Abstract
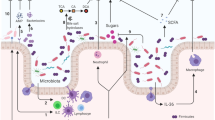
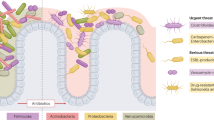
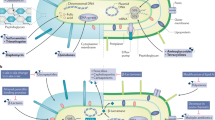
Antibiotics have transformed medicine, saving millions of lives since they were first used to treat a bacterial infection. However, antibiotics administered to target a specific pathogen can also cause collateral damage to the patient’s resident microbial population. These drugs can suppress the growth of commensal species which provide protection against colonization by foreign pathogens, leading to an increased risk of subsequent infection. At the same time, a patient’s microbiota can harbour potential pathogens and, hence, be a source of infection. Antibiotic-induced selection pressure can cause overgrowth of resistant pathogens pre-existing in the patient’s microbiota, leading to hard-to-treat superinfections. In this Review, we explore our current understanding of how antibiotic therapy can facilitate subsequent infections due to both loss of colonization resistance and overgrowth of resistant microorganisms, and how these processes are often interlinked. We discuss both well-known and currently overlooked examples of antibiotic-associated infections at various body sites from various pathogens. Finally, we describe ongoing and new strategies to overcome the collateral damage caused by antibiotics and to limit the risk of antibiotic-associated infections.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Browne, A. J. et al. Global antibiotic consumption and usage in humans, 2000–18: a spatial modelling study. Lancet Planet. Health 5, e893–e904 (2021).
Stewardson, A. J., Huttner, B. & Harbarth, S. At least it won’t hurt: the personal risks of antibiotic exposure. Curr. Opin. Pharmacol. 11, 446–452 (2011).
Blaser, M. J. Antibiotic use and its consequences for the normal microbiome. Science 352, 544–545 (2016).
Hogenauer, C., Hammer, H. F., Krejs, G. J. & Reisinger, E. C. Mechanisms and management of antibiotic‐associated diarrhea. Clin. Infect. Dis. 27, 702–710 (1998).
McFarland, L. V. Epidemiology, risk factors and treatments for antibiotic-associated diarrhea. Dig. Dis. Basel Switz. 16, 292–307 (1998).
Caballero-Flores, G., Pickard, J. M. & Núñez, G. Microbiota-mediated colonization resistance: mechanisms and regulation. Nat. Rev. Microbiol. https://doi.org/10.1038/s41579-022-00833-7 (2022). A comprehensive review on the mechanisms and regulation of colonization resistance.
Sender, R., Fuchs, S. & Milo, R. Revised estimates for the number of human and bacteria cells in the body. PLoS Biol. 14, e1002533 (2016).
Faith, J. J. et al. The long-term stability of the human gut microbiota. Science 341, 1237439 (2013).
Wypych, T. P. & Marsland, B. J. Antibiotics as instigators of microbial dysbiosis: implications for asthma and allergy. Trends Immunol. 39, 697–711 (2018).
Yuan, J. et al. Long-term use of antibiotics and risk of type 2 diabetes in women: a prospective cohort study. Int. J. Epidemiol. 49, 1572–1581 (2020).
Park, S. J. et al. Association between antibiotics use and diabetes incidence in a nationally representative retrospective cohort among Koreans. Sci. Rep. 11, 21681 (2021).
Zhou, W. et al. Longitudinal multi-omics of host–microbe dynamics in prediabetes. Nature 569, 663–671 (2019).
Lynch, S. V. & Pedersen, O. The human intestinal microbiome in health and disease. N. Engl. J. Med. 375, 2369–2379 (2016).
Trasande, L. et al. Infant antibiotic exposures and early-life body mass. Int. J. Obes. 37, 16–23 (2013).
Teng, C., Reveles, K. R., Obodozie-Ofoegbu, O. O. & Frei, C. R. Clostridium difficile infection risk with important antibiotic classes: an analysis of the FDA adverse event reporting system. Int. J. Med. Sci. 16, 630–635 (2019).
Högenauer, C. et al. Klebsiella oxytoca as a causative organism of antibiotic-associated hemorrhagic colitis. N. Engl. J. Med. 355, 2418–2426 (2006).
Shukla, A. & Sobel, J. D. Vulvovaginitis caused by Candida species following antibiotic exposure. Curr. Infect. Dis. Rep. 21, 44 (2019).
Ben-Ami, R. et al. Antibiotic exposure as a risk factor for fluconazole-resistant Candida bloodstream infection. Antimicrob. Agents Chemother. 56, 2518–2523 (2012).
Ichinohe, T. et al. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc. Natl Acad. Sci. USA 108, 5354–5359 (2011).
Blair, J. M. A., Webber, M. A., Baylay, A. J., Ogbolu, D. O. & Piddock, L. J. V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 13, 42–51 (2014).
Bush, K. et al. Tackling antibiotic resistance. Nat. Rev. Microbiol. 9, 894–896 (2011).
Yassour, M. et al. Natural history of the infant gut microbiome and impact of antibiotic treatment on bacterial strain diversity and stability. Sci. Transl. Med. 8, 343ra81 (2016). This longitudinal study of the infant gut microbiome observed transient blooms of specific species and resistance levels during antibiotic treatment.
Stecher, B., Maier, L. & Hardt, W.-D. ‘Blooming’ in the gut: how dysbiosis might contribute to pathogen evolution. Nat. Rev. Microbiol. 11, 277–284 (2013).
Buffie, C. G. et al. Profound alterations of intestinal microbiota following a single dose of clindamycin results in sustained susceptibility to Clostridium difficile-induced colitis. Infect. Immun. 80, 62–73 (2012).
Shen, Z. et al. Emerging carriage of NDM-5 and MCR-1 in Escherichia coli from healthy people in multiple regions in China: a cross sectional observational study. eClinicalMedicine 6, 11–20 (2018).
van Hattem, J. M. et al. Prolonged carriage and potential onward transmission of carbapenemase-producing Enterobacteriaceae in Dutch travelers. Future Microbiol. 11, 857–864 (2016).
Hu, Y. et al. Metagenome-wide analysis of antibiotic resistance genes in a large cohort of human gut microbiota. Nat. Commun. 4, 2151 (2013).
Group, B. M. J. P. Superinfections during antibiotic treatment. Br. Med. J. 1, 537–538 (1952).
Ramirez, J. et al. Antibiotics as major disruptors of gut microbiota. Front. Cell. Infect. Microbiol. 10, 572912 (2020).
Sullivan, Å. Effect of antimicrobial agents on the ecological balance of human microflora. Lancet Infect. Dis. 1, 101–114 (2001). A review of the drug-specific effects of antibiotic on the microbiota, including extra-intestinal microbiota sites.
Yang, L. et al. The varying effects of antibiotics on gut microbiota. AMB Express 11, 116 (2021).
Maier, L. et al. Unravelling the collateral damage of antibiotics on gut bacteria. Nature 599, 120–124 (2021). This study comprehensively screened gut commensals to identify drug combinations active against pathogens but that minimize collateral damage against other species.
Kelly, S. A., Rodgers, A. M., O’Brien, S. C., Donnelly, R. F. & Gilmore, B. F. Gut check time: antibiotic delivery strategies to reduce antimicrobial resistance. Trends Biotechnol. 38, 447–462 (2020).
Dethlefsen, L. & Relman, D. A. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl Acad. Sci. USA 108, 4554–4561 (2011). This study highlights the differences between individuals in the microbiota response and recovery to antibiotics.
Jeffery, I. B., Lynch, D. B. & O’Toole, P. W. Composition and temporal stability of the gut microbiota in older persons. ISME J. 10, 170–182 (2016).
Zimmermann, M., Patil, K. R., Typas, A. & Maier, L. Towards a mechanistic understanding of reciprocal drug–microbiome interactions. Mol. Syst. Biol. 17, e10116 (2021).
Gjonbalaj, M. et al. Antibiotic degradation by commensal microbes shields pathogens. Infect. Immun. 88, e00012–e00020 (2020).
Belkaid, Y. & Hand, T. W. Role of the microbiota in immunity and inflammation. Cell 157, 121–141 (2014).
Byrd, A. L., Belkaid, Y. & Segre, J. A. The human skin microbiome. Nat. Rev. Microbiol. 16, 143–155 (2018).
Elvers, K. T. et al. Antibiotic-induced changes in the human gut microbiota for the most commonly prescribed antibiotics in primary care in the UK: a systematic review. BMJ Open 10, e035677 (2020).
Zimmermann, P. & Curtis, N. The effect of antibiotics on the composition of the intestinal microbiota — a systematic review. J. Infect. 79, 471–489 (2019).
Levison, M. E. & Levison, J. H. Pharmacokinetics and pharmacodynamics of antibacterial agents. Infect. Dis. Clin. North. Am. 23, 791–815 (2009).
Levy, R. M., Huang, E. Y., Roling, D., Leyden, J. J. & Margolis, D. J. Effect of antibiotics on the oropharyngeal flora in patients with acne. Arch. Dermatol. 139, 467–471 (2003).
Kim, S., Covington, A. & Pamer, E. G. The intestinal microbiota: antibiotics, colonization resistance, and enteric pathogens. Immunol. Rev. 279, 90–105 (2017).
Kelly, S. A. et al. Antibiotic therapy and the gut microbiome: investigating the effect of delivery route on gut pathogens. ACS Infect. Dis. 7, 1283–1296 (2021).
Zhang, L., Huang, Y., Zhou, Y., Buckley, T. & Wang, H. H. Antibiotic administration routes significantly influence the levels of antibiotic resistance in gut microbiota. Antimicrob. Agents Chemother. 57, 3659–3666 (2013). A comparison of oral and intravenous antibiotic administration on the spread of antibiotic resistance in the mouse intestine.
Finegold, S. M. Anaerobic infections in humans: an overview. Anaerobe 1, 3–9 (1995).
Eckburg, P. B. et al. Diversity of the human intestinal microbial flora. Science 308, 1635–1638 (2005).
Donskey, C. J. et al. Effect of antibiotic therapy on the density of vancomycin-resistant enterococci in the stool of colonized patients. N. Engl. J. Med. 343, 1925–1932 (2000). This study of patients colonized with vancomycin-resistant enterococci showed overgrowth in the intestine during treatment with various anti-anaerobic antibiotics.
Brook, I., Wexler, H. M. & Goldstein, E. J. C. Antianaerobic antimicrobials: spectrum and susceptibility testing. Clin. Microbiol. Rev. 26, 526–546 (2013).
Taur, Y. et al. Intestinal domination and the risk of bacteremia in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin. Infect. Dis. 55, 905–914 (2012). Intestinal domination by various bacteria is associated with subsequent bacteraemia in patients undergoing haematopoietic stem cell transplantation.
Young, V. B. & Schmidt, T. M. Antibiotic-associated diarrhea accompanied by large-scale alterations in the composition of the fecal microbiota. J. Clin. Microbiol. 42, 1203–1206 (2004).
Wiström, J. et al. Frequency of antibiotic-associated diarrhoea in 2462 antibiotic-treated hospitalized patients: a prospective study. J. Antimicrob. Chemother. 47, 43–50 (2001).
Ma, H. et al. Combined administration of antibiotics increases the incidence of antibiotic-associated diarrhea in critically ill patients. Infect. Drug. Resist. 12, 1047–1054 (2019).
Rashidi, A. et al. Gut microbiota response to antibiotics is personalized and depends on baseline microbiota. Microbiome 9, 211 (2021).
Arvidsson, A., Leijd, B., Nord, C. E. & Angelin, B. Interindividual variability in biliary excretion of ceftriaxone: effects on biliary lipid metabolism and on intestinal microflora. Eur. J. Clin. Invest. 18, 261–266 (1988).
Coyte, K. Z., Schluter, J. & Foster, K. R. The ecology of the microbiome: networks, competition, and stability. Science 350, 663–666 (2015).
Dethlefsen, L., Huse, S., Sogin, M. L. & Relman, D. A. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol. 6, e280 (2008).
Anthony, W. E. et al. Acute and persistent effects of commonly used antibiotics on the gut microbiome and resistome in healthy adults. Cell Rep. 39, 110649 (2022). This study shows short- and long-term effects of antibiotics on the gut microbiota of healthy volunteers.
Yee, A. L. et al. Longitudinal microbiome composition and stability correlate with increased weight and length of very-low-birth-weight infants. mSystems 4, e00229-18 (2019).
Sorg, R. A. et al. Collective resistance in microbial communities by intracellular antibiotic deactivation. PLoS Biol. 14, e2000631 (2016).
Cubillos-Ruiz, A. et al. An engineered live biotherapeutic for the prevention of antibiotic-induced dysbiosis. Nat. Biomed. Eng. 6, 910–921 (2022).
Sun, J. et al. Environmental remodeling of human gut microbiota and antibiotic resistome in livestock farms. Nat. Commun. 11, 1427 (2020).
Foster, K. R., Schluter, J., Coyte, K. Z. & Rakoff-Nahoum, S. The evolution of the host microbiome as an ecosystem on a leash. Nature 548, 43–51 (2017).
Schirmer, M. et al. Linking the human gut microbiome to inflammatory cytokine production capacity. Cell 167, 1125–1136.e8 (2016).
Maier, L. et al. Extensive impact of non-antibiotic drugs on human gut bacteria. Nature 555, 623–628 (2018).
Montandon, S. A. & Jornayvaz, F. R. Effects of antidiabetic drugs on gut microbiota composition. Genes 8, 250 (2017).
Le Bastard, Q. et al. Systematic review: human gut dysbiosis induced by non-antibiotic prescription medications. Aliment. Pharmacol. Ther. 47, 332–345 (2018).
Kwok, C. S. et al. Risk of Clostridium difficile infection with acid suppressing drugs and antibiotics: meta-analysis. Am. J. Gastroenterol. 107, 1011–1019 (2012).
Trifan, A. et al. Proton pump inhibitors therapy and risk of Clostridium difficile infection: systematic review and meta-analysis. World J. Gastroenterol. 23, 6500–6515 (2017).
Caballero-Flores, G., Pickard, J. M., Fukuda, S., Inohara, N. & Núñez, G. An enteric pathogen subverts colonization resistance by evading competition for amino acids in the gut. Cell Host Microbe 28, 526–533.e5 (2020).
Theriot, C. M. et al. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat. Commun. 5, 3114 (2014).
Sassone-Corsi, M. et al. Microcins mediate competition among Enterobacteriaceae in the inflamed gut. Nature 540, 280–283 (2016).
Buffie, C. G. & Pamer, E. G. Microbiota-mediated colonization resistance against intestinal pathogens. Nat. Rev. Immunol. 13, 790–801 (2013).
Ivanov, I. I. et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe 4, 337–349 (2008).
Deshmukh, H. S. et al. The microbiota regulates neutrophil homeostasis and host resistance to Escherichia coli K1 sepsis in neonatal mice. Nat. Med. 20, 524–530 (2014).
Pamer, E. G. Resurrecting the intestinal microbiota to combat antibiotic-resistant pathogens. Science 352, 535–538 (2016).
Freter, R. The fatal enteric cholera infection in the guinea pig, achieved by inhibition of normal enteric flora. J. Infect. Dis. 97, 57–65 (1955). An early study identifying the protection against infection conferred by an intact microbiota.
Miller, C. P., Bohnhoff, M. & Rifkind, D. The effect of an antibiotic on the susceptibility of the mouse’s intestinal tract to Salmonella infection. Trans. Am. Clin. Climatol. Assoc. 68, 51–58 (1957).
Sekirov, I. et al. Antibiotic-induced perturbations of the intestinal microbiota alter host susceptibility to enteric infection. Infect. Immun. 76, 4726–4736 (2008).
Hensgens, M. P. M., Goorhuis, A., Dekkers, O. M. & Kuijper, E. J. Time interval of increased risk for Clostridium difficile infection after exposure to antibiotics. J. Antimicrob. Chemother. 67, 742–748 (2012). A multicenter case–control study to determine the period at risk for CDI after cessation of antibiotics.
Tvede, M. & Rask-Madsen, J. Bacteriotherapy for chronic relapsing Clostridium difficile diarrhoea in six patients. Lancet 333, 1156–1160 (1989).
van Nood, E. et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N. Engl. J. Med. 368, 407–415 (2013).
Buffie, C. G. et al. Precision microbiome restoration of bile acid-mediated resistance to Clostridium difficile. Nature 517, 205–208 (2015). Bile-acid-mediated colonization resistance against C. difficile could be restored by the human gut commensal C. scindens.
Aguirre, A. M. et al. Bile acid-independent protection against Clostridioides difficile infection. PLoS Pathog. 17, e1010015 (2021).
Gregory, A. L., Pensinger, D. A. & Hryckowian, A. J. A short chain fatty acid-centric view of Clostridioides difficile pathogenesis. PLoS Pathog. 17, e1009959 (2021).
Iwata, K. et al. A systematic review for pursuing the presence of antibiotic associated enterocolitis caused by methicillin resistant Staphylococcus aureus. BMC Infect. Dis. 14, 247 (2014).
Lane, A. B., Copeland, N. K., Onmus-Leone, F. & Lawler, J. V. Methicillin-resistant Staphylococcus aureus as a probable cause of antibiotic-associated enterocolitis. Case Rep. Infect. Dis. 2018, e3106305 (2018).
Lichtman, J. S. et al. Host–microbiota interactions in the pathogenesis of antibiotic-associated diseases. Cell Rep. 14, 1049–1061 (2016).
Pavia, A. T. et al. Epidemiologic evidence that prior antimicrobial exposure decreases resistance to infection by antimicrobial-sensitive Salmonella. J. Infect. Dis. 161, 255–260 (1990).
Holmberg, S. D., Osterholm, M. T., Senger, K. A. & Cohen, M. L. Drug-resistant Salmonella from animals fed antimicrobials. N. Engl. J. Med. 311, 617–622 (1984).
Gradel, K. O., Dethlefsen, C., Ejlertsen, T., Schønheyder, H. C. & Nielsen, H. Increased prescription rate of antibiotics prior to non-typhoid Salmonella infections: a one-year nested case–control study. Scand. J. Infect. Dis. 40, 635–641 (2008).
Doorduyn, Y., Van Den Brandhof, W. E., Van Duynhoven, Y. T. H. P., Wannet, W. J. B. & Van Pelt, W. Risk factors for Salmonella Enteritidis and Typhimurium (DT104 and non-DT104) infections in The Netherlands: predominant roles for raw eggs in Enteritidis and sandboxes in Typhimurium infections. Epidemiol. Infect. 134, 617–626 (2006).
Malik, U. et al. Association between prior antibiotic therapy and subsequent risk of community-acquired infections: a systematic review. J. Antimicrob. Chemother. 73, 287–296 (2018).
Humphreys, H. et al. Four country healthcare associated infection prevalence survey 2006: risk factor analysis. J. Hosp. Infect. 69, 249–257 (2008).
Chen, Y. E., Fischbach, M. A. & Belkaid, Y. Skin microbiota–host interactions. Nature 553, 427–436 (2018).
Nakatsuji, T. et al. Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis. Sci. Transl. Med. 9, eaah4680 (2017).
Zipperer, A. et al. Human commensals producing a novel antibiotic impair pathogen colonization. Nature 535, 511–516 (2016).
Belkaid, Y. & Segre, J. A. Dialogue between skin microbiota and immunity. Science 346, 954–959 (2014).
Liu, Q. et al. Staphylococcus epidermidis contributes to healthy maturation of the nasal microbiome by stimulating antimicrobial peptide production. Cell Host Microbe 27, 68–78.e5 (2020).
Man, W. H., de Steenhuijsen Piters, W. A. A. & Bogaert, D. The microbiota of the respiratory tract: gatekeeper to respiratory health. Nat. Rev. Microbiol. 15, 259–270 (2017).
Clark, S. E. Commensal bacteria in the upper respiratory tract regulate susceptibility to infection. Curr. Opin. Immunol. 66, 42–49 (2020).
Santagati, M., Scillato, M., Patanè, F., Aiello, C. & Stefani, S. Bacteriocin-producing oral streptococci and inhibition of respiratory pathogens. FEMS Immunol. Med. Microbiol. 65, 23–31 (2012).
Horn, K. J. et al. Corynebacterium species inhibit Streptococcus pneumoniae colonization and infection of the mouse airway. Front. Microbiol. 12, 804935 (2021).
Thackray, L. B. et al. Oral antibiotic treatment of mice exacerbates the disease severity of multiple flavivirus infections. Cell Rep. 22, 3440–3453.e6 (2018).
Margolis, D. J., Bowe, W. P., Hoffstad, O. & Berlin, J. A. Antibiotic treatment of acne may be associated with upper respiratory tract infections. Arch. Dermatol. 141, 1132–1136 (2005).
Smith, H. S. et al. Antecedent antimicrobial use increases the risk of uncomplicated cystitis in young women. Clin. Infect. Dis. 25, 63–68 (1997).
Robinson, C. J. & Young, V. B. Antibiotic administration alters the community structure of the gastrointestinal micobiota. Gut Microbes 1, 279–284 (2010).
Zacharioudakis, I. M., Zervou, F. N., Pliakos, E. E., Ziakas, P. D. & Mylonakis, E. Colonization with toxinogenic C. difficile upon hospital admission, and risk of infection: a systematic review and meta-analysis. Am. J. Gastroenterol. 110, 381–390 (2015).
Stevens, E. J., Bates, K. A. & King, K. C. Host microbiota can facilitate pathogen infection. PLoS Pathog. 17, e1009514 (2021).
Kluytmans-van den Bergh, M. F. Q. et al. Rectal carriage of extended-spectrum-β-lactamase-producing enterobacteriaceae in hospitalized patients: selective preenrichment increases yield of screening. J. Clin. Microbiol. 53, 2709–2712 (2015).
Sheppard, S. K. Strain wars and the evolution of opportunistic pathogens. Curr. Opin. Microbiol. 67, 102138 (2022).
Donskey, C. J. The role of the intestinal tract as a reservoir and source for transmission of nosocomial pathogens. Clin. Infect. Dis. 39, 219–226 (2004).
Sim, C. K. et al. A mouse model of occult intestinal colonization demonstrating antibiotic-induced outgrowth of carbapenem-resistant Enterobacteriaceae. Microbiome 10, 43 (2022). Mice colonized with resistant bacteria at undetectable levels showed overgrowth following antibiotic treatment.
Maurice, C. F., Haiser, H. J. & Turnbaugh, P. J. Xenobiotics shape the physiology and gene expression of the active human gut microbiome. Cell 152, 39–50 (2013).
Bottery, M. J. et al. Inter-species interactions alter antibiotic efficacy in bacterial communities. ISME J. 16, 812–821 (2022).
Tavernier, S. et al. Community composition determines activity of antibiotics against multispecies biofilms. Antimicrob. Agents Chemother. 61, e00302–e00317 (2017).
Brown, G. D. et al. Hidden killers: human fungal infections. Sci. Transl. Med. 4, 165rv13 (2012).
Mayer, F. L., Wilson, D. & Hube, B. Candida albicans pathogenicity mechanisms. Virulence 4, 119–128 (2013).
Xu, J. et al. Effect of antibiotics on vulvovaginal candidiasis: a MetroNet study. J. Am. Board. Fam. Med. 21, 261–268 (2008).
MacDonald, T. M. et al. The risks of symptomatic vaginal candidiasis after oral antibiotic therapy. Q. J. Med. 86, 419–424 (1993).
Tan, C. T., Xu, X., Qiao, Y. & Wang, Y. A peptidoglycan storm caused by β-lactam antibiotic’s action on host microbiota drives Candida albicans infection. Nat. Commun. 12, 2560 (2021).
Seelig, M. S. The role of antibiotics in the pathogenesis of Candida infections. Am. J. Med. 40, 887–917 (1966).
Takahashi, S. et al. Septic pulmonary embolism caused by Candida albicans after treatment for urinary multidrug-resistant Pseudomonas aeruginosa. J. Infect. Chemother. 14, 436–438 (2008).
Zhai, B. et al. High-resolution mycobiota analysis reveals dynamic intestinal translocation preceding invasive candidiasis. Nat. Med. 26, 59–64 (2020).
Samonis, G. et al. Prospective evaluation of effects of broad-spectrum antibiotics on gastrointestinal yeast colonization of humans. Antimicrob. Agents Chemother. 37, 51–53 (1993).
Spigaglia, P., Mastrantonio, P. & Barbanti, F. in Updates on Clostridium difficile in Europe: Advances in Microbiology, Infectious Diseases and Public Health Volume 8 (eds Mastrantonio, P. & Rupnik, M.) 137–159 (Springer International, 2018). https://doi.org/10.1007/978-3-319-72799-8_9.
Toth, M., Stewart, N. K., Smith, C. & Vakulenko, S. B. Intrinsic class D β-lactamases of Clostridium difficile. mBio 9, e01803–e01818 (2018).
Eyre, D. W. et al. Diverse sources of C. difficile infection identified on whole-genome sequencing. N. Engl. J. Med. 369, 1195–1205 (2013). A study showing that many C. difficile infections are not the result of transmission chains in hospital settings.
Ayres, J. S., Trinidad, N. J. & Vance, R. E. Lethal inflammasome activation by a multidrug-resistant pathobiont upon antibiotic disruption of the microbiota. Nat. Med. 18, 799–806 (2012).
Segura Munoz, R. R. et al. Experimental evaluation of ecological principles to understand and modulate the outcome of bacterial strain competition in gut microbiomes. ISME J. 16, 1594–1604 (2022).
Hardin, G. The competitive exclusion principle. Science 131, 1292–1297 (1960).
Lentsch, V. et al. Combined oral vaccination with niche competition can generate sterilizing immunity against enteropathogenic bacteria. Preprint at bioRxiv https://doi.org/10.1101/2022.07.20.498444 (2022).
Lee, S. M. et al. Bacterial colonization factors control specificity and stability of the gut microbiota. Nature 501, 426–429 (2013).
Murray, B. E., Rensimer, E. R. & Dupont, H. L. Emergence of high-level trimethoprim resistance in fecal Escherichia coli during oral administration of trimethoprim or trimethoprim–sulfamethoxazole. N. Engl. J. Med. 306, 130–135 (1982).
Palleja, A. et al. Recovery of gut microbiota of healthy adults following antibiotic exposure. Nat. Microbiol. 3, 1255–1265 (2018).
Vollaard, E. J., Clasener, H. A. L., van Griethuysen, A. J. A., Janssen, A. J. & Sanders-Reijmers, A. J. Influence of amoxycillin, erythromycin and roxithromycin on colonization resistance and on appearance of secondary colonization in healthy volunteers. J. Antimicrob. Chemother. 20, 131–138 (1987).
Brandl, K. et al. Vancomycin-resistant enterococci exploit antibiotic-induced innate immune deficits. Nature 455, 804–807 (2008).
Ubeda, C. et al. Vancomycin-resistant Enterococcus domination of intestinal microbiota is enabled by antibiotic treatment in mice and precedes bloodstream invasion in humans. J. Clin. Invest. 120, 4332–4341 (2010). This study showed how VRE overgrow in the intestine during antibiotic treatment.
Soares, F. S. et al. Antibiotic-induced pathobiont dissemination accelerates mortality in severe experimental pancreatitis. Front. Immunol. 8, 1890 (2017).
Drummond, R. A. et al. Long-term antibiotic exposure promotes mortality after systemic fungal infection by driving lymphocyte dysfunction and systemic escape of commensal bacteria. Cell Host Microbe 30, 1020–1033.e6 (2022).
Knoop, K. A., McDonald, K. G., Kulkarni, D. H. & Newberry, R. D. Antibiotics promote inflammation through the translocation of native commensal colonic bacteria. Gut 65, 1100–1109 (2016).
Forde, B. M. et al. Population dynamics of an Escherichia coli ST131 lineage during recurrent urinary tract infection. Nat. Commun. 10, 3643 (2019).
Flores-Mireles, A. L., Walker, J. N., Caparon, M. & Hultgren, S. J. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat. Rev. Microbiol. 13, 269–284 (2015).
Wheatley, R. M. et al. Gut to lung translocation and antibiotic mediated selection shape the dynamics of Pseudomonas aeruginosa in an ICU patient. Nat. Commun. 13, 6523 (2022).
Lawley, T. D. et al. Antibiotic treatment of Clostridium difficile carrier mice triggers a supershedder state, spore-mediated transmission, and severe disease in immunocompromised hosts. Infect. Immun. 77, 3661–3669 (2009).
Magill, S. S. et al. Prevalence of antimicrobial use in US acute care hospitals, May–September 2011. J. Am. Med. Asssoc. 312, 1438–1446 (2014).
Stracy, M. et al. Minimizing treatment-induced emergence of antibiotic resistance in bacterial infections. Science 375, 889–894 (2022). Personalized antibiotic recommendations could reduce the emergence of resistance during antibiotic treatment.
Caballero, J. D. et al. Mixed strain pathogen populations accelerate the evolution of antibiotic resistance in patients. Nat. Commun. 14, 4083 (2023).
Tchesnokova, V. L. et al. Pandemic uropathogenic fluoroquinolone-resistant Escherichia coli have enhanced ability to persist in the gut and cause bacteriuria in healthy women. Clin. Infect. Dis. 70, 937–939 (2020).
von Eiff, C., Becker, K., Machka, K., Stammer, H. & Peters, G. Nasal carriage as a source of Staphylococcus aureus bacteremia. N. Engl. J. Med. 344, 11–16 (2001).
Gasparrini, A. J. et al. Persistent metagenomic signatures of early-life hospitalization and antibiotic treatment in the infant gut microbiota and resistome. Nat. Microbiol. 4, 2285–2297 (2019).
Korpela, K. et al. Intestinal microbiome is related to lifetime antibiotic use in Finnish pre-school children. Nat. Commun. 7, 10410 (2016).
Jernberg, C., Löfmark, S., Edlund, C. & Jansson, J. K. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiol. Read. Engl. 156, 3216–3223 (2010).
Wenzler, E., Mulugeta, S. G. & Danziger, L. H. The antimicrobial stewardship approach to combating Clostridium difficile. Antibiotics 4, 198–215 (2015).
Baur, D. et al. Effect of antibiotic stewardship on the incidence of infection and colonisation with antibiotic-resistant bacteria and Clostridium difficile infection: a systematic review and meta-analysis. Lancet Infect. Dis. 17, 990–1001 (2017).
Aldeyab, M. A. et al. An evaluation of the impact of antibiotic stewardship on reducing the use of high-risk antibiotics and its effect on the incidence of Clostridium difficile infection in hospital settings. J. Antimicrob. Chemother. 67, 2988–2996 (2012).
Wiesch, P. A., zur, Kouyos, R., Abel, S., Viechtbauer, W. & Bonhoeffer, S. Cycling empirical antibiotic therapy in hospitals: meta-analysis and models. PLoS Pathog. 10, e1004225 (2014).
Moser, C. et al. Antibiotic therapy as personalized medicine — general considerations and complicating factors. APMIS 127, 361–371 (2019).
Yeh, Y.-C., Huang, T.-H., Yang, S.-C., Chen, C.-C. & Fang, J.-Y. Nano-based drug delivery or targeting to eradicate bacteria for infection mitigation: a review of recent advances. Front. Chem. 8, 286 (2020).
Wang, Y. et al. Targeted delivery of antibiotics to the infected pulmonary tissues using ROS-responsive nanoparticles. J. Nanobiotechnol. 17, 103 (2019).
Yao, J. et al. A pathogen-selective antibiotic minimizes disturbance to the microbiome. Antimicrob. Agents Chemother. 60, 4264–4273 (2016).
Mu, H. et al. Pathogen-targeting glycovesicles as a therapy for salmonellosis. Nat. Commun. 10, 4039 (2019).
Brochado, A. R. et al. Species-specific activity of antibacterial drug combinations. Nature 559, 259–263 (2018).
Gutiérrez, B. & Domingo-Calap, P. Phage therapy in gastrointestinal diseases. Microorganisms 8, 1420 (2020).
Cotter, P. D., Ross, R. P. & Hill, C. Bacteriocins — a viable alternative to antibiotics? Nat. Rev. Microbiol. 11, 95–105 (2013).
Meade, E., Slattery, M. A. & Garvey, M. Bacteriocins, potent antimicrobial peptides and the fight against multi drug resistant species: resistance is futile? Antibiotics 9, 32 (2020).
Hatfull, G. F., Dedrick, R. M. & Schooley, R. T. Phage therapy for antibiotic-resistant bacterial infections. Annu. Rev. Med. 73, 197–211 (2022).
Dedrick, R. M. et al. Engineered bacteriophages for treatment of a patient with a disseminated drug-resistant Mycobacterium abscessus. Nat. Med. 25, 730–733 (2019).
Schooley, R. T. et al. Development and use of personalized bacteriophage-based therapeutic cocktails to treat a patient with a disseminated resistant Acinetobacter baumannii infection. Antimicrob. Agents Chemother. 61, e00954-17 (2017).
Rea, M. C. et al. Thuricin CD, a posttranslationally modified bacteriocin with a narrow spectrum of activity against Clostridium difficile. Proc. Natl Acad. Sci. USA 107, 9352–9357 (2010).
McDonald, L. C. et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin. Infect. Dis. 66, 987–994 (2018).
Yelin, I. et al. Genomic and epidemiological evidence of bacterial transmission from probiotic capsule to blood in ICU patients. Nat. Med. 25, 1728–1732 (2019).
Suez, J. et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 174, 1406–1423.e16 (2018).
Imperial, I. C. V. J. & Ibana, J. A. Addressing the antibiotic resistance problem with probiotics: reducing the risk of its double-edged sword effect. Front. Microbiol. 7, 1983 (2016).
Kassam, Z., Lee, C. H., Yuan, Y. & Hunt, R. H. Fecal microbiota transplantation for Clostridium difficile infection: systematic review and meta-analysis. Am. J. Gastroenterol. 108, 500 (2013).
Pamer, E. G. Fecal microbiota transplantation: effectiveness, complexities, and lingering concerns. Mucosal Immunol. 7, 210–214 (2014).
Taur, Y. et al. Reconstitution of the gut microbiota of antibiotic-treated patients by autologous fecal microbiota transplant. Sci. Transl. Med. 10, eaap9489 (2018).
DeFilipp, Z. et al. Drug-resistant E. coli bacteremia transmitted by fecal microbiota transplant. N. Engl. J. Med. 381, 2043–2050 (2019).
Amrane, S. & Lagier, J.-C. Fecal microbiota transplantation for antibiotic resistant bacteria decolonization. Hum. Microbiome J. 16, 100071 (2020). A comprehensive overview of the application of FMT to decolonize the gut of antibiotic resistance bacteria.
Walker, A. W. et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 5, 220–230 (2011).
Stecher, B. & Hardt, W.-D. Mechanisms controlling pathogen colonization of the gut. Curr. Opin. Microbiol. 14, 82–91 (2011).
Pickard, J. M. & Núñez, G. Pathogen colonization resistance in the gut and its manipulation for improved health. Am. J. Pathol. 189, 1300–1310 (2019).
Russell, A. B., Peterson, S. B. & Mougous, J. D. Type VI secretion system effectors: poisons with a purpose. Nat. Rev. Microbiol. 12, 137–148 (2014).
Brunet, Y. R., Espinosa, L., Harchouni, S., Mignot, T. & Cascales, E. Imaging type VI secretion-mediated bacterial killing. Cell Rep. 3, 36–41 (2013).
Cotter, P. D., Hill, C. & Ross, R. P. Bacteriocins: developing innate immunity for food. Nat. Rev. Microbiol. 3, 777–788 (2005).
Vital, M., Rud, T., Rath, S., Pieper, D. H. & Schlüter, D. Diversity of bacteria exhibiting bile acid-inducible 7α-dehydroxylation genes in the human gut. Comput. Struct. Biotechnol. J. 17, 1016–1019 (2019).
Didelot, X., Walker, A. S., Peto, T. E., Crook, D. W. & Wilson, D. J. Within-host evolution of bacterial pathogens. Nat. Rev. Microbiol. 14, 150–162 (2016).
Ikuta, K. S. et al. Global mortality associated with 33 bacterial pathogens in 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 400, 2221–2248 (2022).
Magruder, M. et al. Gut uropathogen abundance is a risk factor for development of bacteriuria and urinary tract infection. Nat. Commun. 10, 5521 (2019).
Acknowledgements
This research was funded by Wellcome Trust grant 224212/Z/21/Z.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article. L.d.N. and M.S. contributed substantially to discussion of the content. L.d.N. and M.S. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Microbiology thanks Simone Becattini and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Nies, L., Kobras, C.M. & Stracy, M. Antibiotic-induced collateral damage to the microbiota and associated infections. Nat Rev Microbiol 21, 789–804 (2023). https://doi.org/10.1038/s41579-023-00936-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-023-00936-9