Abstract
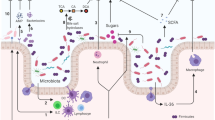
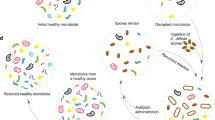
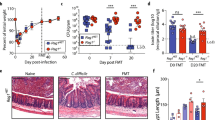
Faecal microbiota transplantation (FMT) has emerged as a remarkably successful treatment for recurrent Clostridioides difficile infection that cannot be cured with antibiotics alone. Understanding the complex biology and pathogenesis of C. difficile infection, which we discuss in this Perspective, is essential for understanding the potential mechanisms by which FMT cures this disease. Although FMT has already entered clinical practice, different microbiota-based products are currently in clinical trials and are vying for regulatory approval. However, all these therapeutics belong to an entirely new class of agents that require the development of a new branch of pharmacology. Characterization of microbiota therapeutics uses novel and rapidly evolving technologies and requires incorporation of microbial ecology concepts. Here, we consider FMT within a pharmacological framework, including its essential elements: formulation, pharmacokinetics and pharmacodynamics. From this viewpoint, multiple gaps in knowledge become apparent, identifying areas that require systematic research. This knowledge is needed to help clinical providers use microbiota therapeutics appropriately and to facilitate development of next-generation microbiota products with improved safety and efficacy. The discussion here is limited to FMT as a representative of microbiota therapeutics and recurrent C. difficile as the indication; however, consideration of the intrinsic basic principles is relevant to this entire class of microbiota-based therapeutics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Costello, E. K., Stagaman, K., Dethlefsen, L., Bohannan, B. J. & Relman, D. A. The application of ecological theory toward an understanding of the human microbiome. Science 336, 1255–1262 (2012).
Adams, J. B. et al. Microbiota transplant therapy and autism: lessons for the clinic. Expert. Rev. Gastroenterol. Hepatol. 13, 1033–1037 (2019).
Dutta, S. K. et al. Parkinson’s disease: the emerging role of gut dysbiosis, antibiotics, probiotics, and fecal microbiota transplantation. J. Neurogastroenterol. Motil. 25, 363–376 (2019).
Oren, A. & Rupnik, M. Clostridium difficile and Clostridioides difficile: two validly published and correct names. Anaerobe 52, 125–126 (2018).
Drekonja, D. et al. Fecal microbiota transplantation for Clostridium difficile infection: a systematic review. Ann. Intern. Med. 162, 630–638 (2015).
Borody, T. J. & Khoruts, A. Fecal microbiota transplantation and emerging applications. Nat. Rev. Gastroenterol. Hepatol. 9, 88–96 (2012).
van Nood, E. et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N. Engl. J. Med. 368, 407–415 (2013).
Hvas, C. L. et al. Fecal microbiota transplantation is superior to fidaxomicin for treatment of recurrent Clostridium difficile infection. Gastroenterology https://doi.org/10.1053/j.gastro.2018.12.019 (2019).
McDonald, L. C. et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin. Infect. Dis. 66, 987–994 (2018).
Mullish, B. H. et al. The use of faecal microbiota transplant as treatment for recurrent or refractory Clostridium difficile infection and other potential indications: joint British Society of Gastroenterology (BSG) and Healthcare Infection Society (HIS) guidelines. J. Hosp. Infect. 100, S1–S31 (2018).
Khoruts, A., Sadowsky, M. J. & Hamilton, M. J. Development of fecal microbiota transplantation suitable for mainstream medicine. Clin. Gastroenterol. Hepatol. https://doi.org/10.1016/j.cgh.2014.11.014 (2014).
Tariq, R., Pardi, D. S., Bartlett, M. G. & Khanna, S. Low cure rates in controlled trials of fecal microbiota transplantation for recurrent Clostridium difficile infection: a systematic review and meta-analysis. Clin. Infect. Dis. 68, 1351–1358 (2019).
Edwards, A. N. & McBride, S. M. Isolating and purifying Clostridium difficile spores. Methods Mol. Biol. 1476, 117–128 (2016).
Gerding, D. N., Muto, C. A. & Owens, R. C. Jr Measures to control and prevent Clostridium difficile infection. Clin. Infect. Dis. 46, S43–S49 (2008).
Deakin, L. J. et al. The Clostridium difficile spo0A gene is a persistence and transmission factor. Infect. Immun. 80, 2704–2711 (2012).
Oliveira, P. H. et al. Epigenomic characterization of Clostridioides difficile finds a conserved DNA methyltransferase that mediates sporulation and pathogenesis. Nat. Microbiol. 5, 166–180 (2020).
Srikhanta, Y. N. et al. Cephamycins inhibit pathogen sporulation and effectively treat recurrent Clostridioides difficile infection. Nat. Microbiol. 4, 2237–2245 (2019).
Shaughnessy, M. K. et al. Environmental contamination in households of patients with recurrent Clostridium difficile infection. Appl. Environ. Microbiol. 82, 2686–2692 (2016).
Brandt, L. J. et al. Long-term follow-up of colonoscopic fecal microbiota transplant for recurrent Clostridium difficile infection. Am. J. Gastroenterol. 107, 1079–1087 (2012).
Foster, S. J. & Johnstone, K. Pulling the trigger: the mechanism of bacterial spore germination. Mol. Microbiol. 4, 137–141 (1990).
Bhattacharjee, D., McAllister, K. N. & Sorg, J. A. Germinants and their receptors in Clostridia. J. Bacteriol. 198, 2767–2775 (2016).
Kochan, T. J. et al. Updates to Clostridium difficile spore germination. J. Bacteriol. 200, e00218-18 (2018).
Weingarden, A. R. et al. Microbiota transplantation restores normal fecal bile acid composition in recurrent Clostridium difficile infection. Am. J. Physiol. Gastrointest. Liver Physiol. 306, G310–G319 (2014).
Mullish, B. H. et al. Microbial bile salt hydrolases mediate the efficacy of faecal microbiota transplant in the treatment of recurrent Clostridioides difficile infection. Gut 68, 1791–1800 (2019).
Sorg, J. A. & Sonenshein, A. L. Inhibiting the initiation of Clostridium difficile spore germination using analogs of chenodeoxycholic acid, a bile acid. J. Bacteriol. 192, 4983–4990 (2010).
Weingarden, A. R. et al. Changes in colonic bile acid composition following fecal microbiota transplantation are sufficient to control Clostridium difficile germination and growth. PLoS ONE 11, e0147210 (2016).
Buffie, C. G. et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature 517, 205–208 (2015).
Theriot, C. M., Bowman, A. A. & Young, V. B. Antibiotic-induced alterations of the gut microbiota alter secondary bile acid production and allow for Clostridium difficile spore germination and outgrowth in the large intestine. mSphere 1, e00045-15 (2016).
Solbach, P. et al. BaiCD gene cluster abundance is negatively correlated with Clostridium difficile infection. PLoS ONE 13, e0196977 (2018).
Studer, N. et al. Functional intestinal bile acid 7alpha-dehydroxylation by Clostridium scindens associated with protection from clostridium difficile infection in a gnotobiotic mouse model. Front. Cell Infect. Microbiol. 6, 191 (2016).
Francis, M. B., Allen, C. A., Shrestha, R. & Sorg, J. A. Bile acid recognition by the Clostridium difficile germinant receptor, CspC, is important for establishing infection. PLoS Pathog. 9, e1003356 (2013).
Leon, J. A. & Tumpson, D. B. Competition between two species for two complementary or substitutable resources. J. Theor. Biol. 50, 185–201 (1975).
Ng, K. M. et al. Microbiota-liberated host sugars facilitate post-antibiotic expansion of enteric pathogens. Nature 502, 96–99 (2013).
Ferreyra, J. A. et al. Gut microbiota-produced succinate promotes C. difficile infection after antibiotic treatment or motility disturbance. Cell Host Microbe 16, 770–777 (2014).
McDonald, J. A. K. et al. Inhibiting growth of Clostridioides difficile by restoring valerate, produced by the intestinal microbiota. Gastroenterology 155, 1495–1507 e1415 (2018).
Sorg, J. A. & Sonenshein, A. L. Bile salts and glycine as cogerminants for Clostridium difficile spores. J. Bacteriol. 190, 2505–2512 (2008).
Kang, J. D. et al. Bile acid 7alpha-dehydroxylating gut bacteria secrete antibiotics that inhibit Clostridium difficile: role of secondary bile acids. Cell Chem. Biol. 26, 27–34.e4 (2019).
Darkoh, C., Plants-Paris, K., Bishoff, D. & DuPont, H. L. Clostridium difficile modulates the gut microbiota by inducing the production of indole, an interkingdom signaling and antimicrobial molecule. mSystems 4, e00346-18 (2019).
Hutton, M. L., Mackin, K. E., Chakravorty, A. & Lyras, D. Small animal models for the study of Clostridium difficile disease pathogenesis. FEMS Microbiol. Lett. 352, 140–149 (2014).
Rousseau, C. et al. Clostridium difficile colonization in early infancy is accompanied by changes in intestinal microbiota composition. J. Clin. Microbiol. 49, 858–865 (2011).
Viscidi, R., Willey, S. & Bartlett, J. G. Isolation rates and toxigenic potential of Clostridium difficile isolates from various patient populations. Gastroenterology 81, 5–9 (1981).
Rousseau, C. et al. Prevalence and diversity of Clostridium difficile strains in infants. J. Med. Microbiol. 60, 1112–1118 (2011).
McFarland, L. V., Mulligan, M. E., Kwok, R. Y. & Stamm, W. E. Nosocomial acquisition of Clostridium difficile infection. N. Engl. J. Med. 320, 204–210 (1989).
Furuya-Kanamori, L. et al. Asymptomatic Clostridium difficile colonization: epidemiology and clinical implications. BMC Infect. Dis. 15, 516 (2015).
Crobach, M. J. T. et al. Understanding Clostridium difficile colonization. Clin. Microbiol. Rev. 31, e00021-17 (2018).
Khanna, S., Shin, A. & Kelly, C. P. Management of Clostridium difficile infection in inflammatory bowel disease: expert review from the Clinical Practice Updates Committee of the AGA institute. Clin. Gastroenterol. Hepatol. 15, 166–174 (2017).
Yakob, L. et al. Mechanisms of hypervirulent Clostridium difficile ribotype 027 displacement of endemic strains: an epidemiological model. Sci. Rep 5, 12666 (2015).
Yahav, J. et al. Helicobacter pylori and Clostridium difficile in cystic fibrosis patients. Dig. Dis. Sci. 51, 2274–2279 (2006).
Monaghan, T. M. et al. High prevalence of subclass-specific binding and neutralizing antibodies against Clostridium difficile toxins in adult cystic fibrosis sera: possible mode of immunoprotection against symptomatic C. difficile infection. Clin. Exp. Gastroenterol. 10, 169–175 (2017).
Kyne, L., Warny, M., Qamar, A. & Kelly, C. P. Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A. N. Engl. J. Med. 342, 390–397 (2000).
Aktories, K., Schwan, C. & Jank, T. Clostridium difficile toxin biology. Annu. Rev. Microbiol. 71, 281–307 (2017).
Huang, J. et al. Clostridium difficile toxins induce VEGF-A and vascular permeability to promote disease pathogenesis. Nat. Microbiol. 4, 269–279 (2019).
Huang, B. et al. Real-time cellular analysis coupled with a specimen enrichment accurately detects and quantifies Clostridium difficile toxins in stool. J. Clin. Microbiol. 52, 1105–1111 (2014).
Cohen, N. A. et al. Clostridium difficile fecal toxin level is associated with disease severity and prognosis. United Eur. Gastroenterol. J. 6, 773–780 (2018).
Janoir, C. Virulence factors of Clostridium difficile and their role during infection. Anaerobe 37, 13–24 (2016).
Deneve, C., Delomenie, C., Barc, M. C., Collignon, A. & Janoir, C. Antibiotics involved in Clostridium difficile-associated disease increase colonization factor gene expression. J. Med. Microbiol. 57, 732–738 (2008).
Zarandi, E. R., Mansouri, S., Nakhaee, N., Sarafzadeh, F. & Moradi, M. Effect of sub-MIC of vancomycin and clindamycin alone and in combination with ceftazidime on Clostridium difficile surface layer protein A (slpA) gene expression. Microb. Pathog. 111, 163–167 (2017).
Ueda, A. & Wood, T. K. Connecting quorum sensing, c-di-GMP, pel polysaccharide, and biofilm formation in pseudomonas aeruginosa through tyrosine phosphatase TpbA (PA3885). PLoS Pathog. 5, e1000483 (2009).
Soavelomandroso, A. P. et al. Biofilm structures in a mono-associated mouse model of Clostridium difficile infection. Front. Microbiol. 8, 2086 (2017).
Semenyuk, E. G. et al. Analysis of bacterial communities during Clostridium difficile infection in the mouse. Infect. Immun. 83, 4383–4391 (2015).
Buckley, A. M., Spencer, J., Candlish, D., Irvine, J. J. & Douce, G. R. Infection of hamsters with the UK Clostridium difficile ribotype 027 outbreak strain R20291. J. Med. Microbiol. 60, 1174–1180 (2011).
Abee, T., Kovacs, A. T., Kuipers, O. P. & van der Veen, S. Biofilm formation and dispersal in Gram-positive bacteria. Curr. Opin. Biotechnol. 22, 172–179 (2011).
Ethapa, T. et al. Multiple factors modulate biofilm formation by the anaerobic pathogen Clostridium difficile. J. Bacteriol. 195, 545–555 (2013).
Pettit, L. J. et al. Functional genomics reveals that Clostridium difficile Spo0A coordinates sporulation, virulence and metabolism. BMC Genomics 15, 160 (2014).
Vaishnava, S. et al. The antibacterial lectin RegIIIgamma promotes the spatial segregation of microbiota and host in the intestine. Science 334, 255–258 (2011).
Hansson, G. C. Role of mucus layers in gut infection and inflammation. Curr. Opin. Microbiol. 15, 57–62 (2012).
Wlodarska, M. et al. Antibiotic treatment alters the colonic mucus layer and predisposes the host to exacerbated Citrobacter rodentium-induced colitis. Infect. Immun. 79, 1536–1545 (2011).
Gustafsson, A., Lund-Tonnesen, S., Berstad, A., Midtvedt, T. & Norin, E. Faecal short-chain fatty acids in patients with antibiotic-associated diarrhoea, before and after faecal enema treatment. Scand. J. Gastroenterol. 33, 721–727 (1998).
Antharam, V. C. et al. Intestinal dysbiosis and depletion of butyrogenic bacteria in Clostridium difficile infection and nosocomial diarrhea. J. Clin. Microbiol. 51, 2884–2892 (2013).
Fachi, J. L. et al. Butyrate protects mice from Clostridium difficile-induced colitis through an HIF-1-dependent mechanism. Cell Rep. 27, 750–761.e7 (2019).
Theriot, C. M. et al. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat. Commun. 5, 3114 (2014).
Seekatz, A. M. et al. Restoration of short chain fatty acid and bile acid metabolism following fecal microbiota transplantation in patients with recurrent Clostridium difficile infection. Anaerobe 53, 64–73 (2018).
Abt, M. C. et al. Innate immune defenses mediated by two ILC subsets are critical for protection against acute Clostridium difficile infection. Cell Host Microbe 18, 27–37 (2015).
Rees, W. D. & Steiner, T. S. Adaptive immune response to Clostridium difficile infection: a perspective for prevention and therapy. Eur. J. Immunol. 48, 398–406 (2018).
Kyne, L., Warny, M., Qamar, A. & Kelly, C. P. Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet 357, 189–193 (2001).
Bauer, M. P., Nibbering, P. H., Poxton, I. R., Kuijper, E. J. & van Dissel, J. T. Humoral immune response as predictor of recurrence in Clostridium difficile infection. Clin. Microbiol. Infect. 20, 1323–1328 (2014).
van Opstal, E. et al. Vancomycin treatment alters humoral immunity and intestinal microbiota in an aged mouse model of Clostridium difficile infection. J. Infect. Dis. 214, 130–139 (2016).
Littman, D. R. Do the microbiota influence vaccines and protective immunity to pathogens? If so, is there potential for efficacious microbiota-based vaccines? Cold Spring Harb. Perspect. Biol. 10, a029355 (2018).
Khoruts, A. Is fecal microbiota transplantation a temporary patch for treatment of Clostridium difficile infection or a new frontier of therapeutics? Expert. Rev. Gastroenterol. Hepatol. 12, 435–438 (2018).
Hoffmann, D. et al. Improving regulation of microbiota transplants. Science 358, 1390–1391 (2017).
Jorgensen, S. M. D. et al. Banking feces: a new frontier for public blood banks? Transfusion 59, 2776–2782 (2019).
Theis, K. R. et al. Getting the hologenome concept right: an eco-evolutionary framework for hosts and their microbiomes. mSystems 1, e00028-16 (2016).
Bakken, J. S. et al. Treating Clostridium difficile infection with fecal microbiota transplantation. Clin. Gastroenterol. Hepatol. 9, 1044–1049 (2011).
Cammarota, G. et al. European consensus conference on faecal microbiota transplantation in clinical practice. Gut 66, 569–580 (2017).
Mullish, B. H., Quraishi, M. N., Segal, J. P., Williams, H. R. T. & Goldenberg, S. D. Introduction to the joint British Society of Gastroenterology (BSG) and Healthcare Infection Society (HIS) faecal microbiota transplant guidelines. J. Hosp. Infect. 100, 130–132 (2018).
Cammarota, G. et al. International consensus conference on stool banking for faecal microbiota transplantation in clinical practice. Gut 68, 2111–2121 (2019).
Litvak, Y., Byndloss, M. X. & Baumler, A. J. Colonocyte metabolism shapes the gut microbiota. Science 362, eaat9076 (2018).
Costello, S. P. et al. Effect of fecal microbiota transplantation on 8-week remission in patients with ulcerative colitis: a randomized clinical trial. JAMA 321, 156–164 (2019).
Feltham, R. K., Power, A. K., Pell, P. A. & Sneath, P. A. A simple method for storage of bacteria at −76 degrees C. J. Appl. Bacteriol. 44, 313–316 (1978).
Postgate, J. R. & Hunter, J. R. On the survival of frozen bacteria. J. Gen. Microbiol. 26, 367–378 (1961).
Fuller, B. J. Cryoprotectants: the essential antifreezes to protect life in the frozen state. Cryo Lett. 25, 375–388 (2004).
Hubalek, Z. Protectants used in the cryopreservation of microorganisms. Cryobiology 46, 205–229 (2003).
Pegg, D. E. Principles of cryopreservation. Methods Mol. Biol. 368, 39–57 (2007).
Hamilton, M. J., Weingarden, A. R., Sadowsky, M. J. & Khoruts, A. Standardized frozen preparation for transplantation of fecal microbiota for recurrent Clostridium difficile infection. Am. J. Gastroenterol. 107, 761–767 (2012).
Lee, C. H. et al. Frozen vs fresh fecal microbiota transplantation and clinical resolution of diarrhea in patients with recurrent Clostridium difficile infection: a randomized clinical trial. JAMA 315, 142–149 (2016).
Youngster, I. et al. Oral, capsulized, frozen fecal microbiota transplantation for relapsing Clostridium difficile infection. JAMA 312, 1772–1778 (2014).
Jiang, Z. D. et al. Randomised clinical trial: faecal microbiota transplantation for recurrent Clostridum difficile infection — fresh, or frozen, or lyophilised microbiota from a small pool of healthy donors delivered by colonoscopy. Aliment. Pharmacol. Ther. 45, 899–908 (2017).
Ramai, D., Zakhia, K., Ofosu, A., Ofori, E. & Reddy, M. Fecal microbiota transplantation: donor relation, fresh or frozen, delivery methods, cost-effectiveness. Ann. Gastroenterol. 32, 30–38 (2019).
Kao, D. et al. Effect of oral capsule- vs colonoscopy-delivered fecal microbiota transplantation on recurrent Clostridium difficile infection: a randomized clinical trial. JAMA 318, 1985–1993 (2017).
Goldenberg, S. D. et al. Comparison of different strategies for providing fecal microbiota transplantation to treat patients with recurrent Clostridium difficile infection in two English hospitals: a review. Infect. Dis. Ther. 7, 71–86 (2018).
Benedict, R. G. et al. Preservation of microorganisms by freeze-drying. I. Cell supernatant, Naylor-Smith solution, and salts of various acids as stabilizers for Serratia marcescens. Appl. Microbiol. 6, 401–407 (1958).
Heckly, R. J. Preservation of microorganisms. Adv. Appl. Microbiol 24, 1–53 (1978).
Wagman, J. Evidence of cytoplasmic membrane injury in the drying of bacteria. J. Bacteriol 80, 558–564 (1960).
Roth, B. L., Poot, M., Yue, S. T. & Millard, P. J. Bacterial viability and antibiotic susceptibility testing with SYTOX green nucleic acid stain. Appl. Environ. Microbiol. 63, 2421–2431 (1997).
Boulos, L., Prevost, M., Barbeau, B., Coallier, J. & Desjardins, R. LIVE/DEAD BacLight: application of a new rapid staining method for direct enumeration of viable and total bacteria in drinking water. J. Microbiol. Methods 37, 77–86 (1999).
Staley, C. et al. Successful resolution of recurrent Clostridium difficile infection using freeze-dried, encapsulated fecal microbiota; pragmatic cohort study. Am. J. Gastroenterol. 112, 940–947 (2017).
Miller, D. S. et al. A simple and inexpensive enteric-coated capsule for delivery of acid-labile macromolecules to the small intestine. J. Zhejiang Univ. Sci. B 16, 586–592 (2015).
Khoruts, A. et al. Inflammatory bowel disease affects the outcome of fecal microbiota transplantation for recurrent Clostridium difficile infection. Clin. Gastroenterol. Hepatol. 14, 1433–1438 (2016).
DeFilipp, Z. et al. Drug-resistant E. coli bacteremia transmitted by fecal microbiota transplant. N. Engl. J. Med 381, 2043–2050 (2019).
Eiseman, B., Silen, W., Bascom, G. S. & Kauvar, A. J. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery 44, 854–859 (1958).
Khoruts, A., Dicksved, J., Jansson, J. K. & Sadowsky, M. J. Changes in the composition of the human fecal microbiome after bacteriotherapy for recurrent Clostridium difficile-associated diarrhea. J. Clin. Gastroenterol. 44, 354–360 (2010).
Hamilton, M. J., Weingarden, A. R., Unno, T., Khoruts, A. & Sadowsky, M. J. High-throughput DNA sequence analysis reveals stable engraftment of gut microbiota following transplantation of previously frozen fecal bacteria. Gut Microbes 4, 125–135 (2013).
Seekatz, A. M. et al. Recovery of the gut microbiome following fecal microbiota transplantation. mBio 5, e00893–e00914 (2014).
Chang, J. Y. et al. Decreased diversity of the fecal microbiome in recurrent Clostridium difficile-associated diarrhea. J. Infect. Dis. 197, 435–438 (2008).
Weingarden, A. et al. Dynamic changes in short- and long-term bacterial composition following fecal microbiota transplantation for recurrent Clostridium difficile infection. Microbiome 3, 10 (2015).
Jalanka, J. et al. Long-term effects on luminal and mucosal microbiota and commonly acquired taxa in faecal microbiota transplantation for recurrent Clostridium difficile infection. BMC Med. 14, 155 (2016).
Kelly, C. R. et al. Effect of fecal microbiota transplantation on recurrence in multiply recurrent Clostridium difficile infection: a randomized trial. Ann. Intern. Med. 165, 609–616 (2016).
Smillie, C. S. et al. Strain tracking reveals the determinants of bacterial engraftment in the human gut following fecal microbiota transplantation. Cell Host Microbe 23, 229–240.e5 (2018).
Li, S. S. et al. Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science 352, 586–589 (2016).
Broecker, F., Klumpp, J. & Moelling, K. Long-term microbiota and virome in a Zurich patient after fecal transplantation against Clostridium difficile infection. Ann. N. Y. Acad. Sci. 1372, 29–41 (2016).
Youngster, I. et al. Fecal microbiota transplant for relapsing Clostridium difficile infection using a frozen inoculum from unrelated donors: a randomized, open-label, controlled pilot study. Clin. Infect. Dis. 58, 1515–1522 (2014).
Angelberger, S. et al. Temporal bacterial community dynamics vary among ulcerative colitis patients after fecal microbiota transplantation. Am. J. Gastroenterol. 108, 1620–1630 (2013).
Ridaura, V. K. et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341, 1241214 (2013).
Staley, C., Kelly, C. R., Brandt, L. J., Khoruts, A. & Sadowsky, M. J. Complete microbiota engraftment is not essential for recovery from recurrent Clostridium difficile infection following fecal microbiota transplantation. mBio 7, e01965-16 (2016).
Staley, C. et al. Community dynamics drive punctuated engraftment of the fecal microbiome following transplantation using freeze-dried, encapsulated fecal microbiota. Gut Microbes 8, 276–288 (2017).
Staley, C. et al. Predicting recurrence of Clostridium difficile infection following encapsulated fecal microbiota transplantation. Microbiome 6, 166 (2018).
Knights, D. et al. Bayesian community-wide culture-independent microbial source tracking. Nat. Methods 8, 761–763 (2011).
Shenhav, L. et al. FEAST: fast expectation-maximization for microbial source tracking. Nat. Methods 16, 627–632 (2019).
Fischer, M. et al. Predictors of early failure after fecal microbiota transplantation for the therapy of Clostridium difficile infection: a multicenter study. Am. J. Gastroenterol. 111, 1024–1031 (2016).
Allegretti, J. R. et al. Classifying fecal microbiota transplantation failure: an observational study examining timing and characteristics of fecal microbiota transplantation failures. Clin. Gastroenterol. Hepatol. 16, 1832–1833 (2018).
Shankar, V. et al. Species and genus level resolution analysis of gut microbiota in Clostridium difficile patients following fecal microbiota transplantation. Microbiome 2, 13 (2014).
Fuentes, S. et al. Reset of a critically disturbed microbial ecosystem: faecal transplant in recurrent Clostridium difficile infection. ISME J. 8, 1621–1633 (2014).
Staley, C. et al. Durable long-term bacterial engraftment following encapsulated fecal microbiota transplantation to treat Clostridium difficile infection. mBio 10, e01586-19 (2019).
Tvede, M. & Rask-Madsen, J. Bacteriotherapy for chronic relapsing Clostridium difficile diarrhoea in six patients. Lancet 1, 1156–1160 (1989).
Louie, T. J. et al. Fidaxomicin versus vancomycin for Clostridium difficile infection. N. Engl. J. Med 364, 422–431 (2011).
Khanna, S. et al. A novel microbiome therapeutic increases gut microbial diversity and prevents recurrent Clostridium difficile infection. J. Infect. Dis. 214, 173–181 (2016).
Businesswire. Seres Therapeutics announces interim results from SER-109 phase 2 ECOSPOR study in multiply recurrent Clostridium difficile infection. http://www.businesswire.com/news/home/20160729005385/en/Seres-Therapeutics-Announces-Interim-Results-SER-109-Phase (2016).
Peck, B. C. E., Shanahan, M. T., Singh, A. P. & Sethupathy, P. Gut microbial influences on the mammalian intestinal stem cell niche. Stem Cell Int. 2017, 1–17 (2017).
Teigen, L. M. et al. Dietary factors in sulfur metabolism and pathogenesis of ulcerative colitis. Nutrients 11, 931 (2019).
Zoetendal, E. G. et al. Mucosa-associated bacteria in the human gastrointestinal tract are uniformly distributed along the colon and differ from the community recovered from feces. Appl. Environ. Microbiol. 68, 3401–3407 (2002).
Newman, K. M., Rank, K. M., Vaughn, B. P. & Khoruts, A. Treatment of recurrent Clostridium difficile infection using fecal microbiota transplantation in patients with inflammatory bowel disease. Gut Microbes 8, 303–309 (2017).
Khanna, S. et al. Changes in microbial ecology after fecal microbiota transplantation for recurrent C. difficile infection affected by underlying inflammatory bowel disease. Microbiome 5, 55 (2017).
Allegretti, J. R. et al. Risk of Clostridium difficile infection with systemic antimicrobial therapy following successful fecal microbiota transplant: should we recommend anti-Clostridium difficile antibiotic prophylaxis? Dig. Dis. Sci. 64, 1668–1671 (2019).
Wadhwa, A. et al. High risk of post-infectious irritable bowel syndrome in patients with Clostridium difficile infection. Aliment. Pharmacol. Ther. 44, 576–582 (2016).
Gutierrez, R. L., Riddle, M. S. & Porter, C. K. Increased risk of functional gastrointestinal sequelae after Clostridium difficile infection among active duty United States military personnel (1998–2010). Gastroenterology 149, 1408–1414 (2015).
Jalanka, J. et al. The long-term effects of faecal microbiota transplantation for gastrointestinal symptoms and general health in patients with recurrent Clostridium difficile infection. Aliment. Pharmacol. Ther. 47, 371–379 (2018).
Broecker, F. et al. Long-term changes of bacterial and viral compositions in the intestine of a recovered Clostridium difficile patient after fecal microbiota transplantation. Cold Spring Harb. Mol. Case Stud. 2, a000448 (2016).
Sorg, J. A. & Sonenshein, A. L. Chenodeoxycholate is an inhibitor of Clostridium difficile spore germination. J. Bacteriol. 191, 1115–1117 (2009).
Monaghan, T. et al. Effective fecal microbiota transplantation for recurrent Clostridioides difficile infection in humans is associated with increased signalling in the bile acid-farnesoid X receptor-fibroblast growth factor pathway. Gut Microbes 10, 142–148 (2019).
Geng, S. et al. Faecal microbiota transplantation reduces susceptibility to epithelial injury and modulates tryptophan metabolism of the microbial community in a piglet model. J. Crohns Colitis 12, 1359–1374 (2018).
Levy, M. et al. Microbiota-modulated metabolites shape the intestinal microenvironment by regulating NLRP6 inflammasome signaling. Cell 163, 1428–1443 (2015).
Zhang, M., Borovikova, L. V., Wang, H., Metz, C. & Tracey, K. J. Spermine inhibition of monocyte activation and inflammation. Mol. Med. 5, 595–605 (1999).
Kim, S. G. et al. Microbiota-derived lantibiotic restores resistance against vancomycin-resistant Enterococcus. Nature 572, 665–669 (2019).
Van De Kamer, J. H., Ten Bokkel Huinink, H. & Weyers, H. A. Rapid method for the determination of fat in feces. J. Biol. Chem. 177, 347–355 (1949).
Monaghan, T. M. et al. Decreased complexity of serum N-glycan structures associates with successful fecal microbiota transplantation for recurrent Clostridioides difficile infection. Gastroenterology 157, 1676–1678 e1673 (2019).
Konturek, P. C. et al. Successful therapy of Clostridium difficile infection with fecal microbiota transplantation. J. Physiol. Pharmacol. 67, 859–866 (2016).
Nuding, S., Frasch, T., Schaller, M., Stange, E. F. & Zabel, L. T. Synergistic effects of antimicrobial peptides and antibiotics against Clostridium difficile. Antimicrob. Agents Chemother. 58, 5719–5725 (2014).
Terveer, E. M. et al. Human transmission of blastocystis by fecal microbiota transplantation without development of gastrointestinal symptoms in recipients. Clin. Infect. Dis. https://doi.org/10.1093/cid/ciz1122 (2019).
Fischer, M. et al. Fecal microbiota transplant in severe and severe-complicated Clostridium difficile: a promising treatment approach. Gut Microbes 8, 289–302 (2016).
Krajicek, E., Bohm, M., Sagi, S. & Fischer, M. Fulminant Clostridium difficile infection cured by fecal microbiota transplantation in a bone marrow transplant recipient with critical neutropenia. ACG Case Rep. J. 6, e00198 (2019).
US Food and Drug Administration. Important safety alert regarding use of fecal microbiota for transplantation and risk of serious adverse reactions due to transmission of multi-drug resistant organisms. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/important-safety-alert-regarding-use-fecal-microbiota-transplantation-and-risk-serious-adverse (2019).
Spanogiannopoulos, P., Bess, E. N., Carmody, R. N. & Turnbaugh, P. J. The microbial pharmacists within us: a metagenomic view of xenobiotic metabolism. Nat. Rev. Microbiol. 14, 273–287 (2016).
Hryckowian, A. J. et al. Microbiota-accessible carbohydrates suppress Clostridium difficile infection in a murine model. Nat. Microbiol. 3, 662–669 (2018).
Collins, J. et al. Dietary trehalose enhances virulence of epidemic Clostridium difficile. Nature 553, 291–294 (2018).
Lay, C. L., Dridi, L., Bergeron, M. G., Ouellette, M. & Fliss, I. L. Nisin is an effective inhibitor of Clostridium difficile vegetative cells and spore germination. J. Med. Microbiol. 65, 169–175 (2016).
Khoruts, A. Targeting the microbiome: from probiotics to fecal microbiota transplantation. Genome Med. 10, 80 (2018).
De Simone, C. The unregulated probiotic market. Clin. Gastroenterol. Hepatol. 17, 809–817 (2019).
Mercer, M. et al. How patients view probiotics: findings from a multicenter study of patients with inflammatory bowel disease and irritable bowel syndrome. J. Clin. Gastroenterol. 46, 138–144 (2012).
Rodgers, B., Kirley, K. & Mounsey, A. PURLs: prescribing an antibiotic? Pair it with probiotics. J. Fam. Pract. 62, 148–150 (2013).
Zmora, N. et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174, 1388–1405.e21 (2018).
Suez, J. et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 174, 1406–1423.e16 (2018).
Borody, T. J., Warren, E. F., Leis, S., Surace, R. & Ashman, O. Treatment of ulcerative colitis using fecal bacteriotherapy. J. Clin. Gastroenterol. 37, 42–47 (2003).
Khoruts, A. & Brandt, L. J. Fecal microbiota transplant: a rose by any other name. Am. J. Gastroenterol. 114, 1176 (2019).
US National Library of Medicine. Intestinal microbiota transplantation ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/results?cond=&term=intestinal+microbiota+transplantation&cntry=&state=&city=&dist=.e.g.,NCT03426683,NCT03648086,NCT03429439,NCT03437876 (2020).
Australian Therapeutic Goods Administration. Faecal microbiota transplant (FMT) product regulation. https://www.tga.gov.au/publication/faecal-microbiota-transplant-fmt-product-regulation (2020).
Khoruts, A., Hoffmann, D. E. & Palumbo, F. The impact of regulatory policies on the future of fecal microbiota transplantation. J. Law. Med. Ethics 47, 482–504 (2020).
Ott, S. J. et al. Efficacy of sterile fecal filtrate transfer for treating patients with Clostridium difficile infection. Gastroenterology 152, 799–811.e7 (2017).
Draper, L. A. et al. Long-term colonisation with donor bacteriophages following successful faecal microbial transplantation. Microbiome 6, 220 (2018).
Zuo, T. et al. Bacteriophage transfer during faecal microbiota transplantation in Clostridium difficile infection is associated with treatment outcome. Gut 67, 634–643 (2017).
Park, H. et al. The success of fecal microbial transplantation in Clostridium difficile infection correlates with bacteriophage relative abundance in the donor: a retrospective cohort study. Gut Microbes 10, 676–687 (2019).
Limon, J. J., Skalski, J. H. & Underhill, D. M. Commensal fungi in health and disease. Cell Host Microbe 22, 156–165 (2017).
Markey, L. et al. Pre-colonization with the commensal fungus Candida albicans reduces murine susceptibility to Clostridium difficile infection. Gut Microbes 9, 497–509 (2018).
Zuo, T. et al. Gut fungal dysbiosis correlates with reduced efficacy of fecal microbiota transplantation in Clostridium difficile infection. Nat. Commun. 9, 3663 (2018).
Moyes, D. L. & Naglik, J. R. The mycobiome: influencing IBD severity. Cell Host Microbe 11, 551–552 (2012).
Kostic, A. D., Xavier, R. J. & Gevers, D. The microbiome in inflammatory bowel disease: current status and the future ahead. Gastroenterology 146, 1489–1499 (2014).
Acknowledgements
A.K. and M.J.S. acknowledge that this work was supported in part by the US Department of Defense grant W81XWH-17-1-0636.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
A.K. and M.J.S. have patents related to the preparation of faecal microbiota for transplantation. C.S. declares no competing interests.
Additional information
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks J. Allegretti, G. Cammarota, B. Mullish and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khoruts, A., Staley, C. & Sadowsky, M.J. Faecal microbiota transplantation for Clostridioides difficile: mechanisms and pharmacology. Nat Rev Gastroenterol Hepatol 18, 67–80 (2021). https://doi.org/10.1038/s41575-020-0350-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-020-0350-4
This article is cited by
-
Microbiota–gut–brain axis and its therapeutic applications in neurodegenerative diseases
Signal Transduction and Targeted Therapy (2024)
-
Current perspectives on fecal microbiota transplantation in inflammatory bowel disease
Indian Journal of Gastroenterology (2024)
-
Successful Treatment of Recurrent Clostridioides difficile Infection Using a Novel, Drinkable, Oral Formulation of Fecal Microbiota
Digestive Diseases and Sciences (2024)
-
Bacillus velezensis DSM 33864 reduces Clostridioides difficile colonization without disturbing commensal gut microbiota composition
Scientific Reports (2023)
-
Impact of environment on transmission of antibiotic-resistant superbugs in humans and strategies to lower dissemination of antibiotic resistance
Folia Microbiologica (2023)