Abstract
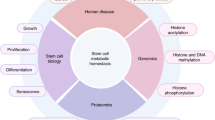
Metabolic diseases, including obesity, diabetes mellitus and cardiovascular disease, are a major threat to health in the modern world, but efforts to understand the underlying mechanisms and develop rational treatments are limited by the lack of appropriate human model systems. Notably, advances in stem cell and organoid technology allow the generation of cellular models that replicate the histological, molecular and physiological properties of human organs. Combined with marked improvements in gene editing tools, human stem cells and organoids provide unprecedented systems for studying mechanisms of metabolic diseases. Here, we review progress made over the past decade in the generation and use of stem cell-derived metabolic cell types and organoids in metabolic disease research, especially obesity and liver diseases. In particular, we discuss the limitations of animal models and the advantages of stem cells and organoids, including their application to metabolic diseases. We also discuss mechanisms of drug action, understanding the efficacy and toxicity of existing therapies, screening for new treatments and pursuing personalized therapies. We highlight the potential of combining stem cell-derived organoids with gene editing and functional genomics to revolutionize the approach to finding treatments for metabolic diseases.
Key points
-
Animal models of metabolic diseases are limited by unique human biology.
-
Human adipose and liver organoid models can be generated from pluripotent stem cells and healthy or diseased tissues.
-
Adipose stem cells and organoids can model white and brown adipogenesis and the progression of obesity.
-
Hepatic organoids can be used to model acquired metabolic diseases of the liver.
-
Human stem cells and organoid models have utility in drug discovery and precision medicine for metabolic diseases.
Similar content being viewed by others
Introduction
The obesity pandemic constitutes a major public health concern and contributes to numerous chronic diseases, including diabetes mellitus, cardiovascular diseases, liver diseases and some cancers1. Obesity reflects an increase in adipose tissue, which is the main energy storage organ in the body2. Adipose tissue has a major role in the regulation of whole-body energy homeostasis, and both a surfeit (as in obesity) as well as a dearth of adipose tissue, known as lipodystrophy, are tightly related to the development of type 2 diabetes mellitus (T2DM) and other metabolic disorders3,4.
There are three main types of adipocytes: lipid-filled white adipocytes, metabolically healthy inducible beige (or brite) adipocytes and thermogenic and mitochondrial-enriched brown adipocytes2,5. Understanding the ontogeny of the different adipocyte populations is crucial to developing new therapeutic strategies to combat obesity and T2DM. Most studies have focused on studying adipose tissue using rodent models, which have identified numerous potential therapeutic targets for treating obesity and metabolic diseases6,7. However, these studies have been difficult to translate into humans due to the major differences between human physiology and animal model systems. In particular, the origin, identity and heterogeneity of brown adipose tissue (BAT) differ between mouse models and humans. For example, the activity of human BAT is driven by the β2 adrenergic receptor, in contrast to the β3 adrenergic receptor in adipose tissue of mice8. Also, there is a lack of an appropriate mouse model that mimics normal human BAT development9, as mice are typically housed at temperatures below their thermoneutral zone, while humans usually live in thermoneutral conditions. Thus, there is a great need to generate mature human adipocytes, or adipose organoid systems that model the complexity and crosstalk of different cell types within adipose tissue, to develop new therapeutic strategies for adipose-related metabolic diseases.
Another important metabolic organ is the liver, which is composed of hepatocytes, cholangiocytes and non-parenchymal cells that together perform important metabolic, detoxification, endocrine and exocrine functions required for organismal homeostasis10. The prevalence of liver diseases is rising across the globe, largely related to obesity but also due to genetic, infectious, toxic or malignant aetiologies11. In obesity, insulin resistance drives lipid accumulation in the liver due to increased de novo hepatic lipogenesis as well as increased adipose tissue lipolysis and adipokine secretion, which modulate liver metabolism12,13. Similarly, hepatokines such as fibroblast growth factor 21 and liver-derived exosomes regulate adipose physiology, such as insulin sensitivity, inflammation and thermogenesis14,15.
Thus, obesity is highly related to non-alcoholic fatty liver disease (NAFLD), which can progress from steatosis to more advanced non-alcoholic steatohepatitis (NASH) that is characterized by progressive inflammation and fibrosis16,17. NASH is a precursor of hepatocellular carcinoma as well as liver failure, which is a leading indication for liver transplantation18. However, there is currently no FDA-approved therapy for NASH19. Numerous cellular models and animal models have been used to explore the underlying mechanisms of liver development and pathogenesis, but little progress has been made towards developing successful therapeutic strategies for liver diseases, especially NASH, due to the limitations of traditional cellular and animal models.
Since the establishment of human embryonic stem cells (hESCs) and induced pluripotent stem cell (iPSC) technology20,21,22, various disease-relevant cell types can be generated from these human pluripotent stem cells (hPSCs) by stepwise differentiation protocols that mimic in vivo organogenesis23. In the past decade, the modelling of human physiology and disease has benefited from the development of organoids. An organoid is defined by the HPB (hepatic, pancreatic and biliary) Organoid Consortium as a three-dimensional structure derived from stem cells, progenitor cells and/or differentiated cells that self-organize through cell–cell and cell–matrix interactions to recapitulate aspects of the native tissue structure and function in vitro24, unlike traditional cellular and animal models (Supplementary Table 1). In this Review, we focus on progress made over the past decade in the generation and use of stem cell-derived adipose and liver organoids that have promise in furthering our understanding of basic mechanisms and facilitating the development of new and personalized therapies for metabolic diseases.
The challenges of animal models
Many biological processes are evolutionarily conserved between humans and animals, especially mammals. In keeping with this fact, animal model systems have been of huge benefit for biomedical research and translational medicine25,26. These models provide unique insights into the pathophysiology and progression of disease, and often reveal novel therapeutic targets for diseases. Moreover, they exhibit a degree of feasibility of environmental and genetic manipulation that is not realistic in humans, and provide a platform to test drug efficacy in preclinical studies. However, the results of preclinical drug safety or efficacy studies often lead to the failure of clinical trials, the so called ‘valley of death’ for drug develpment27. Indeed, many clinical trials of drug candidates that were promising in preclinical models of obesity, T2DM and NASH have been terminated in the past 20 years28,29,30.
The failures in translating animal research into the clinic are largely due to the species specificity of genomes and epigenomes, and differences in development, metabolism and drug responses between animal models and humans. For instance, most mouse models are inbred, but each human individual has a unique genomic background. Patient-specific disease onset, progression and drug responses are poorly modelled using animal models31. Additionally, there are large differences in the development, structure and function of animal organs compared with human organs32. Moreover, animals used in preclincal studies are usually smaller, faster-growing, and with a shorter life expectancy than humans. They also show great dissimilarities in metabolism, such as energy expenditure33. There are large differences in drug absorption, distribution and metabolism that could explain the discrepancies between drug efficacy and toxicity between humans and animal models34. Thus, there is a great need to develop new models that recapitulate many aspects of human pathophysiology.
The promise of human stem cells
One of the great obstacles to developing new treatments for human disease is the scarcity of suitable preclinical models. In this context, the use of human stem cells to model human disease is very promising. iPSC technology overcomes ethical concerns around using hESCs that are derived from human embryos and allows the generation of patient-specific pluripotent stem cells, which provide a promising platform for drug discovery and cell therapy in vitro. The road to precision medicine has also benefited from the biobanking of patient-specific iPSCs. Combined with state-of-art gene editing technology, such as CRISPR–Cas9 or base editing tools35,36,37, isogenic iPSC lines can be generated to systematically understand the mechanisms of disease initiation and progression38. It can also be used to interrogate the biological function of disease risk alleles implicated by genome-wide association studies (GWAS), which do not have the power to determine the underlying target genes and cell types39.
Emergence of human organoid technology
As 2D cellular models do not capture human pathophysiology well, 3D organ culture technology (referred to as organoids) has been developed and widely utilized in biomedical research in the past decade40,41. Since the first organoid methodology was developed from intestine leucine-rich repeat-containing G protein-coupled receptor 5 (LGR5)-positive stem cells42, the field of organoid biology has made considerable progress, and was recognized as the Method of the Year in 2017 by Nature Methods43. Organoids closely resemble their native tissue or organ in terms of their gene profile, tissue architecture and function. Organoids can contain a single epithelial cell type only (epithelial organoids), multiple cell types of a single organ (multi-tissue organoids) or even reconstitute multiple organs (multi-organ organoids)24. They thus provide a promising platform for developmental biology, disease modelling, drug screening and cell therapy. Currently, the majority of human organs, such as the brain, liver, adipose tissue, pancreas, heart, kidney, gut, stomach and lung, have their own in vitro organoid models derived either from hPSCs, adult tissue stem cells (ASCs) or even fully differentiated primary cells42,44,45,46,47,48,49,50,51,52.
The principle of developing hPSC-derived organoids involves mimicking the organogenesis that occurs during embryo development in vitro. Based on previous knowledge of organ development in model animals, existing hPSC differentiation protocols and emerging datasets of human early embryo development, appropriate biochemical cues are applied at certain time windows to direct the differentiation of hPSCs into an organ of interest in a stepwise manner. When embedded in Matrigel as a scaffold, they self-organize into organoids, which have a similar gene expression profile, cellular composition, tissue architecture and function to the target organ, although the maturation level of hPSC-derived organoids still requires further improvement51.
Development of human organoid models
Inspired by the limitations of animal models, a variety of human cell and tissue models have been developed, including 2D immortalized cell lines, patient-derived xenografts, stem cell-derived cell types and organoids. Each model has its own benefits and drawbacks (Supplementary Table 1). For example, 2D human cell lines can be easily maintained and expanded and adapt to various genetic manipulation tools, which make them ideal for studying mechanisms of diseases progression as well as for drug screening. However, these 2D models do not recapitulate human tissue characteristics, such as cell–cell interaction and cell–niche interaction, which are critical aspects of human biology53. Moreover, while patient-derived xenografts do recapitulate the complexity of the native tissue, this system is time-consuming to establish, expensive, and not amenable to high-throughput studies54. Thus there is great interest in hPSCs and organoids, which have been used to advance basic biomedical knowledge and applied largely to regenerative medicine over the past 20 years. In this Review, we focus on the progress in the past 10 years in generating organoids from two important metabolic tissues, adipose tissue and liver, which play substantial roles in maintaining metabolic health. Progress in organoids related to other metabolic tissues, such as the pancreas, has been intensively reviewed elsewhere47,55.
hPSC-derived models of adipose tissue
hPSC-derived adipocytes
hPSC-derived adipocytes provide a unique model to study metabolic diseases such as obesity and T2DM and to address questions that cannot be answered by animal models alone. Several groups have established protocols to effectively induce hPSCs to differentiate into white adipocytes in a stepwise manner56,57,58,59,60,61 (Fig. 1), through ectopic expression of adipocyte lineage determination factors or using specific signal molecules that are important for adipocyte development in vitro, as discussed in a recent review62.
a | Different methodologies can be used to guide the differentiation of white or brown adipocytes from human pluripotent stem cells58,60,61. b | Adipose tissue stromal vascular fraction (SVF) can be cultured in hang drop or low-attachment plates to grow as spheroids72. Moreover, when embedded in Matrigel, SVF cells can develop into vascularized adipose organoids or immune cell-contained adipose organoids under optimal culture conditions50,74,76. Furthermore, tissue engineering, bioprinting and organ-on-chip technology can help create more complex and mature adipose organoids that facilitate the study of adipose development, diseases and crosstalk with other organs77,78,79,80,81. BAT, brown adipose tissue; hPSC, human pluripotent stem cell; MPC, mesenchymal progenitor cell.
An example of these initial techniques is the use of embryoid bodies to generate adipocytes. The embryoid bodies were first formed from hPSCs in suspension and then differentiated into mesenchymal stem cells (MSCs), which were further induced to generate white adipocytes by defined medium and/or specific transcriptional factors, such as the nuclear receptor peroxisome proliferator activated receptor (PPARγ). These adipocytes expressed mature adipocyte markers and exhibited functional properties such as lipid catabolism and insulin responsiveness58. Interestingly, brown adipocytes were also effectively created by treating the hPSC-derived MSCs with two more transcriptional factors CCAAT–enhancer-binding protein-β (CEBPB) and PR domain containing 16 (PRDM16)58,63, which are key regulators of brown adipocyte development and function64. Similarly, overexpression of PAX3, a paraxial mesoderm marker, effectively induced hPSCs into brown adipocytes57. Since the induced adipocytes generated ectopic fat pads resembling BAT in terms of morphology and function when transplanted into mice, these cells provide a valuable resource for potential cell therapy to combat obesity and T2DM58. However, the risks involved in transplanting cells containing transgenes limits their therapeutic potential, such as immune disruption and tumour formation. Thus integration-free methods for brown adipocyte generation are needed.
Inspired by the close link between bone marrow adipogenesis and haematopoiesis65, a protocol for a high-efficiency (>90%) differentiation of hPSCs into brown adipocytes was successfully developed using specific growth factors that promote a haematopoietic identity without the addition of exogenous genes60. These hPSC-derived brown adipocytes expressed BAT-specific genes and showed thermogenic activation upon β3-adrenergic receptor stimulation. Excitingly, transplantation of hPSC-derived brown adipocytes into mice rescued the glucose intolerance caused by transplantation of white adipocytes60. However, the differentiation efficiencies of brown adipocytes substantially differed among human iPSC (hiPSC) lines, which limited their clinical applications. In the past 5 years, however, three groups used previous advances in the understanding of the developmental pathways leading to differentiation of brown or beige adipocytes in mice and humans to develop robust systems to induce brown or beige adipocytes56,61,66. Specifically, upon recognition that brown adipocytes arose from paraxial mesoderm progenitor cells, a multistep protocol was developed to recapitulate brown adipocyte development from hPSCs61. These hPSC-derived brown adipocytes are metabolically active and responsive to cAMP-dependent signalling. Importantly, transplantation of these hPSC-derived brown adipocytes reduced circulating levels of glucose in hyperglycaemic mice compared with the levels in mice receiving paraxial mesoderm progenitor cells, possibly through elevated whole-body energy expenditure61.
Although these studies suggest that hPSC-derived white adipocytes and brown and/or beige adipocytes have potential as tools for disease modelling and therapeutic development, adipose tissue is a highly heterogeneous organ that consists of many other cell types besides adipocytes, such as immune cells, neurons and endothelial cells67. Thus, understanding the various cell–cell interactions within adipose tissues will be critical for delineating the mechanisms of adipose development, activation or dysfunction and will be important for developing new therapeutic targets for metabolic diseases. However, cell–cell interactions cannot be adequately recapitulated in 2D hPSC-derived adipocytes.
Simple adipose organoids
In practice, the term ‘adipose organoid’ has often been used interchangeably with another term, ‘adipose spheroid’, although there are substantial differences between these culture systems68. Adipose spheroids are usually simple clusters of immortal or primary cell types derived from adipose tissue that form into spheroids due to cell aggregation68, while adipose organoids are self-organized from adipose progenitor cells or hPSCs, and have structures and functions that resemble those of adipose tissue. Adipose organoids and spheroids exhibit greater utility than classic adipose cellular models such as ceiling and/or Transwell cultures and PSC-derived adipocytes in many scenarios, including drug screening and disease modelling, due to their greater morphological and functional resemblance to adipose tissue. In the following sections, we discuss the generation and applications of adipose organoids without distinguishing between adipose organoids and adipose spheroids.
In an effort to compare adipose depot-specific adipogenesis and functions, 3D culture systems have been used to differentiate preadipocytes in vitro from the various adipose tissue depots69. When cultured within a collagen hydrogel, preadipocytes from visceral depots were differentiated into mature adipocytes to the same extent as subcutaneous preadipocytes, with maintenance of depot-specific gene expression signatures69. In subsequent studies, human adipose-derived stem cells were cultured on elastin-like polypeptide (ELP)–polyethyleneimine (PEI) copolymer-coated plates, because the PEI segment encourages spheroid formation and the ELP segment allows surface attachment of the formed spheroids. This culture system allowed the generation of functional adipocytes that responded well to fatty acid treatment70. This study demonstrates the feasibility of using human primary cells to generate patient-specific adipose organoids for disease modelling, drug screening and personalized medicine.
However, the broad use of collagen, hydrogel, ELP–PEI or other matrix components that help with organoid formation limits the clinical applications of these organoids due to undefined components of these matrix systems71. Thus a scaffold-free method was developed to generate 3D adipose spheroids for metabolism research and drug discovery72. Preadipocytes self-organized into spheroids in hanging drops and were then maintained in low attachment plates for long-term culture. Interestingly, these differentiated adipose spheroids were sensitive to environmental toxins, upregulating cytokine production and downregulating adiponectin secretion in response to polychlorinated biphenyls72. Importantly, due to the uniform property of these adipose spheroids and the scaffold-free nature of this culture system, high-throughput screening was successfully performed to identify modulators of adipogenesis72. 3D spheroids derived from BAT using a similar strategy were also found to retain expression of brown adipocyte markers better than 2D cultures derived from the same tissue73. Unfortunately, although this organized, compact tissue was rich in collagen fibrils, it contained only a few small clusters of UCP1+ cells73. Thus, more robust human adipose organoids need to be developed, especially since no protocol to date has been sufficient to model complex adipose-related metabolic diseases using hPSCs. The development of integration-free, efficient and reproducible protocols of hPSC-derived adipocytes will provide a great basis for the generation of adipose organoids directly from hPSCs.
Complex adipose organoids
Ideal adipose organoids would consist of every in vivo component of adipose tissue, including adipocytes, connective tissue, vasculature and nerves. Many efforts have been made to generate adipose organoids that model cell–cell interaction and cell–environment communication within adipose tissue (Fig. 1). To model the inflammatory response of adipose tissue, primary stromal vascular fraction (SVF) cells that maintain resident immune cells were differentiated into scaffold-free 3D adipose tissue organoids without further addition of growth factors74. Interestingly, these immune cell-containing adipose organoids responded to pro-inflammatory and anti-inflammatory signals, providing a great tool for immune–metabolic research. Of note, these adipose organoids only included macrophages and mast cells. Therefore, assessing the role of other immune cell types, such as T cells, which play an important role in adipose homeostasis75 will require additional models to be developed in the future.
Adipose vascularization and innervation allow communication between adipose tissue and other tissues, which is difficult to model in vitro using 2D cellular models. A methodology was therefore developed to generate vascularized adipose spheroids, in which endogenous endothelial cells self-organized into endothelial networks among SVF cells of human subcutaneous white adipose tissue50. Upon specific differentiation signal cues, the adipose spheroids were induced into mature adipocytes and, interestingly, contained a dense vascular network. Importantly, these adipose spheroids were able to connect their own vascularization to the vascular system of the host mice and to maintain the adipocytes’ survival when engrafted into immunodeficient mice50, suggesting their potential for clinical transplantation. Notably, in this methodology, endogenous endothelial cells were contained within adipose organoids that recreated the native environment, which would explain why the adipose organoids were well maintained both in vitro and in vivo. Another strategy to generate vascularized organoids is to incorporate exogenous endothelial cells into organoids by co-culture. Human adipose-derived stem cells and endothelial cells were co-cultured for 2 weeks in 3D aqueous silk scaffolds, which led to the formation of a continuous endothelial lumen. When differentiated, these adipose organoids secreted leptin and lipids76. Thus, patient-specific vascularized adipose organoids provide a promising model to develop new drugs to treat obesity by modulation of the adipose vasculature.
Besides the organoid platforms described so far, there are also numerous bioengineering approaches to reconstruct the adipose tissue in vitro, which have been described in detail elsewhere68,77,78,79,80,81. The combinations of organ-on-chip, bioprinting and tissue engineering technologies strongly promote the generation of functional adipose tissue in vitro to model complex metabolic diseases (Box 1).
Development of liver organoid models
Adult stem cell-derived liver organoids
Since the development of the first intestinal organoids established from LGR5+ stem cells42, similar approaches have been applied to generate organoids from other tissue stem cells. In the mouse liver, expression of previously undetectable LGR5+ stem cells increased upon treatment with carbon tetrachloride (a hepatotoxic chemical) and were associated with the robust activation of Wnt signalling82. Culturing these LGR5+ stem cells with the Wnt agonist R-spondin in a matrix generated cyst-like structures, which were able to further differentiate into functional hepatocytes and repopulate the liver upon transplantation into Fah−/− mice82. Similarly, human adult liver organoids were derived from EpCAM+ biliary cells within the liver tissue, and maintained genomic stability after long-term expansion (>1 year)83, thus providing a promising tool for liver disease modelling, drug discovery and regenerative medicine (Fig. 2). However, these biliary organoids expanded mainly as homogeneous epithelial stem cells that required further differentiation to generate functional hepatocytes or cholangiocytes in vitro or in vivo83.
a | Liver organoids established from LGR5+ or EpCAM+ bipotent liver progenitor cells82,83 or directly from primary hepatocytes48,84 or from fibroblasts86. b | Various liver organoid models can be generated from pluripotent stem cells under different culture conditions. These models include liver buds88,90, hepatic organoids100, cholangiocyte organoids93,94,95, hepatobiliary organoids97,98,99, multicellular liver organoids101,102 and hepato–biliary–pancreatic organoids103, in order of increasing structural and functional complexity. ESC, embryonic stem cell; iPSC, induced pluripotent stem cell.
Liver organoids derived from primary hepatocytes
As hepatocytes have high proliferative potential in vivo, two independent studies developed unique methodology to expand hepatocyte organoids48,84. In one study, hepatocyte organoids were successfully generated directly from primary mouse hepatocytes and fetal human hepatocytes, which grew in a ‘bunch-of-grapes’ structure typical of hepatocyte organoids, in contrast to the spherical cystic structure of biliary organoids48. These hepatocyte organoids showed a gene expression signature similar to that of proliferating hepatocytes after partial hepatectomy, and exhibited similar metabolic function to primary hepatocytes48. Another model utilized inflammatory signals, which play an important role in liver regeneration85, to efficiently create hepatocyte organoids from mice84. These organoids exhibited similar gene expression profiles to proliferating hepatocytes and extensively repopulated the injured liver of Fah−/− mice, although whether the inflammatory signals promoted the formation and expansion of human hepatocyte organoids was not tested84. It will be important to improve the culture conditions further, possibly by combining the above two protocols48,84 to more efficiently expand hepatocyte organoids even from the adult human liver. Hepatocyte organoids have also been successfully directly generated from fibroblasts using defined transcription factors86. Nevertheless, in contrast to ASC-derived or primary hepatocyte-derived organoids, iPSCs, which can be patient-specific and continually expanded, allow the robust generation of liver organoids for modelling liver diseases and precision medicine87 (Fig. 2).
hPSC-derived liver buds
The self-renewing capacity and developmental potential of hPSCs has facilitated the development of various liver organoid models, from simple epithelial organoids to multi-organ organoids24. The first hPSC-derived liver organoid methodology was developed to generate liver buds88, in which hiPSC-derived hepatic endoderm cells were co-cultured with human umbilical vein endothelial cells and human MSCs, aiming to recapitulate organogenetic environments during embryonic liver development89. The multiple cell types interacted and self-organized into a 3D liver bud on a soft matrix and exhibited similar gene expression signatures to that of liver buds of mouse embryo88. Interestingly, functional and vascularized liver tissue was generated when these in vitro liver buds were engrafted into immunodeficient mice, and even rescued the drug-induced lethal liver failure88. The same group subsequently modified the protocol and massively generated liver organoids entirely from human iPSC-derived endoderm, endothelial and mesenchymal progenitors90. These organoids provide potential cell resources for regenerative medicine by using HLA-matched iPSCs and clinical grade differentiation medium. However, the complex co-culture system and immature state of these liver buds limits their application88,90.
Cholangiocyte organoids
In other studies, specific cues were utilized to direct the differentiation of hPSCs towards cholangiocyte organoids, hepatobiliary organoids or hepatic organoids consisting of multiple liver cell types91. In the case of cholangiocyte organoids, hPSCs were guided to differentiate into definitive endoderm, hepatoblasts and cholangiocytes in a stepwise manner with the appropriate developmental cues, such as notch signalling92. After embedding in matrix, hPSC-derived cholangiocytes self-organized into cholangiocyte organoids, which showed cystic and ductal structures and expressed typical mature biliary marker genes93,94,95. Furthermore, hPSC-derived cholangiocyte organoids exhibited epithelial functions, including cystic fibrosis transmembrane conductance regulator-mediated fluid secretion94. Since region-specific cholangiocyte organoids derived from primary human cholangiocytes regenerated the biliary tree when injected into the intrahepatic ducts of livers from deceased organ donors using ex vivo normothermic perfusion technology96, cholangiocyte organoids exhibit great potential for disease modelling and regenerative medicine of liver diseases.
hPSC-derived hepatobiliary organoids
Similarly, several groups have generated hepatobiliary organoids that consist of hepatocytes and cholangiocytes and exhibit bile duct-like structures97,98,99. These organoids recapitulate key aspects of early hepatogenesis, displaying hepatic gene expression signatures and key functions of hepatobiliary cells, and were used to characterize the effect of different JAG1 mutations in liver disease development97. A 2019 study optimized a protocol using sorted hiPSC-derived EpCAM+ endodermal cells to robustly generate hepatic organoids that could be stably expanded for more than 1 year with persistent expression of hepatic marker genes but decreased expression of cholangiocyte marker genes over long-term culture100. To examine if functional bile canaliculi networks could form in hepatobiliary organoids, a novel hepatobiliary organoid method was developed in 3D suspension culture using a stepwise differentiation strategy with optimized culture media99. These organoids self-organized a system of functional bile canaliculi, recapitulating the bile transport network in the liver tissue, which provides a useful platform to model both drug-induced and disease-induced cholestasis. The avoidance of matrix in this protocol broadens the potential application of these hepatobiliary organoids to high-throughput drug screening and cell therapy99.
hPSC-derived complex liver organoids
To further recapture the complexity of liver tissue, hPSC-derived liver organoids that consist of multiple liver cell types, such as hepatocyte-like cells, stellate-like cells and Kupffer-like cells, were developed101. After treatment with free fatty acids (FFA) for 5 days, these organoids recapitulated the key features of steatohepatitis, exhibiting inflammatory and fibrosis responses101. As the derivation efficiency of these multicellular liver organoids is low, the same group further optimized the protocol to markedly increase organoid numbers and, more importantly, to generate polarized immature hepatocytes with bile canaliculi-like architecture102. Using this unique model, high-throughput screening was performed to evaluate the effects of various drugs on cholestasis102. Key advantages of this strategy include the establishment of intermediate foregut progenitors, which can be isolated and cryopreserved. Moreover, the multiple cell types were induced simultaneously under the same culture conditions, thus delineating the propensity for hPSCs to differentiate into multicellular liver organoids will further shed light on early human embryonic liver development.
To model the higher complexity of organ communications, hepatic–biliary–pancreatic (HBP) organoids were formed autonomously without introducing additional signal cues when co-culturing SOX2+ anterior spheroids adjacent to CDX2+ posterior spheroids derived from hPSCs103. Long-term-cultured HBP organoids developed into segregated multi-organ anlages composed of hepatic and pancreatic tissues with connecting ducts103. HBP organoids thus provide an easily accessible model for the study of complex human endoderm organogenesis. In future, even more complex and mature organoid models, by combining with tissue engineering and organ-on-chip technologies (Box 1), are likely to be developed to recapitulate tissue–tissue interaction and organ–organ communication during the development of metabolic diseases104,105,106.
Applications in metabolic research
With the emergence of human organoid technology, it is possible to generate patient-specific organoid models that structurally and functionally mimic various tissues. This advance opens up new opportunities for dissecting underlying mechanisms of metabolic diseases and developing personalized drugs, since the iPSC-derived or ASC-derived organoids retain the genetic background of the respective individual carrying specific disease-causing mutations107. Moreover, by combining state-of-the-art gene editing tools and human stem cell-derived organoids, many disease-related genes and mutations, identified either by GWAS or rare genetic variants, can be directly examined in iPSC-derived organoids, in which disease-causing mutations can be introduced or reversed108. Hereafter, we discuss the applications of hPSCs and organoid culture systems in adipose-related diseases and liver diseases (Fig. 3).
a | Liver organoids can be used to model many liver diseases, such as inherited liver diseases and non-alcoholic fatty liver disease (NAFLD), as well as virus–host interactions. Adipose organoids provide a platform to study adipogenesis, lipogenesis, lipolysis and the adipose tissue response to insulin. b | The establishment of a large cohort of organoids can serve as a biobank for use in precision medicine. c | Reproducible and scalable organoids allow high-throughput drug screening. d | Organoids can be used to predict drug toxicity. e | Drug responses in human organoids can be measured based on the structure, gene profile and metabolic properties of organoids. f | Patient-derived organoids exhibit great power in predicting individual-specific drug responses.
Applications of adipose organoids
Adipose organoids for modelling obesity
Over-nutrition in modern society results in lipid accumulation and redistribution, which further impairs glucose and lipid metabolism and can cause obesity, T2DM and cardiovascular disease109. Notably, while visceral adipose tissue mass is positively associated with risk of metabolic disease, the accumulation of subcutaneous adipose tissue in the body is beneficial for health110,111. Thus, understanding the depot-specific characteristics of adipose tissues could provide mechanistic insights into obesity and T2DM. However, this effort is hindered by the lack of human depot-specific adipocyte models, in part because visceral preadipocytes differentiate poorly compared with the robust differentiation of subcutaneous preadipocytes112. To overcome this barrier, 3D culture systems have been used to differentiate visceral and subcutaneous preadipocytes into mature adipocytes that retain depot-specific characteristics, such as gene expression profile, thermogenesis, lipolysis and cytokine secretion69. These well-differentiated adipose tissue organoid systems represent promising models to study adipogenesis and obesity.
Modelling adipose homeostasis
Adipose tissue is highly heterogeneous, consisting not only of adipocytes, but also of connective tissue, vasculature, immune cells and nerves67. As noted above, macrophage-containing adipose organoids respond to inflammatory signals more robustly than 2D adipocytes74, but it will be important to develop adipose organoids containing diverse immune cell types that better recapitulate the interaction between adipocytes and immune cells. Moreover, angiogenesis and adipogenesis develop interdependently through direct cell–cell contact and secreted signal cues113,114. Vascularized adipose organoids50,76 provide a model to delineate the connections between adipogenesis and angiogenesis.
Another layer of regulation of adipose tissue is innervation by sympathetic nerve fibres, which control adipocyte lipolysis and thermogenesis through the secretion of noradrenaline115. Therefore, developing adipose organoids containing neural cells might promote the maturation and metabolic function of adipose organoids, and induce the browning of adipose organoids, which could be a potential cell therapy for the treatment of obesity. Furthermore, the organ-on-chip technology provides an opportunity to study the communication between adipose tissue, liver, pancreas, muscle and brain116 (Box 2). For example, the first human-on-a-chip model that was composed of human hepatocytes and adipose tissue provided a great platform to explore the role of adipocyte lipolysis and insulin resistance in NAFLD117. More importantly, this unique model revealed the discrepancy of metformin efficacy in treating NAFLD between preclinical animal model and patients. This study highlighted the value of human-on-a-chip models as a system to recapitulate the communication between relevant tissues, not just the individual tissue response.
Modelling genetic causes of adipose dysfunction
GWAS have pointed to many genes in the central nervous system that play an important role in obesity susceptibility, while genes expressed in adipose tissue have tended to be critical for adipose tissue distribution118,119. Although many genetic variants have been linked to the dysfunction of lipid metabolism and adipogenesis120, the majority are found in non-coding regions of the genome without identification of the associated protein-coding genes, nor the cell type in which these genetic variants are functional. Stem cell-derived adipocyte models and gene editing tools have the potential to address these critical questions. Patient-specific adipocytes can be differentiated from multiple lines of iPSCs, and used to validate GWAS variants in metabolic disease121. For example, several genes involved in lipid metabolism, insulin signalling and glucose metabolism have been interrogated in hESC-derived adipocytes, including the role of SORT1 in regulating insulin-responsive glucose uptake, the impact of AKT2 mutations on triglyceride accumulation and the function of PLIN1 in lipolysis122. Taken together, the state-of-the-art of stem cell models and gene editing tools allow us to validate the role of genetic variants on metabolic diseases.
Applications of liver organoids
Liver tissue is a heterogeneous organ that contains many cell types including hepatocytes, cholangiocytes, Kupffer cells and endothelial cells123. Various liver diseases are caused by the interplay between genetic factors and the environment, which involves the dysfunction of cell–cell interactions and cell–environment communication within liver tissues124,125,126. Thus the development of multicellular liver organoids would be valuable for the modelling of liver diseases that exhibit impaired cell–cell interaction and tissue structural alteration, such as alcoholic liver disease (ALD) and NAFLD.
Modelling alcoholic liver disease
High levels of alcohol consumption have resulted in an epidemic of ALD worldwide, which exhibits a wide range of liver pathologies from simple steatosis to steatohepatitis, fibrosis, cirrhosis and even hepatocellular carcinoma127. No treatment for ALD has been approved by the FDA or other agencies128, partially due to the difficulties of accessing human liver tissue and establishing reliable in vitro models of the human condition. In 2019, a multicellular liver organoid system was developed by co-culturing human fetal liver mesenchymal cells with hESC-derived hepatic organoids. These liver organoids exhibited structural complexity, functional maturity and lipid accumulation and expressed ethanol metabolism-associated enzymes, with accompanying fibrosis and inflammation, after 7 days of ethanol treatment129. Mechanistically, oxidative stress and mitochondrial dysfunction were noted in these organoids, similar to the proposed mechanism of ALD129, proving that this system provides a unique tool to study and develop new drugs to prevent or cure ALD. However immune cells such as Kupffer cells were missing in this system, which needs optimization.
Modelling non-alcoholic fatty liver disease
Pharmacological targeting strategies for treating NAFLD and NASH are greatly hindered by a shortage of liver biopsies from patients with NAFLD and a lack of appropriate cellular and animal models. In 2019, a hPSC-derived multicellular liver organoid system consisting of multiple cell types including hepatic-like cells, stellate-like cells and Kupffer-like cells was subjected to FFA-induced accumulation of intracellular lipids, increased inflammatory responses and progression of fibrosis, thereby recapitulating the pathological features of NAFLD101. Organoids were also derived from iPSCs of patients with Wolman disease, a lysosomal acid lipase deficiency which leads to an exaggerated steatohepatitis phenotype, and can be rescued by treatment with FGF19 (ref.101). Moreover, hepatobiliary organoids with organized and functional bile canaliculi were found to be sensitive to treatment with FFA, which induced gene expression signatures similar to those of livers with NASH and triggered a decay of bile canaliculi network and ductal reactions, as seen in patients with NASH99. Furthermore, a vascularized liver organoid system developed from hiPSC-derived hepatoblasts, MSCs, hepatic stellate cells and endothelial cells exhibited hepatic cellular spatial organization and metabolic functions. This organoid system developed steatosis, inflammation and fibrosis after FFA treatment, faithfully mimicking NAFLD pathology130.
In addition to hPSC-derived organoid models, bipotent ductal organoids derived directly from the liver of patients with NASH displayed impaired growth capacity, decreased albumin production, increased lipid accumulation and increased cytochrome P450 metabolism compared with organoids derived from healthy individuals, thus recapitulating the hallmarks of NASH in humans131. Consistently, liver organoids derived from mice with NASH mimicked the progression of the disease, with the organoids exhibiting pro-inflammatory responses and the emergence of fibrosis132. As deriving these liver organoids directly from liver tissue requires only a small amount of starting material, which can be obtained by needle biopsy, patient-specific liver organoids open an avenue for precision medicine of NAFLD disease.
Importantly, individuals exhibit differential susceptibility to steatosis development and progression, due to the interplay between genetics and environment124,133. GWAS have revealed numerous polymorphisms associated with NAFLD development4. Patatin-like phospholipase domain-containing protein 3 (PNPLA3) Ile148Met is the most well-described variant134. 3D spheroids composed of an immortalized hepatocyte cell line (HepG2) and hepatic stellate cells (LX-2), both homozygous for the PNPLA3 Ile148Met variant, accumulated lipids and collagen upon exposure to FFA. This phenotype could be rescued by incubating the spheroids with liraglutide, elafibranor or momelotinib135,136, all drugs that are being investigated as potential treatments for NAFLD. Similar approaches were used to study the mechanisms underlying the association of transmembrane 6 superfamily member 2 (TM6SF2) Glu167Lys and membrane-bound O-acyltransferase 7 (MBOAT7) with NAFLD137,138. A large cohort of organoids from patients with different genetic backgrounds would be ideal to evaluate the direct effect of genetic variation on the progression of NAFLD to NASH in vitro, without the overlay of environmental factors.
Circadian rhythms in metabolic organoids
Over-nutrition disrupts circadian metabolic rhythms, and dysfunction of the circadian clock leads to various metabolic diseases139. This knowledge largely stems from rodent studies, however, as obtaining timed tissue samples from patients is a huge challenge. As humans are diurnal whereas rodents are nocturnal140, there is a great need to understand the regulation and function of circadian clocks in humans, and organoids have great potential for this purpose141. The core clock does not function in hiPSCs and hiPSC-derived immature human intestinal organoids, but immature human intestinal organoids show robust circadian rhythms after differentiation into mature human intestinal enteroids142. Interestingly, these enteroids display changes in the necrotic cell death response to Clostridium difficile toxin B in a circadian phase-dependent manner. Another study revealed that the entrainment of hESC-derived islets to daily feeding and fasting cycles promoted islet metabolic maturation51. Thus the presence of circadian rhythms is a marker of the maturity and functionality of hPSC-derived organoids, and organoids can be used to evaluate human circadian biology.
Drug discovery and precision medicine
With the development of reproducible, complex and functional human stem cell-derived metabolic cell types and organoids, advances have been made to use these models for testing drug toxicity, drug response and high-throughput screening in metabolic disease (Fig. 3). Furthermore, this technology allows biobanking of patient stem cell-derived cell types or organoids, thus providing a platform for precision or personalized medicine.
Testing drug efficacy
The resemblance of liver organoids to liver tissue makes them an attractive model for screening the effects of preclinical or clinically used drugs on liver phenotypes prior to testing in patients. For example, a hepatic organoid system that exhibits lipid accumulation upon FFA treatment has been used to probe the effects of the drugs metformin and l-carnitine, which are used to treat T2DM143. Similarly, FGF19 treatment resulted in the reduced lipid accumulation and increased survivability in the liver organoids from patients with Wolman disease compared with untreated liver organoids101. In contrast, magnesium treatment, which is a highly controversial clinical therapy for treating T2DM and liver metabolic discorders144, has proven ineffective in this 3D system101. Moreover, sorafenib was shown to reduce steatosis-induced fibrogenesis in a human 3D co-culture model of NAFLD145. Thus, liver organoids represent an innovative approach to the development of new treatments and the testing of drug responses ex vivo.
High-throughput screening
The reproducibility and scalability of stem cell-derived cell types and organoids make them suitable for high-throughput screens146. In particular, organoids are accessible for imaging and standard automated liquid handling with pipetting workstations and can therefore be adapted for use in standardized and well-established automated chemical screening pipelines. Numerous studies have utilized this high-throughput screening system to search for new drugs to treat metabolic diseases. For instance, highly uniform adipose spheroids in ultra-low attachment 384-well plates were used for high-throughput screening to identify compounds promoting adipogenesis72. Additionally, many groups have identified small molecules that promote the browning of adipocytes by high-throughput screening using hPSC-derived adipocyte models58,147,148,149. Some of these compounds have also shown promising effects on the induction of brown and beige adipocyte and the promotion of energy expenditure in mouse models148,149.
Besides the advantage of utilizing stem cell-derived hepatocytes and organoids in high-throughput hepatotoxic drug screening102, these models have also been used to search for new therapies for metabolic diseases. For example, the use of hepatocyte-like cells differentiated from iPSCs from patients with homozygous familial hypercholesterolaemia revealed that cardiac glycosides reduce the production of apolipoprotein B, the essential core protein of VLDL, in patient hepatocytes. Similarly, hepatocytes differentiated from iPSCs of patients with mitochondrial DNA depletion syndrome were used to demonstrate that NAD can improve mitochondrial function in patient hepatocyte-like cells, providing a potential treatment strategy150. Moreover, a feline liver organoid system was used to identify promising candidates for the treatment of hepatic lipidosis151. Furthermore, the 3D human hepatic organoid model was also used as a screening platform for small-molecule inhibitors in the preclinical NAFLD and/or NASH settings136.
Precision medicine
Advanced iPSCs and organoid technologies allow the generation of disease-relevant cell types and organoids with individual-specific genetic information, which enable the development of personalized medicine. In practice, individual responses to drugs differ, with some patients experiencing beneficial effects while others experience no effect or even adverse effects. This variation is largely due to numerous genetic differences between humans152,153.
Thiazolidinedione drugs target the transcriptional activity of PPARγ to reverse insulin resistance in T2DM, but adverse effects limit their clinical use154. Furthermore, 20–30% of patients with T2DM fail to respond to thiazolidinediones155. To understand the underlying mechanism behind this variation in response, human ASC-derived adipocytes from people with obesity were generated to test the response of rosiglitazone, a thiazolidinedione drug in clinical use156, which has been withdrawn from the market in many countries due to adverse cardiovascular effects that were under debate. Functional genomic analysis revealed that genetic variants affected PPARγ function and caused patient-specific drug responses156. In particular, a specific single nucleotide polymorphism rs4743771 modulated the PPARγ target gene ABCA1, whose protein product regulates reverse cholesterol transport157, in a patient-specific manner. Genetic variation at rs4743771 controlled the effect of rosiglitazone on cholesterol metabolism in patient adipocytes, and was predictive of serum cholesterol increases in people with T2DM who were treated with rosiglitazone156.
Similarly, adipose stem cell-derived adipocytes and iPSC-derived hepatocytes from multiple individuals were also used to model individual-specific responses to dexamethasone158, a widely used anti-inflammation drug that has also proved to be effective in treating severe COVID-19 infection159. With these unique cellular models, many genetic variants were found to modulate dexamethasone response through affecting the function of the glucocorticoid receptor158. Remarkably, these genetic variations were highly associated with increases in serum levels of glucose, lipids and body mass in patients on dexamethasone therapy158. Thus human stem cell-derived adipocytes and hepatocytes can be used to identify genetic variants that predispose individuals to adverse drug effects and allow personalized predictions of therapeutic index.
Due to their self-renewable properties, organoid models could also serve as cryopreserved biobanks of healthy and diseased individuals. In this context, liver organoid biobanks, such as HUB Organoids founded by the Hubrecht Institute, have the potential to play important roles in the development of new therapeutic drugs and personalized treatments, as well as in the identification of novel diagnostic markers for many liver diseases160,161. Indeed, the progression of NAFLD varies among humans124,133, and the development of liver organoid models from different genetic backgrounds could identify individual risk factors and new biomarkers for NAFLD progression. Individual-specific drug responses could also be tested in these liver organoid models. Moreover, liver organoid biobanks could also serve as a platform for predicting individual-specific drug-induced liver injury, the main underlying factor in acute liver failure162,163.
Gene therapy and regenerative medicine
Advanced gene editing tools have shown potential in preventing or rescuing phenotypes of genetic diseases by repairing or inactivating mutations in cellular models or in animal models164. Many efforts have been made to generate healthy isogenic disease-relevant cell types or organoids to treat genetic metabolic diseases by combining modern genome editing and stem cell or organoid technology165,166. The ability to generate patient-specific cell types or organoids allows autologous transplantation to ensure better survival of grafts without any immune rejection, particularly with the potential use of gene editing to develop HLA-matched hiPSC-derived cells or organoids167 (Fig. 4).
a | Rescue of disease-relevant organoids using CRISPR–Cas9 or other gene-editing tools, and autologous transplantation of gene-edited organoids into individuals with metabolic diseases. b | The establishment of HLA-matched induced pluripotent stem cells (iPSCs) can be utilized to restrict immune rejection of transplanted allogeneic organoids. c | Transplantation of iPSC-derived brown adipocytes to treat individuals with obesity by enhancing energy expenditure.
With the goal of treating obesity, human brown-like cells were generated by engineering human white preadipocytes using CRISPR–Cas9 to activate endogenous UCP1 expression, and transplantation of these cells into obese mice led to sustained improvement in glucose tolerance and insulin sensitivity, as well as increased energy expenditure168. Similarly, implantation into obese mice of human brown-like adipocytes in which the thermogenic suppressor gene NRIP1 was deleted using CRISPR–Cas9 decreased adiposity and levels of liver triglycerides while enhancing glucose tolerance169. CRISPR–Cas9 and base editing have also been used to revert a disease-causing mutation to wild-type in patient-derived hepatic organoids to rescue the phenotype of Alagille syndrome and Wilson disease97,170, providing a powerful therapeutic approach for liver metabolic diseases.
At present, orthotopic liver transplantation is the only effective treatment for late-stage liver failure, but this option is limited by a lack of proper and heathy donors, as well as the risk of immune rejection171. The expansion and differentiation potential of liver organoids with complex structures and metabolic functions makes these a potential alternative cell resource for transplantation (Fig. 4). Various hepatic organoid models have been shown to efficiently engraft into the liver of recipient mice and repopulate injured livers48,84,88,90.
Limitations of organoids
Organoid technology has many advantages in modelling metabolic diseases and the potential to revolutionize metabolic research. However, this methodology is still in its infancy and various challenges and limitations need to be addressed (Box 3).
Heterogeneity and reproducibility
Due to intrinsic cell fate determination and self-organization of stem cell-derived organoids, organoids are often heterogeneous in terms of size, shape and cell components. This heterogeneity can occur whether the organoids are derived from different patients, established in different laboratories, or even obtained from the same stem cells. This lack of standardization creates huge difficulties and reduces the enthusiasm for using organoids in metabolic disease modelling and drug discovery. Further standardization and automation of these procedures with defined culture media and starting cell types are required to reduce the variability and improve the reproducibility of the system. Notably, an hPSC-derived adipose organoid system has not yet been successfully established, possibly due to low efficiency and the time-consuming, complex process of differentiating hPSCs into adipocytes. Future efforts with optimized differentiation strategies and tissue engineering technologies are needed to generate hPSC-derived adipose organoids to model obesity and related diseases. In addition, single-cell transcriptome and epigenome analysis can be used to ensure the quality and uniformity of organoids used in metabolic research.
Structural and functional limitations
Current adipose organoids and liver organoids often lack specific cell types found in the native tissue and fail to recapitulate the full functional repertoire of target organs. For example, most liver organoid models contain only one or two cell types, and are missing functional bile canaliculi networks91. Even the best multicellular liver organoid models lack the normal liver zonation patterns. Furthermore, due to the epigenetic barrier of hPSC differentiation into mature cells and the limited time span of organoid culture, hPSC-derived organoids usually fail to functionally mature beyond the fetal stage. Introduction of a vascular system, which is essential for gas and nutrient exchange, and utilization of a tissue engineering and air–liquid interface culture system, might allow the maturation of hPSC-derived organoids that faithfully mimic the structure and function of adipose and liver tissues.
The influence of Matrigel
One drawback in organoid research is the reliance on Matrigel, which is derived from mouse sarcoma cells and of which most of the components are undefined, contributing to the variability and poor reproducibility of organoids. Moreover, including Matrigel in the culture system increases the difficulties of organoid passaging, genetic manipulation, genome-wide genetic screening and high-throughput drug screening due to physical obstacles, in addition to posing challenges in assessing the metabolic functions of adipose and liver organoids. Ongoing development of synthetic and versatile scaffolds will reduce cost, increase reproducibility and promote the translatability of organoids for the study of metabolic diseases in the future172,173.
Conclusions
Translational metabolic research has been largely constrained by the lack of accurate and reliable in vitro models that faithfully reflect human physiology. Liver organoids and adipose organoids, whether derived from hPSCs, healthy tissue or diseased tissue, are demonstrating great potential to provide excellent platforms to study the development and progression of, and potential treatments for, human metabolic diseases. The use of individual-derived stem cells adds to the excitement by facilitating personalized approaches to therapy.
Current organoid technology is imperfect, and improvements in quality and quantity of organoids, as well as even more accurate cellular composition of stem cell-derived tissues, are required for progress to continue apace. The continued development of organoid technology will further bridge the gap between basic research and translational medicine.
References
Caballero, B. The global epidemic of obesity: an overview. Epidemiol. Rev. 29, 1–5 (2007).
Rosen, E. D. & Spiegelman, B. M. Adipocytes as regulators of energy balance and glucose homeostasis. Nature 444, 847–853 (2006).
Joffe, B. I., Panz, V. R. & Raal, F. J. From lipodystrophy syndromes to diabetes mellitus. Lancet 357, 1379–1381 (2001).
Al-Goblan, A. S., Al-Alfi, M. A. & Khan, M. Z. Mechanism linking diabetes mellitus and obesity. Diabetes Metab. Syndr. Obes. 7, 587–591 (2014).
Wang, W. & Seale, P. Control of brown and beige fat development. Nat. Rev. Mol. Cell Biol. 17, 691–702 (2016).
Berger, J. P., Akiyama, T. E. & Meinke, P. T. PPARs: therapeutic targets for metabolic disease. Trends Pharmacol. Sci. 26, 244–251 (2005).
Kurylowicz, A. In search of new therapeutic targets in obesity treatment: sirtuins. Int. J. Mol. Sci. 17, 572 (2016).
Blondin, D. P. et al. Human brown adipocyte thermogenesis is driven by β2-AR stimulation. Cell Metab. 32, 287–300.e7 (2020).
de Jong, J. M. A. et al. Human brown adipose tissue is phenocopied by classical brown adipose tissue in physiologically humanized mice. Nat. Metab. 1, 830–843 (2019).
Stanger, B. Z. Cellular homeostasis and repair in the mammalian liver. Annu. Rev. Physiol. 77, 179–200 (2015).
Zhou, W. C., Zhang, Q. B. & Qiao, L. Pathogenesis of liver cirrhosis. World J. Gastroenterol. 20, 7312–7324 (2014).
Olefsky, J. M. Fat talks, liver and muscle listen. Cell 134, 914–916 (2008).
Perry, R. J., Samuel, V. T., Petersen, K. F. & Shulman, G. I. The role of hepatic lipids in hepatic insulin resistance and type 2 diabetes. Nature 510, 84–91 (2014).
Castillo-Armengol, J., Fajas, L. & Lopez-Mejia, I. C. Inter-organ communication: a gatekeeper for metabolic health. EMBO Rep. 20, e47903 (2019).
Jensen-Cody, S. O. & Potthoff, M. J. Hepatokines and metabolism: deciphering communication from the liver. Mol. Metab. 44, 101138 (2021).
Golabi, P. et al. Burden of non-alcoholic fatty liver disease in Asia, the Middle East and North Africa: data from Global Burden of Disease 2009–2019. J. Hepatol. 75, 795–809 (2021).
Michelotti, G. A., Machado, M. V. & Diehl, A. M. NAFLD, NASH and liver cancer. Nat. Rev. Gastroenterol. Hepatol. 10, 656–665 (2013).
Zou, B. et al. Prevalence, characteristics and mortality outcomes of obese, nonobese and lean NAFLD in the United States, 1999-2016. J. Intern. Med. 288, 139–151 (2020).
Vuppalanchi, R., Noureddin, M., Alkhouri, N. & Sanyal, A. J. Therapeutic pipeline in nonalcoholic steatohepatitis. Nat. Rev. Gastroenterol. Hepatol. 18, 373–392 (2021).
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Sharma, A., Sances, S., Workman, M. J. & Svendsen, C. N. Multi-lineage human iPSC-derived platforms for disease modeling and drug discovery. Cell Stem Cell 26, 309–329 (2020).
Marsee, A. et al. Building consensus on definition and nomenclature of hepatic, pancreatic, and biliary organoids. Cell Stem Cell 28, 816–832 (2021).
Bolker, J. A. Animal models in translational research: rosetta stone or stumbling block? Bioessays 39, 1700089 (2017).
Horrobin, D. F. Modern biomedical research: an internally self-consistent universe with little contact with medical reality? Nat. Rev. Drug Discov. 2, 151–154 (2003).
Butler, D. Translational research: crossing the valley of death. Nature 453, 840–842 (2008).
Ramos, M. J., Bandiera, L., Menolascina, F. & Fallowfield, J. A. In vitro models for non-alcoholic fatty liver disease: emerging platforms and their applications. iScience 25, 103549 (2022).
Colca, J. R. Discontinued drug therapies to treat diabetes in 2014. Expert. Opin. Investig. Drugs 24, 1241–1245 (2015).
Colca, J. R. Discontinued drugs in 2008: endocrine and metabolic. Expert. Opin. Investig. Drugs 18, 1243–1255 (2009).
Costantino, S. et al. Epigenetics and precision medicine in cardiovascular patients: from basic concepts to the clinical arena. Eur. Heart J. 39, 4150–4158 (2018).
Krishnan, A. et al. A detailed comparison of mouse and human cardiac development. Pediatr. Res. 76, 500–507 (2014).
Blais, E. M. et al. Reconciled rat and human metabolic networks for comparative toxicogenomics and biomarker predictions. Nat. Commun. 8, 14250 (2017).
Martignoni, M., Groothuis, G. M. & de Kanter, R. Species differences between mouse, rat, dog, monkey and human CYP-mediated drug metabolism, inhibition and induction. Expert. Opin. Drug Metab. Toxicol. 2, 875–894 (2006).
Platt, R. J. et al. CRISPR-Cas9 knockin mice for genome editing and cancer modeling. Cell 159, 440–455 (2014).
Abudayyeh, O. O. et al. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science 353, aaf5573 (2016).
Gaudelli, N. M. et al. Programmable base editing of A*T to G*C in genomic DNA without DNA cleavage. Nature 551, 464–471 (2017).
Zeng, H. et al. An isogenic human ESC platform for functional evaluation of genome-wide-association-study-identified diabetes genes and drug discovery. Cell Stem Cell 19, 326–340 (2016).
Sullivan, K. M. & Susztak, K. Unravelling the complex genetics of common kidney diseases: from variants to mechanisms. Nat. Rev. Nephrol. 16, 628–640 (2020).
Bredenoord, A. L., Clevers, H. & Knoblich, J. A. Human tissues in a dish: the research and ethical implications of organoid technology. Science 355, eaaf9414 (2017).
Lancaster, M. A. & Knoblich, J. A. Organogenesis in a dish: modeling development and disease using organoid technologies. Science 345, 1247125 (2014).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
[No authors listed]. Method of the year 2017: organoids. Nat. Methods 15, 1 (2018).
Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
Miller, A. J. et al. Generation of lung organoids from human pluripotent stem cells in vitro. Nat. Protoc. 14, 518–540 (2019).
Morizane, R. et al. Nephron organoids derived from human pluripotent stem cells model kidney development and injury. Nat. Biotechnol. 33, 1193–1200 (2015).
Zhang, X., Ma, Z., Song, E. & Xu, T. Islet organoid as a promising model for diabetes. Protein Cell 13, 239–257 (2021).
Hu, H. et al. Long-term expansion of functional mouse and human hepatocytes as 3D organoids. Cell 175, 1591–1606.e19 (2018).
Hofbauer, P. et al. Cardioids reveal self-organizing principles of human cardiogenesis. Cell 184, 3299–3317.e22 (2021).
Muller, S. et al. Human adipose stromal-vascular fraction self-organizes to form vascularized adipose tissue in 3D cultures. Sci. Rep. 9, 7250 (2019).
Alvarez-Dominguez, J. R. et al. Circadian entrainment triggers maturation of human in vitro islets. Cell Stem Cell 26, 108–122.e10 (2020).
Crespo, M. et al. Colonic organoids derived from human induced pluripotent stem cells for modeling colorectal cancer and drug testing. Nat. Med. 23, 878–884 (2017).
Armingol, E., Officer, A., Harismendy, O. & Lewis, N. E. Deciphering cell-cell interactions and communication from gene expression. Nat. Rev. Genet. 22, 71–88 (2021).
Jung, J., Seol, H. S. & Chang, S. The generation and application of patient-derived xenograft model for cancer research. Cancer Res. Treat. 50, 1–10 (2018).
Bakhti, M., Bottcher, A. & Lickert, H. Modelling the endocrine pancreas in health and disease. Nat. Rev. Endocrinol. 15, 155–171 (2019).
Guenantin, A. C. et al. Functional human beige adipocytes from induced pluripotent stem cells. Diabetes 66, 1470–1478 (2017).
Mohsen-Kanson, T. et al. Differentiation of human induced pluripotent stem cells into brown and white adipocytes: role of Pax3. Stem Cell 32, 1459–1467 (2014).
Ahfeldt, T. et al. Programming human pluripotent stem cells into white and brown adipocytes. Nat. Cell Biol. 14, 209–219 (2012).
Xiong, C. et al. Derivation of adipocytes from human embryonic stem cells. Stem Cell Dev. 14, 671–675 (2005).
Nishio, M. et al. Production of functional classical brown adipocytes from human pluripotent stem cells using specific hemopoietin cocktail without gene transfer. Cell Metab. 16, 394–406 (2012).
Zhang, L. et al. Generation of functional brown adipocytes from human pluripotent stem cells via progression through a paraxial mesoderm state. Cell Stem Cell 27, 784–797.e11 (2020).
Brown, A. C. Brown adipocytes from induced pluripotent stem cells-how far have we come? Ann. N. Y. Acad. Sci. 1463, 9–22 (2020).
Mukherjee, S. et al. Derivation and characterization of a UCP1 reporter human ES cell line. Stem Cell Res. 30, 12–21 (2018).
Kajimura, S. et al. Initiation of myoblast to brown fat switch by a PRDM16-C/EBP-β transcriptional complex. Nature 460, 1154–1158 (2009).
Wang, H., Leng, Y. & Gong, Y. Bone marrow fat and hematopoiesis. Front. Endocrinol. 9, 694 (2018).
Su, S. et al. A renewable source of human beige adipocytes for development of therapies to treat metabolic syndrome. Cell Rep. 25, 3215–3228.e9 (2018).
Corvera, S. Cellular heterogeneity in adipose tissues. Annu. Rev. Physiol. 83, 257–278 (2021).
Bahmad, H. F. et al. Modeling adipogenesis: current and future perspective. Cells 9, 2326 (2020).
Emont, M. P. et al. Using a 3D culture system to differentiate visceral adipocytes in vitro. Endocrinology 156, 4761–4768 (2015).
Turner, P. A., Gurumurthy, B., Bailey, J. L., Elks, C. M. & Janorkar, A. V. Adipogenic differentiation of human adipose-derived stem cells grown as spheroids. Process. Biochem. 59, 312–320 (2017).
Kozlowski, M. T., Crook, C. J. & Ku, H. T. Towards organoid culture without Matrigel. Commun. Biol. 4, 1387 (2021).
Klingelhutz, A. J. et al. Scaffold-free generation of uniform adipose spheroids for metabolism research and drug discovery. Sci. Rep. 8, 523 (2018).
Di Franco, A. et al. Searching for classical brown fat in humans: development of a novel human fetal brown stem cell model. Stem Cell 34, 1679–1691 (2016).
Taylor, J. et al. Generation of immune cell containing adipose organoids for in vitro analysis of immune metabolism. Sci. Rep. 10, 21104 (2020).
Cipolletta, D. et al. PPAR-γ is a major driver of the accumulation and phenotype of adipose tissue Treg cells. Nature 486, 549–553 (2012).
Kang, J. H., Gimble, J. M. & Kaplan, D. L. In vitro 3D model for human vascularized adipose tissue. Tissue Eng. Part. A 15, 2227–2236 (2009).
Davidenko, N., Campbell, J. J., Thian, E. S., Watson, C. J. & Cameron, R. E. Collagen-hyaluronic acid scaffolds for adipose tissue engineering. Acta Biomater. 6, 3957–3968 (2010).
Kuss, M. et al. Effects of tunable, 3D-bioprinted hydrogels on human brown adipocyte behavior and metabolic function. Acta Biomater. 71, 486–495 (2018).
McCarthy, M. et al. Fat-on-a-chip models for research and discovery in obesity and its metabolic comorbidities. Tissue Eng. Part. B Rev. 26, 586–595 (2020).
Murphy, C. S., Liaw, L. & Reagan, M. R. In vitro tissue-engineered adipose constructs for modeling disease. BMC Biomed. Eng. 1, 27 (2019).
Tseng, H., Daquinag, A. C., Souza, G. R. & Kolonin, M. G. Three-dimensional magnetic levitation culture system simulating white adipose tissue. Methods Mol. Biol. 1773, 147–154 (2018).
Huch, M. et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 494, 247–250 (2013).
Huch, M. et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160, 299–312 (2015).
Peng, W. C. et al. Inflammatory cytokine TNFα promotes the long-term expansion of primary hepatocytes in 3D culture. Cell 175, 1607–1619.e15 (2018).
Campana, L., Esser, H., Huch, M. & Forbes, S. Liver regeneration and inflammation: from fundamental science to clinical applications. Nat. Rev. Mol. Cell Biol. 22, 608–624 (2021).
Sun, L. et al. Modelling liver cancer initiation with organoids derived from directly reprogrammed human hepatocytes. Nat. Cell Biol. 21, 1015–1026 (2019).
Prior, N., Inacio, P. & Huch, M. Liver organoids: from basic research to therapeutic applications. Gut 68, 2228–2237 (2019).
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013).
Ober, E. A. & Lemaigre, F. P. Development of the liver: insights into organ and tissue morphogenesis. J. Hepatol. 68, 1049–1062 (2018).
Takebe, T. et al. Massive and reproducible production of liver buds entirely from human pluripotent stem cells. Cell Rep. 21, 2661–2670 (2017).
Lam, D., Dan, Y. Y., Chan, Y. S. & Ng, H. H. Emerging liver organoid platforms and technologies. Cell Regen. 10, 27 (2021).
Zong, Y. et al. Notch signaling controls liver development by regulating biliary differentiation. Development 136, 1727–1739 (2009).
Dianat, N. et al. Generation of functional cholangiocyte-like cells from human pluripotent stem cells and HepaRG cells. Hepatology 60, 700–714 (2014).
Ogawa, M. et al. Directed differentiation of cholangiocytes from human pluripotent stem cells. Nat. Biotechnol. 33, 853–861 (2015).
Sampaziotis, F. et al. Directed differentiation of human induced pluripotent stem cells into functional cholangiocyte-like cells. Nat. Protoc. 12, 814–827 (2017).
Sampaziotis, F. et al. Cholangiocyte organoids can repair bile ducts after transplantation in the human liver. Science 371, 839–846 (2021).
Guan, Y. et al. Human hepatic organoids for the analysis of human genetic diseases. JCI Insight 2, e94954 (2017).
Wu, F. et al. Generation of hepatobiliary organoids from human induced pluripotent stem cells. J. Hepatol. 70, 1145–1158 (2019).
Ramli, M. N. B. et al. Human pluripotent stem cell-derived organoids as models of liver disease. Gastroenterology 159, 1471–1486.e12 (2020).
Akbari, S. et al. Robust, long-term culture of endoderm-derived hepatic organoids for disease modeling. Stem Cell Rep. 13, 627–641 (2019).
Ouchi, R. et al. Modeling steatohepatitis in humans with pluripotent stem cell-derived organoids. Cell Metab. 30, 374–384.e6 (2019).
Shinozawa, T. et al. High-fidelity drug-induced liver injury screen using human pluripotent stem cell-derived organoids. Gastroenterology 160, 831–846.e10 (2021).
Koike, H. et al. Modelling human hepato-biliary-pancreatic organogenesis from the foregut-midgut boundary. Nature 574, 112–116 (2019).
Xiang, Y. et al. hESC-derived thalamic organoids form reciprocal projections when fused with cortical organoids. Cell Stem Cell 24, 487–497.e7 (2019).
Xiang, Y. et al. Fusion of regionally specified hPSC-derived organoids models human brain development and interneuron migration. Cell Stem Cell 21, 383–398.e7 (2017).
Miura, Y. et al. Generation of human striatal organoids and cortico-striatal assembloids from human pluripotent stem cells. Nat. Biotechnol. 38, 1421–1430 (2020).
Bose, S., Clevers, H. & Shen, X. Promises and challenges of organoid-guided precision medicine. Med 2, 1011–1026 (2021).
Drost, J. et al. Use of CRISPR-modified human stem cell organoids to study the origin of mutational signatures in cancer. Science 358, 234–238 (2017).
Scherer, P. E. & Hill, J. A. Obesity, diabetes, and cardiovascular diseases: a compendium. Circ. Res. 118, 1703–1705 (2016).
Fox, C. S. et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation 116, 39–48 (2007).
Karpe, F. & Pinnick, K. E. Biology of upper-body and lower-body adipose tissue – link to whole-body phenotypes. Nat. Rev. Endocrinol. 11, 90–100 (2015).
Macotela, Y. et al. Intrinsic differences in adipocyte precursor cells from different white fat depots. Diabetes 61, 1691–1699 (2012).
Han, J. et al. The spatiotemporal development of adipose tissue. Development 138, 5027–5037 (2011).
Volz, A. C., Huber, B. & Kluger, P. J. Adipose-derived stem cell differentiation as a basic tool for vascularized adipose tissue engineering. Differentiation 92, 52–64 (2016).
Collins, S. β-Adrenoceptor signaling networks in adipocytes for recruiting stored fat and energy expenditure. Front. Endocrinol. 2, 102 (2011).
Low, L. A., Mummery, C., Berridge, B. R., Austin, C. P. & Tagle, D. A. Organs-on-chips: into the next decade. Nat. Rev. Drug Discov. 20, 345–361 (2021).
Slaughter, V. L. et al. Validation of an adipose-liver human-on-a-chip model of NAFLD for preclinical therapeutic efficacy evaluation. Sci. Rep. 11, 13159 (2021).
Shungin, D. et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 518, 187–196 (2015).
Locke, A. E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206 (2015).
Wang, K. et al. A genome-wide association study on obesity and obesity-related traits. PLoS ONE 6, e18939 (2011).
Warren, C. R. et al. Induced pluripotent stem cell differentiation enables functional validation of GWAS variants in metabolic disease. Cell Stem Cell 20, 547–557.e7 (2017).
Ding, Q. et al. A TALEN genome-editing system for generating human stem cell-based disease models. Cell Stem Cell 12, 238–251 (2013).
Aizarani, N. et al. A human liver cell atlas reveals heterogeneity and epithelial progenitors. Nature 572, 199–204 (2019).
Albhaisi, S. & Sanyal, A. J. Gene-environmental interactions as metabolic drivers of nonalcoholic steatohepatitis. Front. Endocrinol. 12, 665987 (2021).
Deczkowska, A. et al. XCR1(+) type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis. Nat. Med. 27, 1043–1054 (2021).
Seidman, J. S. et al. Niche-specific reprogramming of epigenetic landscapes drives myeloid cell diversity in nonalcoholic steatohepatitis. Immunity 52, 1057–1074.e7 (2020).
Lamas-Paz, A. et al. Alcoholic liver disease: utility of animal models. World J. Gastroenterol. 24, 5063–5075 (2018).
Frazier, T. H., Stocker, A. M., Kershner, N. A., Marsano, L. S. & McClain, C. J. Treatment of alcoholic liver disease. Ther. Adv. Gastroenterol. 4, 63–81 (2011).
Wang, S. et al. Human ESC-derived expandable hepatic organoids enable therapeutic liver repopulation and pathophysiological modeling of alcoholic liver injury. Cell Res. 29, 1009–1026 (2019).
Tsang, H. Y., Yi Lo, P. H. & Ho Lee, K. K. Generation of liver organoids from human induced pluripotent stem cells as liver fibrosis and steatosis models. bioRxiv https://doi.org/10.1101/2021.06.29.450347 (2021).
McCarron, S. et al. Functional characterization of organoids derived from irreversibly damaged liver of patients with NASH. Hepatology 74, 1825–1844 (2021).
Elbadawy, M. et al. Efficacy of primary liver organoid culture from different stages of non-alcoholic steatohepatitis (NASH) mouse model. Biomaterials 237, 119823 (2020).
Younossi, Z. M. et al. Global epidemiology of nonalcoholic fatty liver disease–meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64, 73–84 (2016).
Romeo, S. et al. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat. Genet. 40, 1461–1465 (2008).
Pingitore, P. et al. Human multilineage 3D spheroids as a model of liver steatosis and fibrosis. Int. J. Mol. Sci. 20, 1629 (2019).
Schwartz, B. E. et al. Discovery and targeting of the signaling controls of PNPLA3 to effectively reduce transcription, expression, and function in pre-clinical NAFLD/NASH settings. Cells 9, 2247 (2020).
Prill, S. et al. The TM6SF2 E167K genetic variant induces lipid biosynthesis and reduces apolipoprotein B secretion in human hepatic 3D spheroids. Sci. Rep. 9, 11585 (2019).
Tanaka, Y. et al. LPIAT1/MBOAT7 depletion increases triglyceride synthesis fueled by high phosphatidylinositol turnover. Gut 70, 180–193 (2021).
Bass, J. & Lazar, M. A. Circadian time signatures of fitness and disease. Science 354, 994–999 (2016).
Mure, L. S. et al. Diurnal transcriptome atlas of a primate across major neural and peripheral tissues. Science 359, eaao0318 (2018).
Yamada, R. G. & Ueda, H. R. The circadian clock ticks in organoids. EMBO J. 41, e110157 (2022).
Rosselot, A. E. et al. Ontogeny and function of the circadian clock in intestinal organoids. EMBO J. 41, e106973 (2022).
Mun, S. J. et al. Generation of expandable human pluripotent stem cell-derived hepatocyte-like liver organoids. J. Hepatol. 71, 970–985 (2019).
Morais, J. B. S. et al. Effect of magnesium supplementation on insulin resistance in humans: a systematic review. Nutrition 38, 54–60 (2017).
Romualdo, G. R. et al. Sorafenib reduces steatosis-induced fibrogenesis in a human 3D co-culture model of non-alcoholic fatty liver disease. Env. Toxicol. 36, 168–176 (2021).
Ranga, A., Gjorevski, N. & Lutolf, M. P. Drug discovery through stem cell-based organoid models. Adv. Drug Deliv. Rev. 69-70, 19–28 (2014).
Chen, Y. et al. Histone demethylase LSD1 promotes adipocyte differentiation through repressing Wnt signaling. Cell Chem. Biol. 23, 1228–1240 (2016).
Nie, B. et al. Brown adipogenic reprogramming induced by a small molecule. Cell Rep. 18, 624–635 (2017).
Qiu, Y. et al. Screening of FDA-approved drugs identifies sutent as a modulator of UCP1 expression in brown adipose tissue. EBioMedicine 37, 344–355 (2018).
Jing, R. et al. A screen using iPSC-derived hepatocytes reveals NAD(+) as a potential treatment for mtDNA depletion syndrome. Cell Rep. 25, 1469–1484.e5 (2018).
Haaker, M. W. et al. Identification of potential drugs for treatment of hepatic lipidosis in cats using an in vitro feline liver organoid system. J. Vet. Intern. Med. 34, 132–138 (2020).
Dawed, A. Y. et al. CYP2C8 and SLCO1B1 variants and therapeutic response to thiazolidinediones in patients with type 2 diabetes. Diabetes Care 39, 1902–1908 (2016).
Lauschke, V. M. & Ingelman-Sundberg, M. The importance of patient-specific factors for hepatic drug response and toxicity. Int. J. Mol. Sci. 17, 1714 (2016).
Soccio, R. E., Chen, E. R. & Lazar, M. A. Thiazolidinediones and the promise of insulin sensitization in type 2 diabetes. Cell Metab. 20, 573–591 (2014).
Sears, D. D. et al. Mechanisms of human insulin resistance and thiazolidinedione-mediated insulin sensitization. Proc. Natl Acad. Sci. USA 106, 18745–18750 (2009).
Hu, W. et al. Patient adipose stem cell-derived adipocytes reveal genetic variation that predicts antidiabetic drug response. Cell Stem Cell 24, 299–308.e6 (2019).
Oram, J. F. & Lawn, R. M. ABCA1. The gatekeeper for eliminating excess tissue cholesterol. J. Lipid Res. 42, 1173–1179 (2001).
Hu, W. et al. Individual-specific functional epigenomics reveals genetic determinants of adverse metabolic effects of glucocorticoids. Cell Metab. 33, 1592–1609.e7 (2021).
Group, R. C. et al. Dexamethasone in hospitalized patients with Covid-19. N. Engl. J. Med. 384, 693–704 (2021).
Drost, J. & Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 18, 407–418 (2018).
van de Wetering, M. et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell 161, 933–945 (2015).
Brooks, A. et al. Liver organoid as a 3D in vitro model for drug validation and toxicity assessment. Pharmacol. Res. 169, 105608 (2021).
Matsui, T. & Shinozawa, T. Human organoids for predictive toxicology research and drug development. Front. Genet. 12, 767621 (2021).
Kennedy, E. M. & Cullen, B. R. Gene editing: a new tool for viral disease. Annu. Rev. Med. 68, 401–411 (2017).
Roper, J. & Yilmaz, O. H. Breakthrough moments: genome editing and organoids. Cell Stem Cell 24, 841–842 (2019).
Melton, D. The promise of stem cell-derived islet replacement therapy. Diabetologia 64, 1030–1036 (2021).
Umekage, M., Sato, Y. & Takasu, N. Overview: an iPS cell stock at CiRA. Inflamm. Regen. 39, 17 (2019).
Wang, C. H. et al. CRISPR-engineered human brown-like adipocytes prevent diet-induced obesity and ameliorate metabolic syndrome in mice. Sci. Transl. Med. 12, eaaz8664 (2020).
Tsagkaraki, E. et al. CRISPR-enhanced human adipocyte browning as cell therapy for metabolic disease. Nat. Commun. 12, 6931 (2021).
Schene, I. F. et al. Prime editing for functional repair in patient-derived disease models. Nat. Commun. 11, 5352 (2020).
Zarrinpar, A. & Busuttil, R. W. Liver transplantation: past, present and future. Nat. Rev. Gastroenterol. Hepatol. 10, 434–440 (2013).
Cruz-Acuna, R. et al. PEG-4MAL hydrogels for human organoid generation, culture, and in vivo delivery. Nat. Protoc. 13, 2102–2119 (2018).
Broguiere, N. et al. Growth of epithelial organoids in a defined hydrogel. Adv. Mater. 30, e1801621 (2018).
Brassard, J. A. & Lutolf, M. P. Engineering stem cell self-organization to build better organoids. Cell Stem Cell 24, 860–876 (2019).
Hofer, M. & Lutolf, M. P. Engineering organoids. Nat. Rev. Mater. 6, 402–420 (2021).
Rogal, J. et al. Autologous human immunocompetent white adipose tissue-on-chip. Adv. Sci. 9, e2104451 (2022).
Rennert, K. et al. A microfluidically perfused three dimensional human liver model. Biomaterials 71, 119–131 (2015).
Lewis, P. L., Green, R. M. & Shah, R. N. 3D-printed gelatin scaffolds of differing pore geometry modulate hepatocyte function and gene expression. Acta Biomater. 69, 63–70 (2018).
Lu, X., Yang, J. & Xiang, Y. Modeling human neurodevelopmental diseases with brain organoids. Cell Regen. 11, 1 (2022).
Andersen, J. et al. Generation of functional human 3D cortico-motor assembloids. Cell 183, 1913–1929.e26 (2020).
Bauer, S. et al. Functional coupling of human pancreatic islets and liver spheroids on-a-chip: towards a novel human ex vivo type 2 diabetes model. Sci. Rep. 7, 14620 (2017).
Jin, Y. et al. Vascularized liver organoids generated using induced hepatic tissue and dynamic liver-specific microenvironment as a drug testing platform. Adv. Funct. Mater. 28, 1801954 (2018).
Viravaidya, K. & Shuler, M. L. Incorporation of 3T3-L1 cells to mimic bioaccumulation in a microscale cell culture analog device for toxicity studies. Biotechnol. Prog. 20, 590–597 (2004).
Miller, P. G. & Shuler, M. L. Design and demonstration of a pumpless 14 compartment microphysiological system. Biotechnol. Bioeng. 113, 2213–2227 (2016).
Acknowledgements
The authors thank M. Kim in the laboratory of M.A.L. for careful proofreading of the manuscript, and other members of the laboratories of M.A.L. and W.H. for valuable discussions and help with the drawing of the first drafts of the figures. The authors also acknowledge the many investigators who have contributed to this area of research and whose work, in many cases, could not be cited owing to the limitation of references allowed in this Review. M.A.L. was supported by the Cox Medical Institute, the JPB Foundation, as well as by National Institutes of Health grants (R01-DK049780). W.H. was supported by American Diabetes Association Training grant #1-18-PDF-132.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
M.A.L. is an advisory board member for Pfizer Inc., a consultant to Madrigal and Calico, and receives support from Pfizer for research not overlapping with the work reported here. W.H. declares no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks T. Sato and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Biobanking
-
A process of collecting and storing biological samples (usually human) for biomedical research.
- Embryoid bodies
-
Three-dimensional aggregates formed in suspension by pluripotent stem cells.
- Mesoderm
-
The middle layer of the three germ layers that develop during gastrulation, and gives rise to the musculoskeletal system, cardiovascular system and the connective tissues.
- Stromal vascular fraction
-
(SVF). A heterogeneous collection of adipose tissue-derived cells which have low fat content and contains mesenchymal stem cells in addition to other cell types.
- Endoderm
-
The inner layer of the three germ layers that develop during gastrulation, and gives rise to the digestive tube and the respiratory tube.
- Ex vivo normothermic perfusion
-
A technique that recreates circulation and allows the restoration of function in an organ prior to transplantation.
- Cholestasis
-
A liver disease characterized by the reduction or stoppage of bile flow.
- Anlages
-
Organs in their earliest stage of development.
- Enteroids
-
Organoids containing multiple intestinal epithelial cell types derived from the intestine.
- Entrainment
-
Adjustment of circadian oscillators to environmental signals.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, W., Lazar, M.A. Modelling metabolic diseases and drug response using stem cells and organoids. Nat Rev Endocrinol 18, 744–759 (2022). https://doi.org/10.1038/s41574-022-00733-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-022-00733-z
This article is cited by
-
Single-cell mapping of lipid metabolites using an infrared probe in human-derived model systems
Nature Communications (2024)