Abstract
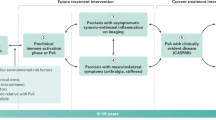
Psoriasis is one of the most common chronic inflammatory skin diseases, affecting 3% of the world’s population, and approximately one-third of patients with psoriasis will eventually transition to having psoriatic arthritis (PsA). The evolution from cutaneous to synovio-entheseal inflammation in these patients presents an opportunity to investigate the critical events linked to arthritis development. The events responsible for progression to PsA are currently unclear. Genetic and clinical–demographic risk factors (most notably familial aggregation and psoriasis sub-phenotypes) provide relevant insights into the variables that promote transition. The specific underlying molecular and cellular mechanisms, however, remain poorly defined. Intriguingly, although targeting the IL-23–IL-17 axis substantially improves psoriasis outcomes, this strategy is not more effective than TNF inhibitors in improving musculoskeletal symptoms in PsA. Major unmet needs in the field of PsA include defining those patients with psoriasis at increased risk of developing arthritis, improving our understanding of the natural history of disease and characterizing the immune, environmental and molecular subclinical events preceding PsA onset. Improving our knowledge of this transition is essential for designing clinical trials with treatments that can delay, attenuate or even prevent the development of PsA in patients with psoriasis.
Key points
-
A revolution in the available treatments for psoriasis has led to beneficial clinical responses in a notable proportion of patients who very often achieve remission.
-
The therapeutic accomplishments in psoriasis, however, are yet to be matched in psoriatic arthritis (PsA), for which no new therapies have so far demonstrated superiority over established therapies.
-
Multiple strategies have been attempted to improve PsA outcomes, including earlier aggressive treatment and trying new targets or combinations of available therapeutics.
-
Once PsA is established, the inflammatory burden of psoriatic disease might not be susceptible to modulation in many patients.
-
Defining patients with psoriasis who are at increased risk of progression to PsA will enable studies to dissect the involvement of genetic, environmental and immune factors in PsA transition.
-
A predictive tool that uses integrated data from patients with psoriasis who are at increased risk of PsA progression should provide a framework for the design of preventive clinical trials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ogdie, A. & Weiss, P. The epidemiology of psoriatic arthritis. Rheum. Dis. Clin. North Am. 41, 545–568 (2015).
Gelfand, J. M. et al. Epidemiology of psoriatic arthritis in the population of the US. J. Am. Acad. Dermatol. 53, 573–577 (2005).
Ritchlin, C. T., Colbert, R. A. & Gladman, D. D. Psoriatic arthritis. N. Engl. J. Med. 376, 957–970 (2017).
Haroon, M., Gallagher, P. & FitzGerald, O. Diagnostic delay of more than 6 months contributes to poor radiographic and functional outcome in psoriatic arthritis. Ann. Rheum. Dis. 74, 1045–1050 (2015).
Gladman, D. D. Early psoriatic arthritis. Rheum. Dis. Clin. North Am. 38, 373–386 (2012).
Tillett, W. et al. Interval between onset of psoriasis and psoriatic arthritis comparing the UK Clinical Practice Research Datalink with a hospital-based cohort. Rheumatology 56, 2109–2113 (2017).
Villani, A. P. et al. Prevalence of undiagnosed psoriatic arthritis among psoriasis patients: systematic review and meta-analysis. J. Am. Acad. Dermatol. 73, 242–248 (2015).
Christophers, E. et al. The risk of psoriatic arthritis remains constant following initial diagnosis of psoriasis among patients seen in European dermatology clinics. J. Eur. Acad. Dermatol. Venereol. 24, 548–554 (2010).
Scher, J. U. The 2018 landscape of RA, PsA, and SpA pathogenesis. Curr. Opin. Rheumatol. 30, 57–58 (2018).
Papp, K. et al. Apremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized, controlled trial (Efficacy and Safety Trial Evaluating the Effects of Apremilast in Psoriasis [ESTEEM] 1). J. Am. Acad. Dermatol. 73, 37–49 (2015).
Armstrong, A. W. et al. From the Medical Board of the National Psoriasis Foundation: treatment targets for plaque psoriasis. J. Am. Acad. Dermatol. 76, 290–298 (2017).
McInnes, I. B. et al. Secukinumab, a human anti-interleukin-17A monoclonal antibody, in patients with psoriatic arthritis (FUTURE 2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 386, 1137–1146 (2015).
Mease, P. J. et al. Ixekizumab, an interleukin-17A specific monoclonal antibody, for the treatment of biologic-naive patients with active psoriatic arthritis: results from the 24-week randomised, double-blind, placebo-controlled and active (adalimumab)-controlled period of the phase III trial SPIRIT-P1. Ann. Rheum. Dis. 76, 79–87 (2017).
Mease, P. J. et al. Adalimumab for the treatment of patients with moderately to severely active psoriatic arthritis: results of a double-blind, randomized, placebo-controlled trial. Arthritis Rheum. 52, 3279–3289 (2005).
Ogdie, A. & Coates, L. The changing face of clinical trials in psoriatic arthritis. Curr. Rheumatol. Rep. 19, 21 (2017).
Banfield, C. et al. The safety, tolerability, pharmacokinetics, and pharmacodynamics of a TYK2/JAK1 inhibitor (PF-06700841) in healthy subjects and patients with plaque psoriasis. J. Clin. Pharmacol. 58, 434–447 (2018).
Xue, X. et al. Pharmacologic modulation of RORgammat translates to efficacy in preclinical and translational models of psoriasis and inflammatory arthritis. Sci. Rep. 6, 37977 (2016).
Mease, P. J. et al. Phase 2 study of ABT-122, a TNF- and IL-17A-targeted dual variable domain immunoglobulin, in psoriatic arthritis with inadequate methotrexate response. Arthritis Rheumatol. 70, 1778–1789 (2018).
Glatt, S. et al. Dual IL-17A and IL-17F neutralisation by bimekizumab in psoriatic arthritis: evidence from preclinical experiments and a randomised placebo-controlled clinical trial that IL-17F contributes to human chronic tissue inflammation. Ann. Rheum. Dis. 77, 523–532 (2018).
Okhovat, J. P. et al. Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network Consortium (PPACMAN) Survey: benefits and challenges of combined rheumatology-dermatology clinics. J. Rheumatol. 44, 693–694 (2017).
Coates, L. C. et al. Comparison of three screening tools to detect psoriatic arthritis in patients with psoriasis (CONTEST study). Br. J. Dermatol. 168, 802–807 (2013).
Siegel, E. L., Orbai, A. M. & Ritchlin, C. T. Targeting extra-articular manifestations in PsA: a closer look at enthesitis and dactylitis. Curr. Opin. Rheumatol. 27, 111–117 (2015).
Carron, P. et al. High rate of drug-free remission after induction therapy with golimumab in early peripheral spondyloarthritis. Arthritis Rheumatol. 70, 1769–1777 (2018).
Deane, K. D. & El-Gabalawy, H. Pathogenesis and prevention of rheumatic disease: focus on preclinical RA and SLE. Nat. Rev. Rheumatol. 10, 212–228 (2014).
Olsen, N. J. & Karp, D. R. Autoantibodies and SLE: the threshold for disease. Nat. Rev. Rheumatol. 10, 181–186 (2014).
Burmester, G. R. & Pope, J. E. Novel treatment strategies in rheumatoid arthritis. Lancet 389, 2338–2348 (2017).
Burgers, L. E., Allaart, C. F., Huizinga, T. W. J. & van der Helm-van Mil, A. H. M. Brief report: clinical trials aiming to prevent rheumatoid arthritis cannot detect prevention without adequate risk stratification: a trial of methotrexate versus placebo in undifferentiated arthritis as an example. Arthritis Rheumatol. 69, 926–931 (2017).
Machold, K. P. et al. The Stop Arthritis Very Early (SAVE) trial, an international multicentre, randomised, double-blind, placebo-controlled trial on glucocorticoids in very early arthritis. Ann. Rheum. Dis. 69, 495–502 (2010).
van Aken, J. et al. Five-year outcomes of probable rheumatoid arthritis treated with methotrexate or placebo during the first year (the PROMPT study). Ann. Rheum. Dis. 73, 396–400 (2014).
Verstappen, S. M. et al. Beneficial effects of a 3-week course of intramuscular glucocorticoid injections in patients with very early inflammatory polyarthritis: results of the STIVEA trial. Ann. Rheum. Dis. 69, 503–509 (2010).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03030118 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02603146 (2018).
Eder, L. et al. The incidence and risk factors for psoriatic arthritis in patients with psoriasis: a prospective cohort study. Arthritis Rheumatol. 68, 915–923 (2016).
Eder, L. et al. Association between environmental factors and onset of psoriatic arthritis in patients with psoriasis. Arthritis Care Res. 63, 1091–1097 (2011).
Eder, L. et al. Incidence of arthritis in a prospective cohort of psoriasis patients. Arthritis Care Res. 63, 619–622 (2011).
Ogdie, A. & Gelfand, J. M. Clinical risk factors for the development of psoriatic arthritis among patients with psoriasis: a review of available evidence. Curr. Rheumatol. Rep. 17, 64 (2015).
Ogdie, A. The preclinical phase of PsA: a challenge for the epidemiologist. Ann. Rheum. Dis. 76, 1481–1483 (2017).
Zhou, S. M. et al. Defining disease phenotypes in primary care electronic health records by a machine learning approach: a case study in identifying rheumatoid arthritis. PLOS ONE 11, e0154515 (2016).
Kristensen, L. E. et al. Societal costs and patients’ experience of health inequities before and after diagnosis of psoriatic arthritis: a Danish cohort study. Ann. Rheum. Dis. 76, 1495–1501 (2017).
Nguyen, U. D. T. et al. Smoking paradox in the development of psoriatic arthritis among patients with psoriasis: a population-based study. Ann. Rheum. Dis. 77, 119–123 (2018).
Cunha, J. S., Qureshi, A. A. & Reginato, A. M. Nail enthesis ultrasound in psoriasis and psoriatic arthritis: a report from the 2016 GRAPPA Annual Meeting. J. Rheumatol. 44, 688–690 (2017).
Eder, L. et al. The development of psoriatic arthritis in patients with psoriasis is preceded by a period of nonspecific musculoskeletal symptoms: a prospective cohort study. Arthritis Rheumatol. 69, 622–629 (2017).
Rouzaud, M. et al. Is there a psoriasis skin phenotype associated with psoriatic arthritis? Systematic literature review. J. Eur. Acad. Dermatol. Venereol. 28 (Suppl. 5), 17–26 (2014).
Wilson, F. C. et al. Incidence and clinical predictors of psoriatic arthritis in patients with psoriasis: a population-based study. Arthritis Rheum. 61, 233–239 (2009).
Aydin, S. Z. et al. Ultrasonographic assessment of nail in psoriatic disease shows a link between onychopathy and distal interphalangeal joint extensor tendon enthesopathy. Dermatology 225, 231–235 (2012).
Castellanos-Gonzalez, M. et al. Nail involvement can predict enthesopathy in patients with psoriasis. J. Dtsch. Dermatol. Ges. 14, 1102–1107 (2016).
Love, T. J., Gudjonsson, J. E., Valdimarsson, H. & Gudbjornsson, B. Small joint involvement in psoriatic arthritis is associated with onycholysis: the Reykjavik Psoriatic Arthritis Study. Scand. J. Rheumatol. 39, 299–302 (2010).
Love, T. J. et al. Obesity and the risk of psoriatic arthritis: a population-based study. Ann. Rheum. Dis. 71, 1273–1277 (2012).
Maejima, H., Taniguchi, T., Watarai, A. & Katsuoka, K. Evaluation of nail disease in psoriatic arthritis by using a modified nail psoriasis severity score index. Int. J. Dermatol. 49, 901–906 (2010).
Ogdie, A. et al. Prevalence and treatment patterns of psoriatic arthritis in the UK. Rheumatology 52, 568–575 (2013).
Soltani-Arabshahi, R. et al. Obesity in early adulthood as a risk factor for psoriatic arthritis. Arch. Dermatol. 146, 721–726 (2010).
Tey, H. L. et al. Risk factors associated with having psoriatic arthritis in patients with cutaneous psoriasis. J. Dermatol. 37, 426–430 (2010).
Yeung, H. et al. Psoriasis severity and the prevalence of major medical comorbidity: a population-based study. JAMA Dermatol. 149, 1173–1179 (2013).
Hajiebrahimi, M. et al. Young patients with risk factors prevalent in the elderly - differences in comorbidity depending on severity of psoriasis: a nationwide cross-sectional study in Swedish health registers. Clin. Epidemiol. 10, 705–715 (2018).
Takeshita, J. et al. Psoriasis and comorbid diseases: epidemiology. J. Am. Acad. Dermatol. 76, 377–390 (2017).
Polachek, A., Li, S., Chandran, V. & Gladman, D. D. Clinical enthesitis in a prospective longitudinal psoriatic arthritis cohort: incidence, prevalence, characteristics, and outcome. Arthritis Care Res. 69, 1685–1691 (2017).
Deane, K. D., Norris, J. M. & Holers, V. M. Preclinical rheumatoid arthritis: identification, evaluation, and future directions for investigation. Rheum. Dis. Clin. North Am. 36, 213–241 (2010).
van Steenbergen, H. W. et al. Characterising arthralgia in the preclinical phase of rheumatoid arthritis using MRI. Ann. Rheum. Dis. 74, 1225–1232 (2015).
Li, W., Han, J. & Qureshi, A. A. Obesity and risk of incident psoriatic arthritis in US women. Ann. Rheum. Dis. 71, 1267–1772 (2012).
Wu, S., Li, W. Q., Han, J., Sun, Q. & Qureshi, A. A. Hypercholesterolemia and risk of incident psoriasis and psoriatic arthritis in US women. Arthritis Rheumatol. 66, 304–310 (2014).
Egeberg, A. et al. Association of psoriatic disease with uveitis: a Danish nationwide cohort study. JAMA Dermatol. 151, 1200–1205 (2015).
Lewinson, R. T. et al. Depression is associated with an increased risk of psoriatic arthritis among patients with psoriasis: a population-based study. J. Invest. Dermatol. 137, 828–835 (2017).
Tsuruta, N., Imafuku, S. & Narisawa, Y. Hyperuricemia is an independent risk factor for psoriatic arthritis in psoriatic patients. J. Dermatol. 44, 1349–1352 (2017).
Ogdie, A. et al. Weight change before and after diagnosis in patients with psoriatic arthritis, rheumatoid arthritis, and ankylosing spondylitis [abstract]. Arthritis Rheumatol. 69 (Suppl. 10), 2524 (2017).
Kaushik, S. B. & Lebwohl, M. G. Psoriasis: which therapy for which patient psoriasis comorbidities and preferred systemic agents. J. Am. Acad. Dermatol. 80, 27–40 (2019).
Wu, W., Debbaneh, M., Moslehi, H., Koo, J. & Liao, W. Tonsillectomy as a treatment for psoriasis: a review. J. Dermatolog. Treat. 25, 482–486 (2014).
Thorarensen, S. M. et al. Physical trauma recorded in primary care is associated with the onset of psoriatic arthritis among patients with psoriasis. Ann. Rheum. Dis. 76, 521–525 (2017).
Hsieh, J., Kadavath, S. & Efthimiou, P. Can traumatic injury trigger psoriatic arthritis? A review of the literature. Clin. Rheumatol. 33, 601–608 (2014).
Punzi, L. et al. Clinical, laboratory and immunogenetic aspects of post-traumatic psoriatic arthritis: a study of 25 patients. Clin. Exp. Rheumatol. 16, 277–281 (1998).
Tinazzi, I. et al. ‘Deep Koebner’ phenomenon of the flexor tendon-associated accessory pulleys as a novel factor in tenosynovitis and dactylitis in psoriatic arthritis. Ann. Rheum. Dis. 77, 922–925 (2018).
Ng, J., Tan, A. L. & McGonagle, D. Unifocal psoriatic arthritis development in identical twins following site specific injury: evidence supporting biomechanical triggering events in genetically susceptible hosts. Ann. Rheum. Dis. 74, 948–949 (2015).
Jacques, P. et al. Proof of concept: enthesitis and new bone formation in spondyloarthritis are driven by mechanical strain and stromal cells. Ann. Rheum. Dis. 73, 437–445 (2014).
Van Mechelen, M. & Lories, R. J. Microtrauma: no longer to be ignored in spondyloarthritis? Curr. Opin. Rheumatol. 28, 176–180 (2016).
Stojanovich, L. Stress and autoimmunity. Autoimmun. Rev. 9, A271–A276 (2010).
Song, H. et al. Association of stress-related disorders with subsequent autoimmune disease. JAMA 319, 2388–2400 (2018).
Naldi, L. et al. Cigarette smoking, body mass index, and stressful life events as risk factors for psoriasis: results from an Italian case-control study. J. Invest. Dermatol. 125, 61–67 (2005).
Eder, L. et al. The association between smoking and the development of psoriatic arthritis among psoriasis patients. Ann. Rheum. Dis. 71, 219–224 (2012).
Li, W., Han, J. & Qureshi, A. A. Smoking and risk of incident psoriatic arthritis in US women. Ann. Rheum. Dis. 71, 804–808 (2012).
Armstrong, A. W., Harskamp, C. T., Dhillon, J. S. & Armstrong, E. J. Psoriasis and smoking: a systematic review and meta-analysis. Br. J. Dermatol. 170, 304–314 (2014).
Wu, S., Cho, E., Li, W. Q., Han, J. & Qureshi, A. A. Alcohol intake and risk of incident psoriatic arthritis in women. J. Rheumatol. 42, 835–840 (2015).
Yilmaz Tasdelen, O., Yurdakul, F. G., Duran, S. & Bodur, H. Isotretinoin-induced arthritis mimicking both rheumatoid arthritis and axial spondyloarthritis. Int. J. Rheum. Dis. 18, 466–469 (2015).
Elnady, B. D., Desouky, D., Elkhouly, T., Dawoud, N. & Kewan, H. Isotretinoin induced seronegative spondyloarthropathy: magnetic resonance imaging as diagnostic and prognostic biomarker [abstract]. Arthritis Rheumatol. 69 (Suppl. 10), 1569 (2017).
Henning, P., Conaway, H. H. & Lerner, U. H. Retinoid receptors in bone and their role in bone remodeling. Front. Endocrinol. (Lausanne) 6, 31 (2015).
Wu, S., Han, J. & Qureshi, A. A. Use of aspirin, non-steroidal anti-inflammatory drugs, and acetaminophen (paracetamol), and risk of psoriasis and psoriatic arthritis: a cohort study. Acta Derm. Venereol. 95, 217–223 (2015).
Karason, A., Love, T. J. & Gudbjornsson, B. A strong heritability of psoriatic arthritis over four generations — the Reykjavik Psoriatic Arthritis Study. Rheumatology 48, 1424–1428 (2009).
Chandran, V. et al. Familial aggregation of psoriatic arthritis. Ann. Rheum. Dis. 68, 664–667 (2009).
FitzGerald, O., Haroon, M., Giles, J. T. & Winchester, R. Concepts of pathogenesis in psoriatic arthritis: genotype determines clinical phenotype. Arthritis Res. Ther. 17, 115 (2015).
Wordsworth, P. & Bell, J. Polygenic susceptibility in rheumatoid arthritis. Ann. Rheum. Dis. 50, 343–346 (1991).
Alarcon-Segovia, D. et al. Familial aggregation of systemic lupus erythematosus, rheumatoid arthritis, and other autoimmune diseases in 1,177 lupus patients from the GLADEL cohort. Arthritis Rheum. 52, 1138–1147 (2005).
Rahman, P. & Elder, J. T. Genetic epidemiology of psoriasis and psoriatic arthritis. Ann. Rheum. Dis. 64 (Suppl. 2), ii37–ii39; discussion ii40–ii41 (2005).
Myers, A., Kay, L. J., Lynch, S. A. & Walker, D. J. Recurrence risk for psoriasis and psoriatic arthritis within sibships. Rheumatology 44, 773–776 (2005).
Bowes, J. et al. Dense genotyping of immune-related susceptibility loci reveals new insights into the genetics of psoriatic arthritis. Nat. Commun. 6, 6046 (2015).
Okada, Y. et al. Fine mapping major histocompatibility complex associations in psoriasis and its clinical subtypes. Am. J. Hum. Genet. 95, 162–172 (2014).
Stuart, P. E. et al. Genome-wide association analysis of psoriatic arthritis and cutaneous psoriasis reveals differences in their genetic architecture. Am. J. Hum. Genet. 97, 816–836 (2015).
Bowes, J. et al. PTPN22 is associated with susceptibility to psoriatic arthritis but not psoriasis: evidence for a further PsA-specific risk locus. Ann. Rheum. Dis. 74, 1882–1885 (2015).
Huffmeier, U. et al. Common variants at TRAF3IP2 are associated with susceptibility to psoriatic arthritis and psoriasis. Nat. Genet. 42, 996–999 (2010).
Aterido, A. et al. Genetic variation at the glycosaminoglycan metabolism pathway contributes to the risk of psoriatic arthritis but not psoriasis. Ann. Rheum. Dis. https://doi.org/10.1136/annrheumdis-2018-214158 (2018).
Patrick, M. T. et al. Genetic signature to provide robust risk assessment of psoriatic arthritis development in psoriasis patients. Nat. Commun. 9, 4178 (2018).
Farh, K. K. et al. Genetic and epigenetic fine mapping of causal autoimmune disease variants. Nature 518, 337–343 (2015).
Hanson, A. & Brown, M. A. Genetics and the causes of ankylosing spondylitis. Rheum. Dis. Clin. North Am. 43, 401–414 (2017).
Li, Z. et al. Epigenetic and gene expression analysis of ankylosing spondylitis-associated loci implicate immune cells and the gut in the disease pathogenesis. Genes Immun. 18, 135–143 (2017).
Brancato, L., Itescu, S., Skovron, M. L., Solomon, G. & Winchester, R. Aspects of the spectrum, prevalence and disease susceptibility determinants of Reiter’s syndrome and related disorders associated with HIV infection. Rheumatol. Int. 9, 137–141 (1989).
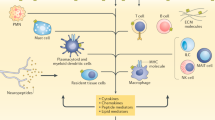
Fitzgerald, O. & Winchester, R. Editorial: emerging evidence for critical involvement of the interleukin-17 pathway in both psoriasis and psoriatic arthritis. Arthritis Rheumatol. 66, 1077–1080 (2014).
Taams, L. S., Steel, K. J. A., Srenathan, U., Burns, L. A. & Kirkham, B. W. IL-17 in the immunopathogenesis of spondyloarthritis. Nat. Rev. Rheumatol. 14, 453–466 (2018).
Leijten, E. F. et al. Brief report: enrichment of activated group 3 innate lymphoid cells in psoriatic arthritis synovial fluid. Arthritis Rheumatol. 67, 2673–2678 (2015).
Menon, B. et al. Interleukin-17+CD8+ T cells are enriched in the joints of patients with psoriatic arthritis and correlate with disease activity and joint damage progression. Arthritis Rheumatol. 66, 1272–1281 (2014).
Leonardi, C. et al. Anti-interleukin-17 monoclonal antibody ixekizumab in chronic plaque psoriasis. N. Engl. J. Med. 366, 1190–1199 (2012).
Raghavan, S. et al. FOXP3 expression in blood, synovial fluid and synovial tissue during inflammatory arthritis and intra-articular corticosteroid treatment. Ann. Rheum. Dis. 68, 1908–1915 (2009).
Flores-Borja, F., Jury, E. C., Mauri, C. & Ehrenstein, M. R. Defects in CTLA-4 are associated with abnormal regulatory T cell function in rheumatoid arthritis. Proc. Natl Acad. Sci. USA 105, 19396–19401 (2008).
Ryder, L. R. et al. FoxP3 mRNA splice forms in synovial CD4+ T cells in rheumatoid arthritis and psoriatic arthritis. APMIS 120, 387–396 (2012).
Sugiyama, H. et al. Dysfunctional blood and target tissue CD4+CD25high regulatory T cells in psoriasis: mechanism underlying unrestrained pathogenic effector T cell proliferation. J. Immunol. 174, 164–173 (2005).
Yun, W. J. et al. Role of CD4CD25FOXP3 regulatory T cells in psoriasis. Ann. Dermatol. 22, 397–403 (2010).
Han, L. et al. Phenotypical analysis of ectoenzymes CD39/CD73 and adenosine receptor 2A in CD4+ CD25(high) Foxp3+ regulatory T cells in psoriasis. Australas. J. Dermatol. 59, e31–e38 (2018).
Cao, D., van Vollenhoven, R., Klareskog, L., Trollmo, C. & Malmstrom, V. CD25brightCD4+ regulatory T cells are enriched in inflamed joints of patients with chronic rheumatic disease. Arthritis Res. Ther. 6, R335–R346 (2004).
Szentpetery, A. et al. Abatacept reduces synovial regulatory T cell expression in patients with psoriatic arthritis. Arthritis Res. Ther. 19, 158 (2017).
Calcinotto, A. et al. IL-23 secreted by myeloid cells drives castration-resistant prostate cancer. Nature 559, 363–369 (2018).
Crane, J. L. & Cao, X. Bone marrow mesenchymal stem cells and TGF-beta signaling in bone remodeling. J. Clin. Invest. 124, 466–472 (2014).
van Buul, G. M. et al. Mesenchymal stem cells secrete factors that inhibit inflammatory processes in short-term osteoarthritic synovium and cartilage explant culture. Osteoarthr. Cartil. 20, 1186–1196 (2012).
Rehaume, L. M. et al. ZAP-70 Genotype disrupts the relationship between microbiota and host, leading to spondyloarthritis and ileitis in SKG mice. Arthritis Rheumatol. 66 2780–2792 (2014).
Scher, J. U., Littman, D. R. & Abramson, S. B. Microbiome in inflammatory arthritis and human rheumatic diseases. Arthritis Rheumatol. 68 35–45 (2016).
Lin, P. et al. HLA-B27 and Human β2-microglobulin affect the gut microbiota of transgenic rats. PLOS ONE 9 e105684 (2014).
Mielants, H. et al. The evolution of spondyloarthropathies in relation to gut histology. II. Histological aspects. J. Rheumatol. 22, 2273–2278 (1995).
Scarpa, R. et al. Microscopic inflammatory changes in colon of patients with both active psoriasis and psoriatic arthritis without bowel symptoms. J. Rheumatol. 27, 1241–1246 (2000).
Ciccia, F. et al. Interleukin-9 overexpression and Th9 polarization characterize the inflamed gut, the synovial tissue, and the peripheral blood of patients with psoriatic arthritis. Arthritis Rheumatol. 68, 1922–1931 (2016).
Li, W. Q., Han, J. L., Chan, A. T. & Qureshi, A. A. Psoriasis, psoriatic arthritis and increased risk of incident Crohn’s disease in US women. Ann. Rheum. Dis. 72, 1200–1205 (2013).
Scher, J. U. et al. Decreased bacterial diversity characterizes the altered gut microbiota in patients with psoriatic arthritis, resembling dysbiosis in inflammatory bowel disease. Arthritis Rheumatol. 67, 128–139 (2015).
Breban, M. et al. Faecal microbiota study reveals specific dysbiosis in spondyloarthritis. Ann. Rheum. Dis. 76, 1614–1622 (2017).
Costello, M. E. et al. Brief report: intestinal dysbiosis in ankylosing spondylitis. Arthritis Rheumatol. 67, 686–691 (2015).
Manasson, J. et al. Gut microbiota perturbations in reactive arthritis and postinfectious spondyloarthritis. Arthritis Rheumatol. 70, 242–254 (2018).
Tito, R. Y. et al. Brief report: dialister as a microbial marker of disease activity in spondyloarthritis. Arthritis Rheumatol. 69, 114–121 (2017).
Mitra, A., Raychaudhuri, S. K. & Raychaudhuri, S. P. Functional role of IL-22 in psoriatic arthritis. Arthritis Res. Ther. 14, R65 (2012).
Sherlock, J. P. et al. IL-23 induces spondyloarthropathy by acting on ROR-γt+ CD3+CD4-CD8- entheseal resident T cells. Nat. Med. 18, 1069–1076 (2012).
Chiu, Y. G. et al. Dendritic cell-surface transmembrane protein (DC-STAMP) is a biomarker for osteoclast presursors in psoriatic arthritis. Arthritis Rheum. 58, S945 (2008).
Ritchlin, C. T., Haas-Smith, S. A., Li, P., Hicks, D. G. & Schwarz, E. M. Mechanisms of TNF-alpha- and RANKL-mediated osteoclastogenesis and bone resorption in psoriatic arthritis. J. Clin. Invest. 111, 821–831 (2003).
Sato, K. et al. Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. J. Exp. Med. 203, 2673–2682 (2006).
Chandran, V. & Scher, J. U. Biomarkers in psoriatic arthritis: recent progress. Curr. Rheumatol. Rep. 16, 453 (2014).
Abji, F., Pollock, R. A., Liang, K., Chandran, V. & Gladman, D. D. Brief report: CXCL10 is a possible biomarker for the development of psoriatic arthritis among patients with psoriasis. Arthritis Rheumatol. 68, 2911–2916 (2016).
Cretu, D. et al. Differentiating psoriatic arthritis from psoriasis without psoriatic arthritis using novel serum biomarkers. Arthritis Care Res. 70, 454–461 (2018).
Cretu, D. et al. Quantitative tandem mass-spectrometry of skin tissue reveals putative psoriatic arthritis biomarkers. Clin. Proteomics 12, 1 (2015).
Gisondi, P. et al. Lower limb enthesopathy in patients with psoriasis without clinical signs of arthropathy: a hospital-based case-control study. Ann. Rheum. Dis. 67, 26–30 (2008).
Namey, T. C. & Rosenthall, L. Periarticular uptake of 99mtechnetium diphosphonate in psoriatics: correlation with cutaneous activity. Arthritis Rheum. 19, 607–612 (1976).
Offidani, A., Cellini, A., Valeri, G. & Giovagnoni, A. Subclinical joint involvement in psoriasis: magnetic resonance imaging and X-ray findings. Acta Derm. Venereol. 78, 463–465 (1998).
Simon, D. et al. Analysis of periarticular bone changes in patients with cutaneous psoriasis without associated psoriatic arthritis. Ann. Rheum. Dis. 75, 660–666 (2016).
Araujo, E. G. et al. Effects of ustekinumab versus tumor necrosis factor inhibition on enthesitis: results from the enthesial clearance in psoriatic arthritis (ECLIPSA) study. Semin. Arthritis Rheum. https://doi.org/10.1016/j.semarthrit.2018.05.011 (2018).
Taylor, W. et al. Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum. 54, 2665–2673 (2006).
Tinazzi, I. et al. Preliminary evidence that subclinical enthesopathy may predict psoriatic arthritis in patients with psoriasis. J. Rheumatol. 38, 2691–2692 (2011).
van Steenbergen, H. W., da Silva, J. A. P., Huizinga, T. W. J. & van der Helm-van Mil, A. H. M. Preventing progression from arthralgia to arthritis: targeting the right patients. Nat. Rev. Rheumatol. 14, 32–41 (2018).
Verheul, M. K. et al. Triple positivity for anti–citrullinated protein autoantibodies, rheumatoid factor, and anti–carbamylated protein antibodies conferring high specificity for rheumatoid arthritis: implications for very early identification of at-risk individuals. Arthritis Rheumatol. 70, 1721–1731 (2018).
Ogdie, A. et al. in Pharmacoepidemiology (eds Strom, B. L., Kimmel, S. E. & Hennessy, S.) Vol. 5 (Wiley-Blackwell, Philadelphia, 2012).
Ogdie, A. et al. Validity of psoriatic arthritis and capture of disease modifying antirheumatic drugs in the health improvement network. Pharmacoepidemiol. Drug Saf. 23, 918–922 (2014).
Hernan, M. & Robins, J. Causal Inference (CRC Press, 2018).
Gottlieb, M., Calin, A. & Gale, R. P. Discordance for psoriatic arthropathy in monozygotic twins. Arthritis Rheum. 22, 805–806 (1979).
Pedersen, O. B., Svendsen, A. J., Ejstrup, L., Skytthe, A. & Junker, P. On the heritability of psoriatic arthritis. Disease concordance among monozygotic and dizygotic twins. Ann. Rheum. Dis. 67, 1417–1421 (2008).
Langley, R. G. et al. Secukinumab in plaque psoriasis–results of two phase 3 trials. N. Engl. J. Med. 371, 326–338 (2014).
Baeten, D. et al. Secukinumab, an interleukin-17A inhibitor, in ankylosing spondylitis. N. Engl. J. Med. 373, 2534–2548 (2015)
Gordon, K. B. et al. Phase 3 trials of ixekizumab in moderate-to-severe plaque psoriasis. N. Engl. J. Med. 375, 345–356 (2016).
van der Heijde, D. et al. Ixekizumab, an interleukin-17A antagonist in the treatment of ankylosing spondylitis or radiographic axial spondyloarthritis in patients previously untreated with biological disease-modifying anti-rheumatic drugs (COAST-V): 16 week results of a phase 3 randomised, double-blind, active-controlled and placebo-controlled trial. Lancet 392, 2441–2451 (2018).
Baeten, D. et al. Risankizumab, an IL-23 inhibitor, for ankylosing spondylitis: results of a randomised, double-blind, placebo-controlled, proof-of-concept, dose-finding phase 2 study. Ann. Rheum. Dis. 77, 1295–1302 (2018).
Lambert, N. C. How twin studies help to understand inflammatory joint disease. Joint Bone Spine 83, 637–643 (2016).
The National Psoriasis Foundation. The road to a cure. psoriasis.org https://www.psoriasis.org/researchers/funding (2018).
Gladman, D., Antoni, C., Mease, P., Clegg, D. & Nash, P. Psoriatic arthritis: epidemiology, clinical features, course and outcome. Ann. Rheum. Dis. 64 (Suppl. 2), ii14–ii17 (2005).
Haberman, R. et al. Bridging the gaps in the care of psoriasis and psoriatic arthritis: the role of combined clinics. Curr. Rheumatol. Rep. 20, 76 (2018).
Schneeweiss, M., Merola, J. F., Karlson, E. W. & Solomon, D. H. Rationale and design of the Brigham Cohort for psoriasis and psoriatic arthritis registry (COPPAR). BMC Dermatol. 17, 11 (2017).
PrePARePsA Study. Preventing Psoriatic Arthritis Registry PrePARe http://www.preparepsa.org (2018).
Virginia Commonwealth University. Mid-Atlantic Twin Registry. VCU https://matr.vcu.edu/twins--families/participate-now/ (updated 22 Aug 2018).
Arakawa, A. et al. Melanocyte antigen triggers autoimmunity in human psoriasis. J. Exp. Med. 212, 2203–2212 (2015).
Acknowledgements
The work of J.U.S. is supported by grants from the US National Institute of Arthritis and Musculoskeletal and Skin Diseases (No. K23AR064318 and R03AR072182), The Riley Family Foundation, The Beatriz Snyder Foundation, the National Psoriasis Foundation and the Rheumatology Research Foundation. The work of A.O. is supported by the National Psoriasis Foundation, the Rheumatology Research Foundation and grants from the US National Institute of Arthritis and Musculoskeletal and Skin Diseases (No. K23AR063764 and R01AR072363). The work of C.R. is supported by grants from the National Psoriasis Foundation Discovery and the National Institutes of Health (No. R01AR069000).
Reviewer information
Nature Reviews Rheumatology thanks F. Behrens, D. Elewaut and other anonymous reviewer(s), for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
J.U.S. has served as a consultant for Bristol-Myers Squibb, Janssen, Novartis and UCB and has received grants to the New York University School of Medicine from Novartis and Pfizer. A.O. has served as a consultant for Abbvie, Amgen, Bristol-Myers Squibb, Celgene, Corrona, Lilly, Novartis, Pfizer and Takeda and has received grants to the University of Pennsylvania from Novartis and Pfizer. J.F.M. is a consultant and/or investigator for Abbvie, Amgen, Biogen IDEC, Celgene, Eli Lilly, GlaxoSmithKline, Janssen, Merck, Novartis, Pfizer, Samumed, Sanofi Regeneron and UCB. C.R. has received grants from Abbvie, Amgen and UCB. He is a consultant for Abbvie, Amgen, Janssen, Novartis, Pfizer and UCB.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Scher, J.U., Ogdie, A., Merola, J.F. et al. Preventing psoriatic arthritis: focusing on patients with psoriasis at increased risk of transition. Nat Rev Rheumatol 15, 153–166 (2019). https://doi.org/10.1038/s41584-019-0175-0
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-019-0175-0
This article is cited by
-
Ultrasonographic and power doppler parameters of nails fail to differentiate between onychodystrophy in patients with psoriasis vulgaris or psoriatic arthritis
Advances in Rheumatology (2024)
-
Drug- or Vaccine-Induced/Aggravated Psoriatic Arthritis: A Systematic Review
Dermatology and Therapy (2024)
-
Therapie der Psoriasis vulgaris
Die Dermatologie (2024)
-
Cutaneous lesions in psoriatic arthritis are enriched in chemokine transcriptomic pathways
Arthritis Research & Therapy (2023)
-
Epidemiological analysis of patients with psoriatic arthritis in follow-up at the brazilian Unified Health System
Advances in Rheumatology (2023)