Abstract
Urinary tests have been used as noninvasive, cost-effective tools for screening, diagnosis and monitoring of diseases since ancient times. As we progress through the 21st century, modern analytical platforms have enabled effective measurement of metabolites, with promising results for both a deeper understanding of cancer pathophysiology and, ultimately, clinical translation. The first study to measure metabolomic urinary cancer biomarkers using NMR and mass spectrometry (MS) was published in 2006 and, since then, these techniques have been used to detect cancers of the urological system (kidney, prostate and bladder) and nonurological tumours including those of the breast, ovary, lung, liver, gastrointestinal tract, pancreas, bone and blood. This growing field warrants an assessment of the current status of research developments and recommendations to help systematize future research.
Key points
-
Initial NMR and mass spectrometry (MS) studies of human urine identified biomarkers that can distinguish patients with cancer from healthy controls and outperform many current clinical markers, possibly enabling early detection.
-
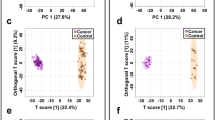
Biomarker panels can be used to identify a single type of cancer, stratify grade and stage, differentiate between multiple cancer types and perform longitudinal evaluations.
-
A similar set of urinary metabolites (hippurate, creatine, tyrosine, citrate, isoleucine, phenylalanine, putrescine, succinate, tryptophan and valine) can indicate malignancy of various organs, possibly reflecting the global metabolic effects of cancer.
-
The lack of specificity means that caution must be exercised and that many biomarkers could be too nonspecific for clinical application.
-
Methodological variations impair comparability of existing studies, highlighting the need for guidelines.
-
The expense of NMR and MS instrumentation means that a centralized testing hub might provide the best solution for eventual clinical implementation of cancer urinary biomarkers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Armstrong, J. A. Urinalysis in Western culture: a brief history. Kidney Int. 71, 384–387 (2007).
Voswinckel, P. From uroscopy to urinalysis. Clin. Chim. Acta 297, 5–16 (2000).
Brian, T. Pisse Prophet, or Certaine Pisse-Pot Lectures (R. Thrale, London, 1637).
Haber, M. H. Pisse prophecy: a brief history of urinalysis. Clin. Lab. Med. 8, 415–430 (1988).
Martin, P. The history of urology. Urol. Cutaneous Rev. 22, 210–214 (1918).
Echeverry, G., Hortin, G. L. & Rai, A. J. Introduction to urinalysis: historical perspectives and clinical application. Methods Mol. Biol. 641, 1–12 (2010).
Pezzuto, F., Buonaguro, L., Buonaguro, F. M. & Tornesello, M. L. The role of circulating free DNA and microRNA in non-invasive diagnosis of HBV- and HCV-related hepatocellular carcinoma. Int. J. Mol. Sci. 19, 1007 (2018).
Lu, T. & Li, J. Clinical applications of urinary cell-free DNA in cancer: current insights and promising future. Am. J. Cancer Res. 7, 2318–2332 (2017).
Nemoto, R., Kato, T., Harada, M., Shibata, K. & Kano, M. Mass screening for urinary tract cancer with urine cytology. J. Cancer Res. Clin. Oncol. 104, 155–159 (1982).
Morrissey, J. J. et al. Evaluation of urine aquaporin-1 and perilipin-2 concentrations as biomarkers to screen for renal cell carcinoma: a prospective cohort study. JAMA Oncol. 1, 204–212 (2015).
Theodorescu, D. et al. Discovery and validation of urinary biomarkers for prostate cancer. Proteomics Clin. Appl. 2, 556–570 (2008).
He, W. S. & Bishop, K. S. The potential use of cell-free-circulating-tumor DNA as a biomarker for prostate cancer. Expert Rev. Mol. Diagn. 16, 839–852 (2016).
Kao, H. W. et al. Urine miR-21-5p as a potential non-invasive biomarker for gastric cancer. Oncotarget 8, 56389–56397 (2017).
Erbes, T. et al. Feasibility of urinary microRNA detection in breast cancer patients and its potential as an innovative non-invasive biomarker. BMC Cancer 15, 193 (2015).
Gasparri, M. L. et al. Beyond circulating microRNA biomarkers: urinary microRNAs in ovarian and breast cancer. Tumour Biol. 39, 1010428317695525 (2017).
Debernardi, S. et al. Noninvasive urinary miRNA biomarkers for early detection of pancreatic adenocarcinoma. Am. J. Cancer Res. 5, 3455–3466 (2015).
Radon, T. P. et al. Identification of a three-biomarker panel in urine for early detection of pancreatic adenocarcinoma. Clin. Cancer Res. 21, 3512–3521 (2015).
Weeks, M. E. et al. Analysis of the urine proteome in patients with pancreatic ductal adenocarcinoma. Proteomics Clin. Appl. 2, 1047–1057 (2008).
Zhang, C. et al. Urine proteome profiling predicts lung cancer from control cases and other tumors. EBioMedicine 30, 120–128 (2018).
Leng, W. et al. Proof-of-concept workflow for establishing reference intervals of human urine proteome for monitoring physiological and pathological changes. EBioMedicine 18, 300–310 (2017).
Horgan, R. P. & Kenny, L. C. ‘Omic’technologies: genomics, transcriptomics, proteomics and metabolomics. Obstet. Gynaecol. 13, 189–195 (2011).
Bathe, O. & Farshidfar, F. From genotype to functional phenotype: unraveling the metabolomic features of colorectal cancer. Genes 5, 536–560 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03727009 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01511653 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01574677 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02781272 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03585114 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02955173 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03091491 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03047616 (2018).
von Minckwitz, G. et al. Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N. Engl. J. Med. 380, 617–628 (2018).
Zhang, Y. L. et al. The prevalence of EGFR mutation in patients with non-small cell lung cancer: a systematic review and meta-analysis. Oncotarget 7, 78985–78993 (2016).
Roberts, L. D., Souza, A. L., Gerszten, R. E. & Clish, C. B. Targeted metabolomics. Curr. Protoc. Mol. Biol. 98, 30.2.1–30.2.24 (2012).
Alonso, A., Marsal, S. & Julia, A. Analytical methods in untargeted metabolomics: state of the art in 2015. Front. Bioeng. Biotechnol. 3, 23 (2015). This paper provides an excellent introduction to the field of metabolomic biomarker research, from sample collection to data analysis, using either NMR spectroscopy or MS.
Shao, Y. et al. Development of urinary pseudotargeted LC-MS-based metabolomics method and its application in hepatocellular carcinoma biomarker discovery. J. Proteome Res. 14, 906–916 (2015).
Moser, H. W., Moser, A. B. & Orr, J. C. Preliminary observations on the occurrence of cholesterol sulfate in man. Biochim. Biophys. Acta 116, 146–155 (1966).
Kruk, J. et al. NMR techniques in metabolomic studies: a quick overview on examples of utilization. Appl. Magn. Reson. 48, 1–21 (2017).
Klein, D. Organic Chemistry (John Wiley & Sons, 2012).
Zerbe, O. & Jurt, S. Applied NMR Spectroscopy for Chemists and Life Scientists (Wiley-VCH, 2013).
Beckonert, O. et al. Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nat. Protoc. 2, 2692–2703 (2007).
Zhang, B., Xie, M., Bruschweiler-Li, L. & Bruschweiler, R. Nanoparticle-assisted metabolomics. Metabolites 8, 21 (2018).
Boiteau, R. M. et al. Structure elucidation of unknown metabolites in metabolomics by combined NMR and MS/MS prediction. Metabolites 8, 8 (2018).
Markley, J. L. et al. The future of NMR-based metabolomics. Curr. Opin. Biotechnol. 43, 34–40 (2017).
Ramaswamy, V. et al. Development of a 13C-optimized 1.5-mm high temperature superconducting NMR probe. J. Magn. Reson. 235, 58–65 (2013).
Ramaswamy, V. et al. Development of a 1H-13C dual-optimized NMR probe based on double-tuned high temperature superconducting resonators. IEEE Trans. Appl. Supercond. 26, 1–5 (2016).
Clendinen, C. S., Pasquel, C., Ajredini, R. & Edison, A. S. 13C NMR metabolomics: INADEQUATE network analysis. Anal. Chem. 87, 5698–5706 (2015).
Keun, H. C. et al. Cryogenic probe 13C NMR spectroscopy of urine for metabonomic studies. Anal. Chem. 74, 4588–4593 (2002).
Dalgliesh, C. E., Horning, E. C., Horning, M. G., Knox, K. L. & Yarger, K. A gas-liquid-chromatographic procedure for separating a wide range of metabolites occuring in urine or tissue extracts. Biochem. J. 101, 792–810 (1966).
Rittenberg, D. & Schoenheimer, R. Studies in protein metabolism VI. Hippuric acid formation studied with the aid of the nitrogen isotope. J. Biol. Chem. 127, 329–331 (1939).
Chan, E. C., Pasikanti, K. K. & Nicholson, J. K. Global urinary metabolic profiling procedures using gas chromatography-mass spectrometry. Nat. Protoc. 6, 1483–1499 (2011).
Want, E. J. et al. Global metabolic profiling of animal and human tissues via UPLC-MS. Nat. Protoc. 8, 17–32 (2013).
Mairinger, T., Causon, T. J. & Hann, S. The potential of ion mobility-mass spectrometry for non-targeted metabolomics. Curr. Opin. Chem. Biol. 42, 9–15 (2018).
Paglia, G. et al. Ion mobility derived collision cross sections to support metabolomics applications. Anal. Chem. 86, 3985–3993 (2014).
Oh, H. B. & Moon, B. Radical-driven peptide backbone dissociation tandem mass spectrometry. Mass Spectrom. Rev. 34, 116–132 (2015).
Mounicou, S., Szpunar, J. & Lobinski, R. Inductively-coupled plasma mass spectrometry in proteomics, metabolomics and metallomics studies. Eur. J. Mass Spectrom. (Chichester) 16, 243–253 (2010).
Wilson, J. M. G. & Jungner, G. Principles and Practice of Screening for Disease (World Health Organization, 1968).
Morrissey, J. J., London, A. N., Luo, J. & Kharasch, E. D. Urinary biomarkers for the early diagnosis of kidney cancer. Mayo Clin. Proc. 85, 413–421 (2010).
Ganti, S. et al. Urinary acylcarnitines are altered in human kidney cancer. Int. J. Cancer 130, 2791–2800 (2012).
Shea, M. W. A proposal for a targeted screening program for renal cancer. Front. Oncol. 3, 207 (2013).
Kim, K. et al. Urine metabolomics analysis for kidney cancer detection and biomarker discovery. Mol. Cell. Proteomics 8, 558–570 (2009). This article describes the importance of uniform urine sample collection time but suggests that the effect of mealtime before specimen collection does not affect findings.
Niziol, J. et al. Metabolomic study of human tissue and urine in clear cell renal carcinoma by LC-HRMS and PLS-DA. Anal. Bioanal. Chem. 410, 3859–3869 (2018).
Falegan, O. S. et al. Urine and serum metabolomics analyses may distinguish between stages of renal cell carcinoma. Metabolites 7, 6 (2017). This novel study combines both NMR and MS to improve differentiation between the urine of patients with renal cell carcinoma of different stages and individuals with benign conditions.
National Cancer Institute. Bladder cancer symptoms, tests, prognosis, and stages (PDQ®)–patient version. NIH https://www.cancer.gov/types/bladder/patient/about-bladder-cancer-pdq (2018).
American Cancer Society. Cancer facts & figures 2017. ACS https://www.cancer.org/Research/Cancer-Facts-Statistics/all-Cancer-Facts-Figures/Cancer-Facts-Figures-2017.Html (2017).
Issaq, H. J. et al. Detection of bladder cancer in human urine by metabolomic profiling using high performance liquid chromatography/mass spectrometry. J. Urol. 179, 2422–2426 (2008).
Pasikanti, K. K. et al. Urinary metabotyping of bladder cancer using two-dimensional gas chromatography time-of-flight mass spectrometry. J. Proteome Res. 12, 3865–3873 (2013).
Pasikanti, K. K. et al. Noninvasive urinary metabonomic diagnosis of human bladder cancer. J. Proteome Res. 9, 2988–2995 (2010).
Huang, Z. et al. Bladder cancer determination via two urinary metabolites: a biomarker pattern approach. Mol. Cell. Proteomics. https://doi.org/10.1074/mcp.M111.007922 (2011).
Wittmann, B. M. et al. Bladder cancer biomarker discovery using global metabolomic profiling of urine. PLOS ONE 9, e115870 (2014).
Srivastava, S. et al. Taurine - a possible fingerprint biomarker in non-muscle invasive bladder cancer: a pilot study by 1H NMR spectroscopy. Cancer Biomark. 6, 11–20 (2010).
Cheng, X. et al. Metabolomics of non-muscle invasive bladder cancer: biomarkers for early detection of bladder cancer. Front. Oncol. 8, 494 (2018).
Shao, C. H. et al. Metabolite marker discovery for the detection of bladder cancer by comparative metabolomics. Oncotarget 8, 38802–38810 (2017).
Liu, X. et al. Investigation of the urinary metabolic variations and the application in bladder cancer biomarker discovery. Int. J. Cancer 143, 408–418 (2018).
Jin, X. et al. Diagnosis of bladder cancer and prediction of survival by urinary metabolomics. Oncotarget 5, 1635–1645 (2014). This study demonstrates high sensitivities and specificities for bladder cancer and detailed identification and interpretation of disease-related metabolites.
Peng, J., Chen, Y. T., Chen, C. L. & Li, L. Development of a universal metabolome-standard method for long-term LC-MS metabolome profiling and its application for bladder cancer urine-metabolite-biomarker discovery. Anal. Chem. 86, 6540–6547 (2014).
Putluri, N. et al. Metabolomic profiling reveals potential markers and bioprocesses altered in bladder cancer progression. Cancer Res. 71, 7376–7386 (2011). This comparison of two different MS modalities measuring the same sample set reveals different metabolomic patterns.
Alberice, J. V. et al. Searching for urine biomarkers of bladder cancer recurrence using a liquid chromatography-mass spectrometry and capillary electrophoresis-mass spectrometry metabolomics approach. J. Chromatogr. A 1318, 163–170 (2013).
Bryan, G. T. The role of urinary tryptophan metabolites in the etiology of bladder cancer. Am. J. Clin. Nutr. 24, 841–847 (1971).
Rose, D. Aspects of tryptophan metabolism in health and disease: a review. J. Clin. Pathol. 25, 17 (1972).
Sreekumar, A. et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 457, 910–914 (2009).
Wu, H. et al. GC/MS-based metabolomic approach to validate the role of urinary sarcosine and target biomarkers for human prostate cancer by microwave-assisted derivatization. Anal. Bioanal. Chem. 401, 635–646 (2011).
Derezinski, P., Klupczynska, A., Sawicki, W., Palka, J. A. & Kokot, Z. J. Amino acid profiles of serum and urine in search for prostate cancer biomarkers: a pilot study. Int. J. Med. Sci. 14, 1–12 (2017).
Struck-Lewicka, W. et al. Urine metabolic fingerprinting using LC-MS and GC-MS reveals metabolite changes in prostate cancer: a pilot study. J. Pharm. Biomed. Anal. 111, 351–361 (2015).
Gamagedara, S. et al. Validation study of urinary metabolites as potential biomarkers for prostate cancer detection. Bioanalysis 4, 1175–1183 (2012).
Bales, J. R., Sadler, P. J., Nicholson, J. K. & Timbrell, J. A. Urinary excretion of acetaminophen and its metabolites as studied by proton NMR spectroscopy. Clin. Chem. 30, 1631–1636 (1984).
Zaragoza, P. et al. Towards the potential use of (1)H NMR spectroscopy in urine samples for prostate cancer detection. Analyst 139, 3875–3878 (2014).
Perez-Rambla, C. et al. Non-invasive urinary metabolomic profiling discriminates prostate cancer from benign prostatic hyperplasia. Metabolomics 13, 52 (2017).
Slupsky, C. M. et al. Urine metabolite analysis offers potential early diagnosis of ovarian and breast cancers. Clin. Cancer Res. 16, 5835–5841 (2010).
Zhang, T. et al. Identification of potential biomarkers for ovarian cancer by urinary metabolomic profiling. J. Proteome Res. 12, 505–512 (2013).
Woo, H. M. et al. Mass spectrometry based metabolomic approaches in urinary biomarker study of women’s cancers. Clin. Chim. Acta 400, 63–69 (2009). The study demonstrates that urinary metabolomic panels can distinguish not just cancer from benign conditions but also one type of cancer from another.
Langer, R. D. et al. Transvaginal ultrasonography compared with endometrial biopsy for the detection of endometrial disease. Postmenopausal estrogen/progestin interventions trial. N. Engl. J. Med. 337, 1792–1798 (1997).
Smith, R. A. et al. American Cancer Society guidelines for the early detection of cancer: update of early detection guidelines for prostate, colorectal, and endometrial cancers. Also: update 2001—testing for early lung cancer detection. CA Cancer J. Clin. 51, 38–75 (2001).
Shao, X. et al. Screening and verifying endometrial carcinoma diagnostic biomarkers based on a urine metabolomic profiling study using UPLC-Q-TOF/MS. Clin. Chim. Acta 463, 200–206 (2016).
Duk, J. M., Aalders, J. G., Fleuren, G. J. & de Bruijn, H. W. CA 125: a useful marker in endometrial carcinoma. Am. J. Obstet. Gynecol. 155, 1097–1102 (1986).
Zhang, X. et al. Associations of self-reported sleep duration and snoring with colorectal cancer risk in men and women. Sleep 36, 681–688 (2013).
Prasad, V., Lenzer, J. & Newman, D. H. Why cancer screening has never been shown to “save lives”—and what we can do about it. BMJ 352, h6080 (2016).
Frickenschmidt, A. et al. Metabonomics in cancer diagnosis: mass spectrometry-based profiling of urinary nucleosides from breast cancer patients. Biomarkers 13, 435–449 (2008).
Cala, M., Aldana, J., Sanchez, J., Guio, J. & Meesters, R. J. W. Urinary metabolite and lipid alterations in Colombian Hispanic women with breast cancer: a pilot study. J. Pharm. Biomed. Anal. 152, 234–241 (2018).
Chen, Y. et al. RRLC-MS/MS-based metabonomics combined with in-depth analysis of metabolic correlation network: finding potential biomarkers for breast cancer. Analyst 134, 2003–2011 (2009).
Cho, S. H. et al. Direct determination of nucleosides in the urine of patients with breast cancer using column-switching liquid chromatography-tandem mass spectrometry. Biomed. Chromatogr. 20, 1229–1236 (2006). This article describes the novel development of a MS technique to quantify nucleosides in urine without interference, a problem experienced by other methods at the time.
Chen, J. et al. Urinary metabolomics for discovering metabolic biomarkers of laryngeal cancer using UPLC-QTOF/MS. J. Pharm. Biomed. Anal. 167, 83–89 (2019).
Beckham, T. H. et al. Targeting sphingolipid metabolism in head and neck cancer: rational therapeutic potentials. Expert Opin. Ther. Targets 14, 529–539 (2010).
Erben, V., Bhardwaj, M., Schrotz-King, P. & Brenner, H. Metabolomics biomarkers for detection of colorectal neoplasms: a systematic review. Cancers (Basel) 10, E246 (2018).
Weir, T. L. et al. Stool microbiome and metabolome differences between colorectal cancer patients and healthy adults. PLOS ONE 8, e70803 (2013).
Liang, Q., Wang, C. & Li, B. Metabolomic analysis using liquid chromatography/mass spectrometry for gastric cancer. Appl. Biochem. Biotechnol. 176, 2170–2184 (2015).
Jung, J. et al. Noninvasive diagnosis and evaluation of curative surgery for gastric cancer by using NMR-based metabolomic profiling. Ann. Surg. Oncol. 21(Suppl. 4), S736–S742 (2014).
Pasechnikov, V., Chukov, S., Fedorov, E., Kikuste, I. & Leja, M. Gastric cancer: prevention, screening and early diagnosis. World J. Gastroenterol. 20, 13842–13862 (2014).
Chen, J. L., Fan, J. & Lu, X. J. CE-MS based on moving reaction boundary method for urinary metabolomic analysis of gastric cancer patients. Electrophoresis 35, 1032–1039 (2014).
Chan, A. W. et al. (1)H-NMR urinary metabolomic profiling for diagnosis of gastric cancer. Br. J. Cancer 114, 59–62 (2016).
Duffy, M. J. et al. Use of faecal markers in screening for colorectal neoplasia: a European group on tumor markers position paper. Int. J. Cancer 128, 3–11 (2011).
Gao, Y. et al. Evaluation of serum CEA, CA19-9, CA72-4, CA125 and ferritin as diagnostic markers and factors of clinical parameters for colorectal cancer. Sci. Rep. 8, 2732 (2018).
Ma, Y. L. et al. Ultra-high performance liquid chromatography-mass spectrometry for the metabolomic analysis of urine in colorectal cancer. Dig. Dis. Sci. 54, 2655–2662 (2009).
Cheng, Y. et al. Distinct urinary metabolic profile of human colorectal cancer. J. Proteome Res. 11, 1354–1363 (2012).
Wang, Z. et al. NMR-based metabolomic techniques identify potential urinary biomarkers for early colorectal cancer detection. Oncotarget 8, 105819–105831 (2017).
Davis, V. W., Schiller, D. E., Eurich, D., Bathe, O. F. & Sawyer, M. B. Pancreatic ductal adenocarcinoma is associated with a distinct urinary metabolomic signature. Ann. Surg. Oncol. 20 (Suppl. 3), 415–423 (2013). This paper shows the excellent performance of urinary metabolomics for the distinction of benign conditions versus pancreatic ductal adenocarcinoma, a cancer with a particularly poor outcome.
Kinross, J. M., Drymousis, P., Jimenez, B. & Frilling, A. Metabonomic profiling: a novel approach in neuroendocrine neoplasias. Surgery 154, 1185–1192 (2013).
Yang, X. et al. Diagnostic value of circulating chromogranin a for neuroendocrine tumors: a systematic review and meta-analysis. PLOS ONE 10, e0124884 (2015).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 68, 7–30 (2018).
Ji, M. et al. Mass screening for liver cancer: results from a demonstration screening project in Zhongshan City, China. Sci. Rep. 8, 12787 (2018).
World Cancer Research Fund. Liver cancer statistics: liver cancer is the sixth most common cancer worldwide. WCRF https://www.wcrf.org/dietandcancer/cancer-trends/liver-cancer-statistics (2018).
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 68, 394–424 (2018).
Shariff, M. I. et al. Hepatocellular carcinoma: current trends in worldwide epidemiology, risk factors, diagnosis and therapeutics. Expert Rev. Gastroenterol. Hepatol. 3, 353–367 (2009).
Akinyemiju, T. et al. The burden of primary liver cancer and underlying etiologies from 1990 to 2015 at the global, regional, and national level: results from the Global Burden of Disease Study 2015. JAMA Oncol. 3, 1683–1691 (2017).
Shariff, M. I. et al. Characterization of urinary biomarkers of hepatocellular carcinoma using magnetic resonance spectroscopy in a Nigerian population. J. Proteome Res. 9, 1096–1103 (2010).
Cox, I. J. et al. Urinary nuclear magnetic resonance spectroscopy of a Bangladeshi cohort with hepatitis-B hepatocellular carcinoma: a biomarker corroboration study. World J. Gastroenterol. 22, 4191–4200 (2016).
Shariff, M. I. et al. Urinary metabolic biomarkers of hepatocellular carcinoma in an Egyptian population: a validation study. J. Proteome Res. 10, 1828–1836 (2011).
Ladep, N. G. et al. Discovery and validation of urinary metabotypes for the diagnosis of hepatocellular carcinoma in West Africans. Hepatology 60, 1291–1301 (2014).
Osman, D., Ali, O., Obada, M., El-Mezayen, H. & El-Said, H. Chromatographic determination of some biomarkers of liver cirrhosis and hepatocellular carcinoma in Egyptian patients. Biomed. Chromatogr. 31, e3893 (2017).
Liu, R. et al. Determination of polyamine metabolome in plasma and urine by ultrahigh performance liquid chromatography-tandem mass spectrometry method: application to identify potential markers for human hepatic cancer. Anal. Chim. Acta 791, 36–45 (2013).
Dai, W. et al. Study of urinary steroid hormone disorders: difference between hepatocellular carcinoma in early stage and cirrhosis. Anal. Bioanal. Chem. 406, 4325–4335 (2014).
Chen, T. et al. Serum and urine metabolite profiling reveals potential biomarkers of human hepatocellular carcinoma. Mol. Cell. Proteomics https://doi.org/10.1074/mcp.M110.004945 (2011).
Chen, J. et al. Metabonomics study of liver cancer based on ultra performance liquid chromatography coupled to mass spectrometry with HILIC and RPLC separations. Anal. Chim. Acta 650, 3–9 (2009).
Wu, H. et al. Metabolomic profiling of human urine in hepatocellular carcinoma patients using gas chromatography/mass spectrometry. Anal. Chim. Acta 648, 98–104 (2009).
Xu, H. et al. Polyamine metabolites profiling for characterization of lung and liver cancer using an LC-tandem MS method with multiple statistical data mining strategies: discovering potential cancer biomarkers in human plasma and urine. Molecules 21, E1040 (2016).
Liang, Q., Liu, H., Wang, C. & Li, B. Phenotypic characterization analysis of human hepatocarcinoma by urine metabolomics approach. Sci. Rep. 6, 19763 (2016). This article shows the great potential of urinary metabolomics for cancer detection at an early stage and contains a thorough pathway analysis.
Warburg, O., Posener, K. & Negelein, E. VIII. The metabolism of cancer cells [German]. Biochem. Z. 152, 129–169 (1924).
Lok, A. S. et al. Des-γ-carboxy prothrombin and α-fetoprotein as biomarkers for the early detection of hepatocellular carcinoma. Gastroenterology 138, 493–502 (2010).
Tzartzeva, K. et al. Surveillance imaging and alpha fetoprotein for early detection of hepatocellular carcinoma in patients with cirrhosis: a meta-analysis. Gastroenterology 154, 1706–1718 (2018).
Berger, E. et al. Can liver cancer be found early? ACS https://www.cancer.org/cancer/liver-cancer/detection-diagnosis-staging/detection.html (2016).
Stewart, B. W. & Wild, C. P. (eds) World Cancer Report 2014 Vol. 3 (International Agency for Research on Cancer, 2017).
Carrola, J. et al. Metabolic signatures of lung cancer in biofluids: NMR-based metabonomics of urine. J. Proteome Res. 10, 221–230 (2011).
An, Z. et al. Integrated ionization approach for RRLC-MS/MS-based metabonomics: finding potential biomarkers for lung cancer. J. Proteome Res. 9, 4071–4081 (2010).
Yang, Q. et al. Urinary metabonomic study of lung cancer by a fully automatic hyphenated hydrophilic interaction/RPLC-MS system. J. Sep. Sci. 33, 1495–1503 (2010).
Zhou, S. et al. Asymmetric dimethylarginine and all-cause mortality: a systematic review and meta-analysis. Sci. Rep. 7, 44692 (2017).
Callejon-Leblic, B., Garcia-Barrera, T., Pereira-Vega, A. & Gomez-Ariza, J. L. Metabolomic study of serum, urine and bronchoalveolar lavage fluid based on gas chromatography mass spectrometry to delve into the pathology of lung cancer. J. Pharm. Biomed. Anal. 163, 122–129 (2019).
Ilias, I., Sahdev, A., Reznek, R. H., Grossman, A. B. & Pacak, K. The optimal imaging of adrenal tumours: a comparison of different methods. Endocr. Relat. Cancer 14, 587–599 (2007).
Arlt, W. et al. Urine steroid metabolomics as a biomarker tool for detecting malignancy in adrenal tumors. J. Clin. Endocrinol. Metab. 96, 3775–3784 (2011).
Zhang, Z. et al. Serum and urinary metabonomic study of human osteosarcoma. J. Proteome Res. 9, 4861–4868 (2010).
Pallister, T. et al. Hippurate as a metabolomic marker of gut microbiome diversity: modulation by diet and relationship to metabolic syndrome. Sci. Rep. 7, 13670 (2017).
Yang, Q. J. et al. Serum and urine metabolomics study reveals a distinct diagnostic model for cancer cachexia. J. Cachexia Sarcopenia Muscle 9, 71–85 (2017). This paper shows that metabolomic biomarker panels can be useful not just for identifying cancer but also for identifying the condition of cachexia, which is associated with malignancies.
Lodi, A. et al. Proton NMR-based metabolite analyses of archived serial paired serum and urine samples from myeloma patients at different stages of disease activity identifies acetylcarnitine as a novel marker of active disease. PLOS ONE 8, e56422 (2013).
Shen, C. et al. Developing urinary metabolomic signatures as early bladder cancer diagnostic markers. OMICS 19, 1–11 (2015).
Polanski, M. & Anderson, N. L. A list of candidate cancer biomarkers for targeted proteomics. Biomark. Insights 2006, 1–48 (2007).
Smolinska, A., Blanchet, L., Buydens, L. M. & Wijmenga, S. S. NMR and pattern recognition methods in metabolomics: from data acquisition to biomarker discovery: a review. Anal. Chim. Acta 750, 82–97 (2012).
Settle, F. A. (ed.) Handbook of Instrumental Techniques for Analytical Chemistry (Prentice Hall, 1997).
Gromski, P. S. et al. A tutorial review: metabolomics and partial least squares-discriminant analysis–a marriage of convenience or a shotgun wedding. Anal. Chim. Acta 879, 10–23 (2015).
Capati, A., Ijare, O. B. & Bezabeh, T. Diagnostic applications of nuclear magnetic resonance-based urinary metabolomics. Magn. Reson. Insights 10, 1–12 (2017).
Trivedi, D. K. & Iles, R. K. Do not just do it, do it right: urinary metabolomics—establishing clinically relevant baselines. Biomed. Chromatogr. 28, 1491–1501 (2014).
Emwas, A. H. et al. Standardizing the experimental conditions for using urine in NMR-based metabolomic studies with a particular focus on diagnostic studies: a review. Metabolomics 11, 872–894 (2015).
de Hoffmann, E. in Kirk-Othmer Encyclopedia of Chemical Technology 4th edn (John Wiley & Sons, 2000).
Castro-Perez, J., Plumb, R. & Granger, J. Maximizing chromatographic resolution of metabolites using ultra performance liquid chromatography (UPLC [TM]). Pharm. Discov. 4, 50–51 (2004).
Wilson, I. D. et al. HPLC-MS-based methods for the study of metabonomics. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 817, 67–76 (2005).
Nassar, A. F., Wu, T., Nassar, S. F. & Wisnewski, A. V. UPLC-MS for metabolomics: a giant step forward in support of pharmaceutical research. Drug Discov. Today 22, 463–470 (2017).
Yu, C. et al. Multi-channel microfluidic chip coupling with mass spectrometry for simultaneous electro-sprays and extraction. Sci. Rep. 7, 17389 (2017).
Turner, D. et al. Cost effectiveness of management strategies for urinary tract infections: results from randomised controlled trial. BMJ 340, c346 (2010).
Bernini, P. et al. Standard operating procedures for pre-analytical handling of blood and urine for metabolomic studies and biobanks. J. Biomol. NMR 49, 231–243 (2011).
Liu, L., Mo, H., Wei, S. & Raftery, D. Quantitative analysis of urea in human urine and serum by 1H nuclear magnetic resonance. Analyst 137, 595–600 (2012).
Giraudeau, P., Silvestre, V. & Akoka, S. Optimizing water suppression for quantitative NMR-based metabolomics: a tutorial review. Metabolomics 11, 1041–1055 (2015).
Hasim, A. et al. Revealing the metabonomic variation of EC using (1)H-NMR spectroscopy and its association with the clinicopathological characteristics. Mol. Biol. Rep. 39, 8955–8964 (2012).
Hao, J. et al. Bayesian deconvolution and quantification of metabolites in complex 1D NMR spectra using BATMAN. Nat. Protoc. 9, 1416 (2014).
Weljie, A. M., Newton, J., Mercier, P., Carlson, E. & Slupsky, C. M. Targeted profiling: quantitative analysis of 1 H NMR metabolomics data. Anal. Chem. 78, 4430–4442 (2006).
Chemonx NMR Suite Version 8.4 (Chenomx Inc., Alberta, Canada).
Sumner, L. W. et al. Proposed minimum reporting standards for chemical analysis Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics 3, 211–221 (2007).
Spicer, R. A., Salek, R. & Steinbeck, C. A decade after the metabolomics standards initiative it’s time for a revision. Sci. Data 4, 170138 (2017).
Protospatharius, T. On Urines (Biblioteca Universitaria Bologna, 15th c.).
Nicholson, J. K. & Lindon, J. C. Systems biology: metabonomics. Nature 455, 1054–1056 (2008).
Coryn, B. The Urine Doctor, holding a matula to examine urine [Print]. Harvard Art Museums https://www.harvardartmuseums.org/art/235931 (17th c.).
Brownlee, C. Lab on a stick. ACS https://www.acs.org/content/dam/acsorg/education/resources/highschool/chemmatters/gc-lab-on-a-stick.pdf (2004).
Guijas, C. et al. METLIN: a technology platform for identifying knowns and unknowns. Anal. Chem. 90, 3156–3164 (2018).
Acknowledgements
The authors kindly acknowledge J. A. Fordham for editorial assistance. A.H. gratefully acknowledges the German Academic Foundation Cusanuswerk, the German Academic Exchange Service and the Max Weber Programme of the State of Bavaria for financial support. Furthermore, A.H. thanks the Department of Diagnostic and Interventional Neuroradiology, University Hospital of Würzburg. S.S.D. gratefully acknowledges the German Academic Scholarship Foundation for financial support.
Review criteria
Representative papers were selected using a PubMed search with the terms “metabolomics” AND “biomarkers” AND “urine” AND “cancer”, which were combined with search strings for the different techniques. For mass spectrometry, those included “mass spectrometry” OR “gas chromatography-mass spectrometry” OR “gc ms” OR “liquid chromatography”. For NMR spectroscopy, the terms “Magnetic Resonance Spectroscopy” OR “Nuclear Magnetic Resonance Spectroscopy” were used. All terms were input with variations in notation and specifications regarding the type of search term MeSH Terms, TIAB and All Fields.
Author information
Authors and Affiliations
Contributions
S.S.D., A.H. and L.A.V. researched data for the article. L.L.C., S.S.D., A.H., L.A.V. and I.A.K. wrote the manuscript. All authors made substantial contributions to discussions of content and reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Uroscopy
-
Medical examination of the urine in order to facilitate diagnosis of a disease or disorder. It is a non-laboratory examination that usually relies on examination of colour, cloudiness and precipitates.
- Pseudotargeted approaches
-
Pseudotargeted approaches combine both untargeted and targeted mass spectrometry methods, thereby aiming to reduce disadvantages of either technique alone. A pseudotargeted metabolomics method contains nontargeted profiling, ion pair picking and multiple reaction monitoring measurement. Consequently, global metabolome information can be retrieved, even if some metabolites are not identified. At the same time, pseudotargeted approaches offer improved reproducibility, simplified data processing and a more expansive linear range than the traditional untargeted metabolomics methods.
- Multiple reaction monitoring
-
(MRM). MRM is an extension of selected reaction monitoring (SRM), a popular tandem mass spectrometry (MS/MS) technique in which the instrument is set up to select the precursor ions at a single mass-to-charge ratio (m/z) value and transmit the product ions at a single m/z value. SRM enables a great degree of selectivity to be achieved and is typically used to detect a single metabolite in a complex mixture (assuming the fragmentation pattern of its molecular ion is known and a suitable fragment ion can be selected). MRM takes advantage of the ability of many modern mass spectrometers to perform multiple MS/MS experiments on a chromatographic timescale, thereby enabling multiple (chromatographically unresolved) metabolites to be identified on the basis of the unique precursor and/or fragment pair for each of them.
- Dynamic nuclear polarization
-
(DNP). A method to hyperpolarize NMR-active nuclei such as 13C or 15N, thereby amplifying the signal. In DNP, the sample is polarized in the presence of microwave-irradiated free radicals. An alternative method, SABRE-RELAY (signal amplification by reversible exchange–relayed coherence transfer), can also be used. In SABRE, a latent singlet spin order of parahydrogen is switched on so that it can polarize a substrate. RELAY is an NMR pulse sequence.
- Principal component analysis
-
(PCA). A popular unsupervised approach for deriving a low-dimensional set of features from a large set of variables. It is often used to identify clusters or outliers.
- Linear discriminant analysis
-
(L-DA). A supervised approach for predicting a categorical response. L-DA is a popular approach when there are more than two response classes.
- Leave-one-out cross-validation
-
A validation approach suitable for small data sets. The data set is repeatedly split into a training set containing all but one observation, and a validation set that contains only that observation and the test error is measured each time.
- Partial least squares discriminant analysis
-
(PLS-DA). A supervised analysis to identify features that contribute most to variation or separation of groups, applicable for separation to several groups. OPLS-DA incorporates an orthogonal signal correction filter and is, therefore, useful for the separation of two groups.
- AUROC
-
(Area under the receiver operating characteristic curve). A graphic for simultaneously displaying the true-positive rate and the false-positive rate of a test. An ideal curve is close to the top left corner; thus, the larger the area under the curve, the better the classifier.
- Least absolute shrinkage and selection operator (LASSO) regularization
-
Also known as regression with L1 regularization. LASSO regulation relies upon a linear model but uses an alternative procedure to estimate the coefficients, making it less flexible and easier to interpret because the response variable will only be related to a small subset of the predictors. The remaining predictors are set to zero.
Rights and permissions
About this article
Cite this article
Dinges, S.S., Hohm, A., Vandergrift, L.A. et al. Cancer metabolomic markers in urine: evidence, techniques and recommendations. Nat Rev Urol 16, 339–362 (2019). https://doi.org/10.1038/s41585-019-0185-3
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-019-0185-3
This article is cited by
-
Investigating the causal associations between metabolic biomarkers and the risk of kidney cancer
Communications Biology (2024)
-
Artificial urinary biomarker probes for diagnosis
Nature Reviews Bioengineering (2024)
-
Urinary microbiome-based metagenomic signature for the noninvasive diagnosis of hepatocellular carcinoma
British Journal of Cancer (2024)
-
High-throughput metabolomics identifies new biomarkers for cervical cancer
Discover Oncology (2024)
-
Urinary biomarkers for hepatocellular carcinoma: current knowledge for clinicians
Cancer Cell International (2023)