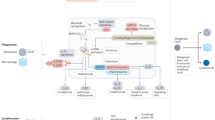
Abstract
With the increase in the number of novel drugs for inflammatory bowel disease (IBD), comparing therapeutic options or strategies has become a key challenge in IBD trials. Head-to-head trials designed and powered to enable formal comparisons are the gold standard in comparative research. Indeed, these trials are requested by some health authorities for evaluating the positioning of new treatments in IBD, as well as helping prescribing physicians to select the most appropriate treatment options for their patients. Despite head-to-head trials including aminosalicylate therapy in IBD having been performed decades ago, the first results of a randomized controlled trial directly comparing biologic agents with different modes of action have only now been published, mainly owing to important methodological issues. This Perspective provides an overview of the past, current and future concepts in IBD trial design, with a detailed focus on the role of comparative research and the challenges and pitfalls in undertaking and interpreting the results from such studies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
05 January 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41575-020-00409-x.
References
Truelove, S. C. & Witts, L. J. Cortisone in ulcerative colitis. BMJ 2, 1041–1048 (1955).
Peyrin-Biroulet, L. et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): determining therapeutic goals for treat-to-target. Am. J. Gastroenterol. 110, 1324–1338 (2015).
Chateau, T. et al. Histological remission in ulcerative colitis. Am. J. Gastroenterol. 115, 179–189 (2020).
Sabino, J., Verstockt, B., Vermeire, S. & Ferrante, M. New biologics and small molecules in inflammatory bowel disease: an update. Therap. Adv. Gastroenterol. 12, 175628481985320 (2019).
Summers, R. W. et al. National Cooperative Crohn’s Disease Study: results of drug treatment. Gastroenterology 77, 847–869 (1979).
Courtney, M. G. et al. Randomised comparison of olsalazine and mesalazine in prevention of relapses in ulcerative colitis. Lancet 339, 1279–1281 (1992).
Kamm, M. A. et al. Once-daily, high-concentration MMX mesalamine in active ulcerative colitis. Gastroenterology 132, 66–75 (2007).
Jeong, D. Y. et al. Induction and maintenance treatment of inflammatory bowel disease: a comprehensive review. Autoimmun. Rev. 18, 439–454 (2019).
Weinblatt, M. E. et al. Head-to-head comparison of subcutaneous abatacept versus adalimumab for rheumatoid arthritis: findings of a phase IIIb, multinational, prospective, randomized study. Arthritis Rheum. 65, 28–38 (2013).
Durelli, L. et al. Every-other-day interferon beta-1b versus once-weekly interferon beta-1a for multiple sclerosis: results of a 2-year prospective randomised multicentre study (INCOMIN). Lancet 359, 1453–1460 (2002).
Gordon, K. B. et al. Efficacy and safety of risankizumab in moderate-to-severe plaque psoriasis (UltIMMa-1 and UltIMMa-2): results from two double-blind, randomised, placebo-controlled and ustekinumab-controlled phase 3 trials. Lancet 392, 650–661 (2018).
Dulai, P. S., Singh, S., Ohno-Machado, L. & Sandborn, W. J. Population health management for inflammatory bowel disease. Gastroenterology 154, 37–45 (2018).
Ma, C. et al. Systematic review with meta-analysis: endoscopic and histologic placebo rates in induction and maintenance trials of ulcerative colitis. Aliment. Pharmacol. Ther. 47, 1578–1596 (2018).
Duijvestein, M. et al. Response to placebo, measured by endoscopic evaluation of Crohn’s disease activity, in a pooled analysis of data from 5 randomized controlled induction trials. Clin. Gastroenterol. Hepatol. https://doi.org/10.1016/j.cgh.2019.08.025 (2019).
Peyrin-Biroulet, L., Lopez, A. & Sandborn, W. Head-to-head comparative studies: challenges and opportunities? J. Crohns Colitis 11, S567–S575 (2017).
Sands, B. E. et al. Vedolizumab versus adalimumab for moderate-to-severe ulcerative colitis. N. Engl. J. Med. 381, 1215–1226 (2019).
Digby-Bell, J. L., Atreya, R., Monteleone, G. & Powell, N. Interrogating host immunity to predict treatment response in inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 17, 9–20 (2020).
Liberati, A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339, b2700 (2009).
Gurevitch, J., Koricheva, J., Nakagawa, S. & Stewart, G. Meta-analysis and the science of research synthesis. Nature 555, 175–182 (2018).
Ioannidis, J. P. A. The mass production of redundant, misleading, and conflicted systematic reviews and meta-analyses. Milbank Q. 94, 485–514 (2016).
Rogozin´ska, E., Marlin, N., Thangaratinam, S., Khan, K. S. & Zamora, J. Meta-analysis using individual participant data from randomised trials: opportunities and limitations created by access to raw data. Evid. Based Med. 22, 157–162 (2017).
Rouse, B., Chaimani, A. & Li, T. Network meta-analysis: an introduction for clinicians. Intern. Emerg. Med. 12, 103–111 (2017).
Ghosh, S. et al. Interpreting registrational clinical trials of biological therapies in adults with inflammatory bowel diseases. Inflamm. Bowel Dis. 22, 2711–2723 (2016).
Corrigan-Curay, J., Sacks, L. & Woodcock, J. Real-world evidence and real-world data for evaluating drug safety and effectiveness. JAMA 320, 867–868 (2018).
Blonde, L., Khunti, K., Harris, S. B., Meizinger, C. & Skolnik, N. S. Interpretation and impact of real-world clinical data for the practicing clinician. Adv. Ther. 35, 1763–1774 (2018).
Olivera, P., Danese, S., Jay, N., Natoli, G. & Peyrin-Biroulet, L. Big data in IBD: a look into the future. Nat. Rev. Gastroenterol. Hepatol. 16, 312–321 (2019).
Ha, C., Ullman, T. A., Siegel, C. A. & Kornbluth, A. Patients enrolled in randomized controlled trials do not represent the inflammatory bowel disease patient population. Clin. Gastroenterol. Hepatol. 10, 1002–1007 (2012).
Pouillon, L. et al. Mucosal healing and long-term outcomes of patients with inflammatory bowel diseases receiving clinic-based vs trough concentration-based dosing of infliximab. Clin. Gastroenterol. Hepatol. 16, 1276–1283 (2018).
Vande Casteele, N. et al. Trough concentrations of infliximab guide dosing for patients with inflammatory bowel disease. Gastroenterology 148, 1320–1329 (2015).
D’Haens, G. et al. Increasing infliximab dose based on symptoms, biomarkers, and serum drug concentrations does not increase clinical, endoscopic, and corticosteroid-free remission in patients with active luminal Crohn’s disease. Gastroenterology 154, 1343–1351 (2018).
Austin, P. C. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar. Behav. Res. 46, 399–424 (2011).
Fleischmann, R., Landewé, S. & Smolen, J. Review of head-to-head study designs in rheumatoid arthritis. Semin. Arthritis Rheum. 46, 279–285 (2016).
Faleck, D. et al. Comparative effectiveness of vedolizumab and TNF-antagonist therapy in ulcerative colitis: a multicentre consortium propensity score-matched analysis [abstract OP026]. J. Crohns Colitis 12, S019 (2018).
Koliani-Pace, J. L. et al. Changes in vedolizumab utilization across US academic centers and community practice are associated with improved effectiveness and disease outcomes. Inflamm. Bowel Dis. 25, 1854–1861 (2019).
Khanna, R. et al. Early combined immunosuppression for the management of Crohn’s disease (REACT): a cluster randomised controlled trial. Lancet 386, 1825–1834 (2015).
Armstrong, K. Methods in comparative effectiveness research. J. Clin. Oncol. 30, 4208–4214 (2012).
Lathyris, D. N., Patsopoulos, N. A., Salanti, G. & Ioannidis, J. P. A. Industry sponsorship and selection of comparators in randomized clinical trials. Eur. J. Clin. Invest. 40, 172–182 (2010).
Flacco, M. E. et al. Head-to-head randomized trials are mostly industry sponsored and almost always favor the industry sponsor. J. Clin. Epidemiol. 68, 811–820 (2015).
Jørgensen, K. K. et al. Switching from originator infliximab to biosimilar CT-P13 compared with maintained treatment with originator infliximab (NOR-SWITCH): a 52-week, randomised, double-blind, non-inferiority trial. Lancet 389, 2304–2316 (2017).
Olivera, P. et al. Physicians’ perspective on the clinical meaningfulness of inflammatory bowel disease trial results: an International Organization for the Study of Inflammatory Bowel Disease (IOIBD) survey. Aliment. Pharmacol. Ther. 47, 773–783 (2018).
Dunn, D. T., Copas, A. J. & Brocklehurst, P. Superiority and non-inferiority: two sides of the same coin? Trials 19, 499 (2018).
Tsui, M., Rehal, S., Jairath, V. & Kahan, B. C. Most noninferiority trials were not designed to preserve active comparator treatment effects. J. Clin. Epidemiol. 110, 82–89 (2019).
Wan, M. T. et al. Head-to-head trials of systemic psoriasis therapies: a systematic review of study design and maximum acceptable treatment differences. J. Eur. Acad. Dermatol. Venereol. 33, 42–55 (2019).
Sandborn, W. J. et al. Induction of clinical and colonoscopic remission of mild-to-moderate ulcerative colitis with budesonide MMX 9 mg: pooled analysis of two phase 3 studies. Aliment. Pharmacol. Ther. 41, 409–418 (2015).
Gross, V. et al. 3g mesalazine granules are superior to 9mg budesonide for achieving remission in active ulcerative colitis: a double-blind, double-dummy, randomised trial. J. Crohns Colitis 5, 129–138 (2011).
Lichtenstein, G. R. et al. Effect of once- or twice-daily MMX mesalamine (SPD476) for the induction of remission of mild to moderately active ulcerative colitis. Clin. Gastroenterol. Hepatol. 5, 95–102 (2007).
Kamm, M. A. et al. Effect of extended MMX mesalamine therapy for acute, mild-to-moderate ulcerative colitis. Inflamm. Bowel Dis. 15, 1–8 (2009).
World Medical Association. WMA Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects. WMA https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/ (2018)
Panaccione, R. et al. Combination therapy with infliximab and azathioprine is superior to monotherapy with either agent in ulcerative colitis. Gastroenterology 146, 392–400 (2014).
Colombel, J. F. et al. Infliximab, azathioprine, or combination therapy for Crohn’s disease. N. Engl. J. Med. 362, 1383–1395 (2010).
Schulz, K. F. & Grimes, D. A. Blinding in randomised trials: hiding who got what. Lancet 359, 696–700 (2002).
Wan, M., Orlu-Gul, M., Legay, H. & Tuleu, C. Blinding in pharmacological trials: the devil is in the details. Arch. Dis. Child. 98, 656–659 (2013).
Williams, J. G. et al. Infliximab versus ciclosporin for steroid-resistant acute severe ulcerative colitis (CONSTRUCT): a mixed methods, open-label, pragmatic randomised trial. Lancet Gastroenterol. Hepatol. 1, 15–24 (2016).
Yoo, D. H. et al. Efficacy and safety of CT-P13 (biosimilar infliximab) in patients with rheumatoid arthritis: comparison between switching from reference infliximab to CT-P13 and continuing CT-P13 in the PLANETRA extension study. Ann. Rheum. Dis. 76, 355–363 (2017).
Park, W. et al. Efficacy and safety of switching from reference infliximab to CT-P13 compared with maintenance of CT-P13 in ankylosing spondylitis: 102-week data from the PLANETAS extension study. Ann. Rheum. Dis. 76, 346–354 (2017).
Colombel, J.-F. et al. Effect of tight control management on Crohn’s disease (CALM): a multicentre, randomised, controlled phase 3 trial. Lancet 390, 2779–2789 (2017).
European Medicines Agency. Guideline on the Development of New Medicinal Products for the Treatment of Ulcerative Colitis. EMA https://www.ema.europa.eu/en/documents/scientific- guideline/guideline-development-new-medicinal-products-treatment-ulcerative-colitis-revision-1_en.pdf (2018).
European Medicines Agency. Guideline on the Development of New Medicinal Products for the Treatment of Crohn’s Disease. EMA https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-development-new-medicinal-products-treatment-crohns-disease-revision-2_en.pdf (2018).
Reinisch, W. et al. Comparison of the EMA and FDA guidelines on ulcerative colitis drug development. Clin. Gastroenterol. Hepatol. 17, 1673–1679.e1 (2019).
Ma, C. et al. Heterogeneity in definitions of efficacy and safety endpoints for clinical trials of Crohn’s disease: a systematic review. Clin. Gastroenterol. Hepatol. 16, 1407–1419 (2018).
Vuitton, L. et al. Defining endoscopic response and remission in ulcerative colitis clinical trials: an international consensus. Aliment. Pharmacol. Ther. 45, 801–813 (2017).
Vuitton, L. et al. IOIBD technical review on endoscopic indices for Crohn’s disease clinical trials. Gut 65, 1447–1455 (2016).
Danese, S. et al. Identification of endpoints for development of antifibrosis drugs for treatment of Crohn’s disease. Gastroenterology 155, 76–87 (2018).
Pittet, V. E. H. et al. Differences in outcomes reported by patients with inflammatory bowel diseases vs their health care professionals. Clin. Gastroenterol. Hepatol. 17, 2050–2059 (2019).
Bryant, R. V. et al. Beyond endoscopic mucosal healing in UC: histological remission better predicts corticosteroid use and hospitalisation over 6 years of follow-up. Gut 65, 408–414 (2016).
D’Haens, G. et al. Challenges to the design, execution, and analysis of randomized controlled trials for inflammatory bowel disease. Gastroenterology 143, 1461–1469 (2012).
Feagan, B. et al. Performance of Crohn’s disease clinical trial endpoints based upon different cutoffs for patient reported outcomes or endoscopic activity: analysis of EXTEND data. Inflamm. Bowel Dis. 24, 932–942 (2018).
Ponsioen, C. Y. et al. Laparoscopic ileocaecal resection versus infliximab for terminal ileitis in Crohn’s disease: a randomised controlled, open-label, multicentre trial. Lancet Gastroenterol. Hepatol. 2, 785–792 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02136069 (2020).
Vande Casteele, N., Herfarth, H., Katz, J., Falck-Ytter, Y. & Singh, S. American Gastroenterological Association Institute technical review on the role of therapeutic drug monitoring in the management of inflammatory bowel diseases. Gastroenterology 153, 835–857.e6 (2017).
Feagan, B. G. et al. Methotrexate in combination with infliximab is no more effective than infliximab alone in patients with Crohn’s disease. Gastroenterology 146, 681–688 (2014).
Laharie, D. et al. Ciclosporin versus infliximab in patients with severe ulcerative colitis refractory to intravenous steroids: a parallel, open-label randomised controlled trial. Lancet 380, 1909–1915 (2012).
Paul, S. et al. Pharmacokinetics of adalimumab in inflammatory bowel diseases: a systematic review and meta-analysis. Inflamm. Bowel Dis. 20, 1288–1295 (2014).
Peyrin-Biroulet, L. et al. Loss of response to vedolizumab and ability of dose intensification to restore response in patients with Crohn’s disease or ulcerative colitis: a systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 17, 838–846 (2019).
Sandborn, W. J. et al. Efficacy and safety of vedolizumab subcutaneous formulation in a randomized trial of patients with ulcerative colitis. Gastroenterology 158, 562–572.e12 (2020).
D’Haens, G. et al. Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn’s disease: an open randomised trial. Lancet 371, 660–667 (2008).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02871635 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02163759 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02171429 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03616821 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03464136 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03662542 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03759288 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/record/NCT03926130 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03466411 (2020).
Verstockt, B. et al. Low TREM1 expression in whole blood predicts anti-TNF response in inflammatory bowel disease. EBioMedicine 40, 733–742 (2019).
Dulai, P. S. et al. Approaches to integrating biomarkers into clinical trials and care pathways as targets for treatment of inflammatory bowel diseases. Gastroenterology 157, 1032–1043.e1 (2019).
Author information
Authors and Affiliations
Contributions
L.P., S.T. and L.P.-B. researched data for and wrote the article. All authors made substantial contributions to discussion of content and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
L.P. received personal fees from Abbvie, Ferring, Takeda; and advisory board fees from Takeda. S.T. received research support from AbbVie, Buhlmann, Celgene, International Organization for the Study of Inflammatory Bowel Disease, Janssen, Lilly, Takeda, UCB, Vifor and Norman Collisson Foundation; consulting fees from AbbVie, Allergan, Amgen, Arena, Asahi, Astellas, Biocare, Biogen, Boehringer Ingelheim, Bristol-Myers Squibb, Buhlmann, Celgene, Chemocentryx, Cosmo, Enterome, Ferring, Giuliani SpA, GSK, Genentech, Gilead, Immunocore, Immunometabolism, Indigo, Janssen, Lexicon, Lilly, Merck, MSD, Neovacs, Novartis, NovoNordisk, NPS Pharmaceuticals, Pfizer, Proximagen, Receptos, Roche, Sandoz, Sensyne, Shire, Sigmoid Pharma, SynDermix, Takeda, Theravance, Tillotts, Topivert, UCB, VHsquared, Vifor and Zeria; speaker fees from AbbVie, Amgen, Biogen, Ferring, Janssen, Shire and Takeda; and declares no stock or share options. P.B. received educational grants from AbbVie, Mundipharma, Pfizer and Janssen; speaker fees from AbbVie, Takeda and Pfizer; and advisory board fees from Hospira, Janssen, MSD, Mundipharma, Roche, Pfizer, Takeda, Sandoz and Pentax. S.D. has served as a speaker, a consultant and an advisory board member for Abbvie, Ferring, Hospira, Johnson & Johnson, Merck, Millennium Takeda, Mundipharma, Pfizer, Tigenix, UCB Pharma and Vifor. L.P.-B. received personal fees from AbbVie, Janssen, Genentech, Ferring, Tillots, Pharmacosmos, Celltrion, Takeda, Boerhinger Ingelheim, Pfizer, Index Pharmaceuticals Sandoz, Celgene, Biogen, Samsung Bioepis, Alma, Sterna, Nestle, Enterome, Allergan, MSD, Roche, Arena, Gilead, Hikma, Amgen, BMS, Vifor, Norgine, Mylan, Lilly, Fresenius Kabi, Oppilan Pharma, Sublimity Therapeutics, Applied Molecular Transport, OSE Immunotherapeutics, Enthera and Theravance; grants from Abbvie, MSD and Takeda; and stock options from CTMA.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
ClinicalTrials.gov: https://www.clinicaltrials.gov/
Supplementary information
Rights and permissions
About this article
Cite this article
Pouillon, L., Travis, S., Bossuyt, P. et al. Head-to-head trials in inflammatory bowel disease: past, present and future. Nat Rev Gastroenterol Hepatol 17, 365–376 (2020). https://doi.org/10.1038/s41575-020-0293-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-020-0293-9
This article is cited by
-
Mucosal IL23A expression predicts the response to Ustekinumab in inflammatory bowel disease
Journal of Gastroenterology (2021)
-
Safety of S1P Modulators in Patients with Immune-Mediated Diseases: A Systematic Review and Meta-Analysis
Drug Safety (2021)
-
TNF-shaped microbiota promotes cancer
Nature Cancer (2020)