Abstract
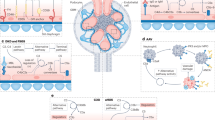
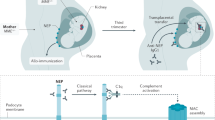
The C3 glomerulopathies are a group of rare kidney diseases characterized by complement dysregulation occurring in the fluid phase and in the glomerular microenvironment, which results in prominent complement C3 deposition in kidney biopsy samples. The two major subgroups of C3 glomerulopathy — dense deposit disease (DDD) and C3 glomerulonephritis (C3GN) — have overlapping clinical and pathological features suggestive of a disease continuum. Dysregulation of the complement alternative pathway is fundamental to the manifestations of C3 glomerulopathy, although terminal pathway dysregulation is also common. Disease is driven by acquired factors in most patients — namely, autoantibodies that target the C3 or C5 convertases. These autoantibodies drive complement dysregulation by increasing the half-life of these vital but normally short-lived enzymes. Genetic variation in complement-related genes is a less frequent cause. No disease-specific treatments are available, although immunosuppressive agents and terminal complement pathway blockers are helpful in some patients. Unfortunately, no treatment is universally effective or curative. In aggregate, the limited data on renal transplantation point to a high risk of disease recurrence (both DDD and C3GN) in allograft recipients. Clinical trials are underway to test the efficacy of several first-generation drugs that target the alternative complement pathway.
Key points
-
C3 glomerulopathies are rare diseases that share an underlying mechanism of complement dysregulation in the fluid phase and glomerular microenvironment.
-
Diagnosis relies solely on renal biopsy immunofluorescence findings; light microscopy findings and complement biomarker profiles are heterogeneous.
-
Acquired drivers, in the form of autoantibodies, are the abnormalities most frequently associated with complement dysregulation.
-
Genetic variants in the C3, CFB, CFH, CFI and CFHR1–CFHR5 genes are potentially causal; both rare and common variants can coexist and are associated with susceptibility to disease.
-
Convertase dysregulation is central to the pathogenesis of C3 glomerulopathy.
-
Conditions such as post-infectious glomerulonephritis cannot be differentiated from C3 glomerulopathy by renal biopsy alone, which can confound early diagnosis and treatment.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pickering, M. C. et al. C3 glomerulopathy: consensus report. Kidney Int. 84, 1079–1089 (2013).
Smith, R. J. H. et al. New approaches to the treatment of dense deposit disease. J. Am. Soc. Nephrol. 18, 2447–2456 (2007).
Bomback, A. S. et al. C3 glomerulonephritis and dense deposit disease share a similar disease course in a large United States cohort of patients with C3 glomerulopathy. Kidney Int. 93, 997–985 (2018).
Medjeral-Thomas, N. R. et al. C3 glomerulopathy: clinicopathologic features and predictors of outcome. Clin. J. Am. Soc. Nephrol. 9, 46–53 (2014).
Servais, A. et al. Acquired and genetic complement abnormalities play a critical role in dense deposit disease and other C3 glomerulopathies. Kidney Int. 82, 454–464 (2012).
Athanasiou, Y. et al. Familial C3 glomerulopathy associated with CFHR5 mutations: clinical characteristics of 91 patients in 16 pedigrees. Clin. J. Am. Soc. Nephrol. 6, 1436–1446 (2011).
Rabasco, C. et al. Effectiveness of mycophenolate mofetil in C3 glomerulonephritis. Kidney Int. 88, 1153–1160 (2015).
Iatropoulos, P. et al. Complement gene variants determine the risk of immunoglobulin-associated MPGN and C3 glomerulopathy and predict long-term renal outcome. Mol. Immunol. 71, 131–142 (2016).
Angelo, J. R., Bell, C. S. & Braun, M. C. Allograft failure in kidney transplant recipients with membranoproliferative glomerulonephritis. Am. J. Kidney Dis. 57, 291–299 (2011).
Zand, L. et al. Clinical findings, pathology, and outcomes of C3GN after kidney transplantation. J. Am. Soc. Nephrol. 25, 1110–1117 (2014).
D’Agati, V. D. & Bomback, A. S. C3 glomerulopathy: what’s in a name? Kidney Int. 82, 379–381 (2012).
Sethi, S. et al. Glomeruli of dense deposit disease contain components of the alternative and terminal complement pathway. Kidney Int. 75, 952–960 (2009).
Sethi, S. et al. Characterization of C3 in C3 glomerulopathy. Nephrol. Dial. Transplant. 32, 459–465 (2017).
Sethi, S. & Fervenza, F. C. Membranoproliferative glomerulonephritis — a new look at an old entity. N. Engl. J. Med. 366, 1119–1131 (2012).
Hou, J. et al. Toward a working definition of C3 glomerulopathy by immunofluorescence. Kidney Int. 85, 450–456 (2014).
Bouatou, Y. et al. Diagnostic accuracy of immunofluorescence versus immunoperoxidase staining to distinguish immune complex-mediated glomerulonephritis and C3 dominant glomerulopathy. Histopathology 72, 601–608 (2018).
Sethi, S. et al. Atypical post-infectious glomerulonephritis is associated with abnormalities in the alternative pathway of complement. Kidney Int. 83, 293–299 (2013).
Khalighi, M. A. et al. Revisiting post-infectious glomerulonephritis in the emerging era of C3 glomerulopathy. Clin. Kidney J. 9, 397–402 (2016).
Sethi, S. et al. A proposal for standardized grading of chronic changes in native kidney biopsy specimens. Kidney Int. 91, 787–789 (2017).
Ricklin, D., Reis, E. S., Mastellos, D. C., Gros, P. & Lambris, J. D. Complement component C3 — the “Swiss army knife” of innate immunity and host defense. Immunol. Rev. 274, 33–58 (2016).
Blatt, A. Z., Pathan, S. & Ferreira, V. P. Properdin: a tightly regulated critical inflammatory modulator. Immunol. Rev. 274, 172–190 (2016).
Wu, J. et al. Structure of complement fragment C3b–factor H and implications for host protection by complement regulators. Nat. Immunol. 10, 728–733 (2009).
Pangburn, M. K., Schreiber, R. D. & Müller-Eberhard, H. J. Human complement C3b inactivator: isolation, characterization, and demonstration of an absolute requirement for the serum protein β1H for cleavage of C3b and C4b in solution. J. Exp. Med. 146, 257–270 (1977).
Whaley, K. & Ruddy, S. Modulation of the alternative complement pathways by β1H globulin. J. Exp. Med. 144, 1147 (1976).
Medjeral-Thomas, N. & Pickering, M. C. The complement factor H-related proteins. Immunol. Rev. 274, 191–201 (2016).
Xiao, X. et al. Familial C3 glomerulonephritis caused by a novel CFHR5–CFHR2 fusion gene. Mol. Immunol. 77, 89–96 (2016).
Goicoechea de Jorge, E. et al. Dimerization of complement factor H-related proteins modulates complement activation in vivo. Proc. Natl Acad. Sci. USA 110, 4685–4690 (2013).
Tortajada, A. et al. C3 glomerulopathy-associated CFHR1 mutation alters FHR oligomerization and complement regulation. J. Clin. Invest. 123, 2434–2446 (2013).
Józsi, M. et al. Factor H-related proteins determine complement-activating surfaces. Trends Immunol. 36, 374–384 (2015).
Høgåsen, K., Jansen, J. H., Mollnes, T. E., Hovdenes, J. & Harboe, M. Hereditary porcine membranoproliferative glomerulonephritis type II is caused by factor H deficiency. J. Clin. Invest. 95, 1054–1061 (1995).
Pickering, M. C. et al. Uncontrolled C3 activation causes membranoproliferative glomerulonephritis in mice deficient in complement factor H. Nat. Genet. 31, 424–428 (2002).
Ruseva, M. M. et al. Loss of properdin exacerbates C3 glomerulopathy resulting from factor H deficiency. J. Am. Soc. Nephrol. 24, 43–52 (2013).
Lesher, A. M. et al. Combination of factor H mutation and properdin deficiency causes severe C3 glomerulonephritis. J. Am. Soc. Nephrol. 24, 53–65 (2013).
Pickering, M. C. et al. Prevention of C5 activation ameliorates spontaneous and experimental glomerulonephritis in factor H-deficient mice. Proc. Natl Acad. Sci. USA 103, 9649–9654 (2006).
Rose, K. L. et al. Factor I is required for the development of membranoproliferative glomerulonephritis in factor H-deficient mice. J. Clin. Invest. 118, 608–618 (2008).
Arkill, K. P. et al. Resolution of the three dimensional structure of components of the glomerular filtration barrier. BMC Nephrol. 15, 24 (2014).
Lennon, R. et al. Global analysis reveals the complexity of the human glomerular extracellular matrix. J. Am. Soc. Nephrol. 25, 939–951 (2014).
Hegermann, J., Lünsdorf, H., Ochs, M. & Haller, H. Visualization of the glomerular endothelial glycocalyx by electron microscopy using cationic colloidal thorium dioxide. Histochem. Cell Biol. 145, 41–51 (2016).
Loeven, M. A. et al. Mutations in complement factor H impair alternative pathway regulation on mouse glomerular endothelial cells in vitro. J. Biol. Chem. 291, 4974–4981 (2016).
Persson, B. D. et al. Structure of the extracellular portion of CD46 provides insights into its interactions with complement proteins and pathogens. PLOS Pathog. 6, e1001122 (2010).
Heinen, S. et al. Factor H-related protein 1 (CFHR-1) inhibits complement C5 convertase activity and terminal complex formation. Blood 114, 2439–2447 (2009).
Eberhardt, H. U. et al. Human factor H-related protein 2 (CFHR2) regulates complement activation. PLOS ONE 18, e78617 (2013).
Chen, Q. et al. Complement factor H-related 5-hybrid proteins anchor properdin and activate complement at self-surfaces. J. Am. Soc. Nephrol. 27, 1413–1425 (2016).
Chen, Q. et al. Complement factor H-related hybrid protein deregulates complement in dense deposit disease. J. Clin. Invest. 124, 145–155 (2014).
Gale, D. P. et al. Identification of a mutation in complement factor H-related protein 5 in patients of Cypriot origin with glomerulonephritis. Lancet 376, 794–801 (2010).
Malik, T. H. et al. A hybrid CFHR3–1 gene causes familial C3 glomerulopathy. J. Am. Soc. Nephrol. 23, 1155–1160 (2012).
Togarsimalemath, S. K. et al. A novel CFHR1–CFHR5 hybrid leads to a familial dominant C3 glomerulopathy. Kidney Int. 92, 876–887 (2017).
Csincsi, Á. I. et al. Factor H-related protein 5 interacts with pentraxin 3 and the extracellular matrix and modulates complement activation. J. Immunol. 194, 4963–4973 (2015).
Larsen, S. Glomerular immune deposits in kidneys from patients with no clinical or light microscopic evidence of glomerulonephritis. Assessment of the influence of autolysis on identification of immunoglobulins and complement. Acta Pathol. Microbiol. Scand. A 87A, 313–319 (1979).
Velosa, J., Miller, K. & Michael, A. F. Immunopathology of the end-stage kidney. Immunoglobulin and complement component deposition in nonimmune disease. Am. J. Pathol. 84, 149–162 (1976).
Leivo, I. & Engvall, E. C3d fragment of complement interacts with laminin and binds to basement membranes of glomerulus and trophoblast. J. Cell. Biol. 103, 1091–1100 (1986).
Bu, F. et al. High-throughput genetic testing for thrombotic microangiopathies and C3 glomerulopathies. J. Am. Soc. Nephrol. 27, 1245–1253 (2016).
Osborne, A. J. et al. Statistical validation of rare complement variants provides insights into the molecular basis of atypical hemolytic uremic syndrome and C3 glomerulopathy. J. Immunol. 200, 2464–2478 (2018).
Noris, M. & Remuzzi, G. Glomerular diseases dependent on complement activation, including atypical hemolytic uremic syndrome, membranoproliferative glomerulonephritis, and C3 glomerulopathy: core curriculum 2015. Am. J. Kidney Dis. 66, 359–375 (2015).
Barbour, T. D. et al. Complement receptor 3 mediates renal protection in experimental C3 glomerulopathy. Kidney Int. 89, 823–832 (2016).
Zhang, Y. et al. Causes of alternative pathway dysregulation in dense deposit disease. Clin. J. Am. Soc. Nephrol. 7, 265–274 (2012).
Marinozzi, M. C. et al. C5 nephritic factors drive the biological phenotype of C3 glomerulopathies. Kidney Int. 92, 1232–1241 (2017).
Blanc, C. et al. Anti-factor H autoantibodies in C3 glomerulopathies and in atypical hemolytic uremic syndrome: one target, two diseases. J. Immunol. 194, 5129–5138 (2015).
Marinozzi, M. C. et al. Anti-factor B and anti-C3b autoantibodies in C3 glomerulopathy and Ig-associated membranoproliferative GN. J. Am. Soc. Nephrol. 28, 1603–1613 (2017).
Zhang, Y. et al. C4 nephritic factors in C3 glomerulopathy: a case series. Am. J. Kidney Dis. 70, 834–843 (2017).
Strobel, S., Zimmering, M., Papp, K., Prechl, J. & Józsi, M. Anti-factor B autoantibody in dense deposit disease. Mol. Immunol. 47, 1476–1483 (2010).
Blom, A. M. et al. Testing the activity of complement convertases in serum/plasma for diagnosis of C4Nef-mediated C3 glomerulonephritis. J. Clin. Immunol. 36, 517–527 (2016).
Meri, S., Koistinen, V., Miettinen, A., Törnroth, T. & Seppälä, I. J. Activation of the alternative pathway of complement by monoclonal lambda light chains in membranoproliferative glomerulonephritis. J. Exp. Med. 175, 939–950 (1992).
Hauer, J. J. et al. Complement biomarkers as predictors of disease outcome in C3 glomerulopathy. Mol. Immunol. 102, 160 (2018).
Ravindran, A., Fervenza, F. C., Smith, R. J. H. & Sethi, S. C3 glomerulopathy associated with monoclonal Ig is a distinct subtype. Kidney Int. 94, 178–186 (2018).
Ravindran, A., Fervenza, F. C., Smith, R. J. H., De Vriese, A. S. & Sethi, S. C3 glomerulopathy: ten years’ experience at Mayo Clinic. Mayo Clin. Proc. 93, 991–1008 (2018).
Nasr, S. H. et al. Dense deposit disease: clinicopathologic study of 32 pediatric and adult patients. Clin. J. Am. Soc. Nephrol. 4, 22–32 (2009).
Servais, A., Noël, L. H., Frémeaux-Bacchi, V. & Lesavre, P. C3 glomerulopathy. Contrib. Nephrol. 181, 185–193 (2013).
Servais, A. et al. Primary glomerulonephritis with isolated C3 deposits: a new entity which shares common genetic risk factors with haemolytic uraemic syndrome. J. Med. Genet. 44, 193–199 (2007).
Chauvet, S. et al. Treatment of B cell disorder improves renal outcome of patients with monoclonal gammopathy-associated C3 glomerulopathy. Blood 129, 1437–1447 (2017).
Lloyd, I. E. et al. C3 glomerulopathy in adults: a distinct patient subset showing frequent association with monoclonal gammopathy and poor renal outcome. Clin. Kidney J. 9, 794–799 (2016).
Goodship, T. H. et al. Atypical hemolytic uremic syndrome and C3 glomerulopathy: conclusions from a “Kidney Disease: Improving Global Outcomes” (KDIGO) controversies conference. Kidney Int. 91, 539–551 (2017).
Maisch, N. M. & Pezzillo, K. K. HMG-CoA reductase inhibitors for the prevention of nephropathy. Ann. Pharmacother. 38, 342–345 (2004).
Nickolas, T. L., Radhakrishnan, J. & Appel, G. B. Hyperlipidemia and thrombotic complications in patients with membranous nephropathy. Semin. Nephrol. 23, 406–411 (2003).
Licht, C. et al. Successful plasma therapy for atypical hemolytic uremic syndrome caused by factor H deficiency owing to a novel mutation in the complement cofactor protein domain 15. Am. J. Kidney Dis. 45, 415–421 (2005).
Kurtz, K. A. & Schlueter, A. J. Management of membranoproliferative glomerulonephritis type II with plasmapheresis. J. Clin. Apher. 17, 135–137 (2002).
Avasare, R. S. et al. Mycophenolate mofetil in combination with steroids for treatment of C3 glomerulopathy: a case series. Clin. J. Am. Soc. Nephrol. 13, 406–413 (2018).
Nester, C. M. & Smith, R. J. Complement inhibition in C3 glomerulopathy. Semin. Immunol. 28, 241–249 (2016).
Bomback, A. S. et al. Eculizumab for dense deposit disease and C3 glomerulonephritis. Clin. J. Am. Soc. Nephrol. 7, 748–7560 (2012).
Le Quintrec, M. et al. Patterns of clinical response to eculizumab in patients with C3 glomerulopathy. Am. J. Kidney Dis. 72, 84–92 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03301467 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03369236 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03459443 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02682407 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03453619 (2018).
Lu, D. F., Moon, M., Lanning, L. D., McCarthy, A. M. & Smith, R. J. H. Clinical features and outcomes of 98 children and adults with dense deposit disease. Pediatr. Nephrol. 27, 773–781 (2012).
Burdett, L. et al. DRB-DQB1 diversity in the analysis of 4727 donors typed by SBT. Hum. Immunol. 64 (Suppl.), S6 (2003).
De Vriese, A. S. et al. Kidney disease caused by dysregulation of the complement alternative pathway: an etiologic approach. J. Am. Soc. Nephrol. 26, 2917–2929 (2015).
Acknowledgements
The authors’ research is supported in part by the US National Institutes of Health grant R01 DK110023 to R.J.H.S. and C.M.N.; by the Kidneeds Foundation, the National Research, Development and Innovation Fund of Hungary (grants K 109055 and K 125219) and the Institutional Excellence Program of the Ministry of Human Capacities of Hungary, all to M.J.; by the National Institute for Health Research Biomedical Research Centre based at Imperial College Healthcare NHS Trust and Imperial College London to M.C.P., who is a Wellcome Trust Senior Fellow in Clinical Science (WT082291MA); by the Spanish Ministerio de Economía y Competitividad/FEDER (grant SAF2015-66287-R), the Autonomous Region of Madrid (grant S2017/BMD-3673) and the Fundación Inocente Inocente to S.R.d.C.; and by the Deutsche Forschungsgemeinschaft (grant DFG CRC 1192) to P.F.Z.
Reviewer information
Nature Reviews Nephrology thanks C. Licht, F. Fervenza and the other anonymous reviewer(s) for their contributions to the peer review of this manuscript.
Author information
Authors and Affiliations
Contributions
R.J.H.S. researched data for the article, made substantial contributions to discussions of the article content, wrote the initial draft of the manuscript and participated in review or editing of the manuscript before submission. G.B.A., C.M.N., D.K., F.F. and G.R. had primary responsibility for the section on treatment; A.M.B., M.J. and J.V.d.V. had primary responsibility for the section on the glomerular microenvironment; J.D.L., V.F.-B., M.N., S.R.d.C., M.C.P. and P.F.Z. had primary responsibility for the sections on complement, genetics and autoantibodies; S.S., H.T.C. and V.D.D. had primary responsibility for the section on pathology. All authors reviewed, edited and approved all drafts of this paper.
Corresponding author
Ethics declarations
Competing interests
R.J.H.S. declares that he is Director of the Molecular Otolaryngology and Renal Research Laboratories (which provides genetic and functional testing for complement-mediated renal diseases). G.A. declares that he acts as a consultant for Achillion, Alexion, Chemocentryx and Omeros and has received research grants from Achillion and Chemocentryx. H.T.C. declares that he acts as a consultant for Alexion and Achillion. J.L. declares that he is the founder of Amyndas Pharmaceuticals, is named as an inventor on patents or patent applications describing the therapeutic use of complement inhibitors (some of which are being developed by Amyndas Pharmaceuticals) and is the inventor of the compstatin analogue licensed to Apellis Pharmaceuticals termed 4(1MeW)7 W (also known as POT4 and APL1) and pegylated derivatives such as APL2. M.N. declares that he has received honoraria for lecturing and participation in advisory boards from Alexion Pharmaceuticals and has received research grants from Chemocentryx and Omeros. M.C.P. declares that he has received research grants from Achillion, Alexion and Ra Pharma and has acted as a consultant for Achillion, Alexion, Chemocentryx and Ra Pharma. G.R. declares that he acts as a consultant for Alnylam, Boehringer Ingelheim, Handok, Hoffmann–La Roche and Janssen Research and Development (he has not accepted any personal remuneration from Alnylam or Hoffmann–La Roche; this compensation is used to support his research and educational activities). C.M.N. declares that he is Associate Director of the Molecular Otolaryngology and Renal Research Laboratories. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Smith, R.J.H., Appel, G.B., Blom, A.M. et al. C3 glomerulopathy — understanding a rare complement-driven renal disease. Nat Rev Nephrol 15, 129–143 (2019). https://doi.org/10.1038/s41581-018-0107-2
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-018-0107-2
This article is cited by
-
A guide to complement biology, pathology and therapeutic opportunity
Nature Reviews Immunology (2024)
-
Pioglitazone, a PPAR-y agonist, as one of the new therapeutic candidates for C3 glomerulopathy
Pediatric Nephrology (2024)
-
Complement gene mutations in children with C3 glomerulopathy: do they affect the response to mycophenolate mofetil?
Pediatric Nephrology (2024)
-
Pembrolizumab induced-C3 glomerulonephritis and RBC cast nephropathy: a case report
BMC Nephrology (2023)
-
Coexistence of cryoglobulinemia and ANCA-associated vasculitis in a chronic brucellosis patient -a case report and literature review
BMC Infectious Diseases (2023)