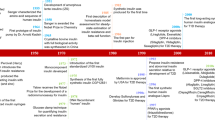
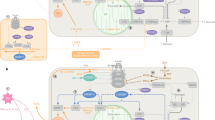
Abstract
Insulin resistance, defined as a defect in insulin-mediated control of glucose metabolism in tissues — prominently in muscle, fat and liver — is one of the earliest manifestations of a constellation of human diseases that includes type 2 diabetes and cardiovascular disease. These diseases are typically associated with intertwined metabolic abnormalities, including obesity, hyperinsulinaemia, hyperglycaemia and hyperlipidaemia. Insulin resistance is caused by a combination of genetic and environmental factors. Recent genetic and biochemical studies suggest a key role for adipose tissue in the development of insulin resistance, potentially by releasing lipids and other circulating factors that promote insulin resistance in other organs. These extracellular factors perturb the intracellular concentration of a range of intermediates, including ceramide and other lipids, leading to defects in responsiveness of cells to insulin. Such intermediates may cause insulin resistance by inhibiting one or more of the proximal components in the signalling cascade downstream of insulin (insulin receptor, insulin receptor substrate (IRS) proteins or AKT). However, there is now evidence to support the view that insulin resistance is a heterogeneous disorder that may variably arise in a range of metabolic tissues and that the mechanism for this effect likely involves a unified insulin resistance pathway that affects a distal step in the insulin action pathway that is more closely linked to the terminal biological response. Identifying these targets is of major importance, as it will reveal potential new targets for treatments of diseases associated with insulin resistance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Root, H. F. Insulin resistance and bronze diabetes. N. Engl. J. Med. 201, 201–206 (1929).
Himsworth, H. P. Diabetes mellitus: its differentiation into insulin-sensitive and insulin-insensitive types. 1936. Int. J. Epidemiol. 42, 1594–1598 (2013).
Petersen, M. C. & Shulman, G. I. Mechanisms of insulin action and insulin resistance. Physiol. Rev. 98, 2133–2223 (2018).
Barber, T. M., Kyrou, I., Randeva, H. S. & Weickert, M. O. Mechanisms of insulin resistance at the crossroad of obesity with associated metabolic abnormalities and cognitive dysfunction. Int. J. Mol. Sci. 22, 546 (2021).
Philipson, L. H. Harnessing heterogeneity in type 2 diabetes mellitus. Nat. Rev. Endocrinol. 16, 79–80 (2020).
Ahlqvist, E. et al. Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol. 6, 361–369 (2018).
Erion, K. A. & Corkey, B. E. Hyperinsulinemia: a cause of obesity? Curr. Obes. Rep. 6, 178–186 (2017).
Clemmensen, C. et al. Gut-brain cross-talk in metabolic control. Cell 168, 758–774 (2017).
Rizza, R. A., Mandarino, L. J., Genest, J., Baker, B. A. & Gerich, J. E. Production of insulin resistance by hyperinsulinaemia in man. Diabetologia 28, 70–75 (1985).
Ferrannini, E. et al. Influence of ethnicity and familial diabetes on glucose tolerance and insulin action: a physiological analysis. J. Clin. Endocrinol. Metab. 88, 3251–3257 (2003).
Kashyap, S. R. et al. Discordant effects of a chronic physiological increase in plasma FFA on insulin signaling in healthy subjects with or without a family history of type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 287, E537–E546 (2004).
Lillioja, S. et al. Insulin resistance and insulin secretory dysfunction as precursors of non-insulin-dependent diabetes mellitus. Prospective studies of Pima Indians. N. Engl. J. Med. 329, 1988–1992 (1993).
Vaag, A., Henriksen, J. E. & Beck-Nielsen, H. Decreased insulin activation of glycogen synthase in skeletal muscles in young nonobese Caucasian first-degree relatives of patients with non-insulin-dependent diabetes mellitus. J. Clin. Invest. 89, 782–788 (1992).
Hollenbeck, C. & Reaven, G. M. Variations in insulin-stimulated glucose uptake in healthy individuals with normal glucose tolerance. J. Clin. Endocrinol. Metab. 64, 1169–1173 (1987).
Ghaben, A. L. & Scherer, P. E. Adipogenesis and metabolic health. Nat. Rev. Mol. Cell Biol. 20, 242–258 (2019).
Jacob, S. et al. Association of increased intramyocellular lipid content with insulin resistance in lean nondiabetic offspring of type 2 diabetic subjects. Diabetes 48, 1113–1119 (1999).
Lim, K., Haider, A., Adams, C., Sleigh, A. & Savage, D. Lipodystrophy: a paradigm for understanding the consequences of ‘overloading’ adipose tissue. Physiol. Rev. https://doi.org/10.1152/physrev.00032.2020 (2020).
Blüher, M. Metabolically healthy obesity. Endocr. Rev. 41, 405–420 (2020).
Pontiroli, A. E., Alberetto, M., Capra, F. & Pozza, G. The glucose clamp technique for the study of patients with hypoglycemia: insulin resistance as a feature of insulinoma. J. Endocrinol. Invest. 13, 241–245 (1990).
Marbán, S. L. & Roth, J. Transgenic hyperinsulinemia: a mouse model of insulin resistance and glucose intolerance without obesity. in Lessons from Animal Diabetes VI: 75th Anniversary of the Insulin Discovery (ed. Shafrir, E.) 201–224 (Birkhäuser, 1996).
Gray, S. L., Donald, C., Jetha, A., Covey, S. D. & Kieffer, T. J. Hyperinsulinemia precedes insulin resistance in mice lacking pancreatic beta-cell leptin signaling. Endocrinology 151, 4178–4186 (2010).
Alemzadeh, R., Slonim, A. E., Zdanowicz, M. M. & Maturo, J. Modification of insulin resistance by diazoxide in obese Zucker rats. Endocrinology 133, 705–712 (1993).
Pedersen, D. J. et al. A major role of insulin in promoting obesity-associated adipose tissue inflammation. Mol. Metab. 4, 507–518 (2015).
Templeman, N. M. et al. Reduced circulating insulin enhances insulin sensitivity in old mice and extends lifespan. Cell Rep. 20, 451–463 (2017).
Czech, M. P. Insulin action and resistance in obesity and type 2 diabetes. Nat. Med. 23, 804–814 (2017). Recent state-of-the-art review of IR, obesity and T2D.
Tan, S.-X. et al. Selective insulin resistance in adipocytes. J. Biol. Chem. 290, 11337–11348 (2015).
Tonks, K. T. et al. Impaired Akt phosphorylation in insulin-resistant human muscle is accompanied by selective and heterogeneous downstream defects. Diabetologia 56, 875–885 (2013).
Brown, M. S. & Goldstein, J. L. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab. 7, 95–96 (2008). Describes the initial formulation of the concept of selective IR, which has become increasingly accepted as critical to understanding T2D.
Hillgartner, F. B., Salati, L. M. & Goodridge, A. G. Physiological and molecular mechanisms involved in nutritional regulation of fatty acid synthesis. Physiol. Rev. 75, 47–76 (1995).
Hellerstein, M. K., Schwarz, J. M. & Neese, R. A. Regulation of hepatic de novo lipogenesis in humans. Annu. Rev. Nutr. 16, 523–557 (1996).
Titchenell, P. M. et al. Direct hepatocyte insulin signaling is required for lipogenesis but is dispensable for the suppression of glucose production. Cell Metab. 23, 1154–1166 (2016).
Hammarstedt, A., Gogg, S., Hedjazifar, S., Nerstedt, A. & Smith, U. Impaired adipogenesis and dysfunctional adipose tissue in human hypertrophic obesity. Physiol. Rev. 98, 1911–1941 (2018).
Kolterman, O. G., Insel, J., Saekow, M. & Olefsky, J. M. Mechanisms of insulin resistance in human obesity: evidence for receptor and postreceptor defects. J. Clin. Invest. 65, 1272–1284 (1980). One of the original articles resolving defects in insulin action into two separate components.
Jia, Q., Morgan-Bathke, M. E. & Jensen, M. D. Adipose tissue macrophage burden, systemic inflammation, and insulin resistance. Am. J. Physiol. Endocrinol. Metab. 319, E254–E264 (2020).
Turner, N. et al. Distinct patterns of tissue-specific lipid accumulation during the induction of insulin resistance in mice by high-fat feeding. Diabetologia 56, 1638–1648 (2013).
Fazakerley, D. J. et al. Mitochondrial CoQ deficiency is a common driver of mitochondrial oxidants and insulin resistance. eLife 7, e32111 (2018). Comprehensive evidence to show that the levels of CoQ in mitochondria are lower in IR and that supplementation with CoQ reverses IR.
Perry, R. J. et al. Hepatic acetyl CoA links adipose tissue inflammation to hepatic insulin resistance and type 2 diabetes. Cell 160, 745–758 (2015). A series of experiments supporting the idea of non-cell-autonomous regulation of hepatic glucose output through insulin’s regulation of lipolysis.
Rebrin, K., Steil, G. M., Mittelman, S. D. & Bergman, R. N. Causal linkage between insulin suppression of lipolysis and suppression of liver glucose output in dogs. J. Clin. Invest. 98, 741–749 (1996).
Minokoshi, Y., Kahn, C. R. & Kahn, B. B. Tissue-specific ablation of the GLUT4 glucose transporter or the insulin receptor challenges assumptions about insulin action and glucose homeostasis. J. Biol. Chem. 278, 33609–33612 (2003).
Kim, J. K. et al. Glucose toxicity and the development of diabetes in mice with muscle-specific inactivation of GLUT4. J. Clin. Invest. 108, 153–160 (2001).
Gancheva, S., Jelenik, T., Álvarez-Hernández, E. & Roden, M. Interorgan metabolic crosstalk in human insulin resistance. Physiol. Rev. 98, 1371–1415 (2018).
Severinsen, M. C. K. & Pedersen, B. K. Muscle-organ crosstalk: the emerging roles of myokines. Endocr. Rev. 41, 594–609 (2020).
Burchfield, J. G. et al. High dietary fat and sucrose results in an extensive and time-dependent deterioration in health of multiple physiological systems in mice. J. Biol. Chem. 293, 5731–5745 (2018).
Hoehn, K. L. et al. IRS1-independent defects define major nodes of insulin resistance. Cell Metab. 7, 421–433 (2008). An important study challenging the view that IRS1 is the critical biochemical site for IR.
Copps, K. D. & White, M. F. Regulation of insulin sensitivity by serine/threonine phosphorylation of insulin receptor substrate proteins IRS1 and IRS2. Diabetologia 55, 2565–2582 (2012).
Kahn, C. R. et al. The syndromes of insulin resistance and acanthosis nigricans. Insulin-receptor disorders in man. N. Engl. J. Med. 294, 739–745 (1976).
Kono, T. & Barham, F. W. The relationship between the insulin-binding capacity of fat cells and the cellular response to insulin. Studies with intact and trypsin-treated fat cells. J. Biol. Chem. 246, 6210–6216 (1971). One of the first studies to highlight the spareness in insulin receptors in adipocytes giving rise to the notion that defects in insulin receptors are unlikely to cause IR in humans.
Kahn, C. R. Insulin resistance, insulin insensitivity, and insulin unresponsiveness: a necessary distinction. Metabolism 27, 1893–1902 (1978).
Olefsky, J. M., Kolterman, O. G. & Scarlett, J. A. Insulin action and resistance in obesity and noninsulin-dependent type II diabetes mellitus. Am. J. Physiol. 243, E15–E30 (1982).
Camps, M. et al. Evidence for the lack of spare high-affinity insulin receptors in skeletal muscle. Biochem. J. 285, 993–999 (1992).
Gumà, A. et al. Effect of benzyl succinate on insulin receptor function and insulin action in skeletal muscle: further evidence for a lack of spare high-affinity insulin receptors. Mol. Cell. Endocrinol. 91, 29–33 (1993).
Fehlmann, M., Morin, O., Kitabgi, P. & Freychet, P. Insulin and glucagon receptors of isolated rat hepatocytes: comparison between hormone binding and amino acid transport stimulation. Endocrinology 109, 253–261 (1981).
Merry, T. L. et al. Impairment of insulin signalling in peripheral tissue fails to extend murine lifespan. Aging Cell 16, 761–772 (2017).
Czech, M. P. Cellular basis of insulin insensitivity in large rat adipocytes. J. Clin. Invest. 57, 1523–1532 (1976).
Melvin, A., O’Rahilly, S. & Savage, D. B. Genetic syndromes of severe insulin resistance. Curr. Opin. Genet. Dev. 50, 60–67 (2018).
Crouthamel, M.-C. et al. Mechanism and management of AKT inhibitor-induced hyperglycemia. Clin. Cancer Res. 15, 217–225 (2009).
Nandi, A., Kitamura, Y., Kahn, C. R. & Accili, D. Mouse models of insulin resistance. Physiol. Rev. 84, 623–647 (2004).
Jaiswal, N. et al. The role of skeletal muscle Akt in the regulation of muscle mass and glucose homeostasis. Mol. Metab. 28, 1–13 (2019).
Lu, M. et al. Insulin regulates liver metabolism in vivo in the absence of hepatic Akt and Foxo1. Nat. Med. 18, 388–395 (2012). Mouse genetic experiments demonstrating non-cell-autonomous regulation of hepatic metabolism.
Tan, S.-X. et al. Amplification and demultiplexing in insulin-regulated Akt protein kinase pathway in adipocytes. J. Biol. Chem. 287, 6128–6138 (2012).
Ng, Y. et al. Cluster analysis of insulin action in adipocytes reveals a key role for Akt at the plasma membrane. J. Biol. Chem. 285, 2245–2257 (2010).
Larance, M. et al. Global phosphoproteomics identifies a major role for AKT and 14-3-3 in regulating EDC3. Mol. Cell. Proteomics 9, 682–694 (2010).
Trefely, S. et al. Kinome screen identifies PFKFB3 and glucose metabolism as important regulators of the insulin/insulin-like growth factor (IGF)-1 signaling pathway. J. Biol. Chem. 290, 25834–25846 (2015).
Ricort, J. M., Tanti, J. F., Van Obberghen, E. & Le Marchand-Brustel, Y. Alterations in insulin signalling pathway induced by prolonged insulin treatment of 3T3-L1 adipocytes. Diabetologia 38, 1148–1156 (1995).
Kurowski, T. G. et al. Hyperglycemia inhibits insulin activation of Akt/protein kinase B but not phosphatidylinositol 3-kinase in rat skeletal muscle. Diabetes 48, 658–663 (1999).
Oku, A. et al. Inhibitory effect of hyperglycemia on insulin-induced Akt/protein kinase B activation in skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 280, E816–E824 (2001).
Ronnett, G. V., Knutson, V. P. & Lane, M. D. Insulin-induced down-regulation of insulin receptors in 3T3-L1 adipocytes. Altered rate of receptor inactivation. J. Biol. Chem. 257, 4285–4291 (1982).
Draznin, B. Molecular mechanisms of insulin resistance: serine phosphorylation of insulin receptor substrate-1 and increased expression of p85alpha: the two sides of a coin. Diabetes 55, 2392–2397 (2006).
Copps, K. D. et al. Irs1 serine 307 promotes insulin sensitivity in mice. Cell Metab. 11, 84–92 (2010).
Brandon, A. E. et al. Protein kinase C epsilon deletion in adipose tissue, but not in liver, improves glucose tolerance. Cell Metab. 29, 183–191.e7 (2019).
Deblon, N. et al. Chronic mTOR inhibition by rapamycin induces muscle insulin resistance despite weight loss in rats. Br. J. Pharmacol. 165, 2325–2340 (2012).
Lamming, D. W. et al. Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science 335, 1638–1643 (2012).
Friedman, J. E., Caro, J. F., Pories, W. J., Azevedo, J. L. Jr & Dohm, G. L. Glucose metabolism in incubated human muscle: effect of obesity and non-insulin-dependent diabetes mellitus. Metabolism 43, 1047–1054 (1994).
Shulman, G. I. et al. Quantitation of muscle glycogen synthesis in normal subjects and subjects with non-insulin-dependent diabetes by 13C nuclear magnetic resonance spectroscopy. N. Engl. J. Med. 322, 223–228 (1990). Demonstration of muscle IR in humans with T2D.
Baron, A. D., Laakso, M., Brechtel, G. & Edelman, S. V. Reduced capacity and affinity of skeletal muscle for insulin-mediated glucose uptake in noninsulin-dependent diabetic subjects. Effects of insulin therapy. J. Clin. Invest. 87, 1186–1194 (1991).
Friedman, J. E. et al. Restoration of insulin responsiveness in skeletal muscle of morbidly obese patients after weight loss. Effect on muscle glucose transport and glucose transporter GLUT4. J. Clin. Invest. 89, 701–705 (1992).
Meyer, M. M., Levin, K., Grimmsmann, T., Beck-Nielsen, H. & Klein, H. H. Insulin signalling in skeletal muscle of subjects with or without type II-diabetes and first degree relatives of patients with the disease. Diabetologia 45, 813–822 (2002).
Kim, Y. B., Nikoulina, S. E., Ciaraldi, T. P., Henry, R. R. & Kahn, B. B. Normal insulin-dependent activation of Akt/protein kinase B, with diminished activation of phosphoinositide 3-kinase, in muscle in type 2 diabetes. J. Clin. Invest. 104, 733–741 (1999).
Ramos, P. A. et al. Insulin-stimulated muscle glucose uptake and insulin signaling in lean and obese humans. J. Clin. Endocrinol. Metab. 106, e1631–e1646 (2020).
Vind, B. F. et al. Impaired insulin-induced site-specific phosphorylation of TBC1 domain family, member 4 (TBC1D4) in skeletal muscle of type 2 diabetes patients is restored by endurance exercise-training. Diabetologia 54, 157–167 (2011).
Caro, J. F. et al. Insulin receptor kinase in human skeletal muscle from obese subjects with and without noninsulin dependent diabetes. J. Clin. Invest. 79, 1330–1337 (1987).
Garvey, W. T., Maianu, L., Zhu, J. H., Hancock, J. A. & Golichowski, A. M. Multiple defects in the adipocyte glucose transport system cause cellular insulin resistance in gestational diabetes. Heterogeneity in the number and a novel abnormality in subcellular localization of GLUT4 glucose transporters. Diabetes 42, 1773–1785 (1993).
Ryder, J. W. et al. Use of a novel impermeable biotinylated photolabeling reagent to assess insulin- and hypoxia-stimulated cell surface GLUT4 content in skeletal muscle from type 2 diabetic patients. Diabetes 49, 647–654 (2000).
Garvey, W. T. et al. Evidence for defects in the trafficking and translocation of GLUT4 glucose transporters in skeletal muscle as a cause of human insulin resistance. J. Clin. Invest. 101, 2377–2386 (1998).
Gumà, A., Zierath, J. R., Wallberg-Henriksson, H. & Klip, A. Insulin induces translocation of GLUT-4 glucose transporters in human skeletal muscle. Am. J. Physiol. 268, E613–E622 (1995).
Dills, W. L. Jr, McDonough, G. M. & Kingsley, P. B. Glucose-stimulated protein synthesis in rat testis slices: substrate specificity and effects of insulin and substrate analogs. Biol. Reprod. 25, 466–474 (1981).
Chlouverakis, C. The action of glucose on lipolysis. Metabolism 16, 469–472 (1967).
Li, J., Houseknecht, K. L., Stenbit, A. E., Katz, E. B. & Charron, M. J. Reduced glucose uptake precedes insulin signaling defects in adipocytes from heterozygous GLUT4 knockout mice. FASEB J. 14, 1117–1125 (2000).
Shepherd, P. R. & Kahn, B. B. Glucose transporters and insulin action — implications for insulin resistance and diabetes mellitus. N. Engl. J. Med. 341, 248–257 (1999).
Etgen, G. J. Jr et al. Exercise training reverses insulin resistance in muscle by enhanced recruitment of GLUT-4 to the cell surface. Am. J. Physiol. 272, E864–E869 (1997). Study presenting evidence that the defect in glucose transport in IR is specific to insulin action and does not apply to exercise or contraction.
Klip, A. et al. Recruitment of GLUT-4 glucose transporters by insulin in diabetic rat skeletal muscle. Biochem. Biophys. Res. Commun. 172, 728–736 (1990).
Kennedy, J. W. et al. Acute exercise induces GLUT4 translocation in skeletal muscle of normal human subjects and subjects with type 2 diabetes. Diabetes 48, 1192–1197 (1999).
Ploug, T., van Deurs, B., Ai, H., Cushman, S. W. & Ralston, E. Analysis of GLUT4 distribution in whole skeletal muscle fibers: identification of distinct storage compartments that are recruited by insulin and muscle contractions. J. Cell Biol. 142, 1429–1446 (1998). Study demonstrating that insulin and contraction stimulate recruitment of GLUT4 from distinct intracellular pools.
Stöckli, J. et al. The RabGAP TBC1D1 plays a central role in exercise-regulated glucose metabolism in skeletal muscle. Diabetes 64, 1914–1922 (2015).
Humphrey, S. J. et al. Dynamic adipocyte phosphoproteome reveals that Akt directly regulates mTORC2. Cell Metab. 17, 1009–1020 (2013). Important study demonstrating the complexity of protein phosphorylation changes in response to insulin.
Kjøbsted, R. et al. Intact regulation of the AMPK signaling network in response to exercise and insulin in skeletal muscle of male patients with type 2 diabetes: illumination of AMPK activation in recovery from exercise. Diabetes 65, 1219–1230 (2016).
Eguez, L. et al. Full intracellular retention of GLUT4 requires AS160 Rab GTPase activating protein. Cell Metab. 2, 263–272 (2005).
Batista, T. M. et al. A cell-autonomous signature of dysregulated protein phosphorylation underlies muscle insulin resistance in type 2 diabetes. Cell Metab. 32, 844–859.e5 (2020).
Boden, G. et al. Excessive caloric intake acutely causes oxidative stress, GLUT4 carbonylation, and insulin resistance in healthy men. Sci. Transl. Med. 7, 304re7 (2015).
Hauck, A. K., Huang, Y., Hertzel, A. V. & Bernlohr, D. A. Adipose oxidative stress and protein carbonylation. J. Biol. Chem. 294, 1083–1088 (2019).
Hammarstedt, A., Graham, T. E. & Kahn, B. B. Adipose tissue dysregulation and reduced insulin sensitivity in non-obese individuals with enlarged abdominal adipose cells. Diabetol. Metab. Syndr. 4, 42 (2012).
Weyer, C., Foley, J. E., Bogardus, C., Tataranni, P. A. & Pratley, R. E. Enlarged subcutaneous abdominal adipocyte size, but not obesity itself, predicts type II diabetes independent of insulin resistance. Diabetologia 43, 1498–1506 (2000).
Abel, E. D. et al. Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature 409, 729–733 (2001). One of the first studies demonstrating that IR in adipocytes can propriate to other peripheral organs.
Vazirani, R. P. et al. Disruption of adipose Rab10-dependent insulin signaling causes hepatic insulin resistance. Diabetes 65, 1577–1589 (2016).
Poulsen, P. et al. Heritability of insulin secretion, peripheral and hepatic insulin action, and intracellular glucose partitioning in young and old Danish twins. Diabetes 54, 275–283 (2005).
Flannick, J. et al. Exome sequencing of 20,791 cases of type 2 diabetes and 24,440 controls. Nature 570, 71–76 (2019).
Dimas, A. S. et al. Impact of type 2 diabetes susceptibility variants on quantitative glycemic traits reveals mechanistic heterogeneity. Diabetes 63, 2158–2171 (2014).
Lotta, L. A. et al. Integrative genomic analysis implicates limited peripheral adipose storage capacity in the pathogenesis of human insulin resistance. Nat. Genet. 49, 17–26 (2017). An important study identifying 53 genetic loci associated with measures of IR in humans, highlighting a major role for adipogenesis.
Levin, K., Hother-Nielsen, O., Henriksen, J. E. & Beck-Nielsen, H. Effects of troglitazone in young first-degree relatives of patients with type 2 diabetes. Diabetes Care 27, 148–154 (2004).
Chaurasia, B. & Summers, S. A. Ceramides in metabolism: key lipotoxic players. Annu. Rev. Physiol. 83, 303–330 (2021).
Boden, G. Free fatty acids (FFA), a link between obesity and insulin resistance. Front. Biosci. 3, d169–d175 (1998).
Pereira, S. et al. Resveratrol prevents insulin resistance caused by short-term elevation of free fatty acids in vivo. Appl. Physiol. Nutr. Metab. 40, 1129–1136 (2015).
Felber, J. P. & Vannotti, A. Effects of fat infusion on glucose tolerance and insulin plasma levels. Med. Exp. Int. J. Exp. Med. 10, 153–156 (1964).
Wang, L. et al. Adipocyte Gi signaling is essential for maintaining whole-body glucose homeostasis and insulin sensitivity. Nat. Commun. 11, 2995 (2020).
Karpe, F., Dickmann, J. R. & Frayn, K. N. Fatty acids, obesity, and insulin resistance: time for a reevaluation. Diabetes 60, 2441–2449 (2011).
Perseghin, G., Ghosh, S., Gerow, K. & Shulman, G. I. Metabolic defects in lean nondiabetic offspring of NIDDM parents: a cross-sectional study. Diabetes 46, 1001–1009 (1997).
McQuaid, S. E. et al. Downregulation of adipose tissue fatty acid trafficking in obesity: a driver for ectopic fat deposition? Diabetes 60, 47–55 (2011).
Jeffery, E., Church, C. D., Holtrup, B., Colman, L. & Rodeheffer, M. S. Rapid depot-specific activation of adipocyte precursor cells at the onset of obesity. Nat. Cell Biol. 17, 376–385 (2015).
Jeffery, E. et al. The adipose tissue microenvironment regulates depot-specific adipogenesis in obesity. Cell Metab. 24, 142–150 (2016).
Arner, P., Arner, E., Hammarstedt, A. & Smith, U. Genetic predisposition for type 2 diabetes, but not for overweight/obesity, is associated with a restricted adipogenesis. PLoS ONE 6, e18284 (2011).
Ye, J. Regulation of PPARgamma function by TNF-alpha. Biochem. Biophys. Res. Commun. 374, 405–408 (2008).
Shao, M. et al. De novo adipocyte differentiation from Pdgfrβ preadipocytes protects against pathologic visceral adipose expansion in obesity. Nat. Commun. 9, 890 (2018).
Kim, J.-Y. et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J. Clin. Invest. 117, 2621–2637 (2007).
Shepherd, P. R. et al. Adipose cell hyperplasia and enhanced glucose disposal in transgenic mice overexpressing GLUT4 selectively in adipose tissue. J. Biol. Chem. 268, 22243–22246 (1993).
Carobbio, S., Pellegrinelli, V. & Vidal-Puig, A. Adipose tissue function and expandability as determinants of lipotoxicity and the metabolic syndrome. Adv. Exp. Med. Biol. 960, 161–196 (2017).
Czech, M. P. Mechanisms of insulin resistance related to white, beige, and brown adipocytes. Mol. Metab. 34, 27–42 (2020).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 425–432 (1994).
Kahn, C. R., Wang, G. & Lee, K. Y. Altered adipose tissue and adipocyte function in the pathogenesis of metabolic syndrome. J. Clin. Invest. 129, 3990–4000 (2019).
Friedman, J. M. Leptin and the endocrine control of energy balance. Nat. Metab. 1, 754–764 (2019).
Blüher, M. et al. Circulating adiponectin and expression of adiponectin receptors in human skeletal muscle: associations with metabolic parameters and insulin resistance and regulation by physical training. J. Clin. Endocrinol. Metab. 91, 2310–2316 (2006).
Meyer, L. K., Ciaraldi, T. P., Henry, R. R., Wittgrove, A. C. & Phillips, S. A. Adipose tissue depot and cell size dependency of adiponectin synthesis and secretion in human obesity. Adipocyte 2, 217–226 (2013).
Yamauchi, T. et al. Targeted disruption of AdipoR1 and AdipoR2 causes abrogation of adiponectin binding and metabolic actions. Nat. Med. 13, 332–339 (2007).
Chen, Z. et al. Effects of adiponectin on T2DM and glucose homeostasis: a mendelian randomization study. Diabetes Metab. Syndr. Obes. 13, 1771–1784 (2020).
Ortega Moreno, L. et al. Evidence of a causal relationship between high serum adiponectin levels and increased cardiovascular mortality rate in patients with type 2 diabetes. Cardiovasc. Diabetol. 15, 17 (2016).
Yore, M. M. et al. Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects. Cell 159, 318–332 (2014).
Agbu, P. & Carthew, R. W. MicroRNA-mediated regulation of glucose and lipid metabolism. Nat. Rev. Mol. Cell Biol. 22, 425–438 (2021).
Crewe, C., An, Y. A. & Scherer, P. E. The ominous triad of adipose tissue dysfunction: inflammation, fibrosis, and impaired angiogenesis. J. Clin. Invest. 127, 74–82 (2017).
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444, 860–867 (2006).
Kammoun, H. L., Kraakman, M. J. & Febbraio, M. A. Adipose tissue inflammation in glucose metabolism. Rev. Endocr. Metab. Disord. 15, 31–44 (2014).
McNelis, J. C. & Olefsky, J. M. Macrophages, immunity, and metabolic disease. Immunity 41, 36–48 (2014).
Mathis, D. Immunological goings-on in visceral adipose tissue. Cell Metab. 17, 851–859 (2013).
Hotamisligil, G. S. Inflammation, metaflammation and immunometabolic disorders. Nature 542, 177–185 (2017).
Wu, H. & Ballantyne, C. M. Metabolic inflammation and insulin resistance in obesity. Circ. Res. 126, 1549–1564 (2020).
Krogh-Madsen, R., Plomgaard, P., Møller, K., Mittendorfer, B. & Pedersen, B. K. Influence of TNF-alpha and IL-6 infusions on insulin sensitivity and expression of IL-18 in humans. Am. J. Physiol. Endocrinol. Metab. 291, E108–E114 (2006).
Kriketos, A. D. et al. Inflammation, insulin resistance, and adiposity: a study of first-degree relatives of type 2 diabetic subjects. Diabetes Care 27, 2033–2040 (2004).
Shimobayashi, M. et al. Insulin resistance causes inflammation in adipose tissue. J. Clin. Invest. 128, 1538–1550 (2018). A study that places IR ‘upstream’ of inflammation rather than the more conventional other way around.
Lee, Y. S. et al. Inflammation is necessary for long-term but not short-term high-fat diet-induced insulin resistance. Diabetes 60, 2474–2483 (2011).
Rafiq, S. et al. Gene variants influencing measures of inflammation or predisposing to autoimmune and inflammatory diseases are not associated with the risk of type 2 diabetes. Diabetologia 51, 2205–2213 (2008).
Everett, B. M. et al. Anti-inflammatory therapy with canakinumab for the prevention and management of diabetes. J. Am. Coll. Cardiol. 71, 2392–2401 (2018).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Smith, U., Li, Q., Rydén, M. & Spalding, K. L. Cellular senescence and its role in white adipose tissue. Int. J. Obes. 45, 934–943 (2021).
Wernstedt Asterholm, I. et al. Adipocyte inflammation is essential for healthy adipose tissue expansion and remodeling. Cell Metab. 20, 103–118 (2014).
Cox, A. R., Chernis, N., Masschelin, P. M. & Hartig, S. M. Immune cells gate white adipose tissue expansion. Endocrinology 160, 1645–1658 (2019).
Chaurasia, B. & Summers, S. A. Ceramides - lipotoxic inducers of metabolic disorders. Trends Endocrinol. Metab. 26, 538–550 (2015).
Luukkonen, P. K. et al. Hepatic ceramides dissociate steatosis and insulin resistance in patients with non-alcoholic fatty liver disease. J. Hepatol. 64, 1167–1175 (2016).
Kolak, M. et al. Adipose tissue inflammation and increased ceramide content characterize subjects with high liver fat content independent of obesity. Diabetes 56, 1960–1968 (2007).
Coen, P. M. et al. Reduced skeletal muscle oxidative capacity and elevated ceramide but not diacylglycerol content in severe obesity. Obesity 21, 2362–2371 (2013).
Turpin, S. M. et al. Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab. 20, 678–686 (2014). A study that together with Raichur et al. (2014) and Chaurasia et al. (2019) provides critical evidence implicating ceramide and ceramide metabolism in IR in mice.
Stöckli, J. et al. Metabolomic analysis of insulin resistance across different mouse strains and diets. J. Biol. Chem. 292, 19135–19145 (2017).
Raichur, S. et al. CerS2 haploinsufficiency inhibits β-oxidation and confers susceptibility to diet-induced steatohepatitis and insulin resistance. Cell Metab. 20, 687–695 (2014).
Chaurasia, B. et al. Targeting a ceramide double bond improves insulin resistance and hepatic steatosis. Science 365, 386–392 (2019).
Chavez, J. A. et al. A role for ceramide, but not diacylglycerol, in the antagonism of insulin signal transduction by saturated fatty acids. J. Biol. Chem. 278, 10297–10303 (2003).
Villa, N. Y. et al. Sphingolipids function as downstream effectors of a fungal PAQR. Mol. Pharmacol. 75, 866–875 (2009).
Mente, A. et al. Causal relationship between adiponectin and metabolic traits: a Mendelian randomization study in a multiethnic population. PLoS ONE 8, e66808 (2013).
Nawrocki, A. R. et al. Mice lacking adiponectin show decreased hepatic insulin sensitivity and reduced responsiveness to peroxisome proliferator-activated receptor gamma agonists. J. Biol. Chem. 281, 2654–2660 (2006).
Cazzolli, R., Carpenter, L., Biden, T. J. & Schmitz-Peiffer, C. A role for protein phosphatase 2A-like activity, but not atypical protein kinase Czeta, in the inhibition of protein kinase B/Akt and glycogen synthesis by palmitate. Diabetes 50, 2210–2218 (2001).
Fox, T. E. et al. Ceramide recruits and activates protein kinase C zeta (PKC zeta) within structured membrane microdomains. J. Biol. Chem. 282, 12450–12457 (2007).
Chaurasia, B., Talbot, C. L. & Summers, S. A. Adipocyte ceramides-the nexus of inflammation and metabolic disease. Front. Immunol. 11, 576347 (2020).
Lyu, K. et al. A membrane-bound diacylglycerol species induces PKCε-mediated hepatic insulin resistance. Cell Metab. 32, 654–664.e5 (2020).
Lyu, K. et al. Short-term overnutrition induces white adipose tissue insulin resistance through sn-1,2-diacylglycerol/PKCε/insulin receptor Thr1160 phosphorylation. JCI Insight 6, e139946 (2021).
Gassaway, B. M. et al. PKCε contributes to lipid-induced insulin resistance through cross talk with p70S6K and through previously unknown regulators of insulin signaling. Proc. Natl Acad. Sci. USA 115, E8996–E9005 (2018).
Perreault, L. et al. Intracellular localization of diacylglycerols and sphingolipids influences insulin sensitivity and mitochondrial function in human skeletal muscle. JCI Insight 3, e96805 (2018).
Gonzalez-Franquesa, A. & Patti, M.-E. Insulin resistance and mitochondrial dysfunction. Adv. Exp. Med. Biol. 982, 465–520 (2017).
Sangwung, P., Petersen, K. F., Shulman, G. I. & Knowles, J. W. Mitochondrial dysfunction, insulin resistance, and potential genetic implications. Endocrinology 161, bqaa017 (2020).
Houstis, N., Rosen, E. D. & Lander, E. S. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature 440, 944–948 (2006).
Fisher-Wellman, K. H. & Neufer, P. D. Linking mitochondrial bioenergetics to insulin resistance via redox biology. Trends Endocrinol. Metab. 23, 142–153 (2012). An excellent review of the role of mitochondrial bioenergetics and ROS in IR.
Hoehn, K. L. et al. Insulin resistance is a cellular antioxidant defense mechanism. Proc. Natl Acad. Sci. USA 106, 17787–17792 (2009).
Anderson, E. J. et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J. Clin. Invest. 119, 573–581 (2009). One of the more compelling studies implicating mitochondrial H2O2 as a possible cause of IR.
Ingram, K. H. et al. Skeletal muscle lipid peroxidation and insulin resistance in humans. J. Clin. Endocrinol. Metab. 97, E1182–E1186 (2012).
Duplain, H. et al. Stimulation of peroxynitrite catalysis improves insulin sensitivity in high fat diet-fed mice. J. Physiol. 586, 4011–4016 (2008).
Fazakerley, D. J. et al. Mitochondrial oxidative stress causes insulin resistance without disrupting oxidative phosphorylation. J. Biol. Chem. 293, 7315–7328 (2018).
Boveris, A., Oshino, N. & Chance, B. The cellular production of hydrogen peroxide. Biochem. J. 128, 617–630 (1972).
Rees-Milton, K. J. et al. Statin use is associated with insulin resistance in participants of the Canadian Multicentre Osteoporosis Study. J. Endocr. Soc. 4, bvaa057 (2020).
Koves, T. R. et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 7, 45–56 (2008).
Di Paola, M., Cocco, T. & Lorusso, M. Ceramide interaction with the respiratory chain of heart mitochondria. Biochemistry 39, 6660–6668 (2000).
Smith, M. E. et al. Mitochondrial fission mediates ceramide-induced metabolic disruption in skeletal muscle. Biochem. J. 456, 427–439 (2013).
Novgorodov, S. A. et al. Novel pathway of ceramide production in mitochondria: thioesterase and neutral ceramidase produce ceramide from sphingosine and acyl-CoA. J. Biol. Chem. 286, 25352–25362 (2011).
von Haefen, C. et al. Ceramide induces mitochondrial activation and apoptosis via a Bax-dependent pathway in human carcinoma cells. Oncogene 21, 4009–4019 (2002).
Ye, R., Onodera, T. & Scherer, P. E. Lipotoxicity and cell maintenance in obesity and type 2 diabetes. J. Endocr. Soc. 3, 617–631 (2019).
Turpin, S. M. et al. Examination of ‘lipotoxicity’ in skeletal muscle of high-fat fed and ob/ob mice. J. Physiol. 587, 1593–1605 (2009).
Kim, Y.-R. et al. Hepatic triglyceride accumulation via endoplasmic reticulum stress-induced SREBP-1 activation is regulated by ceramide synthases. Exp. Mol. Med. 51, 1–16 (2019).
Boslem, E. et al. A lipidomic screen of palmitate-treated MIN6 β-cells links sphingolipid metabolites with endoplasmic reticulum (ER) stress and impaired protein trafficking. Biochem. J. 435, 267–276 (2011).
Flamment, M., Hajduch, E., Ferré, P. & Foufelle, F. New insights into ER stress-induced insulin resistance. Trends Endocrinol. Metab. 23, 381–390 (2012).
Sumitomo, M. et al. Protein kinase Cdelta amplifies ceramide formation via mitochondrial signaling in prostate cancer cells. J. Clin. Invest. 109, 827–836 (2002).
Lee, H.-Y. et al. Mitochondrial-targeted catalase protects against high-fat diet-induced muscle insulin resistance by decreasing intramuscular lipid accumulation. Diabetes 66, 2072–2081 (2017).
Riojas-Hernández, A. et al. Enhanced oxidative stress sensitizes the mitochondrial permeability transition pore to opening in heart from Zucker Fa/fa rats with type 2 diabetes. Life Sci. 141, 32–43 (2015).
Taddeo, E. P. et al. Opening of the mitochondrial permeability transition pore links mitochondrial dysfunction to insulin resistance in skeletal muscle. Mol. Metab. 3, 124–134 (2014).
Cho, J. et al. Mitochondrial ATP transporter depletion protects mice against liver steatosis and insulin resistance. Nat. Commun. 8, 14477 (2017).
O’Neill, B. T. et al. Differential role of insulin/IGF-1 receptor signaling in muscle growth and glucose homeostasis. Cell Rep. 11, 1220–1235 (2015).
Gastaldelli, A. et al. Effect of physiological hyperinsulinemia on gluconeogenesis in nondiabetic subjects and in type 2 diabetic patients. Diabetes 50, 1807–1812 (2001).
Reaven, G. M., Hollenbeck, C. B. & Chen, Y. D. Relationship between glucose tolerance, insulin secretion, and insulin action in non-obese individuals with varying degrees of glucose tolerance. Diabetologia 32, 52–55 (1989).
Firth, R., Bell, P. & Rizza, R. Insulin action in non-insulin-dependent diabetes mellitus: the relationship between hepatic and extrahepatic insulin resistance and obesity. Metabolism 36, 1091–1095 (1987).
Manning, B. D. & Toker, A. AKT/PKB signaling: navigating the network. Cell 169, 381–405 (2017).
James, D. E., Strube, M. & Mueckler, M. Molecular cloning and characterization of an insulin-regulatable glucose transporter. Nature 338, 83–87 (1989).
Birnbaum, M. J. Identification of a novel gene encoding an insulin-responsive glucose transporter protein. Cell 57, 305–315 (1989).
Bryant, N. J., Govers, R. & James, D. E. Regulated transport of the glucose transporter GLUT4. Nat. Rev. Mol. Cell Biol. 3, 267–277 (2002).
Yeh, J. I., Gulve, E. A., Rameh, L. & Birnbaum, M. J. The effects of wortmannin on rat skeletal muscle. Dissociation of signaling pathways for insulin- and contraction-activated hexose transport. J. Biol. Chem. 270, 2107–2111 (1995).
Sano, H. et al. Rab10, a target of the AS160 Rab GAP, is required for insulin-stimulated translocation of GLUT4 to the adipocyte plasma membrane. Cell Metab. 5, 293–303 (2007).
Stöckli, J., Fazakerley, D. J. & James, D. E. GLUT4 exocytosis. J. Cell Sci. 124, 4147–4159 (2011).
Kirwan, J. P., Solomon, T. P. J., Wojta, D. M., Staten, M. A. & Holloszy, J. O. Effects of 7 days of exercise training on insulin sensitivity and responsiveness in type 2 diabetes mellitus. Am. J. Physiol. Endocrinol. Metab. 297, E151–E156 (2009).
James, D. E., Kraegen, E. W. & Chisholm, D. J. Effects of exercise training on in vivo insulin action in individual tissues of the rat. J. Clin. Invest. 76, 657–666 (1985).
Koh, J.-H. et al. PPARβ is essential for maintaining normal levels of PGC-1α and mitochondria and for the increase in muscle mitochondria induced by exercise. Cell Metab. 25, 1176–1185.e5 (2017).
Alibegovic, A. C. et al. Impact of 9 days of bed rest on hepatic and peripheral insulin action, insulin secretion, and whole-body lipolysis in healthy young male offspring of patients with type 2 diabetes. Diabetes 58, 2749–2756 (2009).
Mikines, K. J., Richter, E. A., Dela, F. & Galbo, H. Seven days of bed rest decrease insulin action on glucose uptake in leg and whole body. J. Appl. Physiol. 70, 1245–1254 (1991).
Krogh-Madsen, R. et al. A 2-wk reduction of ambulatory activity attenuates peripheral insulin sensitivity. J. Appl. Physiol. 108, 1034–1040 (2010).
Grams, J. & Garvey, W. T. Weight loss and the prevention and treatment of type 2 diabetes using lifestyle therapy, pharmacotherapy, and bariatric surgery: mechanisms action. Curr. Obes. Rep. 4, 287–302 (2015).
Lean, M. E. et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet 391, 541–551 (2018).
Magkos, F. et al. Effects of moderate and subsequent progressive weight loss on metabolic function and adipose tissue biology in humans with obesity. Cell Metab. 23, 591–601 (2016).
Bray, G. A. & Bouchard, C. The biology of human overfeeding: a systematic review. Obes. Rev. 21, e13040 (2020).
Smith, G. I. et al. One day of mixed meal overfeeding reduces hepatic insulin sensitivity and increases VLDL particle but not VLDL-triglyceride secretion in overweight and obese men. J. Clin. Endocrinol. Metab. 98, 3454–3462 (2013).
McLaughlin, T. et al. Adipose cell size and regional fat deposition as predictors of metabolic response to overfeeding in insulin-resistant and insulin-sensitive humans. Diabetes 65, 1245–1254 (2016).
Ludwig, D. S., Willett, W. C., Volek, J. S. & Neuhouser, M. L. Dietary fat: from foe to friend? Science 362, 764–770 (2018).
Weickert, M. O. et al. Effects of supplemented isoenergetic diets differing in cereal fiber and protein content on insulin sensitivity in overweight humans. Am. J. Clin. Nutr. 94, 459–471 (2011).
Linn, T. et al. Effect of long-term dietary protein intake on glucose metabolism in humans. Diabetologia 43, 1257–1265 (2000).
Sluijs, I. et al. Dietary intake of total, animal, and vegetable protein and risk of type 2 diabetes in the European Prospective Investigation into Cancer and Nutrition (EPIC)-NL study. Diabetes Care 33, 43–48 (2010).
Rosenbaum, M. et al. Glucose and lipid homeostasis and inflammation in humans following an isocaloric ketogenic diet. Obesity 27, 971–981 (2019).
Roberts, M. N. et al. A ketogenic diet extends longevity and healthspan in adult mice. Cell Metab. 26, 539–546.e5 (2017).
Bisschop, P. H. et al. Dietary fat content alters insulin-mediated glucose metabolism in healthy men. Am. J. Clin. Nutr. 73, 554–559 (2001).
Lundsgaard, A.-M. et al. Mechanisms preserving insulin action during high dietary fat intake. Cell Metab. 29, 50–63.e4 (2019).
Sargrad, K. R., Homko, C., Mozzoli, M. & Boden, G. Effect of high protein vs high carbohydrate intake on insulin sensitivity, body weight, hemoglobin A1c, and blood pressure in patients with type 2 diabetes mellitus. J. Am. Diet. Assoc. 105, 573–580 (2005).
Seidelmann, S. B. et al. Dietary carbohydrate intake and mortality: a prospective cohort study and meta-analysis. Lancet Public. Health 3, e419–e428 (2018).
Randle, P. J. Fuel selection in animals. Biochem. Soc. Trans. 14, 799–806 (1986).
Moltke, I. et al. A common Greenlandic TBC1D4 variant confers muscle insulin resistance and type 2 diabetes. Nature 512, 190–193 (2014). Human genetic evidence supporting a role for the AKT target TBC1D4 in the regulation of muscle glucose transport.
Manousaki, D. et al. Toward precision medicine: TBC1D4 disruption is common among the inuit and leads to underdiagnosis of type 2 diabetes. Diabetes Care 39, 1889–1895 (2016).
Palla, L., Higgins, J. P. T., Wareham, N. J. & Sharp, S. J. Challenges in the use of literature-based meta-analysis to examine gene-environment interactions. Am. J. Epidemiol. 171, 1225–1232 (2010).
Martínez-González, M. A. et al. Benefits of the Mediterranean diet: insights from the PREDIMED study. Prog. Cardiovasc. Dis. 58, 50–60 (2015).
Kilpeläinen, T. O. et al. Physical activity attenuates the influence of FTO variants on obesity risk: a meta-analysis of 218,166 adults and 19,268 children. PLoS Med. 8, e1001116 (2011).
Sonestedt, E. et al. Fat and carbohydrate intake modify the association between genetic variation in the FTO genotype and obesity. Am. J. Clin. Nutr. 90, 1418–1425 (2009).
Heianza, Y. et al. Macronutrient intake-associated FGF21 genotype modifies effects of weight-loss diets on 2-year changes of central adiposity and body composition: the POUNDS lost trial. Diabetes Care 39, 1909–1914 (2016).
Hall, K. D. Challenges of human nutrition research. Science 367, 1298–1300 (2020).
Franz, M. J. et al. Academy of nutrition and dietetics nutrition practice guideline for type 1 and type 2 diabetes in adults: systematic review of evidence for medical nutrition therapy effectiveness and recommendations for integration into the nutrition care process. J. Acad. Nutr. Diet. 117, 1659–1679 (2017).
Powell, K. E. et al. The scientific foundation for the Physical Activity Guidelines for Americans, 2nd Edition. J. Phys. Act. Health 16, 1–11 (2018).
American Diabetes Association. 5. Facilitating behavior change and well-being to improve health outcomes: standards of medical care in diabetes-2020. Diabetes Care 43, S48–S65 (2020).
Pernicova, I. & Korbonits, M. Metformin — mode of action and clinical implications for diabetes and cancer. Nat. Rev. Endocrinol. 10, 143–156 (2014).
American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes — 2020. Diabetes Care 43, S98–S110 (2020).
Packer, M. Potentiation of insulin signaling contributes to heart failure in type 2 diabetes: a hypothesis supported by both mechanistic studies and clinical trials. JACC Basic Transl. Sci. 3, 415–419 (2018).
Cariou, B., Charbonnel, B. & Staels, B. Thiazolidinediones and PPARγ agonists: time for a reassessment. Trends Endocrinol. Metab. 23, 205–215 (2012).
Steinberg, G. R. & Carling, D. AMP-activated protein kinase: the current landscape for drug development. Nat. Rev. Drug Discov. 18, 527–551 (2019).
Myers, R. W. et al. Systemic pan-AMPK activator MK-8722 improves glucose homeostasis but induces cardiac hypertrophy. Science 357, 507–511 (2017).
Esquejo, R. M. et al. Activation of liver AMPK with PF-06409577 corrects NAFLD and lowers cholesterol in rodent and primate preclinical models. EBioMedicine 31, 122–132 (2018).
Cokorinos, E. C. et al. Activation of skeletal muscle AMPK promotes glucose disposal and glucose lowering in non-human primates and mice. Cell Metab. 25, 1147–1159.e10 (2017).
Lee, C. et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 21, 443–454 (2015).
Steneberg, P. et al. PAN-AMPK activator O304 improves glucose homeostasis and microvascular perfusion in mice and type 2 diabetes patients. JCI Insight 3, e99114 (2018).
Dassano, A., Loretelli, C. & Fiorina, P. Idebenone and T2D: A new insulin-sensitizing drug for personalized therapy. Pharmacol. Res. 139, 469–470 (2019).
Harrison, S. A. et al. Insulin sensitizer MSDC-0602K in non-alcoholic steatohepatitis: A randomized, double-blind, placebo-controlled phase IIb study. J. Hepatol. 72, 613–626 (2020).
Acknowledgements
D.E.J. is an Australian Research Council Laureate Fellow. The authors are extremely grateful to S. Klein for his thoughtful discussion of the manuscript and to P. Titchenell, G. Cooney, A. Diaz and M. Nelson for carefully reading and commenting on the manuscript.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Additional information
Competing interests
D.E.J. and J.S. declare no competing interests. M.J.B. is a full-time employee and stockholder of Pfizer Inc. and sits on the Board of Directors of Cerevel Therapeutics.
Peer review information
Nature Reviews Molecular Cell Biology thanks M. Czech, B. Goodpaster, T. Kadowaki, D. Neufer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Hepatic glucose output
-
Refers to the release of glucose from the liver into the bloodstream that is upregulated during fasting and suppressed with insulin. It is mediated via the breakdown of liver glycogen (glycogenolysis) or recycling of breakdown products such as lactate from peripheral tissues (gluconeogenesis).
- Impaired fasting glucose
-
According to the American Diabetes Association, this is defined as fasting plasma glucose levels between 100 and 125 mg dl−1 (between 5.6 and 6.9 mmol l−1).
- Impaired glucose tolerance
-
According to the American Diabetes Association, this is defined as 2-h plasma glucose levels between 140 and 199 mg dl−1 (between 7.8 and 11.0 mmol l−1) measured during a 75-g oral glucose tolerance test.
- Relative hyperinsulinaemia
-
The situation where individuals with type 2 diabetes despite having insufficient insulin secretion to normalize their hyperglycaemia still display significantly higher circulating insulin levels than those observed in healthy people.
- Gluconeogenesis
-
A metabolic pathway that is particularly important in liver metabolism in which glucose is regenerated essentially via reverse glycolysis from a range of substrates, such as lactate, amino acids, glycerol or acetate.
- Metabolic diseases
-
Diseases that exhibit dysregulated metabolism of carbohydrates and lipids. Metabolic diseases include type 2 diabetes, cardiovascular disease and liver disease.
- 14-3-3 proteins
-
A ubiquitously expressed family of proteins comprising seven members in mammals that bind to Ser/Thr phosphorylation motifs and are thought to be one of the major conduits by which kinases such as AKT regulate the function of substrates.
- Retromer
-
A heteropentameric complex of proteins originally discovered in yeast to regulate protein trafficking between endosomes and the trans-Golgi network
- Fat depots
-
Distinct fat depots are named after their location. Subcutaneous fat is the layer of fat found just beneath the skin, whereas visceral or abdominal fat is located in the peritoneal cavity. Subcutaneous fat serves as an energy store, a protective layer of tissue and a potential source of heat for thermoregulation. Visceral fat plays a more important role in releasing fat into the circulation for use by other tissues.
- Lipodystrophy
-
A condition whereby individuals are unable to produce adipose tissue leading to excessive deposition of fat ectopically. The two common forms are congenital, caused by mutations in genes that regulate adipogenesis, or acquired. The most common form of acquired lipodystrophy is caused by older generations of antiretroviral drugs used to treat HIV. Both forms are associated with insulin resistance and other metabolic disorders.
- Insulinomas
-
Small pancreatic tumours that produce and secrete excess insulin into the circulation. This is a rare endocrine disorder that is treated surgically.
- Western diet
-
A diet that generally contains a high content (40–60%) of fat and is rich in simple sugars (sucrose).
- C57BL/6J mice
-
Inbred mouse strain that is the preferred mouse model used in metabolic research to study the effects of a Western diet and for genetic manipulations.
- Metabolic flux
-
The rate at which metabolites are metabolized by intracellular pathways. The metabolic flux is modulated by changing the levels or activity of metabolic enzymes, or by altering the supply or demand of the metabolite.
- TBC1D4
-
A RAB GTPase-activating protein that regulates the GTPase RAB10 and several other RABs. It is phosphorylated by AKT and is integral to insulin regulation of glucose transport in muscle and fat.
- Novel PKCs
-
A subclass of protein kinase C (PKC), a family of Ser/Thr kinases that are activated by diacylglycerols (DAGs) without the need for Ca2+.
- JNK
-
JUN N-terminal kinase is a Ser/Thr kinase that belongs to the family of mitogen-activated protein kinases (MAPKs), which respond to stress stimuli, including cytokines.
- mTOR
-
A Ser/Thr kinase that plays a major role in nutrient sensing in all eukaryotes.
- S6 kinase
-
A Ser/Thr kinase named after its best-known substrate, the ribosomal protein S6. S6 kinase is activated by mTOR, and phosphorylation of its substrate S6 induces protein synthesis.
- PDGF receptor
-
(PDGFR). A receptor that, like the insulin receptor, belongs to the receptor tyrosine kinase family of cell surface receptors. PDGF binding to PDGFR triggers a signal transduction pathway analogous to insulin, involving PI3K and AKT. Importantly, PDGF signalling bypasses IRS and thus provides a tool to test the role of IRS in insulin resistance.
- Carbonylation
-
A protein oxidation reaction that is commonly found on the amino acids lysine and arginine, among other amino acids.
- Lipid peroxidation
-
A process whereby oxidants attack (unsaturated) lipids and via multiple reactions this results in the formation of toxic and/or mutagenic lipid hydroperoxides. The lipid peroxidation process is a chain reaction that propagates until it is terminated by antioxidants.
- Adipokine
-
A molecule, such as a protein, a lipid, a metabolite or a microRNA, that is secreted from adipose tissue to potentially regulate functions in other tissues. It is notable that most adipokines are not produced by adipocytes but derive from stromal cells in the adipose tissue.
- Beige adipocytes
-
A subtype of thermogenic adipocytes that arises in white adipose tissue, most commonly in subcutaneous fat, in response to various stimuli, such as cold exposure, catecholamines, exercise, thiazolidinediones and injury. They are capable of uncoupled mitochondrial respiration via the activity of uncoupling proteins such as UCP1 to produce heat at the expense of ATP, and their emergence has been associated with improved metabolic health.
- Leptin
-
The first adipokine to be discovered, by Friedman and colleagues in 1994. The obese (ob) gene long known to regulate appetite in rodents was found to encode leptin and was subsequently shown to play a key role in metabolic homeostasis and the starvation response in mammals.
- Regulatory T cells
-
A subpopulation of T cells that are involved in immune function.
- C-reactive protein
-
(CRP). A protein secreted primarily by the liver in response to inflammatory cytokines. In the clinic, circulating levels of CRP are a measure of inflammation or infection.
- Genome-wide association studies
-
A statistical method that identifies genetic variants that are associated with a particular trait, disease or phenotype.
- Prediabetes
-
The clinical term to describe individuals who exhibit ‘impaired glucose tolerance’ but do not fulfil the requirements for type 2 diabetes classification.
- Ceramides
-
A class of lipids composed of sphingosine and a fatty acid. Distinct ceramide species contain different fatty acids with various carbon chain lengths, and the ceramides are distinguished and named after the carbon length of the fatty acid.
- AMPK
-
Adenosine 5′-monophosphate (AMP)-activated kinase. AMPK is activated in most cells in response to cellular and/or metabolic stress to buffer cellular energy charge.
- Diacylglycerols
-
(DAGs). Two fatty acid moieties linked to glycerol. DAGs were first identified through their role in binding and activating various members of the protein kinase C (PKC) family.
- Oxidative phosphorylation
-
(OXPHOS). The most notorious function of mitochondria, which is to oxidize various nutrients such as fat and carbohydrate via a series of complex chemical reactions to create the basic energy unit ATP.
- Mitophagy
-
The engulfment of mitochondria by an autophagosome, undergoing autophagy. Dysfunctional mitochondria are targeted for mitophagy for removal and degradation. Mitochondrial fission is necessary for mitophagy.
- Mitochondrial fission
-
A process of mitochondrial fragmentation catalysed by regulated fission machinery. Mitochondrial fission plays an essential role in the removal of damaged mitochondria from cells but has also been associated with reduced bioenergetics.
- Coenzyme Q
-
(CoQ). Also known as ubiquinone, this coenzyme plays an essential role in transporting electrons between various components of the electron transport chain and so is essential to mitochondrial energy production. CoQ also serves as an antioxidant.
- Acylcarnitines
-
A form of fatty acid that are conjugated to carnitine to be transported into the mitochondria for oxidative metabolism.
- Endoplasmic reticulum stress
-
Endoplasmic reticulum (ER) stress is a form of stress in which the ability of the ER to fold proteins is exceeded, leading to the initiation of the ER stress response, an ancient signal transduction pathway that initiates a series of events to help clear the ER of misfolded proteins.
- Sodium–glucose co-transporter 2
-
Also known as SGLT2 or SLC5A2, a transmembrane glucose transporter protein that is predominantly responsible for glucose reabsorption in the kidney.
- Glucagon-like peptide 1
-
(GLP1). A small peptide hormone derived from the proglucagon protein that is secreted from enteroendocrine L cells in the intestine. Among its functions is an incretin activity, which promotes insulin secretion from the pancreas.
- Mitochondrial permeability transition pore
-
(mPTP). A multisubunit protein complex that forms a non-specific channel in the inner mitochondrial membrane. The mPTP opens in response to calcium and/or oxidative stress and allows transit of molecules of up to 1,500 Da from inside mitochondria to the cytosol.
- Palmitate
-
The most prevalent saturated fatty acid found in animals. It is obtained either from dietary sources or by biosynthesis from de novo lipogenesis. It contains an aliphatic tail with a chain length of 16 carbons.
- Insulin-like growth factor 1
-
A hormone that is very similar to insulin that plays a central role in early development in mammals and probably other animals. It is produced and secreted by the liver in response to growth hormone.
Rights and permissions
About this article
Cite this article
James, D.E., Stöckli, J. & Birnbaum, M.J. The aetiology and molecular landscape of insulin resistance. Nat Rev Mol Cell Biol 22, 751–771 (2021). https://doi.org/10.1038/s41580-021-00390-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41580-021-00390-6
This article is cited by
-
Association between the cumulative average triglyceride glucose-body mass index and cardiovascular disease incidence among the middle-aged and older population: a prospective nationwide cohort study in China
Cardiovascular Diabetology (2024)
-
Advances in secondary prevention mechanisms of macrovascular complications in type 2 diabetes mellitus patients: a comprehensive review
European Journal of Medical Research (2024)
-
Bisphenol a downregulates GLUT4 expression by activating aryl hydrocarbon receptor to exacerbate polycystic ovary syndrome
Cell Communication and Signaling (2024)
-
Hyperinsulinemia impairs decidualization via AKT-NR4A1 signaling: new insight into polycystic ovary syndrome (PCOS)-related infertility
Journal of Ovarian Research (2024)
-
Relationship between TyG index and the degree of coronary artery lesions in patients with H-type hypertension
Cardiovascular Diabetology (2024)