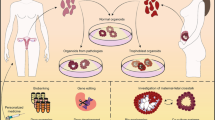
Abstract
Each month during a woman’s reproductive years, the endometrium undergoes vast changes to prepare for a potential pregnancy. Diseases of the endometrium arise for numerous reasons, many of which remain unknown. These endometrial diseases, including endometriosis, adenomyosis, endometrial cancer and Asherman syndrome, affect many women, with an overall lack of efficient or permanent treatment solutions. The challenge lies in understanding the complexity of the endometrium and the extensive changes, orchestrated by ovarian hormones, that occur in multiple cell types over the period of the menstrual cycle. Appropriate model systems that closely mimic the architecture and function of the endometrium and its diseases are needed. The emergence of organoid technology using human cells is enabling a revolution in modelling the endometrium in vitro. The goal of this Review is to provide a focused reference for new models to study the diseases of the endometrium. We provide perspectives on the power of new and emerging models, from organoids to microfluidics, which have opened up a new frontier for studying endometrial diseases.
Key points
-
Endometrial diseases, including Asherman syndrome, endometriosis, adenomyosis and endometrial cancer, represent a major health burden in reproductive-age women and do not have effective therapies.
-
Traditional in vitro culture techniques have long been used to study endometrial diseases, but they are limited by using single cell types and static cultures.
-
Emerging technologies, such as organoids and microphysiological systems, are physiologically relevant and can reproduce many characteristics of native tissues and disease states.
-
Although multiple microphysiological systems that model the endometrium have been developed, most of these have not yet been applied to endometrial diseases.
-
Further innovations in organoid and microfluidic models will enable more accurate endometrial disease modelling and drug testing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Critchley, H. O. D., Maybin, J. A., Armstrong, G. M. & Williams, A. R. W. Physiology of the endometrium and regulation of menstruation. Physiol. Rev. 100, 1149–1179 (2020).
Salamonsen, L. A., Hutchison, J. C. & Gargett, C. E. Cyclical endometrial repair and regeneration. Development 148, dev199577 (2021).
Jain, V., Chodankar, R. R., Maybin, J. A. & Critchley, H. O. D. Uterine bleeding: how understanding endometrial physiology underpins menstrual health. Nat. Rev. Endocrinol. 18, 290–308 (2022).
Conforti, A., Alviggi, C., Mollo, A., De Placido, G. & Magos, A. The management of Asherman syndrome: a review of literature. Reprod. Biol. Endocrinol. 11, 118 (2013).
Dreisler, E. & Kjer, J. J. Asherman’s syndrome: current perspectives on diagnosis and management. Int. J. Women’s Health 11, 191–198 (2019).
Chapron, C. et al. Diagnosing adenomyosis: an integrated clinical and imaging approach. Hum. Reprod. Update 26, 392–411 (2020).
Habiba, M., Gordts, S., Bazot, M., Brosens, I. & Benagiano, G. Exploring the challenges for a new classification of adenomyosis. Reprod. Biomed. Online 40, 569–581 (2020).
Bulun, S. E. Endometriosis. N. Engl. J. Med. 360, 268–279 (2009).
Kim, J. J. & Chapman-Davis, E. Role of progesterone in endometrial cancer. Semin. Reprod. Med. 28, 81–90 (2010).
Fitzgerald, H. C., Schust, D. J. & Spencer, T. E. In vitro models of the human endometrium: evolution and application for women’s health. Biol. Reprod. 104, 282–293 (2021).
Heremans, R., Jan, Z., Timmerman, D. & Vankelecom, H. Organoids of the female reproductive tract: innovative tools to study desired to unwelcome processes. Front. Cell Dev. Biol. 9, 661472 (2021).
Song, Y. & Fazleabas, A. T. Endometrial organoids: a rising star for research on endometrial development and associated diseases. Reprod. Sci. 28, 1626–1636 (2021).
Campo, H. et al. Microphysiological modeling of the human endometrium. Tissue Eng. Part A 26, 759–768 (2020).
Cadena, I., Chen, A., Arvidson, A. & Fogg, K. C. Biomaterial strategies to replicate gynecological tissue. Biomater. Sci. 9, 1117–1134 (2021).
Mancini, V. & Pensabene, V. Organs-on-chip models of the female reproductive system. Bioengineering 6, 103 (2019).
Young, R. E. & Huh, D. D. Organ-on-a-chip technology for the study of the female reproductive system. Adv. Drug Deliv. Rev. 173, 461–478 (2021).
Kelleher, A. M., DeMayo, F. J. & Spencer, T. E. Uterine glands: developmental biology and functional roles in pregnancy. Endocr. Rev. 40, 1424–1445 (2019).
Cooke, P. S., Spencer, T. E., Bartol, F. F. & Hayashi, K. Uterine glands: development, function and experimental model systems. Mol. Hum. Reprod. 19, 547–558 (2013).
Gray, C. A. et al. Developmental biology of uterine glands. Biol. Reprod. 65, 1311–1323 (2001).
Garcia-Alonso, L. et al. Mapping the temporal and spatial dynamics of the human endometrium in vivo and in vitro. Nat. Genet. 53, 1698–1711 (2021).
Wang, W. et al. Single-cell transcriptomic atlas of the human endometrium during the menstrual cycle. Nat. Med. 26, 1644–1653 (2020).
Yamaguchi, M. et al. Spatiotemporal dynamics of clonal selection and diversification in normal endometrial epithelium. Nat. Commun. 13, 943 (2022).
Yamaguchi, M. et al. Three-dimensional understanding of the morphological complexity of the human uterine endometrium. iScience 24, 102258 (2021).
Krikun, G., Mor, G. & Lockwood, C. The immortalization of human endometrial cells. Methods Mol. Med. 121, 79–83 (2006).
Kyo, S. et al. Successful immortalization of endometrial glandular cells with normal structural and functional characteristics. Am. J. Pathol. 163, 2259–2269 (2003).
Park, Y. et al. A novel human endometrial epithelial cell line for modeling gynecological diseases and for drug screening. Lab. Invest. 101, 1505–1512 (2021).
Yuhki, M., Kajitani, T., Mizuno, T., Aoki, Y. & Maruyama, T. Establishment of an immortalized human endometrial stromal cell line with functional responses to ovarian stimuli. Reprod. Biol. Endocrinol. 9, 104 (2011).
Bongso, A. et al. Establishment of human endometrial cell cultures. Hum. Reprod. 3, 705–713 (1988).
Michalski, S. A., Chadchan, S. B., Jungheim, E. S. & Kommagani, R. Isolation of human endometrial stromal cells for in vitro decidualization. J. Vis. Exp. 139, e57684 (2018).
Gellersen, B., Brosens, I. A. & Brosens, J. J. Decidualization of the human endometrium: mechanisms, functions, and clinical perspectives. Semin. Reprod. Med. 25, 445–453 (2007).
Gellersen, B. & Brosens, J. Cyclic AMP and progesterone receptor cross-talk in human endometrium: a decidualizing affair. J. Endocrinol. 178, 357–372 (2003).
Gellersen, B. & Brosens, J. J. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr. Rev. 35, 851–905 (2014).
Cloke, B. et al. The androgen and progesterone receptors regulate distinct gene networks and cellular functions in decidualizing endometrium. Endocrinology 149, 4462–4474 (2008).
Schindler, A. E. et al. Classification and pharmacology of progestins. Maturitas 61, 171–180 (2008).
Kim, J. J., Jaffe, R. C. & Fazleabas, A. T. Comparative studies on the in vitro decidualization process in the baboon (Papio anubis) and human. Biol. Reprod. 59, 160–168 (1998).
Kim, J. J. et al. Regulation of insulin-like growth factor binding protein-1 promoter activity by FKHR and HOXA10 in primate endometrial cells. Biol. Reprod. 68, 24–30 (2003).
Schäfer, W. R. et al. Critical evaluation of human endometrial explants as an ex vivo model system: a molecular approach. Mol. Hum. Reprod. 17, 255–265 (2011).
Blauer, M., Heinonen, P. K., Martikainen, P. M., Tomas, E. & Ylikomi, T. A novel organotypic culture model for normal human endometrium: regulation of epithelial cell proliferation by estradiol and medroxyprogesterone acetate. Hum. Reprod. 20, 864–871 (2005).
Schutte, S. C., James, C. O., Sidell, N. & Taylor, R. N. Tissue-engineered endometrial model for the study of cell–cell interactions. Reprod. Sci. 22, 308–315 (2015).
Zambuto, S. G., Clancy, K. B. H. & Harley, B. A. C. A gelatin hydrogel to study endometrial angiogenesis and trophoblast invasion. Interface Focus. 9, 20190016 (2019).
Li, S. & Ding, L. Endometrial perivascular progenitor cells and uterus regeneration. J. Pers. Med. 11, 477 (2021).
Sweeney, M. & Foldes, G. It takes two: endothelial-perivascular cell cross-talk in vascular development and disease. Front. Cardiovasc. Med. 5, 154 (2018).
Zhu, X. et al. Human endometrial perivascular stem cells exhibit a limited potential to regenerate endometrium after xenotransplantation. Hum. Reprod. 36, 145–159 (2021).
Turco, M. Y. et al. Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium. Nat. Cell Biol. 19, 568–577 (2017).
Boretto, M. et al. Development of organoids from mouse and human endometrium showing endometrial epithelium physiology and long-term expandability. Development 144, 1775–1786 (2017).
Fitzgerald, H. C., Dhakal, P., Behura, S. K., Schust, D. J. & Spencer, T. E. Self-renewing endometrial epithelial organoids of the human uterus. Proc. Natl Acad. Sci. USA 116, 23132–23142 (2019).
Wiwatpanit, T. et al. Scaffold-free endometrial organoids respond to excess androgens associated with polycystic ovarian syndrome. J. Clin. Endocrinol. Metab. 105, 769–780 (2020).
Rawlings, T. M. et al. Modelling the impact of decidual senescence on embryo implantation in human endometrial assembloids. eLife 10, e69603 (2021).
Cindrova-Davies, T. et al. Menstrual flow as a non-invasive source of endometrial organoids. Commun. Biol. 4, 651 (2021).
Filby, C. E. et al. Comparison of organoids from menstrual fluid and hormone-treated endometrium: novel tools for gynecological research. J. Pers. Med. 11, 1314 (2021).
Alzamil, L., Nikolakopoulou, K. & Turco, M. Y. Organoid systems to study the human female reproductive tract and pregnancy. Cell Death Differ. 28, 35–51 (2021).
Watson, D. E., Hunziker, R. & Wikswo, J. P. Fitting tissue chips and microphysiological systems into the grand scheme of medicine, biology, pharmacology, and toxicology. Exp. Biol. Med. 242, 1559–1572 (2017).
Marx, U. et al. Biology-inspired microphysiological systems to advance patient benefit and animal welfare in drug development. ALTEX 37, 365–394 (2020).
Regehr, K. J. et al. Biological implications of polydimethylsiloxane-based microfluidic cell culture. Lab Chip 9, 2132–2139 (2009).
Sung, J. H. et al. Recent advances in body-on-a-chip systems. Anal. Chem. 91, 330–351 (2019).
Edington, C. D. et al. Interconnected microphysiological systems for quantitative biology and pharmacology studies. Sci. Rep. 8, 4530 (2018).
Theobald, J. et al. Liver-kidney-on-chip to study toxicity of drug metabolites. ACS Biomater. Sci. Eng. 4, 78–89 (2018).
Shinha, K. et al. A kinetic pump integrated microfluidic plate (KIM-Plate) with high usability for cell culture-based multiorgan microphysiological systems. Micromachines 12, 1007 (2021).
Rogers, H. B. et al. Dental resins used in 3D printing technologies release ovo-toxic leachates. Chemosphere 270, 129003 (2021).
Gnecco, J. S. et al. Compartmentalized culture of perivascular stroma and endothelial cells in a microfluidic model of the human endometrium. Ann. Biomed. Eng. 45, 1758–1769 (2017).
Gnecco, J. S. et al. Hemodynamic forces enhance decidualization via endothelial-derived prostaglandin E2 and prostacyclin in a microfluidic model of the human endometrium. Hum. Reprod. 34, 702–714 (2019).
Ahn, J. et al. Three-dimensional microengineered vascularised endometrium-on-a-chip. Hum. Reprod. 36, 2720–2731 (2021).
De Bem, T. H. C., Tinning, H., Vasconcelos, E. J. R., Wang, D. & Forde, N. Endometrium on-a-chip reveals insulin- and glucose-induced alterations in the transcriptome and proteomic secretome. Endocrinology 162, bqab054 (2021).
Xiao, S. et al. A microfluidic culture model of the human reproductive tract and 28-day menstrual cycle. Nat. Commun. 8, 14584 (2017).
Park, S. R. et al. Development of a novel dual reproductive organ on a chip: recapitulating bidirectional endocrine crosstalk between the uterine endometrium and the ovary. Biofabrication 13, 015001 (2021).
Yu, D., Wong, Y. M., Cheong, Y., Xia, E. & Li, T. C. Asherman syndrome–one century later. Fertil. Steril. 89, 759–779 (2008).
Foix, A., Bruno, R. O., Davison, T. & Lema, B. The pathology of postcurettage intrauterine adhesions. Am. J. Obstet. Gynecol. 96, 1027–1033 (1966).
Schenker, J. G. & Margalioth, E. J. Intrauterine adhesions: an updated appraisal. Fertil. Steril. 37, 593–610 (1982).
Asherman, J. G. Amenorrhoea traumatica (atretica). J. Obstet. Gynaecol. Br. Emp. 55, 23–30 (1948).
Wang, J. et al. Application of bone marrow-derived mesenchymal stem cells in the treatment of intrauterine adhesions in rats. Cell Physiol. Biochem. 39, 1553–1560 (2016).
Gargett, C. E. & Healy, D. L. Generating receptive endometrium in Asherman’s syndrome. J. Hum. Reprod. Sci. 4, 49–52 (2011).
Cervello, I. et al. Human CD133+ bone marrow-derived stem cells promote endometrial proliferation in a murine model of Asherman syndrome. Fertil. Steril. 104, 1552–1560.e1–3 (2015).
Zhang, S., Li, P., Yuan, Z. & Tan, J. Platelet-rich plasma improves therapeutic effects of menstrual blood-derived stromal cells in rat model of intrauterine adhesion. Stem Cell Res. Ther. 10, 61 (2019).
Xu, L. et al. Umbilical cord-derived mesenchymal stem cells on scaffolds facilitate collagen degradation via upregulation of MMP-9 in rat uterine scars. Stem Cell Res. Ther. 8, 84 (2017).
Jing, Z., Qiong, Z., Yonggang, W. & Yanping, L. Rat bone marrow mesenchymal stem cells improve regeneration of thin endometrium in rat. Fertil. Steril. 101, 587–594 (2014).
Domnina, A. et al. Human mesenchymal stem cells in spheroids improve fertility in model animals with damaged endometrium. Stem Cell Res. Ther. 9, 50 (2018).
Jiang, X. et al. Endometrial membrane organoids from human embryonic stem cell combined with the 3D Matrigel for endometrium regeneration in asherman syndrome. Bioact. Mater. 6, 3935–3946 (2021).
de Miguel-Gomez, L. et al. Comparison of different sources of platelet-rich plasma as treatment option for infertility-causing endometrial pathologies. Fertil. Steril. 115, 490–500 (2021).
Liu, F. et al. Hyaluronic acid hydrogel integrated with mesenchymal stem cell-secretome to treat endometrial injury in a rat model of Asherman’s syndrome. Adv. Healthc. Mater. 8, e1900411 (2019).
Giudice, L. C. & Kao, L. C. Endometriosis. Lancet 364, 1789–1799 (2004).
Sampson, J. A. Metastatic or embolic endometriosis, due to the menstrual dissemination of endometrial tissue into the venous circulation. Am. J. Pathol. 3, 93–110.43 (1927).
Bulun, S. E. et al. Progesterone resistance in endometriosis: link to failure to metabolize estradiol. Mol. Cell Endocrinol. 248, 94–103 (2006).
Bulun, S. E. et al. Endometriosis. Endocr. Rev. 40, 1048–1079 (2019).
Yin, P. et al. Progesterone receptor regulates Bcl-2 gene expression through direct binding to its promoter region in uterine leiomyoma cells. J. Clin. Endocrinol. Metab. 92, 4459–4466 (2007).
Yin, X., Pavone, M. E., Lu, Z., Wei, J. & Kim, J. J. Increased activation of the PI3K/AKT pathway compromises decidualization of stromal cells from endometriosis. J. Clin. Endocrinol. Metab. 97, E35–E43 (2012).
Hey-Cunningham, A. J. et al. Angiogenesis, lymphangiogenesis and neurogenesis in endometriosis. Front. Biosci. 5, 1033–1056 (2013).
D’Hooghe, T. M., Bambra, C. S., Cornillie, F. J., Isahakia, M. & Koninckx, P. R. Prevalence and laparoscopic appearance of spontaneous endometriosis in the baboon (Papio anubis, Papio cynocephalus). Biol. Reprod. 45, 411–416 (1991).
MacKenzie, W. F. & Casey, H. W. Animal model of human disease. Endometriosis. Animal model: endometriosis in rhesus monkeys. Am. J. Pathol. 80, 341–344 (1975).
Gu, Z. Y., Jia, S. Z. & Leng, J. H. Establishment of endometriotic models: the past and future. Chin. Med. J. 133, 1703–1710 (2020).
Sulaiman, H., Dawson, L., Laurent, G. J., Bellingan, G. J. & Herrick, S. E. Role of plasminogen activators in peritoneal adhesion formation. Biochem. Soc. Trans. 30, 126–131 (2002).
Fasciani, A. et al. Three-dimensional in vitro culture of endometrial explants mimics the early stages of endometriosis. Fertil. Steril. 80, 1137–1143 (2003).
Esfandiari, N. et al. Expression of glycodelin and cyclooxygenase-2 in human endometrial tissue following three-dimensional culture. Am. J. Reprod. Immunol. 57, 49–54 (2007).
Esfandiari, N. et al. Effect of a statin on an in vitro model of endometriosis. Fertil. Steril. 87, 257–262 (2007).
Prechapanich, J. et al. Effect of a dienogest for an experimental three-dimensional endometrial culture model for endometriosis. Med. Mol. Morphol. 47, 189–195 (2013).
Esfandiari, N., Nazemian, Z. & Casper, R. F. Three-dimensional culture of endometrial cells: an in vitro model of endometriosis. Am. J. Reprod. Immunol. 60, 283–289 (2008).
Maas, J. W. M. et al. The chick embryo chorioallantoic membrane as a model to investigate the angiogenic properties of human endometrium. Gynecol. Obstet. Invest. 48, 108–112 (1999).
Nap, A. W. et al. Angiostatic agents prevent the development of endometriosis-like lesions in the chicken chorioallantoic membrane. Fertil. Steril. 83, 793–795 (2005).
Nap, A. W. et al. Oral contraceptives prevent the development of endometriosis in the chicken chorioallantoic membrane model. Contraception 78, 257–265 (2008).
Groothuis, P. G. et al. Adhesion of human endometrial fragments to peritoneum in vitro. Fertil. Steril. 71, 1119–1124 (1999).
Koks, C. A. M., Groothuis, P. G., Dunselman, G. A. J., Goeij, A. F. P. M. D. & Evers, J. L. H. Adhesion of shed menstrual tissue in an in-vitro model using amnion and peritoneum: a light and electron microscopic study. Hum. Reprod. 14, 816–822 (1999).
Witz, C. A., Monotoya-Rodriguez, I. A. & Schenken, R. S. Whole explants of peritoneum and endometrium: a novel model of the early endometriosis lesion. Fertil. Steril. 71, 56–60 (1999).
van der Linden, P. J. Q., Goeij, A. F. P. Md, Dunselman, G. A. J., Erkens, H. W. H. & Evers, J. L. H. Amniotic membrane as an in vitro model for endometrium–extracellular matrix interactions. Gynecol. Obstet. Invest. 45, 7–11 (1998).
Lee, Y. J. & Yi, K. W. Bone marrow-derived stem cells contribute to regeneration of the endometrium. Clin. Exp. Reprod. Med. 45, 149–153 (2018).
Chen, P., Mamillapalli, R., Habata, S. & Taylor, H. S. Endometriosis cell proliferation induced by bone marrow mesenchymal stem cells. Reprod. Sci. 28, 426–434 (2020).
Zhang, W. et al. 17β-estradiol promotes bone marrow mesenchymal stem cell migration mediated by chemokine upregulation. Biochem. Biophys. Res. Commun. 530, 381–388 (2020).
de Miguel-Gomez, L. et al. Stem cells and the endometrium: from the discovery of adult stem cells to pre-clinical models. Cells 10, 595 (2021).
Kucia, M. et al. Bone marrow as a home of heterogenous populations of nonhematopoietic stem cells. Leukemia 19, 1118–1127 (2005).
Symons, L. K. et al. The immunopathophysiology of endometriosis. Trends Mol. Med. 24, 748–762 (2018).
Mei, J., Chang, K. K. & Sun, H. X. Immunosuppressive macrophages induced by IDO1 promote the growth of endometrial stromal cells in endometriosis. Mol. Med. Rep. 15, 2255–2260 (2017).
Mei, J. et al. Indoleamine 2,3-dioxygenase-1 (IDO1) in human endometrial stromal cells induces macrophage tolerance through interleukin-33 in the progression of endometriosis. Int. J. Clin. Exp. Pathol. 7, 2743–2757 (2014).
Wang, Y. et al. Combined 17β-estradiol with TCDD promotes M2 polarization of macrophages in the endometriotic milieu with aid of the interaction between endometrial stromal cells and macrophages. PLoS ONE 10, e0125559 (2015).
Bongso, A., Fong, C. Y. & Zhao, H. Q. Effects of peritoneal macrophages from women with endometriosis on endometrial cellular proliferation in an in vitro coculture model. Fertil. Steril. 72, 533–538 (1999).
Shao, J. et al. Macrophages promote the growth and invasion of endometrial stromal cells by downregulating IL-24 in endometriosis. Reproduction 152, 673–682 (2016).
Chan, R. W. S., Lee, C. L., Ng, E. H. Y. & Yeung, W. S. B. Co-culture with macrophages enhances the clonogenic and invasion activity of endometriotic stromal cells. Cell Prolif. 50, e12330 (2017).
Zhou, W.-J., Hou, X.-X., Wang, X.-Q. & Li, D.-J. The CCL17- CCR4 axis between endometrial stromal cells and macrophages contributes to the high levels of IL-6 in ectopic milieu. Am. J. Reprod. Immunol. 78, e12644 (2017).
Mei, J., Zhou, W.-J., Li, S.-Y., Li, M.-Q. & Sun, H.-X. Interleukin-22 secreted by ectopic endometrial stromal cells and natural killer cells promotes the recruitment of macrophages through promoting CCL2 secretion. Am. J. Reprod. Immunol. 82, e13166 (2019).
Yu, J.-J. et al. IL15 promotes growth and invasion of endometrial stromal cells and inhibits killing activity of NK cells in endometriosis. Reproduction 152, 151–160 (2016).
Yang, H.-L. et al. The crosstalk between endometrial stromal cells and macrophages impairs cytotoxicity of NK cells in endometriosis by secreting IL-10 and TGF-β. Reproduction 154, 815–825 (2017).
Brueggmann, D. et al. Novel three-dimensional in vitro models of ovarian endometriosis. J. Ovarian Res. 7, 17 (2014).
Boretto, M. et al. Patient-derived organoids from endometrial disease capture clinical heterogeneity and are amenable to drug screening. Nat. Cell Biol. 21, 1041–1051 (2019).
Esfandiari, F. et al. Insight into epigenetics of human endometriosis organoids: DNA methylation analysis of HOX genes and their cofactors. Fertil. Steril. 115, 125–137 (2021).
Esfandiari, F. et al. Disturbed progesterone signalling in an advanced preclinical model of endometriosis. Reprod. Biomed. Online 43, 139–147 (2021).
Luddi, A. et al. Organoids of human endometrium: a powerful in vitro model for the endometrium–embryo cross-talk at the implantation site. Cells 9, 1121 (2020).
Stejskalova, A. et al. Collagen I triggers directional migration, invasion and matrix remodeling of stroma cells in a 3D spheroid model of endometriosis. Sci. Rep. 11, 4115 (2021).
Chen, Z. et al. Co-cultured endometrial stromal cells and peritoneal mesothelial cells for an in vitro model of endometriosis. Integr. Biol. 4, 1090–1095 (2012).
Chen, C. H. et al. Multiplexed protease activity assay for low-volume clinical samples using droplet-based microfluidics and its application to endometriosis. J. Am. Chem. Soc. 135, 1645–1648 (2013).
Altayyeb, A. et al. Characterization of mechanical signature of eutopic endometrial stromal cells of endometriosis patients. Reprod. Sci. 27, 364–374 (2020).
Kim, J. et al. Acquired contractile ability in human endometrial stromal cells by passive loading of cyclic tensile stretch. Sci. Rep. 10, 9014 (2020).
Harada, M. et al. Mechanical stretch stimulates interleukin-8 production in endometrial stromal cells: possible implications in endometrium-related events. J. Clin. Endocrinol. Metab. 90, 1144–1148 (2005).
Harada, M. et al. Mechanical stretch upregulates IGFBP-1 secretion from decidualized endometrial stromal cells. Am. J. Physiol. Endocrinol. Metab. 290, E268–E272 (2006).
Elad, D. et al. Tissue engineered endometrial barrier exposed to peristaltic flow shear stresses. Apl. Bioeng. 4, 26107 (2020).
Bulletti, C. et al. Characteristics of uterine contractility during menses in women with mild to moderate endometriosis. Fertil. Steril. 77, 1156–1161 (2002).
Yu, O. et al. Adenomyosis incidence, prevalence and treatment: United States population-based study 2006–2015. Am. J. Obstet. Gynecol. 223, 94.e1–94.e10 (2020).
An, M. et al. Different macrophages equally induce EMT in endometria of adenomyosis and normal. Reproduction 154, 79–92 (2017).
An, M. et al. Interaction of macrophages and endometrial cells induces epithelial–mesenchymal transition-like processes in adenomyosis. Biol. Reprod. 96, 46–57 (2017).
Wang, B., Yang, Y., Deng, X., Ban, Y. & Chao, L. Interaction of M2 macrophages and endometrial cells induces downregulation of GRIM-19 in endometria of adenomyosis. Reprod. BioMedicine Online 41, 790–800 (2020).
Mehasseb, M. K., Taylor, A. H., Pringle, J. H., Bell, S. C. & Habiba, M. Enhanced invasion of stromal cells from adenomyosis in a three-dimensional coculture model is augmented by the presence of myocytes from affected uteri. Fertil. Steril. 94, 2547–2551 (2010).
Taylor, A. H., Kalathy, V. & Habiba, M. Estradiol and tamoxifen enhance invasion of endometrial stromal cells in a three-dimensional coculture model of adenomyosis. Fertil. Steril. 101, 288–293 (2014).
Gnecco, J. S. et al. Physiomimetic models of adenomyosis. Semin. Reprod. Med. 38, 179–196 (2020).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 68, 7–30 (2018).
Lortet-Tieulent, J., Ferlay, J., Bray, F. & Jemal, A. International patterns and trends in endometrial cancer incidence, 1978–2013. J. Natl Cancer Inst. 110, 354–361 (2018).
Gallup, D. G. & Stock, R. J. Adenocarcinoma of the endometrium in women 40 years of age or younger. Obstet. Gynecol. 64, 417–420 (1984).
Cancer Genome Atlas Research, N. et al. Integrated genomic characterization of endometrial carcinoma. Nature 497, 67–73 (2013).
Van Nyen, T., Moiola, C. P., Colas, E., Annibali, D. & Amant, F. Modeling endometrial cancer: past, present, and future. Int. J. Mol. Sci. 19, 2348 (2018).
Skok, K. et al. Endometrial cancer and its cell lines. Mol. Biol. Rep. 47, 1399–1411 (2020).
Friel, A. M. et al. Mouse models of uterine corpus tumors: clinical significance and utility. Front. Biosci. 2, 882–905 (2010).
Girda, E., Huang, E. C., Leiserowitz, G. S. & Smith, L. H. The use of endometrial cancer patient-derived organoid culture for drug sensitivity testing is feasible. Int. J. Gynecol. Cancer 27, 1701–1707 (2017).
Maru, Y., Tanaka, N., Itami, M. & Hippo, Y. Efficient use of patient-derived organoids as a preclinical model for gynecologic tumors. Gynecol. Oncol. 154, 189–198 (2019).
Tamura, H. et al. Evaluation of anticancer agents using patient-derived tumor organoids characteristically similar to source tissues. Oncol. Rep. 40, 635–646 (2018).
Chitcholtan, K., Sykes, P. H. & Evans, J. J. The resistance of intracellular mediators to doxorubicin and cisplatin are distinct in 3D and 2D endometrial cancer. J. Transl. Med. 10, 38 (2012).
Bi, J. et al. Successful patient-derived organoid culture of gynecologic cancers for disease modeling and drug sensitivity testing. Cancers 13, 2901 (2021).
Chen, J. et al. An organoid-based drug screening identified a menin-MLL inhibitor for endometrial cancer through regulating the HIF pathway. Cancer Gene Ther. 28, 112–125 (2020).
Jeon, J. S. et al. Human 3D vascularized organotypic microfluidic assays to study breast cancer cell extravasation. Proc. Natl Acad. Sci. USA 112, 214–219 (2015).
Saha, B. et al. OvCa-Chip microsystem recreates vascular endothelium-mediated platelet extravasation in ovarian cancer. Blood Adv. 4, 3329–3342 (2020).
Liu, Y. & Cao, X. Characteristics and significance of the pre-metastatic niche. Cancer Cell 30, 668–681 (2016).
Laschke, M. W. & Menger, M. D. Basic mechanisms of vascularization in endometriosis and their clinical implications. Hum. Reprod. Update 24, 207–224 (2018).
Yetkin-Arik, B. et al. Angiogenesis in gynecological cancers and the options for anti-angiogenesis therapy. Biochim. Biophys. Acta Rev. Cancer 1875, 188446 (2021).
Campo, H. et al. Tissue-specific decellularized endometrial substratum mimicking different physiological conditions influences in vitro embryo development in a rabbit model. Acta Biomater. 89, 126–138 (2019).
Hernandez-Gordillo, V. et al. Fully synthetic matrices for in vitro culture of primary human intestinal enteroids and endometrial organoids. Biomaterials 254, 120125 (2020).
Alawadhi, F., Du, H., Cakmak, H. & Taylor, H. S. Bone marrow-derived stem cell (BMDSC) transplantation improves fertility in a murine model of Asherman’s syndrome. PLoS ONE 9, e96662 (2014).
Taniguchi, F., Wibisono, H., Mon Khine, Y. & Harada, T. Animal models for research on endometriosis. Front. Biosci. 13, 37–53 (2021).
Marquardt, R. M., Jeong, J. W. & Fazleabas, A. T. Animal models of adenomyosis. Semin. Reprod. Med. 38, 168–178 (2020).
Bruner-Tran, K. L., Mokshagundam, S., Herington, J. L., Ding, T. & Osteen, K. G. Rodent models of experimental endometriosis: identifying mechanisms of disease and therapeutic targets. Curr. Women’s Health Rev. 14, 173–188 (2018).
Bazoobandi, S. et al. Induction of Asherman’s syndrome in rabbit. J. Reprod. Infertil. 17, 10–16 (2016).
Feng, Q. et al. Establishment of an animal model of intrauterine adhesions after surgical abortion and curettage in pregnant rats. Ann. Transl. Med. 8, 56 (2020).
Merrill, J. A. Spontaneous endometriosis in the Kenya baboon (Papio doguera). Am. J. Obstet. Gynecol. 101, 569–570 (1968).
D’Hooghe, T. M. & Debrock, S. Endometriosis, retrograde menstruation and peritoneal inflammation in women and in baboons. Hum. Reprod. Update 8, 84–88 (2002).
Fazleabas, A. T., Brudney, A., Gurates, B., Chai, D. & Bulun, S. A modified baboon model for endometriosis. Ann. N. Y. Acad. Sci. 955, 308–317 (2002).
Vernon, M. W. & Wilson, E. A. Studies on the surgical induction of endometriosis in the rat. Fertil. Steril. 44, 684–694 (1985).
Kim, T. H. et al. Activated AKT pathway promotes establishment of endometriosis. Endocrinology 155, 1921–1930 (2014).
Eaton, J. L., Unno, K., Caraveo, M., Lu, Z. & Kim, J. J. Increased AKT or MEK1/2 activity influences progesterone receptor levels and localization in endometriosis. J. Clin. Endocrinol. Metab. 98, E1871–E1879 (2013).
Habiba, M. In Uterine Adenomyosis (eds Habiba, M. & Benagiano, G.) (Springer International Publishing, 2016).
Daikoku, T. et al. Conditional loss of uterine Pten unfailingly and rapidly induces endometrial cancer in mice. Cancer Res. 68, 5619–5627 (2008).
Maru, Y. & Hippo, Y. Two-way development of the genetic model for endometrial tumorigenesis in mice: current and future perspectives. Front. Genet. 12, 798628 (2021).
Moiola, C. P. et al. Patient-derived xenograft models for endometrial cancer research. Int. J. Mol. Sci. 19, 2431 (2018).
Collins, A. et al. Patient-derived explants, xenografts and organoids: 3-dimensional patient-relevant pre-clinical models in endometrial cancer. Gynecol. Oncol. 156, 251–259 (2020).
Bonazzi, V. F. et al. Patient-derived xenograft models capture genomic heterogeneity in endometrial cancer. Genome Med. 14, 3 (2022).
Unno, K. et al. Establishment of human patient-derived endometrial cancer xenografts in NOD scid gamma mice for the study of invasion and metastasis. PLoS ONE 9, e116064 (2014).
Deligdisch, L. Hormonal pathology of the endometrium. Mod. Pathol. 13, 285–294 (2000).
Guttinger, A. & Critchley, H. O. Endometrial effects of intrauterine levonorgestrel. Contraception 75, S93–S98 (2007).
Mihm, M., Gangooly, S. & Muttukrishna, S. The normal menstrual cycle in women. Anim. Reprod. Sci. 124, 229–236 (2011).
Taran, F. A., Stewart, E. A. & Brucker, S. Adenomyosis: epidemiology, risk factors, clinical phenotype and surgical and interventional alternatives to hysterectomy. Geburtshilfe Frauenheilkd. 73, 924–931 (2013).
Mowers, E. L. et al. Prevalence of endometriosis during abdominal or laparoscopic hysterectomy for chronic pelvic pain. Obstet. Gynecol. 127, 1045–1053 (2016).
Miyazaki, K. et al. Generation of progesterone-responsive endometrial stromal fibroblasts from human induced pluripotent stem cells: role of the WNT/CTNNB1 pathway. Stem Cell Rep. 11, 1136–1155 (2018).
Cheung, V. C. et al. Pluripotent stem cell-derived endometrial stromal fibroblasts in a cyclic, hormone-responsive, coculture model of human decidua. Cell Rep. 35, 109138 (2021).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Caroline Gargett, Virginia Pensabene and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Organoids
-
3D cellular aggregates that self-assemble and form structural units that partially resemble the organ in both structure and function.
- Microphysiological systems
-
Integrative, microfabricated platforms designed to recapitulate functional units of human organs in vitro; also known as organ-on-a-chip technology.
- Thermoplastics
-
A class of polymer that can be permanently deformed through the application of heat.
- Curettage
-
Scraping or removal of endometrial tissue for diagnostic or therapeutic purposes.
- Spheroids
-
3D, simple clusters of cells that lack self-assembly or organization.
- Sampson’s retrograde menstruation theory
-
Theory to explain the aetiology of endometriosis in which retrograde flow of sloughed endometrial cells during menstruation occurs through the fallopian tubes into the pelvic cavity promoting the establishment of ectopic lesions.
- Chorioallantoic membrane
-
(CAM). A membrane in bird eggs that is the site of gas exchange for the embryo.
- Transwell
-
A commercially available Boyden chamber device with a microporous membrane that inserts into a standard cell culture well and provides a second compartment.
- Micropatterned
-
Seeding and controlling the geometry and location of cells at the microscale level.
- Müllerian remnants
-
Pluripotent remains of the embryonic precursor to the uterus, which could undergo metaplasia into endometrial tissue during the development of adenomyosis.
- Biobanking
-
Creating a repository of biological samples for use in future research.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Murphy, A.R., Campo, H. & Kim, J.J. Strategies for modelling endometrial diseases. Nat Rev Endocrinol 18, 727–743 (2022). https://doi.org/10.1038/s41574-022-00725-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-022-00725-z
This article is cited by
-
Reversing Uteropathies Including Cancer-Like Changes in Mice by Transplanting Mesenchymal Stromal Cells or XAR Treatment
Stem Cell Reviews and Reports (2024)
-
Revolutionizing the female reproductive system research using microfluidic chip platform
Journal of Nanobiotechnology (2023)