Abstract
Adult neurogenesis in the dentate gyrus of the hippocampus is highly regulated by environmental influences, and functionally implicated in behavioural responses to stress and antidepressants1,2,3,4. However, how adult-born neurons regulate dentate gyrus information processing to protect from stress-induced anxiety-like behaviour is unknown. Here we show in mice that neurogenesis confers resilience to chronic stress by inhibiting the activity of mature granule cells in the ventral dentate gyrus (vDG), a subregion that is implicated in mood regulation. We found that chemogenetic inhibition of adult-born neurons in the vDG promotes susceptibility to social defeat stress, whereas increasing neurogenesis confers resilience to chronic stress. By using in vivo calcium imaging to record neuronal activity from large cell populations in the vDG, we show that increased neurogenesis results in a decrease in the activity of stress-responsive cells that are active preferentially during attacks or while mice explore anxiogenic environments. These effects on dentate gyrus activity are necessary and sufficient for stress resilience, as direct silencing of the vDG confers resilience whereas excitation promotes susceptibility. Our results suggest that the activity of the vDG may be a key factor in determining individual levels of vulnerability to stress and related psychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Snyder, J. S., Soumier, A., Brewer, M., Pickel, J. & Cameron, H. A. Adult hippocampal neurogenesis buffers stress responses and depressive behaviour. Nature 476, 458–461 (2011).
Santarelli, L. et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 301, 805–809 (2003).
Boldrini, M. et al. Antidepressants increase neural progenitor cells in the human hippocampus. Neuropsychopharmacology 34, 2376–2389 (2009).
Lucassen, P. J., Stumpel, M. W., Wang, Q. & Aronica, E. Decreased numbers of progenitor cells but no response to antidepressant drugs in the hippocampus of elderly depressed patients. Neuropharmacology 58, 940–949 (2010).
Strange, B. A., Witter, M. P., Lein, E. S. & Moser, E. I. Functional organization of the hippocampal longitudinal axis. Nat. Rev. Neurosci. 15, 655–669 (2014).
Anacker, C. & Hen, R. Adult hippocampal neurogenesis and cognitive flexibility — linking memory and mood. Nat. Rev. Neurosci. 18, 335–346 (2017).
Kheirbek, M. A. et al. Differential control of learning and anxiety along the dorsoventral axis of the dentate gyrus. Neuron 77, 955–968 (2013).
Spalding, K. L. et al. Dynamics of hippocampal neurogenesis in adult humans. Cell 153, 1219–1227 (2013).
Boldrini, M. et al. Human hippocampal neurogenesis persists throughout aging. Cell Stem Cell 22, 589–599.e5 (2018).
Sorrells, S. F. et al. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature 555, 377–381 (2018).
Danielson, N. B. et al. Distinct contribution of adult-born hippocampal granule cells to context encoding. Neuron 90, 101–112 (2016).
Denny, C. A., Burghardt, N. S., Schachter, D. M., Hen, R. & Drew, M. R. 4- to 6-week-old adult-born hippocampal neurons influence novelty-evoked exploration and contextual fear conditioning. Hippocampus 22, 1188–1201 (2012).
Temprana, S. G. et al. Delayed coupling to feedback inhibition during a critical period for the integration of adult-born granule cells. Neuron 85, 116–130 (2015).
David, D. J. et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron 62, 479–493 (2009).
Hill, A. S., Sahay, A. & Hen, R. Increasing adult hippocampal neurogenesis is sufficient to reduce anxiety and depression-like behaviors. Neuropsychopharmacology 40, 2368–2378 (2015).
Culig, L. et al. Increasing adult hippocampal neurogenesis in mice after exposure to unpredictable chronic mild stress may counteract some of the effects of stress. Neuropharmacology 126, 179–189 (2017).
Dranovsky, A. et al. Experience dictates stem cell fate in the adult hippocampus. Neuron 70, 908–923 (2011).
Ray, R. S. et al. Impaired respiratory and body temperature control upon acute serotonergic neuron inhibition. Science 333, 637–642 (2011).
Wook Koo, J. et al. Essential role of mesolimbic brain-derived neurotrophic factor in chronic social stress-induced depressive behaviors. Biol. Psychiatry 80, 469–478 (2016).
Kempermann, G., Kuhn, H. G. & Gage, F. H. More hippocampal neurons in adult mice living in an enriched environment. Nature 386, 493–495 (1997).
van Praag, H., Kempermann, G. & Gage, F. H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci. 2, 266–270 (1999).
Sahay, A. et al. Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature 472, 466–470 (2011).
Krishnan, V. et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 131, 391–404 (2007).
Burghardt, N. S., Park, E. H., Hen, R. & Fenton, A. A. Adult-born hippocampal neurons promote cognitive flexibility in mice. Hippocampus 22, 1795–1808 (2012).
Ikrar, T. et al. Adult neurogenesis modifies excitability of the dentate gyrus. Front. Neural Circuits 7, 204 (2013).
Drew, L. J. et al. Activation of local inhibitory circuits in the dentate gyrus by adult-born neurons. Hippocampus 26, 763–778 (2016).
Jimenez, J. C. et al. Anxiety cells in a hippocampal-hypothalamic circuit. Neuron 97, 670–683 (2018).
Padilla-Coreano, N. et al. Direct ventral hippocampal-prefrontal input is required for anxiety-related neural activity and behavior. Neuron 89, 857–866 (2016).
Bagot, R. C. et al. Ventral hippocampal afferents to the nucleus accumbens regulate susceptibility to depression. Nat. Commun. 6, 7062 (2015).
Golden, S. A., Covington, H. E., III, Berton, O. & Russo, S. J. A standardized protocol for repeated social defeat stress in mice. Nat. Protoc. 6, 1183–1191 (2011).
Anacker, C. et al. Neuroanatomic differences associated with stress susceptibility and resilience. Biol. Psychiatry 79, 840–849 (2016).
Resendez, S. L. et al. Visualization of cortical, subcortical and deep brain neural circuit dynamics during naturalistic mammalian behavior with head-mounted microscopes and chronically implanted lenses. Nat. Protoc. 11, 566–597 (2016).
Wu, M. V. & Hen, R. Functional dissociation of adult-born neurons along the dorsoventral axis of the dentate gyrus. Hippocampus 24, 751–761 (2014).
Ghosh, K. K. et al. Miniaturized integration of a fluorescence microscope. Nat. Methods 8, 871–878 (2011).
Ziv, Y. et al. Long-term dynamics of CA1 hippocampal place codes. Nat. Neurosci. 16, 264–266 (2013).
Mukamel, E. A., Nimmerjahn, A. & Schnitzer, M. J. Automated analysis of cellular signals from large-scale calcium imaging data. Neuron 63, 747–760 (2009).
Mongiat, L. A., Espósito, M. S., Lombardi, G. & Schinder, A. F. Reliable activation of immature neurons in the adult hippocampus. PLoS ONE 4, e5320 (2009).
Dieni, C. V., Nietz, A. K., Panichi, R., Wadiche, J. I. & Overstreet-Wadiche, L. Distinct determinants of sparse activation during granule cell maturation. J. Neurosci. 33, 19131–19142 (2013).
Acknowledgements
This work was funded by the National Institutes of Health (K99 MH108719 to C.A; K01 AG054765 to V.M.L.; R37 MH068542, R01 MH083862, R01 NS081203 to R.H.), the Hope for Depression Research Foundation (HDRF RGA-13-003 to R.H.), NYSTEM (C029157 to R.H.) and the German Research Foundation (AN1006/1-1 to C.A.). G.S.S. and B.C. were supported by the Doctoral Program in Neurobiology and Behavior at Columbia University. J.C.J. is a Howard Hughes Medical Institute Gilliam Fellow and Columbia University MSTP student. We thank S. Dymecki for providing us with the Cre-responsive loxP-stop-mCherry-loxP-hM4Di mouse line.
Reviewer information
Nature thanks P. Frankland and J. Snyder for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
C.A. and R.H. designed the research and wrote the paper. C.A., A.M., B.C. and R.S. conducted behavioural experiments, surgeries, immunohistochemistry and cell counting. V.M.L. conducted in vitro electrophysiological experiments. C.A., G.S.S., J.C.J. and B.C. analysed in vivo imaging data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Silencing adult-born neurons in i-hM4Di mice.
a, Mice expressing a TMX-inducible CreERT2 recombinase expressed under the control of the Nestin gene promoter17 were crossed to mice expressing loxP-STOP-mCherry-loxP-hM4Di under the control of the CAG promoter18. The eYFP reporter gene was used to visualize recombination by crossing Nestin-CreERT2+/−;loxP-stop-mCherry-loxP-hM4Di mice to loxP-stop-loxP-eYFP mice (Nestin-CreERT2+/−;stop-mCherry fl/−-hM4Di;stopfl/−-eYFP). b, Cre-mediated recombination in Dcx-positive adult-born neurons was visualized using eYFP/ Dcx co-labelling. DAPI, 4ʹ,6-diamidino-2-phenylindole; eYFP, enhanced yellow fluorescent protein. c, Biocytin was injected into abGCs during in vitro whole-cell recordings. Expression of hM4Di in recorded abGCs was confirmed by biocytin+/mCherry− immunofluorescence. d, Schematic illustrating 500-ms current-step injections into abGCs. Representative recordings from abGCs before and after bath application of CNO (5 μM) are shown. Scale bars: 20 mV, 100 ms. e, Bath application of CNO decreases spiking of hM4Di-expressing abGCs (paired, two-tailed t-test, *P = 0.024, n = 5). f, CNO decreases input resistance of abGCs (paired, one-tailed t-test, *P = 0.037, n = 5). g, Representative resting membrane potential trace of abGCs upon bath application of CNO. Scale bars: 2 mV, 100 ms. h, CNO decreases resting membrane potential of abGCs (paired, one-tailed t-test, **P = 0.006, n = 5); Scale bars: 5 mV, 1 min. Error bars, ±s.e.m.
Extended Data Fig. 2 i-hM4Di mice and iBax mice behaviour: time in corner of the social interaction test and time in centre of the open field.
a–d, i-hM4Di mice. a, Silencing abGCs in Cre+ i-hM4Di mice increased the time spent in the corner of the social interaction test after subthreshold defeat (interaction F1,33 = 23.0, ***P < 0.0001, genotype effect F1,33 = 17.96, ***P = 0.0002, stress effect F1,33 = 26.7, ***P < 0.0001, post hoc test, ***P < 0.0001). b, No difference in total distance travelled in the social interaction test (interaction F1,33 = 0.17, P = 0.68; genotype effect F1,33 = 2.5, P = 0.12; stress effect F1,33 = 1.6, P = 0.22) c, Time spent in the centre of the open field after subthreshold defeat (interaction F1,33 = 0.57, P = 0.46, genotype effect F1,33 = 4.63, *P = 0.04, stress effect F1,33 = 2.14, P = 0.15; planned comparison t-test: Cre− defeat versus Cre+ defeat, *P = 0.045). d, No difference in total distance travelled in the open field (interaction F1,33 = 0.007, P = 0.93; genotype effect F1,33 = 0.79, P = 0.38; stress effect F1,33 = 3.22, P = 0.08). n = 9, 9, 9, 10. e–h, iBax mice. e, Chronic defeat (10 days) increased the amount of time that Cre− mice spent in the corner during the social interaction test. This effect was absent in Cre+ iBax mice (interaction F1,50 = 7.9, **P = 0.007; genotype effect F1,50 = 6.3, *P = 0.016; stress effect F1,50 = 1.7, P = 0.19; post hoc test, ***P = 0.0003). f, Chronic defeat decreased the total distance travelled in the social interaction test without genotype effects (interaction F1,50 = 0.5, P = 0.48; genotype effect F1,50 = 0.00001, P = 0.99; stress effect F1,50 = 4.4, *P = 0.04). g, Chronic defeat decreased the time Cre− mice spent in the centre of the open field. This effect was absent in Cre+ iBax mice (interaction F1,50 = 4.5, *P = 0.039; genotype effect, F1,50 = 6.9, *P = 0.01; stress effect, F1,50 = 4.8, *P = 0.03; post hoc test, **P = 0.001). h, Chronic defeat decreased the total distance travelled in the open field without genotype effects (interaction F1,50 = 0.12, P = 0.73; genotype effect, F1,50 = 0.39, P = 0.54; stress effect F1,50 = 25.2, ***P < 0.0001). n = 14, 11, 14, 15. Error bars, ±s.e.m.
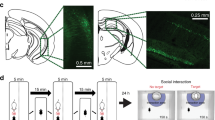
Extended Data Fig. 3 iBax mice show higher levels of adult-born cell survival and ventral X-ray irradiation abolishes stress resilience.
a, Experimental timeline for cell survival assessment using BrdU incorporation. b, The number of cells that have incorporated BrdU at 8 weeks after BrdU injection is increased in undefeated mice and in defeated Cre+ iBax mice compared to Cre− control mice (interaction F1,11 = 0.69, P = 0.43; genotype effect F1,11 = 138.9, ***P < 0.0001; stress effect F1,11 = 4.8, P = 0.05; planned comparison t-test, control Cre− versus Cre+, ***P < 0.001; defeat Cre− versus Cre+, **P = 0.002; n = 5, 3, 3, 4). c, Experimental timeline for X-ray irradiation. d, Schematic indicating sliding window position of a protective lead shield during X-ray irradiation of the vDG. e, Ventral X-ray irradiation eliminated Dcx+ cells in the vDG in Cre− and Cre+ mice. nCre − sham = 5, nCre + sham = 4, nCre − Xray = 5, nCre + Xray = 5. f, Representative images of Dcx expression in the dorsal dentate gyrus and the ventral dentate gyrus in X-ray irradiated mice. g, Cre+ iBax mice showed longer social interaction times than Cre− mice after chronic defeat. This effect was abolished by ventral X-ray irradiation (interaction F1,33 = 3.8, P = 0.06; genotype effect F1,33 = 4.5, *P = 0.04; X-ray effect, F1,33 = 8.4, **P = 0.007; planned comparison t-test, Cre− sham versus Cre+ sham, *P = 0.04; Cre+ sham versus Cre+ X-ray, **P = 0.008; n = 9, 9, 10, 9). h, Ventral X-ray irradiation increased time spent in the corner in both genotypes (interaction F1,33 = 0.38, P = 0.54; genotype effect, F1,33 = 1.8, P = 0.18; X-ray effect, F1,33 = 11.6, **P = 0.0018; planned comparison t-test, Cre− sham versus Cre− X-ray, *P = 0.04; Cre+ sham versus Cre+ X-ray, *P = 0.02; n = 9, 9, 10, 9). i, Cre+ iBax mice show increased centre/total distance in the open field compared with Cre− mice. Ventral X-ray irradiation abolished this effect (interaction F1,33 = 2.9, P = 0.1; genotype effect, F1,33 = 6.1, *P = 0.019; X-ray effect, F1,33 = 4.8, *P = 0.036; planned comparison t-test, Cre− sham versus Cre+ sham, *P = 0.01; Cre+ sham versus Cre+ X-ray, *P = 0.01; n = 9, 9, 10, 9). j, Cre+ iBax mice show increased time spent in the centre of the open field than Cre− mice. Ventral X-ray irradiation abolished this effect (interaction F1,33 = 4.2, *P = 0.049; genotype effect F1,33 = 3.7, P = 0.06; X-ray effect, F1,33 = 2.2, P = 0.15; post hoc test, Cre − sham versus Cre + sham, **P = 0.008; Cre + sham versus Cre + X-ray, *P = 0.02; n = 9, 9, 10, 9). Error bars, ±s.e.m.
Extended Data Fig. 4 iBax mice: Number of attacks and length of attacks during social defeat, and behaviour after 10 days of chronic social defeat for all imaged mice.
a, b, No difference in average duration of attacks on the first day of social defeat (a; unpaired, two-tailed t-test, P = 0.25; n = 7, 7), or the last day of social defeat (b; P = 0.56; n = 7, 7). c, d, No difference in the average number of attacks on the first day of social defeat (c; P = 0.76; n = 7, 7), or the last day of social defeat (d; P = 0.85; n = 7, 7). e, Representative heat map of time spent in the social interaction test during the first trial (without CD1 mouse). f, No differences in time in the interaction zone during the first trial (unpaired, two-tailed t-test, P = 0.47; n = 7, 7). g, No differences in time in the corner zone during the first trial (P = 0.46; n = 7, 7). h, No differences in velocity during the first trial (P = 0.43; n = 7, 7). i, Representative heat map of time spent in the social interaction test during the second trial (with CD1). j, Cre+ iBax mice spent longer in the interaction zone during the second trial than Cre− mice (*P = 0.049; n = 7, 7). k, Cre+ iBax mice spent less time in the corner zone during the second trial than Cre− mice (P = 0.1; n = 7, 7). l, No differences in velocity during the second trial (P = 0.65; n = 7, 7). m, Representative heat map of time spent in the open field test. n, Time spent in the centre of the open field (P = 0.18; \({n}_{{{\rm{Cre}}}^{-}}\) = 6, \({n}_{{{\rm{Cre}}}^{+}}\) = 5). o, No differences in velocity in the open field (P = 0.56; \({n}_{{{\rm{Cre}}}^{-}}\) = 6, \({n}_{{{\rm{Cre}}}^{+}}\) = 5). Error bars, ±s.e.m.
Extended Data Fig. 5 Ca2+ transient rates of non-selective cells, AUC rates, and amplitude rates of significant Ca2+ transients on social defeat day 1 and day 10.
a, Scatter plot showing the Ca2+ transient rates of individual neurons during attack versus no-attack periods. Colours based on selectivity for attacks (orange), no-attack (blue), and non-selectivity (grey) determined by exceeding shuffle distribution. For presentation purposes, the scatter plot shows rates up to 0.12 events per second. Ten cells showed rates greater than 0.12 events per second and were included in the cell selectivity analysis (see Source Data). b, c, No differences in Ca2+ transient rates of non-selective cells on day 1 (b) or day 10 (c). d–i, AUC rate analysis of significant Ca2+ transients. d, No differences in AUC rates between Cre− mice and Cre+ iBax mice on day 1 (no-attack: Mann–Whitney U = 38,448, P = 0.37; attack: U = 40,070, P = 0.94; \({n}_{{{\rm{Cre}}}^{-}}\) = 327, \({n}_{{{\rm{Cre}}}^{+}}\) = 246). e, No difference in AUC rates of attack-selective cells on day 1 (U = 1,093, P = 0.46; \({n}_{{{\rm{Cre}}}^{-}}\) = 57, \({n}_{{{\rm{Cre}}}^{+}}\) = 42). f, No difference in AUC rates of no-attack-selective cells on day 1 (U = 6,065, P = 0.76; \({n}_{{{\rm{Cre}}}^{-}}\) = 115, \({n}_{{{\rm{Cre}}}^{+}}\) = 108). g, AUC rates are increased only in Cre− mice during attack periods on day 10 (Wilcoxon matched-pairs signed rank, ***\({P}_{{{\rm{Cre}}}^{-}}\) = 0.0006; \({n}_{{{\rm{Cre}}}^{-}}\) = 620; \({P}_{{{\rm{Cre}}}^{+}}\) = 0.44; \({n}_{{{\rm{Cre}}}^{+}}\) = 592). Cre+ iBax mice show lower attack responses than Cre− mice (U = 165,092, **P = 0.002). h, AUC rates of attack-selective cells are lower in Cre+ iBax mice than in Cre− mice on day 10 (U = 12,162, ***P < 0.0001; \({n}_{{{\rm{Cre}}}^{-}}\) = 211, \({n}_{{{\rm{Cre}}}^{+}}\) = 169). i, No difference in AUC rates of no-attack-selective cells on day 10 (U = 11,127, P = 0.37; \({n}_{{{\rm{Cre}}}^{-}}\) = 169, \({n}_{{{\rm{Cre}}}^{+}}\) = 140). j–o, Amplitude rate analysis of significant Ca2+ transients. j, No differences in amplitude rates between Cre− mice and Cre+ iBax mice on day 1 (no-attack: Mann–Whitney U = 38,693, P = 0.44; attack: U = 38,741, P = 0.43; \({n}_{{{\rm{Cre}}}^{-}}\) = 327, \({n}_{{{\rm{Cre}}}^{+}}\) = 246). k, No difference in amplitude rates of attack-selective cells on day 1 (U = 1,132, P = 0.65; \({n}_{{{\rm{Cre}}}^{-}}\) = 57, \({n}_{{{\rm{Cre}}}^{+}}\) = 42). l, No difference in amplitude rates of no-attack-selective cells on day 1 (U = 5,767, P = 0.36; \({n}_{{{\rm{Cre}}}^{-}}\) = 115, \({n}_{{{\rm{Cre}}}^{+}}\) = 108). m, Amplitude rates are increased during attack periods on day 10 (Wilcoxon matched-pairs signed rank, ***\({P}_{{{\rm{Cre}}}^{-}}\) < 0.0001; \({n}_{{{\rm{Cre}}}^{-}}\) = 620; **\({P}_{{{\rm{Cre}}}^{+}}\) = 0.0019; \({n}_{{{\rm{Cre}}}^{+}}\) = 592). Cre+ iBax mice show lower attack responses than Cre− mice (U = 170,024, *P = 0.0246). n, Amplitude rates of attack-selective cells are lower in Cre+ iBax mice than in Cre− mice on day 10 (U = 12,936, ***P < 0.0001; \({n}_{{{\rm{Cre}}}^{-}}\) = 211, \({n}_{{{\rm{Cre}}}^{+}}\) = 169). o, No difference in amplitude rates of no-attack-selective cells on day 10 (U = 11,518, P = 0.69; \({n}_{{{\rm{Cre}}}^{-}}\) = 169, \({n}_{{{\rm{Cre}}}^{+}}\) = 140). Error bars, ±s.e.m.
Extended Data Fig. 6 Cumulative frequency distribution plots of Ca2+ transients.
a, No differences in the distributions of Ca2+ transient event rates were observed between Cre− mice and Cre+ iBax mice during attack periods on day 1. \({n}_{{{\rm{Cre}}}^{-}}\) = 460, \({n}_{{{\rm{Cre}}}^{+}}\) = 246. b, Distributions of Ca2+ transient event rates of attack-selective cells are different between Cre− mice and Cre+ iBax mice on day 1. \({n}_{{{\rm{Cre}}}^{-}}\) = 80, \({n}_{{{\rm{Cre}}}^{+}}\) = 42. c, Distributions of Ca2+ transient event rates are significantly different between Cre− mice and Cre+ iBax mice during attack periods on day 10. \({n}_{{{\rm{Cre}}}^{-}}\) = 620, \({n}_{{{\rm{Cre}}}^{+}}\) = 592. d, Distributions of Ca2+ transient event rates of attack-selective cells are significantly different between Cre− mice and Cre+ iBax mice on day 10. \({n}_{{{\rm{Cre}}}^{-}}\) = 211, \({n}_{{{\rm{Cre}}}^{+}}\) = 169. e, No differences in the distributions of Ca2+ transient AUC rates were observed between Cre− mice and Cre+ iBax mice during attack periods on day 1. \({n}_{{{\rm{Cre}}}^{-}}\) = 327, \({n}_{{{\rm{Cre}}}^{+}}\) = 246. f, No differences in the distributions Ca2+ transient AUC rates of attack-selective cells were observed between Cre− mice and Cre+ iBax mice on day 1. \({n}_{{{\rm{Cre}}}^{-}}\) = 57, \({n}_{{{\rm{Cre}}}^{+}}\) = 42. g, Distributions of Ca2+ transient AUC rates are significantly different between Cre− mice and Cre+ iBax mice during attack periods on day 10. \({n}_{{{\rm{Cre}}}^{-}}\) = 620, \({n}_{{{\rm{Cre}}}^{+}}\) = 592. h, Distributions of Ca2+ transient AUC rates of attack-selective cells are significantly different between Cre− mice and Cre+ iBax mice on day 10. \({n}_{{{\rm{Cre}}}^{-}}\) = 211, \({n}_{{{\rm{Cre}}}^{+}}\) = 169. KS, Kolmogorov–Smirnov test P value (shown for significant differences only).
Extended Data Fig. 7 iBax mice: decreased vDG activity during stress-induced social avoidance in the social interaction test.
a, Experimental timeline. b, Ca2+ transient rates in the social interaction (SI) zone during trial 1 (\({n}_{{{\rm{Cre}}}^{-}}\) = 546, \({n}_{{{\rm{Cre}}}^{+}}\) = 233). c, Ca2+ transient rates in the corner zones during trial 1 (\({n}_{{{\rm{Cre}}}^{-}}\) = 530, \({n}_{{{\rm{Cre}}}^{+}}\) = 329). d, Cre+ iBax mice show lower Ca2+ transient rates than Cre− mice in the social interaction zone during trial 2 (Mann–Whitney U = 67,155, *P = 0.018; \({n}_{{{\rm{Cre}}}^{-}}\) = 483, \({n}_{{{\rm{Cre}}}^{+}}\) = 308). e, Cre+ iBax mice show higher Ca2+ transient rates than Cre− mice in the corner zones during trial 2 (U = 22,585, ***P < 0.0001; \({n}_{{{\rm{Cre}}}^{-}}\) = 301, \({n}_{{{\rm{Cre}}}^{+}}\) = 206). f, Cell selectivity pie charts for trial 1. g, No differences in the Ca2+ transient rates of social-interaction-zone-selective cells are seen during exploration of the social interaction zone in trial 1 (\({n}_{{{\rm{Cre}}}^{-}}\) = 187, \({n}_{{{\rm{Cre}}}^{+}}\) = 79). h, No differences in the Ca2+ transient rates of corner-selective cells are seen during corner exploration in trial (\({n}_{{{\rm{Cre}}}^{-}}\) = 119, \({n}_{{{\rm{Cre}}}^{+}}\) = 63). i, Cell selectivity pie charts for trial 2. j, Social-interaction-zone-selective cells show lower Ca2+ transient rates in Cre+ iBax mice than in Cre− mice during exploration of the social interaction zone in trial 2 (U = 2,218, ***P < 0.0001; \({n}_{{{\rm{Cre}}}^{-}}\) = 97, \({n}_{{{\rm{Cre}}}^{+}}\) = 85). k, Corner-selective cells show higher Ca2+ transient rates in Cre+ iBax mice than in Cre− mice during corner exploration in trial 2 (U = 1,465, ***P = 0.0002; \({n}_{{{\rm{Cre}}}^{-}}\) = 110, \({n}_{{{\rm{Cre}}}^{+}}\) = 43). Error bars, ±s.e.m.
Extended Data Fig. 8 iBax mice: decreased vDG activity during stress-induced anxiety-like behaviour in the open field test.
a, Experimental timeline. b, Cre+ iBax mice show lower Ca2+ transient rates than Cre− mice during open field exploration (Mann–Whitney U = 130,884, ***P < 0.0001; \({n}_{{{\rm{Cre}}}^{-}}\) = 859 cells, \({n}_{{{\rm{Cre}}}^{+}}\) = 494 cells). c, Ca2+ transient rates are plotted for each 5-cm distance bin away from the centre of the open field. Cre− mice exhibit increased Ca2+ transient rates in the centre compared to the periphery (Kruskal–Wallis H = −22.57, ***P < 0.001; Dunn’s test: 0–5 cm versus 25–30 cm distance from centre point: **P = 0.002; 5–10 cm versus 25–30 cm distance from centre point: *P = 0.026). Cre+ iBax mice did not exhibit increased Ca2+ transient rates in the centre compared to the periphery (H = 0.93, P = 0.97). d, Cell selectivity pie charts. e, Centre-selective cells show lower Ca2+ transient rates in Cre+ iBax mice than in Cre− mice during centre exploration (Mann–Whitney U = 13,892, ***P < 0.001, \({n}_{{{\rm{Cre}}}^{-}}\) = 302, \({n}_{{{\rm{Cre}}}^{+}}\) = 122). f, Periphery-selective cells show lower Ca2+ transient rates in Cre+ iBax mice than in Cre− mice during periphery exploration (U = 34,033, ***P < 0.001, \({n}_{{{\rm{Cre}}}^{-}}\) = 450, \({n}_{{{\rm{Cre}}}^{+}}\) = 241). Error bars, ±s.e.m.
Extended Data Fig. 9 Manipulating mature granule cell activity counteracts the effect of adult hippocampal neurogenesis on stress resilience.
a, Experimental design for subchronic social defeat stress (5 days) while inhibiting mature granule cells in i-hM4Di mice. b, c, Inhibiting mature granule cells with CNO counteracts the pro-susceptibility phenotype of Cre+ i-hM4Di mice in the social interaction test (b; interaction F1,46 = 4.3, *P = 0.04; genotype F1,46 = 1.8, P = 0.18; CamKII-hM4Di F1,46 = 9.5,**P = 0.004; post hoc test (Cre− mCherry versus Cre+ mCherry), *P = 0.02; post hoc test (Cre+ mCherry versus Cre+ hM4Di),***P = 0.0006; n = 12, 12, 12, 14) and in the open field test (c; interaction F1,46 = 5.57, *P = 0.02; genotype F1,46 = 0.64, P = 0.43; CamKII-hM4Di F1,46 = 0.56, P = 0.46; post hoc test (Cre− mCherry versus Cre+ mCherry), *P = 0.033; post hoc test (Cre+ mCherry versus Cre+ hM4Di),*P = 0.03; n = 12, 12, 12, 14). d, Experimental design for chronic social defeat stress (10 days) while exciting mature granule cells in iBax mice. e, f, Exciting mature granule cells with CNO counteracts the pro-resilience phenotype of Cre+ iBax mice in the social interaction test (e; interaction F1,46 = 5.63, *P = 0.02; genotype F1,46 = 5.52, *P = 0.023; CNO F1,46 = 0.57, P = 0.45; post hoc test (Cre− vehicle versus Cre+ vehicle), **P = 0.003; post hoc test (Cre+ vehicle versus Cre+ CNO), *P = 0.021; n = 10, 11, 12, 17) and in the open field test (f; interaction F1,46 = 0.5, P = 0.49; genotype F1,46 = 4.1, P = 0.05; CNO F1,46 = 7.71, **P = 0.008; planned comparisons: unpaired t-test (Cre− vehicle versus Cre+ vehicle), P = 0.1; unpaired t-test (Cre+ vehicle versus Cre+ CNO), *P = 0.02; n = 10, 11, 12, 17. Error bars, ±s.e.m.
Extended Data Fig. 10 Acute activation of vDG mature granule cells results in a modest increase in anxiety during open field exploration and does not activate the lateral septum.
a, Experimental design for acute stimulation of vDG mature granule cells during open field exploration. b, Exciting mature granule cells with CNO during open field exploration decreases centre distance travelled as a proportion of the total distance (unpaired t-test; *P = 0.039; n = 8, 10). c, No difference was observed for the total time spent in the centre (P = 0.72; n = 8, 10). d, No difference in c-fos expression was observed in the lateral septum (LS) of mice in which the vDG was stimulated during open field exploration using CNO-mediated activation of hM3Dq (P = 0.82; n = 6, 4). e, Representative images of c-fos expression in the lateral septum upon injection with vehicle (VEH) or CNO. f, Increased c-fos expression was observed in the lateral septum of mice upon optogenetic activation of the vDG (POMC-Cre;ChR2), which has been shown to decrease anxiety-like behaviour8 (*P = 0.01; n = 6, 3). g, Representative images of c-fos expression in the lateral septum upon light activation of ChR2 (Cre+; POMC-Cre;ChR2). Error bars, ±s.e.m.
Supplementary information
Source Data
Rights and permissions
About this article
Cite this article
Anacker, C., Luna, V.M., Stevens, G. et al. Hippocampal neurogenesis confers stress resilience by inhibiting the ventral dentate gyrus. Nature 559, 98–102 (2018). https://doi.org/10.1038/s41586-018-0262-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0262-4
This article is cited by
-
Neuronal Stem Cells from Late-Onset Alzheimer Patients Show Altered Regulation of Sirtuin 1 Depending on Apolipoprotein E Indicating Disturbed Stem Cell Plasticity
Molecular Neurobiology (2024)
-
Aging Modulates the Ability of Quiescent Radial Glia-Like Stem Cells in the Hippocampal Dentate Gyrus to be Recruited into Division by Pro-neurogenic Stimuli
Molecular Neurobiology (2024)
-
Effect of 2-Week Naringin Supplementation on Neurogenesis and BDNF Levels in Ischemia–Reperfusion Model of Rats
NeuroMolecular Medicine (2024)
-
Induced Mesenchymal Stem Cells-Small Extracellular Vesicles Alleviate Post-stroke Cognitive Impairment by Rejuvenating Senescence of Neural Stem Cells
Journal of Molecular Neuroscience (2024)
-
Upregulation of carbonic anhydrase 1 beneficial for depressive disorder
Acta Neuropathologica Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.