 675–680 (1990).
Katsouras, I. et al. The negative piezoelectric effect of the ferroelectric polymer poly (vinylidene fluoride). Nat. Mater. 15, 78–84 (2016).
Broadhurst, M. G. & Davis, G. T. Physical basis for piezoelectricity in PVDF. Ferroelectrics 60, 3–13 (1984).
Tashiro, K., Takano, K., Kobayashi, M., Chatani, Y. & Tadokoro, H. Structure and ferroelectric phase transition of vinylidene fluoride-trifluoroethylene copolymers: 2. VDF 55% copolymer. Polymer 25, 195–208 (1984).
Bellet-Amalric, E. & Legrand, J. F. Crystalline structures and phase transition of the ferroelectric P(VDF-TrFE) copolymers, a neutron diffraction study. Eur. Phys. J. B 3, 225–236 (1998).
Lovinger, A. J. & Cais, R. E. Structure and morphology of poly(trifluoroethylene). Macromolecules 17, 1939–1945 (1984).
Bohlén, M. & Bolton, K. Conformational studies of poly(vinylidene fluoride), poly(trifluoroethylene) and poly(vinylidene fluoride-co-trifluoroethylene) using density functional theory. Phys. Chem. Chem. Phys. 16, 12929–12939 (2014).
Wicker, A., Berge, B. & Lajzerowicz, J. Nonlinear optical investigation of the bulk ferroelectric polarization in a vinylidene fluoride/trifluoroethylene copolymer. J. Appl. Phys. 66, 342–349 (1989).
Soulestin, T., Ladmiral, V., Lannuzel, T., Domingues Dos Santos, F. & Améduri, B. Importance of microstructure control for designing new electroactive terpolymers based on vinylidene fluoride and trifluoroethylene. Macromolecules 48, 7861–7871 (2015).
Yagi, T. & Tatemoto, M. A fluorine-19 NMR study of the microstructure of vinylindene fluoride-trifluuoroethylene copolymers. Polym. J. 11, 429–436 (1979).
Hasegawa, R., Takahashi, Y., Chatani, Y. & Tadokoro, H. Crystal structures of three crystalline forms of poly(vinylidene fluoride). Polym. J. 3, 600–610 (1972).
Kobayashi, M., Tashiro, K. & Tadokoro, H. Molecular vibrations of three crystal forms of poly(vinylidene fluoride). Macromolecules 8, 158–171 (1975).
Kim, K. J., Reynolds, N. M. & Hsu, S. L. Spectroscopic analysis of the crystalline and amorphous phases in a vinylidene fluoride/trifluoroethylene copolymer. Macromolecules 22, 4395–4401 (1989).
Gomes, J., Serrado Nunes, J., Sencadas, V. & Lanceros-Mendez, S. Influence of the β-phase content and degree of crystallinity on the piezo- and ferroelectric properties of poly(vinylidene fluoride). Smart Mater. Struct. 19, 065010 (2010).
Furukawa, T., Ohuchi, M., Chiba, A. & Datela, M. Dielectric relaxations and molecular motions in homopolymers and copolymers of vinylidene fluoride and trifluoroethylene. Macromolecules 17, 1384–1390 (1984).
Giannozzi, P. et al. QUANTUM ESPRESSO: a modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 21, 395502 (2009).
Lee, K., Murray, É. D., Kong, L., Lundqvist, B. I. & Langreth, D. C. Higher-accuracy van der Waals density functional. Phys. Rev. B 82, 081101 (2010).
Pack, J. D. & Monkhorst, H. J. ‘Special points for Brillouin-zone integrations’—a reply. Phys. Rev. B 16, 1748–1749 (1977).
Broadhurst, M. G., Davis, G. T., McKinney, J. E. & Collins, R. E. Piezoelectricity and pyroelectricity in polyvinylidene fluoride–a model. J. Appl. Phys. 49, 4992–4997 (1978).
Lines, M. E. & Glass, A. M. Principles and Applications of Ferroelectrics and Related Materials (Oxford Univ. Press, Oxford, 1977).
Jaffe, B., Cook, W. R. & Jaffe, H. Piezoelectric Ceramics (Academic Press, London, 1971).
Bystrov, V. S. et al. Molecular modeling of the piezoelectric effect in the ferroelectric polymer poly(vinylidene fluoride) (PVDF). J. Mol. Model. 19, 3591–3602 (2013).
Acknowledgements
This research was funded by the US Office of Naval Research (grants N000141612082 and N000141612459). The supercomputer time at the National Center for Supercomputing Applications (NSF OCI-0725070 and ACI-1238993) was provided by NSF grant ACI-1615114. Y.L. thanks N. Wonderling, J. Stapleton and J. Long for technical assistance.
Reviewer information
Nature thanks T. Kimura, C. Park and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Y.L. and Q.W. designed the research. W.X. was responsible for the polymer synthesis and NMR measurements. Y.L. prepared the polymer films and collected XRD and DMA data. H.A. performed FTIR measurements. Y.L. and H.A. carried out DSC and dielectric measurements. Y.L., H.A. and W.X. measured P–E loops and electromechanical properties. B.Z., W.L. and J.B. performed the first-principle calculations and analysis. Y.L. and Q.W. wrote the manuscript and all authors revised the manuscript. Q.W. supervised the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Crystallographic structures of different phases in the phase diagram of P(VDF-TrFE) copolymers.
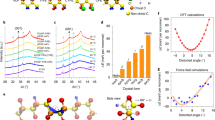
a, b, Side (a) and top (b) view of the trans-planar phase. c, d, Side (c) and top (d) view of the 3/1-helical phase. e, Side view of the gauche dihedral angle ϕ in the 3/1-helical phase. f, Top view of the regular 3/1-helical phase (ϕ = 60°). g, Top view of the (pseudo-)disordered 3/1-helical phase (ϕ1, ϕ2 ≠ 60°). Intramolecular disorder occurs in the (pseudo-)disordered 3/1-helical phase in terms of random departure from the gauche dihedral angle of 60° in the regular form (f). The 3/1-helical phase becomes energetically more stable than the trans-planar phase as the VDF content decreases in the vicinity of 50 mol% (Fig. 4a). Moreover, intramolecular disorder develops mainly in the 3/1-helical phase at the critical VDF content of 55 mol%, while the fraction of the regular trans-planar phase decreases. With further decrease of the VDF content (49 mol% ≤ CVDF ≤ 55 mol%), the fraction of the disordered 3/1-helical phase increases substantially and strongly competes with the trans-planar phase (we call this phase coexistence of the pseudo-disordered and ordered phases, or a morphotropic phase in the phase diagram). Here the morphotropic phase (49 mol% ≤ CVDF ≤ 55 mol%) differs from the disordered paraelectric phase owing to the existence of a phase transition (Fig. 3). As the VDF content decreases below 49 mol%, only the disordered 3/1-helical phase (namely, the pseudo-disordered phase) is energetically preferred. In this case, the pseudo-disordered phase has nearly the same structure as the disordered paraelectric phase owing to the absence of a phase transition (Fig. 3), and differs from both the morphotropic and the VDF-rich compositions. We note that we use the structure of PVDF to simplify this discussion. For the case of P(VDF-TrFE), the substitution of some H atoms by some F atoms leads to different chain tacticities of P(VDF-TrFE) (Fig. 1a), which requires a slight generalization of the above description.
Extended Data Fig. 2 Analysis of 19F NMR spectra of P(VDF-TrFE) copolymers.
a, Assignments of 19F NMR signals of P(VDF-TrFE). The monomers indicated by the 19F NMR signals are undelined. H–H, head to head; H–T, head to tail; T–T, tail to tail. b, Unconditional probabilities of different regiosequences as a function of VDF content. P(VDF-TrFE) is dominated by normal H–T isoregic sequences (about 76.7%–78.3%) with almost constant H–H/T–T regiodefects and non-regioisomers (marked as ‘others’). The slight variation in regioirregular sequences indicates that neither regiodefects nor non-regioisomers are responsible for the observed conformational competition. Instead, we identify the dominant contribution to be the dramatic tacticity change upon the formation of the MPB-like transition. c, Unconditional probabilities of normal H–T sequences consisting of the VDF-VDF, VDF-TrFE and TrFE-TrFE segments. With decreasing VDF content, the TrFE-TrFE units grow substantially, becoming even larger than their VDF-TrFE counterparts (which remain nearly constant for CVDF = 45–65 mol%) for CVDF < 49 mol%, and this change is accompanied by a remarkable decrease in the VDF-VDF units. These results clearly indicate that the polymer chain becomes more PTrFE-like for the copolymers with TrFE-rich compositions. Our findings disagree with previous results31, which showed the H–T VDF-TrFE sequence to be predominant for the copolymers (for CVDF ≈ 30–75 mol%). The poor NMR resolution (56.5 MHz) of the previous measurement led to the absence of many resonance peaks (above 130 p.p.m.). Moreover, only the -CF2- resonance area was considered, and the contributions from the -CHF- resonance area were disregarded31. Consequently, the regioregularity was not appropriately described and the chain tacticity was not analysed. Error bars in b and c represent standard deviations obtained from at least three measurements using different samples and are typically smaller than the symbols. d, The -CFH- resonances. All the triad assignments are indicated by grey arrows.
Extended Data Fig. 3 FTIR spectra of P(VDF-TrFE) copolymers at room temperature.
a, Raw data for selected polymer compositions (CVDF = 70 mol%, 65 mol%, 55 mol%, 49 mol% and 45 mol%). The intensities of the characteristic bands corresponding to the TGT\(\bar{{\rm{G}}}\) conformation near 614 cm−1 and to the 3/1-helical conformation at around 507 cm−1 increase considerably with decreasing VDF content. Because of the extremely low band intensity of the TGT\(\bar{{\rm{G}}}\) conformation, we mainly consider the conformational interconvention between the all-trans and 3/1-helical conformations. b, Zoom-in of the grey regions in a. The left panel shows the band characteristic of the all-trans conformation at around 1,290 cm−1 and the right panel presents the band characteristic of the 3/1-helical conformation at around 507 cm−1. c, d, FTIR spectra of the magnified regions around 1,290 cm−1 and 507 cm−1, with appropriate offset adjustments for clarity.
Extended Data Fig. 4 DSC and DMA results for P(VDF-TrFE) copolymers.
a, b, Heat flow versus temperature. c, Latent heat. d, Melting enthalpy. e, Crystallinity. f, tan(δ) peak. tan(δ) is defined as the ratio of the loss modulus to the storage modulus, measured using oscillatory shear stress tests, and can be used to evaluate the ratio between the viscous and elastic components per cycle of sample deformation. A very broad exothermic peak is found for CVDF < 49 mol% (a), which is accompanied by a dramatic reduction in latent heat (c). As CVDF decreases from 55 mol% to 49 mol%, the relative change in crystallinity (e) and the shift of the transition temperature are limited to about 2% and 6%, respectively, which cannot explain the observed approximately 60% enhancement in d33 achieved at CVDF = 50 mol%. We find that tan(δ) decreases considerably with increasing VDF content (e), which implies that P(VDF-TrFE) copolymers with high VDF content are more elastic. This might be due to lattice contraction with increasing VDF content (Fig. 1e), which imposes restrictions against molecular mobility. We measured the elastic modulus, known as Young’s modulus Y, to provide a measure of stiffness for our polymers (Extended Data Fig. 7e). The Y value of the P(VDF-TrFE) copolymers increases substantially with increasing VDF content, mediated by an intermediate (grey) region (49 mol% ≤ CVDF ≤ 55 mol%). The compositional evolutions of the latent heat and tan(δ) reveal the existence of an intermediate transition region (grey). Error bars in c–f represent standard deviations obtained from at least three measurements using different samples.
Extended Data Fig. 5 Temperature-dependent dielectric constant and loss of P(VDF-TrFE) copolymers.
a–d, Dielectric constant as a function of temperature for P(VDF-TrFE) copolymers with CVDF = 45 mol% (a) CVDF = 49 mol% (b), CVDF = 55 mol% (c) and CVDF = 65 mol% (d). a–c show relaxor behaviours, whereas d shows the typical dielectric response of normal ferroelectrics, in which the dielectric peak is independent of frequency. The dielectric response near Tmax results from molecular motions in the crystalline regions rather than in the amorphous regions32. Therefore, both normal-ferroelectric and relaxor properties coexist in the crystalline regions of P(VDF-TrFE) within the transition region (49 mol% ≤ CVDF ≤ 55 mol%). The discovery of relaxor behaviour not only explains the emergence of the peak at low 2θ in the XRD pattern (Fig. 1d) but also allows the correlation of its origin with intramolecular disorder. The arrows in a–d are guides for the eyes.
Extended Data Fig. 6 Polarization hysteresis in P(VDF-TrFE) copolymers at room temperature.
a, Polarization versus electric field, measured using a triangular a.c. electric field of 100 Hz. A polarization hysteresis curve—characteristic of a normal ferroelectric—is shown for CVDF ≥ 49 mol%, whereas an antiferroelectric-like hysteresis loop is observed for CVDF < 49 mol%. b–d, Maximum polarization Pm (b), remanent polarization Pr (c) and coercive field Ec (d) as a function of VDF content, measured at 1 Hz and 100 Hz. Considerable reduction in Pr is found in c (CVDF < 49 mol%), which is indicative of the disappearance of long-range ferroelectric order. Error bars in b–d represent standard deviations obtained from at least three measurements using different samples.
Extended Data Fig. 7 Electrostrictive properties and theoretical descriptions of the piezoelectric effect in P(VDF-TrFE) copolymers.
a, Strain–electric field (S3–E) curves. b, Strain–polarization (S3–P) curves. c, S3–P2. Typical P(VDF-TrFE) compositions are shown in a–c for clarity. d, Q33 as a function of VDF content. e, Young’s modulus Y as a function of VDF content. f, The magnitude of the piezoelectric voltage constant, g33, as a function of VDF content at room temperature. g33 is determined according to g33 = d33/(εrε0), where εr and ε0 are the relative and vacuum permittivities, respectively. The lines in d–f are guides for the eyes. The light-green shaded areas in e and f indicate the transition region. g, Comparison between experimental and theoretical g33 values. h–l, Electrostrictive data for P(VDF-TrFE) copolymer at 50/50 mol% measured at 70 °C. h, P–E loops. i, Electric-field-induced strain. j, S3–P curves. k, S3–P2curves. l, Electric-field-induced strain at an electric field of 25 MV m−1. The calculated d33 is −68.3 pC N−1 using Q33 = −4.18 m4 C−2 (k), which is slightly larger in magnitude than the experimental data (−63.5 ± 3.2 pC N−1) for P(VDF-TrFE) with CVDF = 50 mol%. A detailed discussion about the data shown here can be found in Supplementary Information. Error bars in d–f represent standard deviations obtained from at least ten measurements using different samples.
Extended Data Fig. 8 X-ray patterns of P(VDF-TrFE) copolymers at various temperatures.
a–h, X-ray patterns of P(VDF-TrFE) copolymers with selected VDF contents (a and e, CVDF = 45 mol%; b and f, CVDF = 49 mol%; c and g, CVDF = 55 mol%; d and h, CVDF = 65 mol%) upon heating (a–d) and cooling (e–h). ‘O’, ‘D’ and ‘PD’ indicate the ordered, disordered and pseudo-disordered phases (Extended Data Fig. 1), respectively. For CVDF < 49 mol%, the peaks shift towards a lower 2θ at an even rate upon heating (a) and cooling (e), indicating the absence of a phase transition. For CVDF ≥ 49 mol%, a change in shift rates is observed near the phase transition temperature (b–d, f–h), indicating the emergence of the order–disorder phase transition. Moreover, the order of the phase transition can be determined by analysing the thermal hysteresis at the transition temperature from the heating and cooling processes.
Extended Data Fig. 9 Infrared absorbance bands of P(VDF-TrFE) copolymers at around 1,290 cm−1 and 507 cm−1, measured at various temperatures.
a–h, Infrared absorbance curves obtained during heating for copolymers with CVDF = 45 mol% (a, e), CVDF = 49 mol% (b, f), CVDF = 55 mol% (c, g) and CVDF = 65 mol% (d, h). i–p, Corresponding curves obtained during cooling for CVDF = 45 mol% (i, m), CVDF = 49 mol% (j, n), CVDF = 55 mol% (k, o) and CVDF = 65 mol% (l, p). ‘O’, ‘D’ and ‘PD’ represent the ordered, disordered and pseudo-disordered phases (Extended Data Fig. 1), respectively. For CVDF < 49 mol%, the characteristic bands that correspond to both the all-trans and 3/1-helical conformations shrink or expand uniformly upon heating (a, e) or cooling (i, m), implying no phase transition. For CVDF ≥ 49 mol%, two distinct regimes can be clearly resolved (as marked by grey arrows), signifying the presence of the order–disorder phase transition. Thermal hysteresis is negligible for 49 mol% ≤ CVDF ≤ 55 mol% (second-order phase transition), whereas substantial thermal hysteresis is observed for P(VDF-TrFE) with a VDF contant of 65 mol% (first-order phase transition).
Extended Data Fig. 10 Rietveld analysis for P(VDF-TrFE) copolymer with CVDF = 55 mol%.
a, XRD pattern in the 2θ range [10°, 50°], measured at room temperature. The indexation was done according to the monoclinic space group Cc. a, b and c are the refined lattice parameters and βM is the monoclinic angle. b, c, Fitting results. Apparent disagreements are observed for the (200,110) and (310) peaks (a), leading to a large agreement factor of R = 25.54% (b, c), which is within the range (about 23%–29%) reported in a previous study25. This poor fitting result is only slightly improved (about 23.03%; Supplementary Fig. 8) by considering chain deflection32. These results suggest that simply considering the planar-zigzag-based chain structure (namely, the ‘cooled’ phase model25) cannot yield a reasonable fit for the XRD data near the transition region. Additionally, no reasonable fit was obtained by considering other space groups, such as Cm or Cm2m (not shown here). Indeed, a large agreement factor (about 10%–30%) was usually achieved in previous structural refinements25,26,32 in PVDF and its copolymers. One of the main reasons for this disagreement is that polymer crystallography generally corresponds to the ideal condition, disregarding concomitant structural defects (disorder) and complex morphology (a composite of amorphous, crystalline and intermediate regions) that exist in real polymers. Typically, the Bragg peaks are very broad at high values of diffraction angle 2θ and the number of available reflections is limited for morphotropic compositions, which considerably increases the difficulty of crystallographic analysis. Obviously, further studies—both theoretical and experimental—of the crystal structure of P(VDF-TrFE) near the transition region are required.
Supplementary information
Supplementary Information
This file contains Supplementary Information sections 1–8, which includes Supplementary Figures 1–24, Supplementary Tables 1 and 2 and Supplementary References.
Rights and permissions
About this article
Cite this article
Liu, Y., Aziguli, H., Zhang, B. et al. Ferroelectric polymers exhibiting behaviour reminiscent of a morphotropic phase boundary. Nature 562, 96–100 (2018). https://doi.org/10.1038/s41586-018-0550-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0550-z
Keywords
This article is cited by
-
Polymer nanocomposite dielectrics for capacitive energy storage
Nature Nanotechnology (2024)
-
Giant piezoelectricity driven by Thouless pump in conjugated polymers
npj Computational Materials (2024)
-
Electrically and mechanically driven rotation of polar spirals in a relaxor ferroelectric polymer
Nature Communications (2024)
-
Tertiary orientation structures enhance the piezoelectricity of MXene/PVDF nanocomposite
Nano Research (2024)
-
Room-temperature ferroelectricity in magnetically ordered CoH2SeO4 flakes
Science China Materials (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.