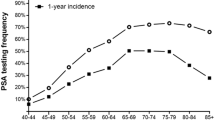
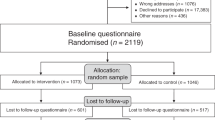
Abstract
Decisions around prostate-specific antigen screening require a patient-centred approach, considering the benefits and risks of potential harm. Using shared decision-making (SDM) can improve men’s knowledge and reduce decisional conflict. SDM is supported by evidence, but can be difficult to implement in clinical settings. An inclusive definition of SDM was used in order to determine the prevalence of SDM in prostate cancer screening decisions. Despite consensus among guidelines endorsing SDM practice, the prevalence of SDM occurring before the decision to undergo or forgo prostate-specific antigen testing varied between 11% and 98%, and was higher in studies in which SDM was self-reported by physicians than in patient-reported recollections and observed practices. The influence of trust and continuity in physician–patient relationships were identified as facilitators of SDM, whereas common barriers included limited appointment times and poor health literacy. Decision aids, which can help physicians to convey health information within a limited time frame and give patients increased autonomy over decisions, are underused and were not shown to clearly influence whether SDM occurs. Future studies should focus on methods to facilitate the use of SDM in clinical settings.
Key points
-
Shared decision-making (SDM) about prostate-specific antigen screening should be collaborative between patients and physicians, and should consist of eliciting patients’ preferences, providing evidence-based information about risks and benefits, and reaching a values-concordant choice.
-
The use of SDM for prostate cancer screening is suggested by guideline groups, but SDM remains underused.
-
Facilitators to SDM include a consistent clinician–provider relationship, trust in the clinician, having a partner, and high education level.
-
Barriers to SDM include limited appointment times, insufficient knowledge, poor health literacy, any barrier to communication, and physician beliefs about screening.
-
Decision aids can help to improve patients’ knowledge and facilitate SDM, but are rarely used in clinical practice.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Charles, C., Gafni, A. & Whelan, T. Shared decision-making in the medical encounter: what does it mean? (or it takes at least two to tango). Soc. Sci. Med. 44, 681–692 (1997).
Makarov, D. V. et al. AUA white paper on implementation of shared decision making into urological practice. Urol. Pract. 3, 355–363 (2016).
Chan, E. C. & Sulmasy, D. P. What should men know about prostate-specific antigen screening before giving informed consent? Am. J. Med. 105, 266–274 (1998).
Barry, M. J. & Edgman-Levitan, S. Shared decision making — pinnacle of patient-centered care. N. Engl. J. Med. 366, 780–781 (2012).
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Franlund, M. et al. Results from 22 years of followup in the Goteborg randomized population-based prostate cancer screening trial. J. Urol. 208, 292–300 (2022).
Hugosson, J. et al. A 16-yr follow-up of the European randomized study of screening for prostate cancer. Eur. Urol. 76, 43–51 (2019).
Tsodikov, A. et al. Reconciling the effects of screening on prostate cancer mortality in the ERSPC and PLCO trials. Ann. Intern. Med. 167, 449–455 (2017).
Cantor, S. B., Volk, R. J., Cass, A. R., Gilani, J. & Spann, S. J. Psychological benefits of prostate cancer screening: the role of reassurance. Health Expect. 5, 104–113 (2002).
Heijnsdijk, E. A. et al. Quality-of-life effects of prostate-specific antigen screening. N. Engl. J. Med. 367, 595–605 (2012).
Shill, D. K., Roobol, M. J., Ehdaie, B., Vickers, A. J. & Carlsson, S. V. Active surveillance for prostate cancer. Transl. Androl. Urol. 10, 2809–2819 (2021).
Carlsson, S., Aus, G., Wessman, C. & Hugosson, J. Anxiety associated with prostate cancer screening with special reference to men with a positive screening test (elevated PSA) — results from a prospective, population-based, randomised study. Eur. J. Cancer 43, 2109–2116 (2007).
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Prostate Cancer Early Detection (Version 2.2023). NCCN https://www.nccn.org/professionals/physician_gls/pdf/prostate_detection.pdf (2023).
Wei, J. T. et al. Early detection of prostate cancer: AUA/SUO guideline part I: prostate cancer screening. J .Urol. 210, 45–53 (2023).
Grossman, D. C. et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA 319, 1901–1913 (2018).
EAU Guidelines. Edn. presented at the EAU Annual Congress Milan 2023. ISBN 978-94-92671-19-6. http://uroweb.org/guidelines/compilations-of-all-guidelines/ (EAU Guidelines Office, Arnhem, The Netherlands, 2023).
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. (The National Academies Press, Washington, DC, 2001).
Carter, H. B. et al. Early detection of prostate cancer: AUA guideline. J. Urol. 190, 419–426 (2013).
Hoffman, R. M. et al. Prostate cancer screening decisions: results from the National Survey of Medical Decisions (DECISIONS study). Arch. Intern. Med. 169, 1611–1618 (2009).
Elwyn, G. et al. Shared decision making: a model for clinical practice. J. Gen. Intern. Med. 27, 1361–1367 (2012).
Amin, H. S. et al. Does shared decision making increase prostate screening uptake in countries with a low prevalence of prostate cancer? Afr. Health Sci. 20, 1870–1874, (2020).
Aminsharifi, A. et al. Primary care perspective and implementation of a multidisciplinary, institutional prostate cancer screening algorithm embedded in the electronic health record. Urol. Oncol. 36, 502.e501–502.e506 (2018).
Choi, S. K. et al. Talking about your prostate: perspectives from providers and community members. J. Cancer Educ. 33, 1052–1060 (2018).
Owens, O. L., Kim, S. & Tavakoli, A. S. Are decision aids leading to shared prostate cancer screening decisions among African-American men?: iDecide. Cancer Causes Control 30, 713–719 (2019).
Shungu, N., Diaz, V. A., Perkins, S. & Kulshreshtha, A. Physician attitudes and self-reported practices toward prostate cancer screening in Black and white men. Fam. Med. 54, 30–37 (2022).
Eiriksdottir, V. K., Baldursdottir, B., Fridriksson, J. O. & Valdimarsdottir, H. B. How much information do Icelandic men receive on pros and cons of prostate-specific antigen testing prior to undergoing testing? Am. J. Mens Health 16, 15579883221097805 (2022).
Allen, J. D. et al. Does shared decision-making for prostate cancer screening among African American men happen? it depends on who you ask. J. Racial Ethn. Health Disparities 9, 1225–1233 (2022).
Tiedje, D. et al. Decision aid and cost compensation influence uptake of PSA-based early detection without affecting decisional conflict: a cluster randomised trial. Sci. Rep. 11, 23503 (2021).
Sedgwick, P. & Greenwood, N. Understanding the Hawthorne effect. BMJ 351, h4672 (2015).
Rim, S. H. et al. Primary care providers’ intended use of decision aids for prostate-specific antigen testing for prostate cancer screening. J. Cancer Educ. 34, 666–670 (2019).
Kushalnagar, P., Hill, C., Carrizales, S. & Sadler, G. R. Prostate-specimen antigen (PSA) screening and shared decision making among deaf and hearing male patients. J. Cancer Educ. 35, 28–35 (2020).
Klaassen, Z. et al. The association between physician trust and prostate-specific antigen screening: implications for shared decision-making. Can. Urol. Assoc. J. 12, 395–400 (2018).
Mainous, A. G. III, Rooks, B. J., Mercado, E. S. & Carek, P. J. Patient provider continuity and prostate specific antigen testing: impact of continuity on receipt of a non-recommended test. Front. Med. 8, 622541 (2021).
Cooper, D. L., Rollins, L., Slocumb, T. & Rivers, B. M. Are men making informed decisions according to the prostate-specific antigen test guidelines? Analysis of the 2015 Behavioral Risk Factor Surveillance System. Am. J. Mens Health 13, 1557988319834843 (2019).
Shungu, N. & Sterba, K. R. Barriers and facilitators to informed decision-making about prostate cancer screening among Black men. J. Am. Board Fam. Med. 34, 925–936 (2021).
Woods-Burnham, L. et al. Physician consultations, prostate cancer knowledge, and PSA screening of African American men in the era of shared decision-making. Am. J. Mens Health 12, 751–759 (2018).
Fedewa, S. A. et al. Recent patterns in shared decision making for prostate-specific antigen testing in the United States. Ann. Fam. Med. 16, 139–144 (2018).
Ross, L. E., Hall, I. J., Howard, D. L., Rim, S. H. & Richardson, L. C. Primary care physicians beliefs about prostate-specific antigen evidence uncertainty, screening efficacy, and test use. J. Natl. Med. Assoc. 110, 491–500 (2018).
Nguyen, D.-D. et al. Impact of health literacy on shared decision making for prostate-specific antigen screening in the United States. Cancer 127, 249–256 (2021).
Paredes, A. Z. et al. Influence of English proficiency on patient-provider communication and shared decision-making. Surgery 163, 1220–1225 (2018).
Hoffman, S. S. et al. Examination of prostate-specific antigen (PSA) screening in military and civilian men: analysis of the 2018 Behavioral Risk Factor Surveillance System. Cancer Causes Control 33, 393–402 (2022).
Muscat, D. M. et al. Addressing health literacy in patient decision aids: an update from the International Patient Decision Aid Standards. Med. Decis. Making 41, 848–869 (2021).
Bergengren, O. et al. 2022 update on prostate cancer epidemiology and risk factors-a systematic review. Eur. Urol. 84, 191–206 (2023).
Mahal, B. A. et al. Prostate cancer racial disparities: a systematic review by the prostate cancer foundation panel. Eur. Urol. Oncol. 5, 18–29 (2022).
Burnett, A. L., Nyame, Y. A. & Mitchell, E. Disparities in prostate cancer. J. Natl. Med. Assoc. 115, S38–S45 (2023).
Crittendon, D. R., LaNoue, M. & George, B. Does perceived racism affect prostate cancer screening rates and patient-provider shared discussions among Black and white men? J. Health Care Poor Underserved 33, 5–19 (2022).
Allen, J. D. et al. African American women’s involvement in promoting informed decision-making for prostate cancer screening among their partners/spouses. Am. J. Mens Health 12, 884–893 (2018).
Miller, D. B., Markt, S. C., Nguyen, C. T. & Coleman, O. C. Prostate cancer screening and young Black men: can early communication avoid later health disparities? J. Cancer Educ. 37, 1460–1465 (2022).
Wolf, M. S. et al. Literacy, race, and PSA level among low-income men newly diagnosed with prostate cancer. Urology 68, 89–93 (2006).
Muvuka, B. et al. Health literacy in African-American communities: barriers and strategies. Health Lit. Res. Pract. 4, e138–e143 (2020).
Eneanya, N. D. et al. Health literacy and education as mediators of racial disparities in patient activation within an elderly patient cohort. J. Health Care Poor Underserved 27, 1427–1440 (2016).
Lombardo, J. et al. Perceptions of and barriers to cancer screening by the sexual and gender minority community: a glimpse into the health care disparity. Cancer Causes Control 33, 559–582 (2022).
Quinn, G. P. et al. Cancer and lesbian, gay, bisexual, transgender/transsexual, and queer/questioning (LGBTQ) populations. CA Cancer J. Clin. 65, 384–400 (2015).
Ma, S. J. et al. Prostate cancer screening patterns among sexual and gender minority individuals. Eur. Urol. 79, 588–592 (2021).
Jiang, C., Fedewa, S. A., Wen, Y., Jemal, A. & Han, X. Shared decision making and prostate-specific antigen based prostate cancer screening following the 2018 update of USPSTF screening guideline. Prostate Cancer Prostatic Dis. 24, 77–80 (2021).
Li, J. et al. Prostate-specific antigen testing initiation and shared decision-making: findings from the 2000 and 2015 National Health Interview Surveys. J. Am. Board Fam. Med. 31, 658–662 (2018).
Misra-Hebert, A. D. et al. Testing of a tool for prostate cancer screening discussions in primary care. Front. Oncol. 8, 238 (2018).
Dierks, T., Heijnsdijk, E. A. M., Korfage, I. J., Roobol, M. J. & de Koning, H. J. Informed decision-making based on a leaflet in the context of prostate cancer screening. Patient Educ. Couns. 102, 1483–1489 (2019).
Pickles, K., Carter, S. M., Rychetnik, L., McCaffery, K. & Entwistle, V. A. Primary goals, information-giving and men’s understanding: a qualitative study of Australian and UK doctors’ varied communication about PSA screening. BMJ Open 8, e018009 (2018).
Legare, F., Leblanc, A., Robitaille, H. & Turcotte, S. The decisional conflict scale: moving from the individual to the dyad level. Z. Evid. Fortbild. Qual. Gesundhwes. 106, 247–252 (2012).
Carlsson, S. V., Vickers, A. J., Gonsky, J. P., Hay, J. L. & Hu, J. C. Problems with numbers in decision aids for prostate-specific antigen screening: a critical review. Eur. Urol. 79, 330–333 (2021).
Ivlev, I., Jerabkova, S., Mishra, M., Cook, L. A. & Eden, K. B. Prostate cancer screening patient decision aids: a systematic review and meta-analysis. Am. J. Prev. Med. 55, 896–907 (2018).
Martínez-González, N. A. et al. Shared decision-making for prostate cancer screening and treatment: a systematic review of randomised controlled trials. Swiss Med Wkly 148, w14584 (2018).
McAlpine, K., Lewis, K. B., Trevena, L. J. & Stacey, D. What is the effectiveness of patient decision aids for cancer-related decisions? A systematic review subanalysis. JCO Clin. Cancer Inform. 2, 1–13 (2018).
Riikonen, J. M. et al. Decision aids for prostate cancer screening choice: a systematic review and meta-analysis. JAMA Intern. Med. 179, 1072–1082, (2019).
Vickers, A. J. Decisional conflict, regret, and the burden of rational decision making. Med Decis. Making 37, 3–5 (2017).
Acknowledgements
We sincerely thank Johanna Goldberg, research informationist, for assistance with the literature search. We express our gratitude to Dr. Laura Liberman, Office of Faculty Development, Memorial Sloan Kettering Cancer Center, New York, USA, for the 2021 Summer Clinical Oncology Research Experience (SCORE) Program that enabled D.K.S.’s work on this study. The work of K.R.P., D.K.S., M.A. and S.V.C. on this paper was supported in part by funding from the National Institutes of Health/National Cancer Institute (P30 CA008748). S.V.C. was supported by NIH/NCI grant (K22 CA234400). K.R.P. was supported in part by the Ruth L. Kirschstein National Research Service Award (T32 CA082088).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
S.V.C. has received travel reimbursement and an honorarium from Ipsen and has served on an advisory board for Prostatype Genomics. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Urology thanks H. van der Poel and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pekala, K.R., Shill, D.K., Austria, M. et al. Shared decision-making before prostate cancer screening decisions. Nat Rev Urol (2024). https://doi.org/10.1038/s41585-023-00840-0
Accepted:
Published:
DOI: https://doi.org/10.1038/s41585-023-00840-0