Abstract
Despite widespread clinical use of antimalarial drugs such as hydroxychloroquine and chloroquine in the treatment of rheumatoid arthritis (RA), systemic lupus erythematosus (SLE) and other inflammatory rheumatic diseases, insights into the mechanism of action of these drugs are still emerging. Hydroxychloroquine and chloroquine are weak bases and have a characteristic ‘deep’ volume of distribution and a half-life of around 50 days. These drugs interfere with lysosomal activity and autophagy, interact with membrane stability and alter signalling pathways and transcriptional activity, which can result in inhibition of cytokine production and modulation of certain co-stimulatory molecules. These modes of action, together with the drug’s chemical properties, might explain the clinical efficacy and well-known adverse effects (such as retinopathy) of these drugs. The unknown dose–response relationships of these drugs and the lack of definitions of the minimum dose needed for clinical efficacy and what doses are toxic pose challenges to clinical practice. Further challenges include patient non-adherence and possible context-dependent variations in blood drug levels. Available mechanistic data give insights into the immunomodulatory potency of hydroxychloroquine and provide the rationale to search for more potent and/or selective inhibitors.
Key points
-
The DMARDs hydroxychloroquine and chloroquine are weak bases that accumulate in acidic compartments such as lysosomes and inflamed (acidic) tissues.
-
Both hydroxychloroquine and chloroquine have a large volume distribution and a long half-life, consistent with their slow onset of action and prolonged effects after drug discontinuation.
-
At the molecular level, hydroxychloroquine and chloroquine interfere with lysosomal activity and autophagy, disrupt membrane stability and alter signalling pathways and transcriptional activity.
-
At the cellular level, direct and indirect mechanisms of these drugs inhibit immune activation by reducing Toll-like receptor signalling and cytokine production and, in T cells, reducing CD154 expression.
-
An increased risk of retinopathy resulted in updated ophthalmology guidelines that recommended a maximal daily dose of 5.0 mg/kg actual body weight for hydroxychloroquine; however, insufficient efficacy data support this recommendation.
-
Future research should address whether specific targeting of lysosome and/or autophagosome activity has potential for the treatment of rheumatic diseases.
Similar content being viewed by others
Introduction
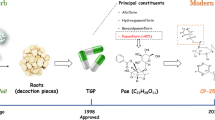
The antimalarial drugs hydroxychloroquine and chloroquine are DMARDs introduced serendipitously and empirically for the treatment of various rheumatic diseases (Fig. 1). Neither chloroquine nor hydroxychloroquine underwent conventional drug development, but their use has become a part of current treatment guidelines for rheumatoid arthritis (RA)1, systemic lupus erythematosus (SLE)2,3,4, antiphospholipid syndrome (APS)5 and primary Sjögren syndrome6,7.
After the very early use of plant extracts for treating malaria, antimalarial drugs were synthesized154 and subsequently approved for medical use155,156. Since the 1940s, chloroquine and hydroxychloroquine have been used for the treatment of rheumatic diseases. New derivatives of these compounds that target autophagosome activity are in development.
Systematic reviews8,9 of randomized controlled and observational studies of antimalarial drugs in SLE, with particular emphasis on hydroxychloroquine, have found strong evidence that this molecule has an immunomodulatory capacity, including the ability to prevent disease flares and promote long-term survival in SLE and control autoimmune disease activity during pregnancies without evidence of fetotoxic or embryotoxic effects. Evidence suggests that hydroxychloroquine can delay or prevent organ damage10, including bone destruction11, in autoimmunity, and that this drug has antithrombotic effects12. Trials assessing antimalarial drugs in hand osteoarthritis, however, have found that hydroxychloroquine is no more effective than placebo in providing pain relief13,14, implying that although antimalarial drugs are useful for a selection of rheumatic diseases, they are not ‘wonder drugs’.
In addition to having direct immunomodulatory effects, chloroquine and hydrochloroquine can reduce rates of atherosclerosis, improve hyperglycaemia and hyperlipidaemia and protect against infections in patients with inflammatory rheumatic diseases15,16. The underlying mechanisms of these clinical consequences, however, remain largely unknown. An important question is if these observed effects share a common mode of action or result from a variety of distinct processes. The mechanisms of action of hydroxychloroquine and chloroquine remain under continuous study in modern molecular medicine17,18 using advanced tools in computational biology19, synthetic biology20,21,22, immunology23,24, structural biology25,26 and ‘big data’-driven public health science27,28. Detailed studies on the mode of action of hydroxychloroquine are needed to better understand dose–response relationships and safety-related aspects. Such knowledge should guide the development of new therapies that modulate lysosomal activity and interfere with autophagy.
The main aim of this Review is to discuss the mode of action of hydroxychloroquine and chloroquine, including pharmacokinetic and pharmacodynamic properties that potentially explain the clinical efficacy and adverse effects of these antimalarial drugs. For a comprehensive overview of the clinical aspects of hydroxychloroquine and chloroquine in the treatment of certain diseases, we refer the reader to the more specialized literature15,29,30.
Pharmacokinetics
Drug structure and chemistry
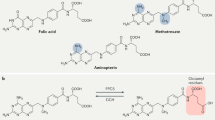
Commonly used antimalarial drugs can be divided into different classes on the basis of their core structure. Hydroxychloroquine and chloroquine belong to a class of drugs known as 4-aminoquinolines, whereas other less frequently used antimalarial drugs belong to other groups (such as the endoperoxidases (artemisinin) or acridines (mepacrine))31. Figure 2 depicts the structure and metabolism of hydroxychloroquine and chloroquine. Both drugs have a flat aromatic core structure and are weak bases due to the presence of a basic side chain. The basic side chain is thought to contribute to the accumulation of these drugs in intracellular compartments, especially lysosomal compartments, which seems to be crucial for their activity and the potential interaction of these drugs with nucleic acids.
a | Hydroxychloroquine and chloroquine belong to a class of drugs known as 4-aminoquinolines. These drugs have a 4-aminoquinoline core structure and a basic side chain. b | Cytochrome P450 (CYP) enzymes mediate dealkylation of chloroquine and hydroxychloroquine. Desethylchloroquine is an immediate downstream product of CYP-mediated dealkylation of both drugs, whereas desethylhydroxychloroquine is a metabolite of only hydroxychloroquine. Bisdesethylchloroquine is a downstream metabolite of both drugs. c | Some of the pharmacokinetic properties of hydroxychloroquine and chloroquine differ. The large volume of distribution and long half-life is characteristic of both drugs; however, these drugs have notably different renal clearance rates. The data in this figure were taken from Costedoat-Chalumeau et al.143 and McChesney40.
Both hydroxychloroquine and chloroquine occur as enantiomers (R and S isomers). (R)-(−)-hydroxychloroquine (the stereochemical ‘rectus’ configuration of hydroxychloroquine) is present at higher concentrations in the blood than (S)-(+)-hydroxychloroquine (the stereochemical ‘sinister’ configuration of hydroxychloroquine)32, suggesting the existence of stereoselective processes in the deposition and/or metabolism of this drug. The efficacy and safety of the drug enantiomers might also differ. However, the (R)-(−) and (S)-(+) isomers of chloroquine have similar effects in vitro33, and the embryotoxicity of chloroquine enantiomers in rats is also equivalent34. Stereoisomer-specific drug formulations have been developed to reduce adverse effects such as risk of retinopathy, but their effects require further clinical investigation35,36 (see below).
Absorption, distribution and elimination
Available data on the pharmacokinetics of hydroxychloroquine and chloroquine (Fig. 2) are largely based on studies of healthy individuals32. The pharmacokinetic characteristics of hydroxychloroquine and chloroquine are complex owing to the large volume of distribution and long half-life of these drugs32. Furthermore, the dose–response relationships and thresholds of toxicity of hydroxychloroquine and chloroquine have yet to be fully delineated.
Hydroxychloroquine is administered as a sulfate, whereas chloroquine is administered as a phosphate salt. Both drugs are usually absorbed in the upper intestinal tract37. The lag time between oral absorption of hydroxychloroquine sulfate (200 mg) and its measurement in the blood is typically 0–0.85 h (mean 0.43 h)37, and both hydroxychloroquine and chloroquine have an overall bioavailability of 0.7–0.8 (ref.38). Some studies have reported marked differences in the pharmacokinetics of hydroxychloroquine and chloroquine in humans38,39; however, these differences can be explained by differences in either the analytical methods applied, the sample source used (that is, plasma versus whole blood), or renal clearance of these drugs38,40.
After absorption, the half-lives of the two drugs are comparably long (40–60 days) owing to a large volume of distribution in the blood (47,257 l for hydroxychloroquine and 65,000 l for chloroquine)39. Both drugs can distribute to aqueous cellular and intercellular compartments, resulting in long mean residence times (~1,300 h for hydroxychloroquine and ~900 h for chloroquine)39. Renal clearance is also an important clinical consideration32, especially in patients with kidney failure, as decreased clearance increases the bioavailability of these drugs (Fig. 2c).
Plasma, blood and serum concentrations of hydroxychloroquine and chloroquine can vary in individual patients (particularly concentrations measured in the serum and whole blood) and between patients (interpatient variability)38. Little information is available concerning drug concentrations in ‘deep’ organs (for example, in lymphoid tissue, immune cells, bone marrow, synovial tissue and other connective tissues) and the pharmacological three-compartment model of drug distribution between tissues cannot be used to predict the drug concentrations in these ‘deep’ organs.
As a weak base, hydroxychloroquine accumulates within acidic vesicles, such as the lysosomal compartment (an important site of action of this drug)41,42. Patients with RA and mild joint symptoms have been reported to have higher blood concentrations of hydroxychloroquine than patients with active seropositive disease43. It is possible that drug level fluctuations could further be influenced by the condition of an individual, such as overt or subclinical inflammation (that is, an acidic milieu) sequestering these drugs.
In 2018, to better understand the complexity of the pharmacokinetics and pharmacodynamics of hydroxychloroquine, researchers used a physiologically based pharmacokinetics model to describe the tissue-specific absorption, distribution, metabolism, excretion and lysosome-specific sequestration of this drug44. The clinical applicability of this model, which requires measurement of plasma concentrations, still needs investigation, but could offer new and important insights into the tissue distribution, dose–response interrelationship and in situ context-related effects of hydroxychloroquine.
Hydroxychloroquine binds strongly to melanin and can deposit in melanin-containing tissues such as the skin and the eyes, which might explain certain tissue-specific mechanisms (such as hydroxychloroquine retinopathy or the efficacy of this drug in the treatment of skin manifestations). Clinical observations suggest that hydroxychloroquine is associated with a lower risk of retinopathy than chloroquine, which might be explained by the lower volume of distribution of hydroxychloroquine compared with chloroquine45.
Drug–drug interactions
The effect of antimalarial drugs on other drugs (and vice versa) is an important clinical consideration. Both chloroquine and hydroxychloroquine are substrates for cytochrome P450 (CYP) enzymes (enzymes responsible for the metabolism of multiple drugs) and hence can interfere with other drugs38,46,47.
CYP enzymes catalyse the dealkylation of chloroquine and hydroxychloroquine to pharmacologically active metabolites38,40,48,49,50 (Fig. 2b). The specific CYP enzymes responsible for the metabolism of various drugs have been investigated using microsomal stability assays or recombinant enzymes48,51,52. CYP2C8, CYP3A4, CYP2D6 and CYP1A1 can metabolize chloroquine48,49,50,52,53. However, the contribution of these isoforms might vary between individuals51,53 and indeed blood concentrations of hydroxychloroquine are reported to vary among individuals54.
In terms of other drugs, concurrent use of chloroquine increases the plasma concentration of digitoxin (a cardiac glycoside) fourfold55. Hence, levels of digitoxin require close monitoring during antimalarial therapy. Hydroxychloroquine influences the levels of metoprolol through prevention of its metabolism by competing for the same CYP enzyme, CYP2D656. As a result, plasma concentrations and the bioavailability of metoprolol increases with hydroxychloroquine therapy. Interestingly, levels of other drugs that are also metabolized by CYP2D6, such dextromethorphan56, are not increased during hydroxychloroquine therapy. As antimalarial drugs are thought to interfere with medications that influence the QT interval, patients on hydroxychloroquine therapy concurrently taking such drugs for the treatment of cardiac comorbidities should also be monitored for the potential risk of cardiac arrhythmia57.
According to ophthalmology recommendations, co-medication of tamoxifen (a selective oestrogen receptor modulatory used to treat breast cancer) with hydroxychloroquine is associated with an increased risk of eye toxicity owing to synergistic inhibition of lysosomal enzymes in retinal epithelial cells58. Thus, combined use of tamoxifen with hydroxychloroquine or chloroquine should be limited to 6 months.
Another relevant drug interaction to consider is the interaction between antimalarial drugs and other DMARDs. Hydroxychloroquine can reduce the gastrointestinal absorption of methotrexate through local pH changes and hence lowers the bioavailability of methotrexate38,59. This effect might explain the decreased risk of methotrexate-associated acute liver adverse effects during co-administration with hydroxychloroquine60. No other interactions between hydroxychloroquine and methotrexate (such as interaction on the enzymatic level47,60) have been reported. Hydroxychloroquine can also increase levels of ciclosporin; hence, levels of ciclosporin should also be closely monitored during combined therapy61.
Some drugs can also interfere with the bioavailability of hydroxychloroquine and chloroquine. For example, agents that increase the pH of gastric acid (for example, proton-pump inhibitors62) might interfere with the oral absorption and oral bioavailability of antimalarial drugs. However, in one study of patients with SLE, the plasma concentrations of hydroxychloroquine between patients taking proton-pump inhibitors and patients not taking proton-pump inhibitors did not differ63. Finally, smoking has previously been suspected to interfere with the bioavailability of hydroxychloroquine64; however, a study in 2017 found no correlation between hydroxychloroquine plasma concentration and smoking status65.
Pregnancy and breastfeeding
Although both chloroquine and hydroxychloroquine cross the placenta, and despite initial concerns relating to the presence of drug-related pigmentations in fetal tissue66, these drugs are not considered to have notable toxic effects on the fetus67. Current guidelines strongly recommend maintaining treatment with hydroxychloroquine during pregnancy in patients with an autoimmune disease68,69,70. Some data suggest that hydroxychloroquine during pregnancy is protective against the induction of congenital heart block71,72, possibly owing to a reduction in expression of a type I interferon (IFN) signature73. Notably, evidence of fetal cardiotoxic effects was absent in one case study74. Hydroxychloroquine can also be transferred in breast milk, but is not associated with adverse events in the child75. Continued therapy after delivery even has the advantage of preventing flares in mothers69. Overall, hydroxychloroquine is considered safe to use during pregnancy and breastfeeding.
Mechanisms of action
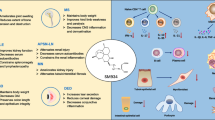
Various modes of action are postulated to explain the therapeutic and/or adverse effects of hydroxychloroquine and chloroquine, most of which have been based on in vitro studies. Notably, the link between these mechanisms and the clinical efficacy and safety observed in vivo have yet to be fully delineated. Antimalarial drugs have direct molecular effects on lysosomal activity, autophagy and signalling pathways (Fig. 3). Data on the effects of these drugs on plasmacytoid dendritic cells (pDCs), B cells, other antigen-presenting cells and T cells are also available (Fig. 4). As with various therapeutic interventions of the immune system (Box 1), the mechanism of action is probably context-dependent (that is, dependent on the inflammatory conditions and/or affected tissues or organs).
a | Hydroxychloroquine (HCQ) enters and accumulates in lysosomes along a pH gradient. In lysosomes, hydroxychloroquine inhibits the degradation of cargo derived externally (via endocytosis or phagocytosis) or internally (via the autophagy pathway) in autolysosomes by increasing the pH to prevent the activity of lysosomal enzymes. Inhibition of lysosomal activity can prevent MHC class II-mediated autoantigen presentation. b | Hydroxychloroquine can also accumulate in endosomes and bind to the minor groove of double-stranded DNA. This drug can inhibit Toll-like receptor (TLR) signalling by altering the pH of endosomes (involved in TLR processing) and/or preventing TLR7 and TLR9 from binding their ligands (RNA and DNA, respectively). Hydroxychloroquine can also inhibit the activity of the nucleic acid sensor cyclic GMP-AMP (cGAMP) synthase (cGAS) by interfering with its binding to cytosolic DNA. By preventing TLR signalling and cGAS–stimulator of interferon genes (STING) signalling, hydroxychloroquine can reduce the production of pro-inflammatory cytokines, including type I interferons.
Hydroxychloroquine (HCQ) can interfere with immune activation at various cellular levels by inhibiting various innate and adaptive immune processes. During autoimmunity, cellular debris can activate Toll-like receptor 7 (TLR7) and TLR7 signalling pathways in plasmacytoid dendritic cells (pDCs) and other immune antigen-presenting cells (APCs), including monocytes, macrophages and B cells, resulting in the activation of multiple cell types and secretion of various pro-inflammatory cytokines. In APCs, hydroxychloroquine potentially interferes with TLR7 and TLR9 ligand binding and TLR signalling (through lysosomal inhibition and reduced MyD88 signalling), which inhibits TLR-mediated cell activation and cytokine production. In APCs, such as pDCs and B cells, this drug also inhibits antigen processing and subsequent MHC class II presentation to T cells, preventing T cell activation, differentiation and expression of co-stimulatory molecules (such as CD154) and also reducing the production of cytokines (such as IL-1, IL-6 and TNF) by both T cells and B cells. BAFF, B-cell activating factor.
Molecular effects
Inhibition of lysosomal activity and autophagy
An important mode of action of chloroquine and hydroxychloroquine is the interference of lysosomal activity and autophagy. It is widely accepted that chloroquine and hydroxychloroquine accumulate in lysosomes (lysosomotropism) and inhibit their function. In vitro, chloroquine can destabilize lysosomal membranes and promote the release of lysosomal enzymes inside cells76. Although evidence of this latter mechanism is scarce, the ability of these drugs to interfere with lysosomal activity has been repeatedly documented77,78,79. Interference of lysosomal activity might inhibit the function of lymphocytes and have immunomodulatory or even anti-inflammatory effects.
One mechanism by which these drugs might have anti-inflammatory effects is by impairing antigen presentation via the lysosomal pathway. Lysosomes contain hydrolytic enzymes and cooperate with other vesicles to digest cargo (such as organelles and material from inside the cell (in a process known as autophagy) or material from outside the cell (via the endocytosis or phagocytosis pathway)). Lysosomes are involved not only in recycling cellular substrates80 but also in antigen processing and MHC class II presentation, indirectly promoting immune activation81. Autophagy is also involved in antigen presentation and immune activation82,83. For example, data from one study suggest that autophagy is important for MHC class II-mediated autoantigen presentation by antigen-presenting cells to CD4+ T cells84. As the pH in lysosomes is optimal for lysosomal enzymes involved in hydrolysis, by increasing the pH of endosomal compartments85, chloroquine and hydroxychloroquine might impair the maturation of lysosomes and autophagosomes and inhibit antigen presentation along the lysosomal pathway (Fig. 3). Overall, the available studies suggest that hydroxychloroquine and chloroquine impair or inhibit lysosomal and autophagosome functions and subsequently immune activation.
Beyond lysosomotropism, efforts to identify exact molecular targets of hydroxychloroquine within the lysosome are also currently underway. One study has identified palmitoyl-protein thioesterase 1 (PPT1), an enzyme involved in the catabolism of lipid-modified proteins, as a potential lysosomal target of chloroquine and chloroquine derivatives86. Hydroxychloroquine can bind and inhibit PPT1 activity86, and, notably, PPT1 is overexpressed in the synovial tissue of patients with RA87. Although an interesting example of ongoing research, confirmatory functional studies and the identification of other molecular targets within the lysosomes are nevertheless warranted.
Inhibition of signalling pathways
Hydroxychloroquine and chloroquine can also interfere with Toll-like receptor (TLR) signalling. For example, changes in endosomal pH can interfere with TLR9 and TLR7 processing88, and, hence, these antimalarial drugs might prevent TLR activation upon extracellular stimuli by mediating changes in the local pH88. Chloroquine or hydroxychloroquine can also directly bind to nucleic acids and hence might block TLR9 signalling at the intracellular level by inhibiting TLR–ligand interactions (steric blockade) (Fig. 3). This latter hypothesis is supported by an analysis based on surface plasmon resonance and fluorescence spectroscopy showing that antimalarial drugs can directly inhibit CpG–TLR9 interactions89,90. In addition to TLR9 signalling, chloroquine can also inhibit RNA-mediated activation of TLR7 signalling91,92. Although the exact modes of action by which these drugs inhibit TLR7 and TLR9 requires further delineation at the molecular level93, inhibition of TLR processing and inhibition of TLR binding are likely central mechanisms of action.
Another potential mode of action of hydroxychloroquine and chloroquine is interference with cyclic GMP-AMP (cGAMP) synthase (cGAS) activity by inhibiting ligand binding94. The cGAS–stimulator of IFN genes (STING) pathway is a major source of the type I IFN response. Cytosolic DNA binds to cGAS and the second messenger cGAMP to mediate STING-dependent transcription of type I IFNs through the transcription factor IFN regulatory factor 3 (IRF3)95,96. Notably, cGAS inhibitors are currently in development for the treatment of inflammatory rheumatic diseases97.
Cellular effects
Cytokine production and immune activation
Hydroxychloroquine and chloroquine can indirectly reduce the production of anti-inflammatory cytokines by various cell types. In vitro, hydroxychloroquine and chloroquine inhibit the production of IL-1, IL-6, TNF and IFNγ by mononuclear cells98 (Fig. 4). Furthermore, treatment with hydroxychloroquine inhibits the production of TNF, IFNα, IL-6 and CCL4 (also known as MIP1β) in pDC and natural killer cell co-cultures stimulated with RNA-containing immune complexes99,100.
TLR signals stimulate the production of cytokines, and hence hydroxychloroquine and chloroquine might inhibit cytokine production by inhibiting TLR pathways. Notably, a small molecule inhibitor of IL-1 receptor-associated kinase 4 (IRAK4; a component of the TLR7 and TLR9 signalling pathways) could reduce production of cytokines by peripheral blood mononuclear cells (PBMCs) more strikingly than hydroxychloroquine101. Upon stimulation, IRAK4 inhibition altered the expression of a larger number of RNA-induced and immune-complex-induced genes than hydroxychloroquine (492 versus 65 genes). This finding suggests that hydroxychloroquine is less effective at inhibiting the production of a wide range of cytokines than the IRAK4 inhibitor. Nevertheless, this study also convincingly shows that hydroxychloroquine has a notable effect on cytokine production and gene expression, including an inhibitory effect on TNF production by PBMCs from patients with SLE101. Indeed, in other studies, treatment with hydroxychloroquine was associated with a reduction in serum levels of IFNα in patients with SLE102. Furthermore, in patients with RA, long-term treatment with hydroxychloroquine (200–400 mg/day) can reduce circulating levels of IL-1 and IL-6 and is associated with improvement in erythrocyte sedimentation rate103,104.
The anti-inflammatory effects of hydroxychloroquine and chloroquine could be explained in part by the upstream interference of immune activation (including inhibition of lysosomal activity). Indeed, treatment with hydroxychloroquine is associated with a dose-dependent downregulation of the co-stimulatory molecule CD154 on CD4+ T cells from patients with SLE, which is accompanied by a decrease in intracellular Ca2+ mobilization and translocation of nuclear factor of activated T cells cytoplasmic 1 (NFATc1) and NFATc2105. However, the direct effect of antimalarial drugs on cytokine production requires further delineation.
Cardiovascular effects
Although hydroxychloroquine is not an anticoagulant, this drug is widely believed to have vascular protective effects and prevent the development of thrombotic complications. This protective effect seems to be most relevant for patients with a secondary coagulopathy owing to systemic inflammation106 and in patients with primary APS107.
Patients with inflammatory rheumatic diseases are at an increased risk of developing cardiovascular complications compared with the general population108,109,110,111. This increased risk is caused by the underlying disease, drugs used to treat the disease (such as NSAIDs, including COX-2 inhibitors112 and high-dose glucocorticoids) and the presence of comorbidities, such as arterial hypertension, hyperlipidaemia, chronic kidney failure and diabetes mellitus. By contrast, treatment with hydroxychloroquine seems to counter these effects and provide long-term benefits by reducing the risk of cardiovascular events, lowering fasting glucose levels113 and reducing hyperlipidaemia15,114. For example, in a study of patients with SLE, combined use of low-dose aspirin and hydroxychloroquine was superior to treatment with aspirin or hydroxychloroquine alone in terms of preventing cardiovascular complications115. However, sufficiently large and controlled studies are needed to quantify the benefit-to-risk profile of hydroxychloroquine in the prevention of cardiovascular complications in patients with rheumatic diseases and other non-rheumatological cohorts116.
Potential mechanisms by which hydroxychloroquine and chloroquine reduce the procoagulatory state in autoinflammatory diseases include inhibition of antiphospholipid antibody binding117 or inhibition of platelet aggregation118,119,120. Notably, in a mouse model of APS, hydroxychloroquine treatment was associated with improvement in endothelial function121,122. The exact molecular mechanisms by which these drugs mediate their antithrombotic effects remain largely unknown.
Therapeutic implications
Adverse effects: drug toxicity
Hydroxychloroquine and chloroquine have a known and good safety profile and are considered to function as immunomodulatory rather than immunosuppressant drugs. The various immune pathways targeted by these drugs (such as TLR7 and TLR9 signalling, T cell receptor (TCR) and B cell receptor (BCR) activation and others discussed above) remain largely preserved (that is, not fully inhibited) during treatment.
Unlike treatment with immunosuppressant drugs such as methotrexate and leflunomide, treatment with hydroxychloroquine or chloroquine is not associated with an increased risk of infectious complications16 or cancer123. The most common adverse effect of these antimalarial drugs are gastrointestinal effects, including nausea, vomiting, diarrhoea and abdominal discomfort124. In addition, several studies have reported the occurrence of hydroxychloroquine-associated myopathy125,126,127,128 and hydroxychloroquine-mediated and/or chloroquine-mediated cardiotoxic effects, including rhythm disorders (such as a prolonged QT interval) and the development of cardiomyopathy in patients with rheumatic diseases129,130,131,132. However, conclusive evidence of cardiotoxicity caused by these drugs is lacking and further pharmacovigilance is required. Impaired renal function can increase the bioavailability of antimalarial drugs and increase the risk of adverse effects.
The most severe complication attributed to antimalarial treatment is the development of retinopathy. These drugs are thought to cause retinal damage by disrupting lysosomal degradation of photoreceptor outer segments in the retinal pigment epithelium (an important step in the visual cycle). Mechanistically, this interference is thought to lead to an increase in lipofuscin in retinal pigment epithelial cells and photoreceptor degradation133. Retinopathy is more commonly associated with chloroquine than with hydroxychloroquine and can result in patients developing circular defects (known as bull’s eye maculopathy) and diametric defects of the retina. Details on the pathophysiology, epidemiology and treatment of hydroxychloroquine retinopathy have been reviewed extensively elsewhere133. Notably, advances in diagnostic procedures in the past few years, and the resulting realization that hydroxychloroquine-related retinopathy is more common that previously realized, have led to the development of new ophthalmology guidelines134,135. Various factors are thought to increase the risk of developing retinopathy during treatment with hydrochloroquine: a drug dose of >5 mg/kg actual body weight per day, prolonged use of the drug (10–25 years), a high cumulative dose (above 600–1,000 g), stage 3–5 chronic kidney disease and comedication with tamoxifen (>6 months)133. The most important predictors of hydroxychloroquine retinopathy are thought to be high-dose and long-term (>5 years) use, but current evidence is limited to retrospective studies, most of which are based on health care records and have substantial limitations (such as limited data on renal impairments)133.
Dosing considerations
According to ophthalmology guidelines, the occurrence of ocular adverse reactions during hydroxychloroquine or chloroquine therapy can be minimized by using the appropriate drug dosage (that is, taking into consideration the actual body weight of the patient) and by using advanced screening modalities such as optical coherence tomography133. However, the dose–response relationships for hydroxychloroquine and chloroquine are still unknown due to their complex pharmacokinetics. Both hydroxychloroquine and chloroquine have a gradual onset of action41,42,136 that might take weeks to reach maximal activity; furthermore, both drugs have a prolonged efficacy that is evident even after drug discontinuation. The complex pharmacokinetics of these drugs could be explained by the long plasma elimination half-lives of hydroxychloroquine, chloroquine and their dealkylated metabolites, as well as their extensive volume of distribution.
Therapeutic drug measurements of hydroxychloroquine in patients with rheumatic diseases have provided a number of important insights. First, low blood concentrations of hydroxychloroquine are predictive of subsequent flares in patients with SLE during hydroxychloroquine treatment137. Second, non-adherence to hydroxychloroquine is a major cause of flares in patients with SLE138. Among 305 patients with SLE who were enrolled in a prospective study, 18.4% were non-adherent to hydroxychloroquine therapy, defined as having hydroxychloroquine concentrations below 200 ng/ml or undetectable concentrations of desethylchloroquine, whereas 23.4% of patients were non-adherent according to self-administered questionnaires138. Risk factors of non-adherence in this population were younger age, absence of steroid co-medication, higher body mass index and unemployment. Thus, non-adherence is an issue and therapeutic drug monitoring might be useful.
In chronic kidney disease, despite the fact that decreased drug clearance can result in increased (and potentially toxic) concentrations of hydroxychloroquine, current guidelines do not include the recommendation to reduce the dosage of hydroxychloroquine in patients with chronic kidney diseases. In a real-world cohort of patients with SLE and end-stage renal disease, 20% of the patients were still taking hydroxychloroquine139. The adverse effects of hydroxychloroquine in the kidney can mimic Fabry’s disease140. In patients with RA, reduced concentrations of hydroxychloroquine and desethylhydroxychloroquine in the blood are associated with increased morning stiffness, pain intensity and positivity for rheumatoid factor43,46. Hence, studies that investigate levels of these drugs and clinical response of patients could help to overcome limited knowledge on the dose–response characteristics within different diseases as well as during active versus quiescent periods.
The latest guidelines from the American Academy of Ophthalmology, the Royal College of Ophthalmology and EULAR2,134,141 recommend the use of a lower dose of hydroxychloroquine (maximum of 5.0 mg/kg actual body weight per day) than previously used for many patients. This maximum dosage is controversial and is largely based on an ophthalmological study by Melles and Marmor142. As this study used pharmacy refill information, the dosing estimates represent actual hydroxychloroquine intake rather than prescribed dosages. Thus, the cut-off of 5 mg/kg in this study probably corresponds to a prescribed dose of 6 mg/kg actual body weight (as discussed elsewhere143).
In cancer studies, the prescribed doses of hydroxychloroquine can reach up to 1,200 mg daily. Despite such high doses, no relevant toxic adverse effects (beside cytopenia) were reported in these studies, even when hydroxychloroquine was combined with other oculotoxic agents144,145,146,147,148. However, the treatment periods for oncological conditions are generally much shorter than for rheumatic diseases, and therefore the cumulative dose is usually lower in patients with cancer than in patients with rheumatic diseases.
A relevant problem with the current ophthalmology guidelines is that the efficacy of the recently recommended maximum dose (5 mg/kg actual body weight per day) has not been evaluated for patients with inflammatory rheumatic disease. The clinical consequences of following this dosing strategy might be manifold. Many past studies evaluating the efficacy of hydroxychloroquine investigated a dosage of at least 6.5 mg/kg ideal body weight per day. As a result, the effective dose during flares might be higher than the recommended maximum dose. Moreover, patients with different diseases might require different dosages of antimalarial drugs.
Current and future directions
It is of utmost importance to further delineate the mode of action of hydroxychloroquine and chloroquine, together with the safety and efficacy of these drugs at the current recommended doses. The identification of molecular targets of these drugs, as exemplified by PPT1, should permit the development of new treatment modalities. Additional knowledge gaps and research topics are listed in Box 2. One current line of investigation is whether these drugs can sufficiently prevent the production of IFNα by blocking TLR7 and TLR9 engagement, as these are important pathways in connective tissue diseases. How these drugs interfere with signalling downstream of nucleic acid sensors, including sensors of double-stranded DNA and RNA (such as TLR9 and TLR7), and the selectivity and size of these effects, also requires further study. Another unique feature of these drugs is their ability to inhibit lysosomal activity and block MHC class II presentation. These processes possibly occur, at least in part, through drug-mediated changes in the pH of autophagosomes and/or lysosomes, which indirectly influence immune activation; however, such a mode of action requires additional validation to aid with future drug development.
An important feature of hydroxychloroquine and chloroquine (and other DMARDs) seems to be their ability to inhibit both innate and adaptive immunity149. However, hydroxychloroquine and chloroquine are less effective at inhibiting the production of cytokines than glucocorticoids, Janus kinase (JAK) inhibitors and biological drugs, and have a delayed onset of action. However, apart from the risk of retinopathy, these antimalarial drugs have a good safety profile, probably because they do not notably interfere with important pathways involved in protection against infection such as BCR or TCR signalling, JAK–STAT-dependent cytokine signalling, TLR4 signalling and certain chemokine signalling pathways. Importantly, interference of TLR7 and TLR9 signalling and MHC class II presentation by antimalarial drugs does not result in an increased risk of infection.
There is a great deal of interest in developing specific inhibitors of autophagy150 for the treatment of cancers, which might have clinical value for rheumatology. Inhibitors of IRAK4 are also under investigation for the treatment of autoimmune diseases151 as well as for the treatment of cancer152. This strategy does not interfere with MHC class II presentation, but might have more potent anticytokine effects than antimalarial drugs.
Finally, oral administration of a non-degradable polymeric form of chloroquine is under investigation for the treatment of inflammatory bowel diseases. Clinical data suggest that this formulation can suppress the production of IL-6 and IL-1β and upregulate the production of IL-2 in the intestine153 as a result of altered mucosal immune homeostasis.
Conclusion
Although hydroxychloroquine and chloroquine are well-known DMARDs that have been used for the treatment of patients with rheumatic diseases for many years, their exact mechanisms of action are only beginning to be understood. Important to the pharmacokinetic, pharmacodynamics and toxic properties of these drugs is their ability to accumulate in acidic compartments such as lysosomes, as well as inflamed (acidic) tissues. The large volume distribution and long half-lives of these drugs can explain some of their clinical characteristics, such as their slow onset of action and prolonged effects after drug discontinuation. At the cellular level, several direct and indirect mechanisms have been described that account for the immunomodulatory effects of hydroxychloroquine and chloroquine. In particular, inhibition of autophagy prevents immune activation of different cell types, which inhibits cytokine production and modulates CD154 expression on T cells. Renal failure and certain drug interactions (such as interactions with tamoxifen, glycosides, methotrexate and ciclosporin) can influence the pharmacokinetics of hydroxychloroquine and chloroquine and hence require consideration. Although hydroxychloroquine and chloroquine are considered safe to use, these drugs are associated with an increased risk of retinopathy. This increased risk has resulted in ophthalmology guidelines recommending a maximal daily dose of 5.0 mg/kg actual body weight for hydroxychloroquine, which is debated among rheumatologists. Ultimately, drug level monitoring studies are needed to reassess the risk to benefit profile of this dosing strategy. Preclinical studies of compounds targeting lysosome and autophagosome activity are underway and should reveal whether such strategies have value in the treatment of systemic autoimmune diseases.
References
Smolen, J. S. et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2013 update. Ann. Rheum. Dis. 73, 492–509 (2014).
Fanouriakis, A. et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann. Rheum. Dis. 78, 736–745 (2019).
Pons-Estel, B. A. et al. First Latin American clinical practice guidelines for the treatment of systemic lupus erythematosus: Latin American Group for the Study of Lupus (GLADEL, Grupo Latino Americano de Estudio del Lupus)-Pan-American League of Associations of Rheumatology (PANLAR). Ann. Rheum. Dis. 77, 1549–1557 (2018).
Gordon, C. et al. The British Society for Rheumatology guideline for the management of systemic lupus erythematosus in adults. Rheumatology 57, e1–e45 (2018).
Tektonidou, M. G. et al. EULAR recommendations for the management of antiphospholipid syndrome in adults. Ann. Rheum. Dis. 78, 1296–1304 (2019).
Brito-Zeron, P., Ramos-Casals, M. & EULAR-SS task force group. Advances in the understanding and treatment of systemic complications in Sjogren’s syndrome. Curr. Opin. Rheumatol. 26, 520–527 (2014).
Vivino, F. B. et al. New treatment guidelines for Sjogren’s disease. Rheum. Dis. Clin. North. Am. 42, 531–551 (2016).
Ruiz-Irastorza, G. et al. Clinical efficacy and side effects of antimalarials in systemic lupus erythematosus: a systematic review. Ann. Rheum. Dis. 69, 20–28 (2010).
Ostensen, M. et al. Pregnancy and reproduction in autoimmune rheumatic diseases. Rheumatology 50, 657–664 (2011).
Akhavan, P. S. et al. The early protective effect of hydroxychloroquine on the risk of cumulative damage in patients with systemic lupus erythematosus. J. Rheumatol. 40, 831–841 (2013).
Mok, C. C., Mak, A. & Ma, K. M. Bone mineral density in postmenopausal Chinese patients with systemic lupus erythematosus. Lupus 14, 106–112 (2005).
Petri, M. Use of hydroxychloroquine to prevent thrombosis in systemic lupus erythematosus and in antiphospholipid antibody-positive patients. Curr. Rheumatol. Rep. 13, 77–80 (2011).
Kingsbury, S. R. et al. Hydroxychloroquine effectiveness in reducing symptoms of hand osteoarthritis: a randomized trial. Ann. Intern. Med. 168, 385–395 (2018).
Lee, W. et al. Efficacy of hydroxychloroquine in hand osteoarthritis: a randomized, double-blind, placebo-controlled trial. Arthritis Care Res. 70, 1320–1325 (2018).
Rempenault, C. et al. Metabolic and cardiovascular benefits of hydroxychloroquine in patients with rheumatoid arthritis: a systematic review and meta-analysis. Ann. Rheum. Dis. 77, 98–103 (2018).
Ruiz-Irastorza, G. et al. Predictors of major infections in systemic lupus erythematosus. Arthritis Res. Ther. 11, R109 (2009).
Flannery, E. L., Chatterjee, A. K. & Winzeler, E. A. Antimalarial drug discovery – approaches and progress towards new medicines. Nat. Rev. Microbiol. 11, 849–862 (2013).
Ridley, R. G. Medical need, scientific opportunity and the drive for antimalarial drugs. Nature 415, 686–693 (2002).
Minie, M. et al. CANDO and the infinite drug discovery frontier. Drug Discov. Today 19, 1353–1363 (2014).
Paddon, C. J. et al. High-level semi-synthetic production of the potent antimalarial artemisinin. Nature 496, 528–532 (2013).
Hale, V. et al. Microbially derived artemisinin: a biotechnology solution to the global problem of access to affordable antimalarial drugs. Am. J. Trop. Med. Hyg. 77, 198–202 (2007).
Mitchell, W. Natural products from synthetic biology. Curr. Opin. Chem. Biol. 15, 505–515 (2011).
Conway, D. J. Paths to a malaria vaccine illuminated by parasite genomics. Trends Genet. 31, 97–107 (2015).
Karunamoorthi, K. Malaria vaccine: a future hope to curtail the global malaria burden. Int. J. Prev. Med. 5, 529–538 (2014).
de Beer, T. A. et al. Antimalarial drug discovery: in silico structural biology and rational drug design. Infect. Disord. Drug Targets 9, 304–318 (2009).
Vedadi, M. et al. Genome-scale protein expression and structural biology of Plasmodium falciparum and related Apicomplexan organisms. Mol. Biochem. Parasitol. 151, 100–110 (2007).
Buckee, C. O. et al. Mobile phones and malaria: modeling human and parasite travel. Travel. Med. Infect. Dis. 11, 15–22 (2013).
Hay, S. I. et al. Big data opportunities for global infectious disease surveillance. PLoS Med. 10, e1001413 (2013).
Ponticelli, C. & Moroni, G. Hydroxychloroquine in systemic lupus erythematosus (SLE). Expert. Opin. Drug Saf. 16, 411–419 (2017).
Wang, S. Q. et al. Is hydroxychloroquine effective in treating primary Sjogren’s syndrome: a systematic review and meta-analysis. BMC Musculoskelet. Disord. 18, 186 (2017).
Manohar, S., Tripathi, M. & Rawat, D. S. 4-aminoquinoline based molecular hybrids as antimalarials: an overview. Curr. Top. Med. Chem. 14, 1706–1733 (2014).
Rainsford, K. D. et al. Therapy and pharmacological properties of hydroxychloroquine and chloroquine in treatment of systemic lupus erythematosus, rheumatoid arthritis and related diseases. Inflammopharmacology 23, 231–269 (2015).
Witiak, D. T. et al. Synthesis and preliminary pharmacological evaluation of asymmetric chloroquine analogues. J. Med. Chem. 24, 712–717 (1981).
Tagoe, C. N. & Ofori-Adjei, D. Effects of chloroquine and its enantiomers on the development of rat embryos in vitro. Teratology 52, 137–142 (1995).
Ducharme, J. et al. Enantioselective disposition of hydroxychloroquine after a single oral dose of the racemate to healthy subjects. Br. J. Clin. Pharmacol. 40, 127–133 (1995).
Tett, S. E. et al. Pharmacokinetics and pharmacodynamics of hydroxychloroquine enantiomers in patients with rheumatoid arthritis receiving multiple doses of racemate. Chirality 6, 355–359 (1994).
Tett, S. E. et al. Bioavailability of hydroxychloroquine tablets in healthy volunteers. Br. J. Clin. Pharmacol. 27, 771–779 (1989).
Furst, D. E. Pharmacokinetics of hydroxychloroquine and chloroquine during treatment of rheumatic diseases. Lupus 5, S11–S15 (1996).
Cutler, D. J., MacIntyre, A. C. & Tett, S. E. Pharmacokinetics and cellular uptake of 4-aminoquinoline antimalarials. Agents Actions Suppl. 24, 142–157 (1988).
McChesney, E. W. Animal toxicity and pharmacokinetics of hydroxychloroquine sulfate. Am. J. Med. 75, 11–18 (1983).
Fishbain, D. A. et al. Do some chronic pain patients with atypical facial pain overvalue and obsess about their pain? Psychosomatics 34, 355–359 (1993).
Tett, S. et al. Insights from pharmacokinetic and pharmacodynamic studies of hydroxychloroquine. Agents Actions Suppl. 44, 145–190 (1993).
Tett, S. E., Day, R. O. & Cutler, D. J. Concentration-effect relationship of hydroxychloroquine in rheumatoid arthritis – a cross sectional study. J. Rheumatol. 20, 1874–1879 (1993).
Collins, K. P., Jackson, K. M. & Gustafson, D. L. Hydroxychloroquine: a physiologically-based pharmacokinetic model in the context of cancer-related autophagy modulation. J. Pharmacol. Exp. Ther. 365, 447–459 (2018).
Rynes, R. I. & Bernstein, H. N. Ophthalmologic safety profile of antimalarial drugs. Lupus 2, S17–S19 (1993).
Munster, T. et al. Hydroxychloroquine concentration-response relationships in patients with rheumatoid arthritis. Arthritis Rheum. 46, 1460–1469 (2002).
Carmichael, S. J., Charles, B. & Tett, S. E. Population pharmacokinetics of hydroxychloroquine in patients with rheumatoid arthritis. Ther. Drug Monit. 25, 671–681 (2003).
Spaldin, V. et al. The effect of enzyme inhibition on the metabolism and activation of tacrine by human liver microsomes. Br. J. Clin. Pharmacol. 38, 15–22 (1994).
Projean, D. et al. In vitro metabolism of chloroquine: identification of CYP2C8, CYP3A4, and CYP2D6 as the main isoforms catalyzing N-desethylchloroquine formation. Drug Metab. Dispos. 31, 748–754 (2003).
Gil, J. P. & Gil Berglund, E. CYP2C8 and antimalaria drug efficacy. Pharmacogenomics 8, 187–198 (2007).
Projean, D. et al. Identification of CYP3A4 and CYP2C8 as the major cytochrome P450 s responsible for morphine N-demethylation in human liver microsomes. Xenobiotica 33, 841–854 (2003).
Kim, K. A. et al. Cytochrome P450 2C8 and CYP3A4/5 are involved in chloroquine metabolism in human liver microsomes. Arch. Pharm. Res. 26, 631–637 (2003).
Li, X. Q. et al. Identification of human cytochrome P(450)s that metabolise anti-parasitic drugs and predictions of in vivo drug hepatic clearance from in vitro data. Eur. J. Clin. Pharmacol. 59, 429–442 (2003).
Lee, J. Y. et al. Association of polymorphisms of cytochrome P450 2D6 with blood hydroxychloroquine levels in patients with systemic lupus erythematosus. Arthritis Rheumatol. 68, 184–190 (2016).
Leden, I. Digoxin-hydroxychloroquine interaction? Acta Med. Scand. 211, 411–412 (1982).
Somer, M. et al. Influence of hydroxychloroquine on the bioavailability of oral metoprolol. Br. J. Clin. Pharmacol. 49, 549–554 (2000).
Kormelink, T. G. et al. Decrease in immunoglobulin free light chains in patients with rheumatoid arthritis upon rituximab (anti-CD20) treatment correlates with decrease in disease activity. Ann. Rheum. Dis. 69, 2137–2144 (2010).
Toimela, T., Tahti, H. & Salminen, L. Retinal pigment epithelium cell culture as a model for evaluation of the toxicity of tamoxifen and chloroquine. Ophthalmic Res. 27, 150–153 (1995).
Bannwarth, B. et al. Clinical pharmacokinetics of low-dose pulse methotrexate in rheumatoid arthritis. Clin. Pharmacokinet. 30, 194–210 (1996).
Carmichael, S. J. et al. Combination therapy with methotrexate and hydroxychloroquine for rheumatoid arthritis increases exposure to methotrexate. J. Rheumatol. 29, 2077–2083 (2002).
van den Borne, B. E. et al. Combination therapy in recent onset rheumatoid arthritis: a randomized double blind trial of the addition of low dose cyclosporine to patients treated with low dose chloroquine. J. Rheumatol. 25, 1493–1498 (1998).
Namazi, M. R. The potential negative impact of proton pump inhibitors on the immunopharmacologic effects of chloroquine and hydroxychloroquine. Lupus 18, 104–105 (2009).
Jallouli, M. et al. Determinants of hydroxychloroquine blood concentration variations in systemic lupus erythematosus. Arthritis Rheumatol. 67, 2176–2184 (2015).
Ezra, N. & Jorizzo, J. Hydroxychloroquine and smoking in patients with cutaneous lupus erythematosus. Clin. Exp. Dermatol. 37, 327–334 (2012).
Yeon Lee, J. et al. Factors related to blood hydroxychloroquine concentration in patients with systemic lupus erythematosus. Arthritis Care Res. 69, 536–542 (2017).
Borden, M. B. & Parke, A. L. Antimalarial drugs in systemic lupus erythematosus: use in pregnancy. Drug Saf. 24, 1055–1063 (2001).
Costedoat-Chalumeau, N. et al. Safety of hydroxychloroquine in pregnant patients with connective tissue diseases. Review of the literature. Autoimmun. Rev. 4, 111–115 (2005).
Teng, Y. K. O. et al. An evidence-based approach to pre-pregnancy counselling for patients with systemic lupus erythematosus. Rheumatology 57, 1707–1720 (2017).
Andreoli, L. et al. EULAR recommendations for women’s health and the management of family planning, assisted reproduction, pregnancy and menopause in patients with systemic lupus erythematosus and/or antiphospholipid syndrome. Ann. Rheum. Dis. 76, 476–485 (2017).
Gotestam Skorpen, C. et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann. Rheum. Dis. 75, 795–810 (2016).
Izmirly, P. M. et al. Maternal use of hydroxychloroquine is associated with a reduced risk of recurrent anti-SSA/Ro-antibody-associated cardiac manifestations of neonatal lupus. Circulation 126, 76–82 (2012).
Saxena, A. et al. Prevention and treatment in utero of autoimmune-associated congenital heart block. Cardiol. Rev. 22, 263–267 (2014).
Lisney, A. R. et al. High maternal expression of SIGLEC1 on monocytes as a surrogate marker of a type I interferon signature is a risk factor for the development of autoimmune congenital heart block. Ann. Rheum. Dis. 76, 1476–1480 (2017).
Friedman, D. et al. No histologic evidence of foetal cardiotoxicity following exposure to maternal hydroxychloroquine. Clin. Exp. Rheumatol. 35, 857–859 (2017).
Sammaritano, L. R. & Bermas, B. L. Rheumatoid arthritis medications and lactation. Curr. Opin. Rheumatol. 26, 354–360 (2014).
Circu, M. et al. Modulating lysosomal function through lysosome membrane permeabilization or autophagy suppression restores sensitivity to cisplatin in refractory non-small-cell lung cancer cells. PLoS One 12, e0184922 (2017).
Mauthe, M. et al. Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion. Autophagy 14, 1435–1455 (2018).
Frustaci, A. et al. Inhibition of cardiomyocyte lysosomal activity in hydroxychloroquine cardiomyopathy. Int. J. Cardiol. 157, 117–119 (2012).
Sundelin, S. P. & Terman, A. Different effects of chloroquine and hydroxychloroquine on lysosomal function in cultured retinal pigment epithelial cells. APMIS 110, 481–489 (2002).
Ballabio, A. & Bonifacino J. S. Lysosomes as dynamic regulators of cell and organismal homeostasis. Nat. Rev. Mol. Cell Biol. 21, 101–118 (2019).
Lotteau, V. et al. Intracellular transport of class II MHC molecules directed by invariant chain. Nature 348, 600–605 (1990).
Ghislat, G. & Lawrence, T. Autophagy in dendritic cells. Cell Mol. Immunol. 15, 944–952 (2018).
Munz, C. Autophagy beyond intracellular MHC class II antigen presentation. Trends Immunol. 37, 755–763 (2016).
Ireland, J. M. & Unanue, E. R. Autophagy in antigen-presenting cells results in presentation of citrullinated peptides to CD4 T cells. J. Exp. Med. 208, 2625–2632 (2011).
Ohkuma, S. & Poole, B. Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc. Natl Acad. Sci. USA 75, 3327–3331 (1978).
Rebecca, V. W. et al. PPT1 promotes tumor growth and is the molecular target of chloroquine derivatives in cancer. Cancer Discov. 9, 220–229 (2019).
Ma, C. et al. Identifying key genes in rheumatoid arthritis by weighted gene co-expression network analysis. Int. J. Rheum. Dis. 20, 971–979 (2017).
Ewald, S. E. et al. The ectodomain of Toll-like receptor 9 is cleaved to generate a functional receptor. Nature 456, 658–662 (2008).
Kuznik, A. et al. Mechanism of endosomal TLR inhibition by antimalarial drugs and imidazoquinolines. J. Immunol. 186, 4794–4804 (2011).
Hacker, H. et al. CpG-DNA-specific activation of antigen-presenting cells requires stress kinase activity and is preceded by non-specific endocytosis and endosomal maturation. EMBO J. 17, 6230–6240 (1998).
Lau, C. M. et al. RNA-associated autoantigens activate B cells by combined B cell antigen receptor/Toll-like receptor 7 engagement. J. Exp. Med. 202, 1171–1177 (2005).
Vollmer, J. et al. Immune stimulation mediated by autoantigen binding sites within small nuclear RNAs involves Toll-like receptors 7 and 8. J. Exp. Med. 202, 1575–1585 (2005).
An, J. et al. Antimalarial drugs as immune modulators: new mechanisms for old drugs. Annu. Rev. Med. 68, 317–330 (2017).
Zhang, X. et al. The cytosolic DNA sensor cGAS forms an oligomeric complex with DNA and undergoes switch-like conformational changes in the activation loop. Cell Rep. 6, 421–430 (2014).
Zhang, X. et al. Cyclic GMP-AMP containing mixed phosphodiester linkages is an endogenous high-affinity ligand for STING. Mol. Cell 51, 226–235 (2013).
Shu, C., Li, X. & Li, P. The mechanism of double-stranded DNA sensing through the cGAS-STING pathway. Cytokine Growth Factor Rev. 25, 641–648 (2014).
An, J. et al. Cutting edge: antimalarial drugs inhibit IFN-β production through blockade of cyclic GMP-AMP synthase-DNA interaction. J. Immunol. 194, 4089–4093 (2015).
van den Borne, B. E. et al. Chloroquine and hydroxychloroquine equally affect tumor necrosis factor-alpha, interleukin 6, and interferon-gamma production by peripheral blood mononuclear cells. J. Rheumatol. 24, 55–60 (1997).
Wallace, D. J. et al. The effect of hydroxychloroquine therapy on serum levels of immunoregulatory molecules in patients with systemic lupus erythematosus. J. Rheumatol. 21, 375–376 (1994).
Wallace, D. J. et al. The relevance of antimalarial therapy with regard to thrombosis, hypercholesterolemia and cytokines in SLE. Lupus 2, S13–S15 (1993).
Hjorton, K. et al. Cytokine production by activated plasmacytoid dendritic cells and natural killer cells is suppressed by an IRAK4 inhibitor. Arthritis Res. Ther. 20, 238 (2018).
Willis, R. et al. Effect of hydroxychloroquine treatment on pro-inflammatory cytokines and disease activity in SLE patients: data from LUMINA (LXXV), a multiethnic US cohort. Lupus 21, 830–835 (2012).
Klinefelter, H. F. & Achurra, A. Effect of gold salts and antimalarials on the rheumatoid factor in rheumatoid arthritis. Scand. J. Rheumatol. 2, 177–182 (1973).
Dixon, J. S. et al. Biochemical indices of response to hydroxychloroquine and sodium aurothiomalate in rheumatoid arthritis. Ann. Rheum. Dis. 40, 480–488 (1981).
Wu, S. F. et al. Hydroxychloroquine inhibits CD154 expression in CD4(+) T lymphocytes of systemic lupus erythematosus through NFAT, but not STAT5, signaling. Arthritis Res. Ther. 19, 183 (2017).
Qushmaq, N. A. & Al-Emadi, S. A. Review on effectiveness of primary prophylaxis in aPLs with and without risk factors for thrombosis: efficacy and safety. ISRN Rheumatol. 2014, 348726 (2014).
Nuri, E. et al. Long-term use of hydroxychloroquine reduces antiphospholipid antibodies levels in patients with primary antiphospholipid syndrome. Immunol. Res. 65, 17–24 (2017).
Dadoun, S. et al. Mortality in rheumatoid arthritis over the last fifty years: systematic review and meta-analysis. Joint Bone Spine 80, 29–33 (2013).
van den Hoek, J. et al. Mortality in patients with rheumatoid arthritis: a 15-year prospective cohort study. Rheumatol. Int. 37, 487–493 (2017).
Avina-Zubieta, J. A. et al. Risk of myocardial infarction and stroke in newly diagnosed systemic lupus erythematosus: a general population-based study. Arthritis Care Res. 69, 849–856. (2017).
Tselios, K., Gladman, D. D., Su, J., Ace, O. & Urowitz, M. B. Evolution of risk factors for atherosclerotic cardiovascular events in systemic lupus erythematosus: a longterm prospective study. J. Rheumatol. 44 1841–1849 (2017).
Padol, I. T. & Hunt, R. H. Association of myocardial infarctions with COX-2 inhibition may be related to immunomodulation towards a Th1 response resulting in atheromatous plaque instability: an evidence-based interpretation. Rheumatology 49, 837–843 (2010).
Hage, M. P., Al-Badri, M. R. & Azar, S. T. A favorable effect of hydroxychloroquine on glucose and lipid metabolism beyond its anti-inflammatory role. Ther. Adv. Endocrinol. Metab. 5, 77–85 (2014).
Cansu, D. U. & Korkmaz, C. Hypoglycaemia induced by hydroxychloroquine in a non-diabetic patient treated for RA. Rheumatology 47, 378–379 (2008).
Fasano, S. et al. Longterm hydroxychloroquine therapy and low-dose aspirin may have an additive effectiveness in the primary prevention of cardiovascular events in patients with systemic lupus erythematosus. J. Rheumatol. 44, 1032–1038 (2017).
Towers, C. G. & Thorburn, A. Therapeutic targeting of autophagy. EBioMedicine 14, 15–23 (2016).
Rand, J. H. et al. Hydroxychloroquine directly reduces the binding of antiphospholipid antibody-β2-glycoprotein I complexes to phospholipid bilayers. Blood 112, 1687–1695 (2008).
Jancinova, V., Nosal, R. & Petrikova, M. On the inhibitory effect of chloroquine on blood platelet aggregation. Thromb. Res. 74, 495–504 (1994).
Bertrand, E. et al. Antiaggregation action of chloroquine. Med. Trop. 50, 143–146 (1990).
Nosal, R., Jancinova, V. & Petrikova, M. Chloroquine inhibits stimulated platelets at the arachidonic acid pathway. Thromb. Res. 77, 531–542 (1995).
Miranda, S. et al. Hydroxychloroquine reverses the prothrombotic state in a mouse model of antiphospholipid syndrome: role of reduced inflammation and endothelial dysfunction. PLoS One 14, e0212614 (2019).
Edwards, M. H. et al. Hydroxychloroquine reverses thrombogenic properties of antiphospholipid antibodies in mice. Circulation 96, 4380–4384 (1997).
Lazarus, M. N. et al. Incidence of cancer in a cohort of patients with primary Sjogren’s syndrome. Rheumatology 45, 1012–1015 (2006).
Srinivasa, A., Tosounidou, S. & Gordon, C. Increased incidence of gastrointestinal side effects in patients taking hydroxychloroquine: a brand-related issue? J. Rheumatol. 44, 398 (2017).
Abdel-Hamid, H., Oddis, C. V. & Lacomis, D. Severe hydroxychloroquine myopathy. Muscle Nerve 38, 1206–1210 (2008).
Jafri, K. et al. Antimalarial myopathy in a systemic lupus erythematosus patient with quadriparesis and seizures: a case-based review. Clin. Rheumatol. 36, 1437–1444 (2017).
Khosa, S. et al. Hydroxychloroquine-induced autophagic vacuolar myopathy with mitochondrial abnormalities. Neuropathology 38, 646–652 (2018).
Stein, M., Bell, M. J. & Ang, L. C. Hydroxychloroquine neuromyotoxicity. J. Rheumatol. 27, 2927–2931 (2000).
Dogar, M. U. et al. Hydroxychloroquine-induced restrictive cardiomyopathy: a case report. Postgrad. Med. J. 94, 185–186 (2018).
Chang, I. C. Y. et al. Hydroxychloroquine-mediated cardiotoxicity with a false-positive 99mTechnetium-labeled pyrophosphate scan for transthyretin-related cardiac amyloidosis. Circ. Cardiovasc. Imaging 11, e007059 (2018).
Chatre, C. et al. Cardiac complications attributed to chloroquine and hydroxychloroquine: a systematic review of the literature. Drug Saf. 41, 919–931 (2018).
Costedoat-Chalumeau, N. et al. Heart conduction disorders related to antimalarials toxicity: an analysis of electrocardiograms in 85 patients treated with hydroxychloroquine for connective tissue diseases. Rheumatology 46, 808–810 (2007).
Jorge, A. et al. Hydroxychloroquine retinopathy – implications of research advances for rheumatology care. Nat. Rev. Rheumatol. 14, 693–703 (2018).
Marmor, M. F. et al. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 Revision). Ophthalmology 123, 1386–1394 (2016).
Yusuf, I. H. et al. The Royal College of Ophthalmologists recommendations on screening for hydroxychloroquine and chloroquine users in the United Kingdom: executive summary. Eye 32, 1168–1173 (2018).
MacIntyre, A. C. & Cutler, D. J. Kinetics of chloroquine uptake into isolated rat hepatocytes. J. Pharm. Sci. 82, 592–600 (1993).
Costedoat-Chalumeau, N. et al. Low blood concentration of hydroxychloroquine is a marker for and predictor of disease exacerbations in patients with systemic lupus erythematosus. Arthritis Rheum. 54, 3284–3290 (2006).
Costedoat-Chalumeau, N. et al. A prospective international study on adherence to treatment in 305 patients with flaring SLE: assessment by drug levels and self-administered questionnaires. Clin. Pharmacol. Ther. 103, 1074–1082 (2018).
Bethel, M. et al. Hydroxychloroquine in patients with systemic lupus erythematosus with end-stage renal disease. J. Investig. Med. 64, 908–910 (2016).
Sperati, C. J. & Rosenberg, A. Z. Hydroxychloroquine-induced mimic of renal Fabry disease. Kidney Int. 94, 634 (2018).
Yusuf, I. H., Lotery, A. J. & Ardern-Jones, M. R. Joint recommendations for retinal screening in long-term users of hydroxychloroquine and chloroquine in the United Kingdom, 2018. Br. J. Dermatol. 179, 995–996 (2018).
Melles, R. B. & Marmor, M. F. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 132, 1453–1460 (2014).
Costedoat-Chalumeau, N., Isenberg, D. & Petri, M. Letter in response to the 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus by Fanouriakis et al. Ann. Rheum. Dis. https://doi.org/10.1136/annrheumdis-2019-215573 (2019).
Karasic, T. B. et al. Effect of gemcitabine and nab-paclitaxel with or without hydroxychloroquine on patients with advanced pancreatic cancer: a phase 2 randomized clinical trial. JAMA Oncol. 5, 993–998 (2019).
Rosenfeld, M. R. et al. A phase I/II trial of hydroxychloroquine in conjunction with radiation therapy and concurrent and adjuvant temozolomide in patients with newly diagnosed glioblastoma multiforme. Autophagy 10, 1359–1368 (2014).
Rangwala, R. et al. Phase I trial of hydroxychloroquine with dose-intense temozolomide in patients with advanced solid tumors and melanoma. Autophagy 10, 1369–1379 (2014).
Rangwala, R. et al. Combined MTOR and autophagy inhibition: phase I trial of hydroxychloroquine and temsirolimus in patients with advanced solid tumors and melanoma. Autophagy 10, 1391–1402 (2014).
Nti, A. A. et al. Frequent subclinical macular changes in combined BRAF/MEK inhibition with high-dose hydroxychloroquine as treatment for advanced metastatic braf mutant melanoma: preliminary results from a phase I/II clinical treatment trial. Retina 39, 502–513 (2019).
Dorner, T. Crossroads of B cell activation in autoimmunity: rationale of targeting B cells. J. Rheumatol. Suppl. 77, 3–11 (2006).
Liang, N. et al. Multifaceted roles of ATM in autophagy: from nonselective autophagy to selective autophagy. Cell Biochem. Funct. 37, 177–184 (2019).
Bahia, M. S. et al. Interleukin-1 receptor associated kinase inhibitors: potential therapeutic agents for inflammatory- and immune-related disorders. Cell Signal. 27, 1039–1055 (2015).
Khanfar, M. A. & Alqtaishat, S. Discovery of potent IRAK-4 inhibitors as potential anti-inflammatory and anticancer agents using structure-based exploration of IRAK-4 pharmacophoric space coupled with QSAR analyses. Comput. Biol. Chem. 79, 147–154 (2019).
Kanvinde, S. et al. Pharmacokinetics and efficacy of orally administered polymeric chloroquine as macromolecular drug in the treatment of inflammatory bowel disease. Acta Biomater. 82, 158–170 (2018).
Messai, A. in Emerging Research on Bioinspired Materials Engineering (ed. Bououdina, M.) 160–196 (IGI Global, 2016).
U.S. Food and Drug Administration. Drugs@FDA: FDA-Approved Drugs Accessdata.fda.gov https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&varApplNo=006002 (2014).
U.S. Food and Drug Administration. Drugs@FDA: FDA-Approved Drugs Accessdata.fda.gov https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=009768 (2014).
Acknowledgements
The authors would like to thank the Clinical Scientist Program of the Berlin Institutes of Health who provided grant support to E.V.S. and the Deutsche Forschungsgemeinschaft who provided grant support to T.D. (projects Do491/7-4 and Do491/10-1). Deutsches Rheumaforschungszentrum (DRFZ) Berlin is funded by the Leibniz society.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Rheumatology thanks K. Elkon and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Enantiomers
-
R and S enantiomers are mirror images of each other, but have different optical activities; enantiomers can interact differently with biomolecules and hence can have different biologic and possibly clinical activities or toxicities.
- Volume of distribution
-
A pharmacokinetic parameter used to describe the distribution of a drug in the body; the volume of distribution is the theoretical volume needed to contain the total amount of an administered drug at the same concentration as that present in the plasma.
- Bioavailability
-
The fraction of an administered dose of an unchanged drug that reaches the circulation; by definition, the bioavailability of an intravenously administered medication is 100%.
- Three-compartment model
-
A model used to predict the rate and extent of distribution of a drug once administered; this model divides the body into a central compartment (compartment 1) and two peripheral compartments (compartments 2 and 3). The central compartment consists of the plasma and tissues where the drug is immediately distributed. The peripheral compartments consist of tissues and cells in which the drug is distributed more slowly.
Rights and permissions
About this article
Cite this article
Schrezenmeier, E., Dörner, T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol 16, 155–166 (2020). https://doi.org/10.1038/s41584-020-0372-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-020-0372-x
This article is cited by
-
Drug repurposing-based nanoplatform via modulating autophagy to enhance chemo-phototherapy against colorectal cancer
Journal of Nanobiotechnology (2024)
-
Rheumatological complaints in H syndrome: from inflammatory profiling to target treatment in a case study
Pediatric Rheumatology (2024)
-
Effects of hydroxychloroquine therapy on choroidal volume and choroidal vascularity index
Eye (2024)
-
Vascular damage in systemic lupus erythematosus
Nature Reviews Nephrology (2024)
-
Cyclical palmitoylation regulates TLR9 signalling and systemic autoimmunity in mice
Nature Communications (2024)